Abstract
Objective: Several magnetic resonance imaging (MRI) and postmortem studies have supported the role of the thalamus in the pathophysiology of schizophrenia. Interestingly, a recent small diffusion weighted imaging (DWI) study showed abnormal thalamic microstructure in patients with schizophrenia. The objective of our study was to use structural MRI and DWI to explore for the first time both thalamic volumes and integrity in schizophrenia.
Methods: We measured thalamic volumes and apparent diffusion coefficient (ADC) measures bilaterally in 71 patients with schizophrenia, representative of those living in the geographically defined catchment area of South Verona (i.e., 100 000 inhabitants), and 75 individuals without schizophrenia. The presence of the adhesio interthalamica was also detected.
Results: We found no significant differences in thalamus size between patients with schizophrenia and participants in the control group, with only a trend for decreased left volumes. No abnormal frequency of the adhesio interthalamica was found. In contrast, significantly increased thalamic ADC values were shown in schizophrenia patients. Age significantly inversely correlated with thalamic volumes in both groups and correlated positively with posterior ADCs in patients with schizophrenia. No significant associations between clinical variables and either volumes or ADC values were reported.
Conclusion: Widespread altered microstructure integrity and partially preserved thalamus size were found in schizophrenia patients. Therefore, subtle thalamic structural abnormalities are present in schizophrenia, even with maintained volumes. This may result from disruption at the cytoarchitecture level, ultimately supporting corticothalamic misconnection. Future imaging studies should further explore thalamic tissue coherence and its role for cognitive disturbances in patients at high risk for schizophrenia and in first-degree relatives.
Introduction
Schizophrenia is characterized by impairment of cognitive domains involved in higher cognitive functions such as thought processing and perception.1 It has been suggested that dysfunctional cortico–subcortical connections are involved in the pathophysiology and psychopathology of the disease.2,3 In this regard, the thalamus is a key region for frontotemporal communication and is crucial for modulating emotion and cognition in humans.4 Indeed, it is a major subcortical relay station that filters incoming primary sensory input and modulates processed cortical information through reciprocal corticothalamic connections.5 The thalamus is composed of several topographically organized nuclei, each with precise cortical projections and function.6,7 In particular, the anterior nuclei are connected to the limbic regions and play an important role in memory formation, learning and emotional experience. The medial group of nuclei project primarily to the prefrontal cortex, and lesions in these nuclei cause deficits in working and episodic memory. The lateral nuclei participate in higherorder somatosensory functions, whereas the posterior nuclei are important in the higher-order visual and auditory functions. Therefore, the thalamus is an important structure in the relay of sensory and motor information, maintaining consciousness and attention through reciprocal connections with the cerebral cortex, cerebellum and brain stem.8
The role of the thalamus for the pathophysiology of schizophrenia is supported by imaging and neuropathologic reports. Postmortem studies have reported reductions in neuronal and oligodendrocyte numbers in the thalamus of patients with schizophrenia.9–13 Also, functional neuroimaging investigations found thalamic hypoactivation during cognitive tasks involving working memory, attention and visual processing in patients with schizophrenia.14–19 Further, several neurochemical studies using proton magnetic resonance spectroscopy showed decreased thalamic concentrations of N-acetylaspartate, which is a putative marker of neuronal viability and function,20 in schizophrenia.21–25 Moreover, structural magnetic resonance imaging (sMRI) studies have found reduced thalamic volumes in schizophrenia.26–32 Conversely, several sMRI studies have found preserved thalamic size in schizophrenia.33–37 Although there are some controversial findings for volume abnormalities, these studies taken together strongly support the role of the thalamus for the pathophysiology of schizophrenia.4
Interestingly, a recent small diffusion weighted imaging (DWI) study showed abnormal thalamic microstructure organization in patients with schizophrenia, particularly in mediodorsal and anterior nuclei, as shown by increased mean diffusivity of water.38 By detecting the microscopic motion of water within the extracellular space, DWI incorporates diffusion gradient pulses within a conventional MR pulse sequence, providing important information on tissue microstructural integrity that cannot be visualized by conventional MRI.39 The apparent diffusion coefficient (ADC) quantifies water tissue movement, representing a critical measure for a detailed investigation of brain tissue organization and providing evidence of disruption when increased.40
The aim of our study was to combine volume and microstructure analysis of the thalamus by using structural MRI and DWI in a large sample of patients with schizophrenia recruited from the geographically defined catchment area of South Verona, Italy (i.e., 100 000 inhabitants). Our hypothesis was that ADC values would be altered in the thalamus of patients with schizophrenia, even in the absence of detectable thalamic volume changes.
