Abstract
Objective: Valproate (VPA) is effectively used in the treatment of bipolar disorder, although the mechanism of action is unclear. In patients with bipolar disorder, 5-hydroxytryptamine (5-HT)–induced intraplatelet calcium (Ca) mobilization has been shown to be enhanced.
Methods: We examined the effect of VPA on 5-HT–induced Ca response in the platelets of normal subjects, in the presence of a calmodulin antagonist W-7 (N-[6-aminohexyl]-5-chloro-1-naphthalenesulfonamide), a protein kinase C (PKC) activator phorbol 12-myristate 13-acetate (PMA) or PKC inhibitors staurosporine and bisindolylmaleimide II.
Results: VPA inhibited the 5-HT–induced Ca response in a concentration-dependent manner. For calmodulin pathways, W-7 enhanced the 5-HT–stimulated Ca response, which was not affected by VPA. For PKC pathways, PMA reduced the Ca response, although both PKC inhibitors had no effect. PMA, staurosporine or bisindolylmaleimide II reversed the inhibitory effect of VPA on the Ca response, while W-7 did not modify it.
Conclusion: These findings suggest the possibility that the mechanism of action of VPA may be partly related to PKC signalling.
Introduction
Recent studies have suggested that abnormalities in intracellular signal transduction processes may be important in the etiology of bipolar disorder and that mood stabilizers such as lithium and valproate (VPA) have common effects on post-receptor functions, which may be responsible for mood stabilizing mechanisms.1,2 Probably reflecting postreceptor signal transduction changes, it has been repeatedly observed that serotonin 5-hydroxytryptamine (5-HT)–induced intraplatelet calcium (Ca) mobilization is enhanced in bipolar disorder.3–7 In recent years, VPA has been demonstrated to possess antimanic properties which, in the treatment of bipolar disorders, are obtained several days after VPA administration.8 VPA has acute effects on inhibitory and excitatory amino acid systems and plasma membrane ion channels in the central nervous system (CNS), which may be associated with the rapid anticonvulsant properties of the drug.9 However, the underlying mechanism for the antimanic effect of VPA is poorly understood.
Recent investigations into the pathophysiology of bipolar disorder have focused on the role of protein kinase C (PKC). Abnormal PKC activities in the platelets or brain from patients with bipolar disorder have been observed.10–12 Some researchers have demonstrated that chronic VPA alters the level and function of PKC in rat neuronal cultured cells and in the brain.13,14 Further, Suzuki and others15 reported the effect of the PKC inhibitor staurosporine on Ca response to 5-HT in platelets from patients with bipolar disorder. Although the 5-HT–induced Ca mobilization was significantly suppressed with the pretreatment of staurosporine in normal control subjects and patients with major depressive disorder, this inhibitory effect of staurosporine was not observed in patients with bipolar disorder. Kusumi and others16 have reported a significant relation between the enhanced Ca response to 5-HT and a good treatment response to such mood stabilizers as VPA or lithium in bipolar disorders. In this context, it is possible that the VPA mechanism(s) of action could be relevant to the mechanism(s) of enhanced Ca mobilization induced by 5-HT in the platelets of bipolar disorder patients, involving the PKC signalling pathway.
Recently we reported that lithium significantly decreased the enhanced Ca response to 5-HT in the presence of a myosin light chain kinase, Ca/calmodulin-dependent enzyme, inhibitor in the platelets of healthy subjects.17 These findings suggest that the enhanced Ca response observed in bipolar disorder may be relevant to the decreased function of calmodulin systems and that the mechanism of action of lithium may include a compensatory effect on calmodulin dysfunctions. We also found that VPA inhibited the 5-HT–induced intraplatelet Ca response in a concentration-dependent manner and exerted its effect for a short time (30 min).17 However, the mechanism of VPA rapid inhibitory effect on the 5-HT–induced Ca response has still been unclear. In this pilot study, to elucidate whether the effect of VPA on intracellular Ca mobilization is involved in calmodulin and PKC systems, we investigated the effect of VPA on 5-HT–induced Ca response in the presence of a calmodulin antagonist or PKC modulators in the platelets of healthy subjects.
Methods
We purchased 5-hydroxytryptamine (HT) creatinine sulfate N-(6-aminohexyl)-5-chloro-1-naphthalensulfonamide (W-7), phorbol 12-myristate 13-acetate (PMA) and bisindolylmaleimide II from Sigma Chemical Co. (St. Louis, Mo.). Sodium valproate was kindly provided by Kyowa Hakko Kogyo Co. (Tokyo, Japan), and we purchased fura-2/acetoxymethyl ester (fura-2/AM) from the Dojindo Laboratory (Kumamoto, Japan).
Measurement of intracellular Ca concentration with VPA
Subjects were 13 healthy male volunteers with a mean age of 31.9 (standard deviation [SD] 4.9) years, free of psychiatric and physical illness, not receiving calcium channel blocking medication, acetylasalicylic acid (ASA) or nonsteroidal anti-inflammatory drugs. All subjects gave written informed consent, which was approved by the Institutional Review Board in Hokkaido University Graduate School of Medicine. Blood samples were taken from the antecubital vein and were anti-coagulated with the addition of acid citrate dextrose (93 mM sodium citrate, 213 mM citric acid, 111 mM glucose). We used human platelets as a possible model of central neurons because they possess several neurotransmitter receptors whose stimulations are followed by intracellular signal transduction systems similar to the CNS.18 The isolation of platelets and the measurement of intracellular Ca concentration were performed as previously described.19 Briefly, platelet rich plasma (PRP) was obtained by twice centrifugation of blood at 200 × g for 10 minutes at room temperature. The PRP was divided into 4 or 5 aliquots and was incubated without VPA, or with one of 3 or 4 concentrations of VPA for 15 minutes at 37°C. Further incubation was then performed with 4 μM fura-2/acetoxymethyl ester for 15 minutes. Platelets were isolated from the PRP by further centrifugation at 600 × g for 15 minutes. The platelet pellet was suspended in Krebs-Ringer HEPES buffer (145 mM NaCl, 5 mM KCl, 1 mM MgSO4, 0.5 mM Na2HPO4, 6 mM glucose, 10mM HEPES, pH 7.4). After 1 mM CaCl2 was added to the suspended platelets, they were prewarmed in a cuvette at 37ºC for 4 minutes with or without a calmodulin antagonist W-7 (30 μM), a PKC activator PMA (1 nM), and PKC inhibitors staurosporine (10 nM) or bisindolylmaleimide II (500 nM). Ten μM 5-HT was added to the incubation medium, and fluorescence was measured on a Hitachi F-2000 fluorometer with excitation at 340 and 380 nm and with emission at 510 nm using a Ca2+-dye dissociation constant (Kd) of 224 nM. Intracellular Ca concentrations were calculated from the ratio of fluorescence intensities at 2 excitation wavelengths in the platelet samples, according to the method of Grynkiewicz and colleagues.20 Ca increase represents that initial peak minus resting level.
Statistics
Data are expressed as the mean (and standard error of the mean [SEM]). The effect of VPA concentrations on basal Ca level and 5-HT–induced Ca response were analyzed with a 1-way analysis of variance (ANOVA) for repeated-measures, followed by post-hoc Dunnett’s test. The effects of VPA and calmodulin/PKC modulators on 5-HT–induced Ca response were examined with a 2-way ANOVA for repeated-measures. The sphericity assumption of the covariance matrix was tested to examine whether a within-subjects test could be valid. When there was a significant interaction between the effect of VPA and the presence of a cal-modulin/PKC modulator, a 1-way ANOVA was used to examine the dose-dependent effect of VPA on the percent inhibition of Ca response by a calmodulin/PKC modulator. Significant differences among VPA concentrations were analyzed with post-hoc Dunnett’s test. A paired t test was used to assess the effect of calmodulin/PKC modulators on the 5-HT–induced Ca response in the absence of VPA.