Methods
Study population
We recruited 71 patients from the geographically defined catchment area of South Verona (i.e., 100 000 inhabitants). All had received a diagnosis of schizophrenia according to the criteria of the Diagnostic and Statistical Manual of Mental Disorders, fourth edition (DSM-IV)41 and were being treated by the South Verona Community-based Mental Health Service and by other clinics reporting to the South Verona Psychiatric Care Register42,43 (Table 1). We used the Item Group Checklist of the Schedules for Clinical Assessment in Neuropsychiatry (IGC-SCAN)44 to obtain the diagnosis of schizophrenia. This instrument was administered by research clinical psychologists who had extensive experience with it. They were required to show interrater reliability both blindly and independently with those of a senior investigator also trained in the instrument by achieving similar diagnoses for at least 8 of 10 assessments. Moreover, we compared the psychopathologic item groups completed by the raters to discuss any major symptom discrepancies. As well, we regularly ensured the reliability of the IGC-SCAN diagnoses by holding consensus meetings with treating psychiatrists and a senior investigator. The Italian version of the SCAN was edited by our group,45 and our investigators attended specific training courses to learn how to administer the IGC-SCAN. Subsequently, diagnoses for schizophrenia according to the DSMIV criteria were corroborated with the clinical consensus of 2 staff psychiatrists. We excluded patients with comorbid psychiatric disorders, epilepsy or other neurologic diseases, alcohol or substance abuse within the 6 months preceding the study or a history of traumatic head injury with loss of consciousness. All but 2 patients were receiving antipsychotic medications at the time of imaging: 24 patients were taking typical antipsychotic medication (14 haloperidol, 3 chlorpromazine, 2 fluphenazine, 2 clotiapine, 2 zuclopenthixol, 1 thioridazine), and 45 patients were taking atypical antipsychotic drugs (25 olanzapine, 10 clozapine, 8 risperidone, 2 quetiapine). We retrieved the patients’ clinical information from psychiatric interviews, the attending psychiatrist and medical charts. The Brief Psychiatric Rating Scale (24-item version)46 was used to characterize clinical symptoms. Again, it was administered by trained research clinical psychologists following the same reliability procedure as outlined above for the IGC-SCAN.
Characteristics of the study groups
We also recruited 75 individuals without schizophrenia (control group) (Table 1). They had no DSM-IV axis I disorders, as determined by an interview modified from the Structured Clinical Interview for DSM-IV nonpatient version (SCID-NP). As well, they had no history of psychiatric disorders among first-degree relatives, no history of alcohol or substance abuse and no current major medical illness. They were recruited from hospital/university staff volunteers or from individuals undergoing imaging for dizziness whose MRI showed no evidence of central nervous system abnormalities when reviewed by the neuroradiologist (R.C.). Any dizziness was due to benign paroxysmal positional vertigo or to nontoxic labyrinthitis. Participants in the control group were scanned only after a full medical history was taken and general neurologic, otoscopic and physical examinations were carried out; they had completely recovered from their dizziness. Also, none of these participants was taking medication, including drugs for nausea or vertigo, during the study.
This study was approved by the Biomedical Ethics Committee of the Azienda Ospedaliera of Verona. All participants provided signed informed consent after they understood all aspects of study participation.
MRI procedure
MRI scans were acquired with a 1.5 T Magnetom Symphony Maestro class syngo MR 2002B (Siemens), and in total, it took about 19 minutes to complete an MR session. A standard head coil was used for radio frequency transmission and reception of the MR signal, and restraining foam pads were used to minimize head motion. T1-weighted images were first obtained to verify the participant’s head position and image quality (TR = 450 ms, TE = 14 ms, flip angle = 90°, FOV = 230 × 230, 18 slices, slice thickness = 5 mm, matrix size = 384 × 512, NEX = 2). Proton density (PD)/T2–weighted images were then acquired (TR = 2500 ms, TE = 24/121 ms, flip angle = 180°, FOV = 230 × 230, 20 slices, slice thickness = 5 mm, matrix size = 410 × 512, NEX = 2) according to an axial plane running parallel to the anterior–posterior (AC–PC) commissures to exclude focal lesions. Subsequently, a coronal 3-dimensional magnetization prepared rapid gradient echo (MP-RAGE) sequence was acquired (TR = 2060 ms, TE = 3.9 ms, flip angle = 15°, FOV = 176 × 235, slice thickness = 1.25 mm, matrix size = 270 × 512, inversion time = 1100) to obtain 144 images covering the entire brain. Diffusion weighted echo-planar images were acquired in the axial plane parallel to the AC–PC line (TR = 3200 ms, TE = 94 ms, FOV = 230 × 230, 20 slices, slice thickness = 5 mm with 1.5 mm gap, matrix size = 128 × 128, echo train length = 5; these parameters were the same for b = 0, b = 1000, and the ADC maps). Specifically, 3 gradients were acquired in 3 orthogonal directions.
Anatomical image analysis
Anatomical imaging data were transferred to a PC workstation and analyzed with the BRAINS2 software developed at the University of Iowa. After the thalamus was identified in the coronal plane, its limits were manually traced.47,48 All thalamic measurements were done by a well-trained rater (O.K.) blinded to the participant’s identity. Interrater reliability of r = 0.96 for the right thalamus and r = 0.95 for the left thalamus was achieved. Thalamic volumes (mL) were obtained by summing the volumes of all relevant slices and were expressed in cubic centimetres.
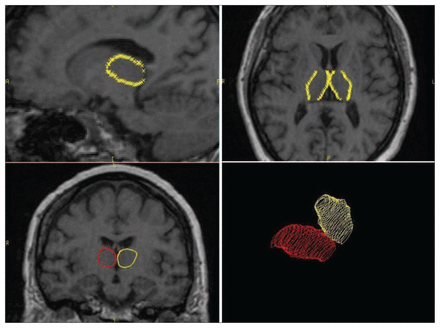
The tracing of the thalamus was performed on the T1-weighted MP-RAGE sequence, beginning at the coronal slice where the anterior pillars of the fornix merge into the mammillary bodies and continuing to the slice in which it was no longer possible to distinguish the thalamus from the surrounding brain matter. The lateral ventricles at the superior border, the red nucleus and the substantia nigra at the inferior border, the posterior limb of the internal capsule at the lateral border separating the thalamus from the adjacent lentiform nucleus and the third ventricle at the medial border demarcated the limits of the thalamus (Fig. 1). The presence of the adhesio interthalamica was also detected.
Anatomical landmarks of the thalamus for detection of volume. The tracing of the thalamus began at the coronal slice where the anterior pillars of the fornix merge into the mammillary bodies and continued to the slice in which it was no longer possible to distinguish the thalamus from the surrounding brain matter.
Intracranial volume (ICV) was traced in the coronal plane along the border of the brain and included the cerebrospinal fluid (CSF), dura mater, sinus, optic chiasma, brainstem, cerebral and cerebellar matter. The inferior border did not extend below the base of the cerebellum. The first and last 10 slices including brain matter were traced, after which 1 of every 5 slices was traced. ICV measurements were obtained by a well-trained rater who achieved interrater reliability of 0.97 (intraclass correlation coefficient), established by blindly tracing 10 randomly selected scans.