Results
Effects of VPA (10–1000 μg/mL) on the 5-HT (10 μM)–induced Ca response were shown in Table 1. Although basal Ca concentration was unchanged, VPA significantly reduced the 5-HT–induced Ca response in a concentration-dependent manner (F4,48 = 64.424, p < 0.0001, n = 13, p < 0.05 for 50 μg/mL, 88.2% of control; p < 0.01 for 100 μg/mL, 80.4% of control; p < 0.01 for 1000 μg/mL, 40.7% of control).
Effects of valproate on 5-HT–induced intraplatelet Ca mobilization
Effects of VPA on 5-HT–induced Ca response in the presence of a calmodulin antagonist
In the absence of VPA, the 5-HT–induced Ca response pretreated with 30 μM W-7 was significantly enhanced, up to 147.8%, compared with the response without W-7 (Fig. 1; t [df 3] = 3.391, p < 0.05, n = 4). The enhanced effect of W-7 on the Ca response was not affected by VPA (10–100 μg/mL); that is, there was no significant interaction between the effect of VPA and the presence of W-7 (F3,12 = 0.155, p = 0.929, n = 4).
Effect of valproate (VPA) on 5-hydroxytryptamine (HT)-induced calcium (Ca) increase in the presence of W-7. Values are the mean and standard error of the mean. There was no significant interaction between the effect of VPA and W-7 on 5-HT–induced Ca response (F3,12 = 0.155, p = 0.929, n = 4).
Effects of VPA on 5-HT–induced Ca response with a PKC stimulator
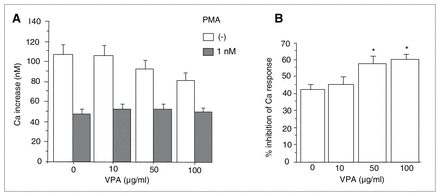
The pretreatment with 1 nM PMA significantly reduced the 5-HT–induced Ca response, compared with no pretreatment, in the absence of VPA (Fig. 2a; t [df 4] = 10.682, p < 0.001, n = 5, 44.8% of Ca increase without PMA). In the presence of PMA, varying VPA concentrations had no effect on the Ca response (Fig. 2a; F3,12 = 1.904, p = 0.18). In other words, pretreatment with PMA reversed the inhibitory effect of VPA on the 5-HT–induced Ca response. There was a significant interaction between the effect of VPA and the presence of PMA on the Ca response (Fig. 2a; F3,16 = 4.495, p = 0.0180, n = 5). To evaluate the dose-dependent effect of VPA on the inhibition of 5-HT–induced Ca response by PMA, we calculated percent inhibition of the Ca response by PMA, which showed Ca increase in the presence of PMA / in the absence of PMA × 100 (%) (Fig. 2b). VPA at 50 and 100 μg/mL significantly reversed the inhibition of 5-HT–induced Ca response by PMA (F3,16 = 8.265, p = 0.0015, n = 5; p < 0.05 v. without VPA).
Effect of valproate (VPA) on 5-hydroxytryptamine (HT)–induced calcium (Ca) increase in the presence of PMA (A). Values are the mean and standard error of the mean. There was a significant interaction between the effect of VPA and phorbol 12-myristate 13-acetate (PMA) on 5-HT–induced Ca response (F3,16 = 4,495, p = 0.018, n = 5). Effect of VPA on the percent inhibition of the Ca response by PMA (B). VPA at 50 and 100 μg/mL significantly reversed the inhibition of 5-HT-induced Ca response by PMA (F3,16 = 8.265, p = 0.0015, n = 5; p < 0.05 versus without VPA).