Diffusion image analysis
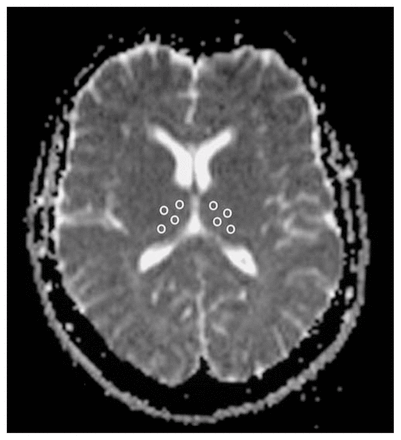
Images were displayed on a commercial Siemens workstation for the postprocessing analyses, including the calculation of ADC values. ADC maps were obtained from the diffusion images, with b = 1000, according to the following equation: –bADC = ln [A(b)/A(0)], where A(b) is the measured echo magnitude, b is the measure of diffusion weighting and A(0) is the echo magnitude without diffusion gradient applied.49 The resulting ADC was expressed in units of 10−5 mm2/s. Circular regions of interest (ROIs) standardized at 5 pixels (corresponding to an area of 0.16 cm2) were placed on both the right and left thalamus in 4 subregions (i.e., anterior, centromedial, centrolateral and posterior) on the non–diffusion weighted (b = 0) echoplanar images (Fig. 2). One axial slice at the level of the anterior and posterior horns of the lateral ventricles was chosen, and adjacent slices were checked to ensure that partial volume effects from CSF were minimized. The anterior, centromedial and posterior ROIs were placed following the medial limits of the thalamus, and the centrolateral ROI was placed close to the lateral border in front of the centromedial ROI. The 4 resulting thalamic subregions approximately corresponded to anatomical subdivisions of the thalamus (i.e., the anterior ROI to the anterior nuclei, the centromedial ROI to the dorsomedial nucleus, the centrolateral ROI to the ventrolateral nuclei and the posterior ROI to the pulvinar). The ROIs were then automatically transferred to the corresponding maps to obtain the ADC of water molecules. Two raters achieved high reliability, as defined by intraclass correlation coefficients over 0.85, established by tracing 8 training scans. Then, the same rater, blind to study hypotheses, group assignment and sociodemographic/clinical data, measured all scans.
Regions of interest (ROIs) placed in the thalamus for detection of the apparent diffusion coefficient (ADC). ROIs were placed in an axial slice at the level of the anterior and posterior horns of the lateral ventricles by following the medial limits of the thalamus for the anterior, centromedial and posterior ROIs. The centrolateral ROI was placed at the lateral border in front of the centromedial ROI.
Statistical analyses
All analyses were conducted with the SPSS for Windows software, version 11.0 (SPSS, Inc.); we set the 2-tailed statistical significance level at p < 0.05. We used a multivariate analysis of covariance (MANCOVA) to compare volumes and ADC values between patients with schizophrenia and participants in the control group, and we used Bonferroni adjustment for multiple comparisons. The assumption that the vector of the measures followed a multivariate normal distribution (Kolmogorov–Smirnov test) and the variancecovariance matrices for all cells were the same (Box M test) was verified. Pearson correlation and partial correlation analyses controlled for age were used to examine possible associations between age and clinical variables, respectively, and thalamic measures.
Results
Volumes and ADC measures
The assumptions for the MANCOVA were verified for both volumes and ADC values. Thalamic size slightly differed between patients with schizophrenia and individuals without schizophrenia (MANCOVA with age, sex and ICV as covariates; p = 0.053), with trends for significantly decreased volumes in the left side (Table 2). In both patients and participants in the control group, the right thalamus was larger than the left, this difference being statistically significant (Student paired t test, t70 = 3.18, p < 0.01; t74 = 2.35, p = 0.02, respectively). Accordingly, we found no significant group differences for the right-to-left volume difference (volumes of right minus left thalamus) and the laterality index (right minus left volumes, divided by the sum of right and left volumes) (MANCOVA with age and sex as covariates; p > 0.05). The adhesio interthalamica was absent in 2 of 75 individuals without schizophrenia (2.67%) and in 6 out of 71 patients with schizophrenia (8.45%), the between-group difference being nonsignificant (χ21 = 2.36, p = 0.12).
Volume and ADC measures for the thalamus in participants in the control group and in patients with schizophrenia
In contrast, patients with schizophrenia had significantly greater ADC values when compared with participants in the control group over all thalamic subregions (MANCOVA with age and sex as covariates; p = 0.03) (Table 2). After Bonferroni adjustment for multiple comparisons, right anterior, right centromedial, right posterior and left centrolateral ADC values were still significantly greater among patients when compared with participants in the control group (p = 0.012, p = 0.005, p = 0.002 and p = 0.05, respectively), with trends for significantly greater measures for right centrolateral and left posterior (p = 0.08 and p = 0.09, respectively). After Bonferroni adjustment, no significant differences were found for left anterior and left centromedial ADCs (p = 0.31 and p = 0.19, respectively).
When the 2 groups were stratified by sex, male patients had significantly greater ADC values in comparison with men in the control group (p < 0.05), with the exception of left anterior ADC values (p = 0.09); whereas female patients with schizophrenia had significantly greater ADC values for the right (p < 0.05) but not the left side (p > 0.05) compared with women in the control group (Student t test).