Effects of VPA on 5-HT–induced Ca response with PKC inhibitors
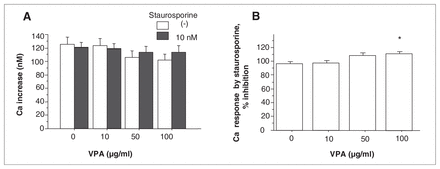
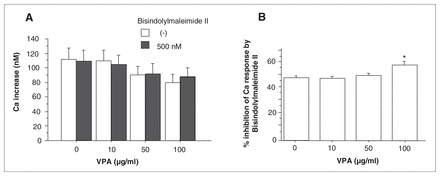
No remarkable change in the 5-HT–induced Ca increase was observed when pretreated with 10 nM staurosporine (Fig. 3a; t (df 4) = 1.532, p = 0.20, n = 5,) or 500 nM bisindolylmaleimide II (Fig. 4a; t [df 4] = 2.161, p = 0.10, n = 5) in the absence of VPA. There was a significant interaction between the effect of VPA and the presence of staurosporine on the Ca response (Fig. 3a; F3,16 = 5.246, p = 0.0103, n = 5). VPA at 100 μg/mL significantly increased the percent inhibition of the 5-HT–induced Ca response by staurosporine (Fig. 3b; F3,16 = 6.156, p = 0.0055, n = 5; p < 0.05 v. without VPA). Similar results were obtained for more specific PKC inhibitor, bisindolylmaleimide II. A significant interaction was observed between the effect of VPA and the presence of bisindolylmaleimide II on the Ca response (Fig. 4a; F3,16 = 3.260, p = 0.0390, n = 5). VPA at 100 μg/mL significantly increased the percent inhibition of the Ca response by bisindolylmaleimide II (Fig. 4b; F3,16 = 8.016, p = 0.0017, n = 5; p < 0.05 v. without VPA).
(A) Effect of VPA on 5-HT–induced calcium (Ca) increase in the presence of staurosporine. Values are the mean (and standard error of the mean) There was a significant interaction between the effect of VPA and staurosporine on 5-HT–induced Ca response (F3,16 = 5.246, p = 0.0103, n = 5). (B) Effect of VPA on the percent inhibition of the Ca response by staurosporine. VPA at 100 μg/mL significantly reversed the inhibition of 5-HT–induced Ca response by staurosporine (F3,16 = 6.156, p = 0.0055, n = 5; p < 0.05 v. without VPA).
Effect of valproate (VPA) on 5-hydroxytryptamine (HT)–induced calcium (Ca) increase in the presence of bisindolylmaleimide II (A). Values are the mean and standard error of the mean. There was a significant interaction between the effect of VPA and bisindolylmaleimide II on 5-HT–induced Ca response (F3,16 = 3.260, p = 0.0390, n = 5). Effect of VPA on the percent inhibition of the Ca response by bisindolylmaleimide II (B). VPA at 100 μg/mL significantly reversed the inhibition of 5-HT–induced Ca response by bisindolylmaleimide II (F3,16 = 8.016, p = 0.0017, n = 5; p < 0.05 versus without VPA).
Discussion
In the present study, we reconfirmed our previous finding17 that the therapeutic concentration of VPA inhibited the 5-HT–induced intraplatelet Ca response in a concentration-dependent manner without affecting basal concentration and exerted its effect for a short time (30 min). Although therapeutic concentration of VPA for bipolar disorders has not been established, some investigators suggest VPA could be efficacious at the serum concentration range of 50–100 μg/mL in patients with acute mania.21,22 This acute effect of VPA on the 5-HT–induced Ca response might be involved in the antimanic properties in the treatment of bipolar disorders, which are obtained several days after VPA administration.8
The 5-HT–induced Ca response was significantly enhanced by pretreatment with 30 μM W-7, which has a specific inhibitory effect on calmodulin.23 Although the mechanisms by which calmodulin antagonist increase the Ca mobilization have not been fully clarified, Ohkubo and others24 showed similar results: W-7 enhanced thromboxane A2-mediated Ca mobilization in rabbit platelets. In the present study, although the enhanced Ca response to 5-HT in the presence of W-7 was decreased when incubated in the presence of 50 or 100 μg/mL VPA, there was no significant interaction between the effect of VPA and the presence of W-7, suggesting that the acute effect of VPA on Ca response may be independent of a calmodulin system, in contrast with the effect of lithium.17 However, it is reported that VPA inhibits the cyclooxygenase pathway25,26 and the synthesis of thromboxane A225 in platelets and brains. Thus, it is possible that the interaction between W-7 and VPA on the Ca response may be cancelled by the mechanisms, via arachidonate cascade.
PMA (1 nM) alone attenuated the 5-HT–induced Ca response, consistent with a previous report27 showing that thrombin-induced intracellular Ca increase was suppressed in PMA-treated human platelets through the accumulation of the PIP2 and the reduction of IP3. These findings indicated that PKC would operate a negative feedback on the degeneration of inositol phospholipids. Therefore, our results might also might be owing to the negative feedback by PMA. In addition, pretreatment of PMA reversed the inhibitory effect of VPA on the 5-HT–induced Ca response, contrary to producing the additional inhibition of Ca response. That is, the degree of the reduced Ca response to 5-HT induced by a PKC activator was diminished by preincubation with VPA in a dose-dependent manner, whereas VPA itself significantly inhibited the 5-HT–induced Ca response. This indicates that VPA may have a counterbalancing effect on the excessive activation of PKC. However, the nonspecific PKC inhibitor staurosporine (10 nM) and the specific PKC inhibitor bisindolylmaleimide II (500 nM) did not modify the 5-HT–induced Ca response, congruent with the previous observation with another PKC inhibitor H-7.28 Regarding the effect of staurosporine on PI signalling, Watson and colleagues29 demonstrated that staurosporine (1 μM) had no effect on the formation of phosphatidic acid and inositol phosphates induced by thrombin in human platelets.
These findings strongly suggest that, in contrast with PMA, the inhibition of PKC either by staurosporine or bisindolylmaleimide II is not related to the receptor-mediated PI signalling leading to Ca mobilization. However, as with the combination of VPA and PMA, the pretreatment of staurosporine or bisindolylmaleimide II, which did not modify the Ca response alone, reversed the inhibitory effect of 100 μg/mL VPA on the 5-HT–induced Ca response. The interaction between the effect of VPA and the presence of PKC inhibitor suggests that the action of VPA on 5-HT–stimulated Ca mobilization is affected at least by PKC inhibition.
Taken together, the therapeutically effective dose of VPA attenuates the Ca response in an acute phase and may modulate PKC systems rather than calmodulin systems. However, the association between these 2 phenomena has not determined in this study. In light of the relation between treatment response to mood stabilizers and the Ca response, we reported in a longitudinal follow-up study that the patients with depression with enhanced Ca response to 5-HT exhibited a good response to mood stabilizers such as VPA and lithium in bipolar disorders. Therefore, further investigations, including an examination of the effect of VPA on the Ca response in platelets of patients with bipolar disorder, are needed to elucidate whether the mechanism of the acute effect of VPA on Ca mobilization is related to therapeutic actions as a mood stabilizer.
In conclusion, we demonstrated that VPA at clinical efficacious concentration itself reduced the 5-HT–induced intraplatelet Ca increase in normal subjects. Both a PKC activator and PKC inihibitors attenuated this inhibitory effect of VPA, while the enhanced effect of a calmodulin antagonist on the Ca response was not affected by VPA. The present findings suggest that the mechanism of the rapid action of VPA may be partly related to a PKC pathway, although there is a possibility that some other mechanisms may be involved.
Acknowledgements
This work was partly supported by grants-in-aid for Soul and diseases-of-the-nervous-system research (T. Koyama) from the Japanese Ministry of Health, Labor and Welfare, and for Scientific Research No. 17591192 (I. Kusumi) from the Japanese Ministry of Education, Culture, Sports, Science and Technology.
Footnotes
Medical subject headings: calcium; valproate; calmodulin; protein kinase c; serotonin; platelet.
Competing interests: None declared.
Contributors: Drs. Kusumi, Koyama and Suzuki designed the study. Drs. Akimoto and Suzuki participated in the acquisition of data, which Drs. Akimoto, Kusumi and Masui analyzed. Drs. Akimoto and Kusumi wrote the article, and Drs. Kusumi, Masui, Koyama and Suzuki critically reviewed it. All authors gave approval for the final version of the article to be published.
- Received April 8, 2006.
- Revision received June 9, 2006.
- Revision received August 15, 2006.
- Revision received August 21, 2006.
- Accepted August 21, 2006.