We also had similar findings when we separately compared schizophrenia patients with participants in the control group from hospital/university staff (n = 38) or with recovered dizziness (n = 37): in patients with schizophrenia, thalamic volumes were preserved and ADC measures increased. Moreover, we found no significant differences between the 2 subgroups of the control group for any thalamic values — either volumes (MANCOVA with age, sex and ICV as covariates; p = 0.86) or ADC measures (MANCOVA with age and sex as covariates; p = 0.21).
Age effects
We found significant inverse correlations between left and right thalamic volumes and age in both the control group (Pearson correlation coefficient = −0.37, p = 0.002; Pearson correlation coefficient = −0.40, p < 0.001, respectively) and patients with schizophrenia (Pearson correlation coefficient = −0.45, p < 0.001; Pearson correlation coefficient = −0.55, p < 0.001, respectively). Also, left and right posterior thalamic ADC values were positively associated with age in patients (Pearson correlation coefficient = 0.32, p = 0.007; Pearson correlation coefficient = 0.36, p = 0.002, respectively) but not in individuals without schizophrenia (Pearson correlation coefficient = 0.11, p = 0.39; Pearson correlation coefficient = 0.21, p = 0.09, respectively). All other thalamic ADC values did not significantly correlate with age in either group (Pearson’s correlation coefficient, p > 0.05).
Clinical variables
No significant associations were reported between clinical variables (age at onset, length of illness, number of prior hospitalizations, Brief Psychiatric Rating Scale scores, antipsychotic lifetime treatment) and thalamic volumes or ADCs (partial correlation controlled for age, p > 0.05). Compared with the control group, patients treated with either typical (n = 24) or atypical (n = 45) antipsychotic drugs had significantly greater ADC measures (MANCOVA with age and sex as covariates, p < 0.05), whereas they did not significantly differ for volumes (MANCOVA with age, sex and ICV as covariates, p > 0.05). We found no significant differences between patients on typical and atypical antipsychotics for either volumes or ADC measures (MANCOVA, p > 0.05).
Discussion
To the best of our knowledge, this is the first study combining the analyses of thalamic volumes and microstructural organization in schizophrenia and reporting widespread altered integrity, as shown by increased ADC values. On the contrary, partially preserved thalamic volumes, at least in the right side, and a normal pattern of right-greater-than-left asymmetry were found in patients with schizophrenia. Nonetheless, a trend for significantly decreased left thalamic volumes was shown, which may become significant with a larger sample. A recent study also showed abnormally increased water diffusivity within the thalamus in 12 patients with schizophrenia, particularly in mediodorsal and anterior nuclei.38 Consistent with our findings, preserved thalamic volumes were found in several prior MRI reports.50 Moreover, a review showed that only 42% of all MRI studies published from 1988 to 2001 found strong-to-moderate evidence of thalamic volume reduction in schizophrenia.51 These conflicting findings on thalamic volumes in schizophrenia may be due to heterogeneous patient populations and different methods of analysis. However, this study suggests that subtle changes in thalamic tissue composition in schizophrenia may not necessarily affect the structural gross volume and should be investigated at a cytoarchitecture level. In this regard, the DWI technique is instrumental. Indeed, it provides evidence of tissue disruption even when conventional volumetric quantification fails to detect size differences between patients and individuals without schizophrenia.52 Specifically, the ADC maps provide a relative representation of the diffusion coefficient in each pixel within the image, where low- and high-intensity values indicate, respectively, low and high microscopic water diffusion.49 In particular, increased ADC values reflect disruption of the composition of the extracellular interneuronal space, which may ultimately lead to altered neuronal and glial cross-talk mediated by neurotransmitters. It has been strongly suggested that abnormalities of neurotransmission systems, such as the dopamine, glutamate or γ-aminobutyric acidergic systems, are the core of the pathophysiology of schizophrenia. Potentially, alterations of thalamic microstructure organization may sustain such cellular communication deficits in schizophrenia in the context of the corticothalamic misconnection theory.53,54 This may ultimately result in impaired thalamic function and biochemical integrity, as shown by recent functional MRI and proton magnetic resonance spectroscopy studies.50
Interestingly, a significant inverse correlation between age and thalamic volumes bilaterally was found in this study for both patients and individuals without schizophrenia, suggesting a shrinkage of thalamic size with aging. Also, a negative effect of age on the integrity of the posterior thalamic subregion was shown in patients with schizophrenia, but not in participants in the control group. In our method, this area largely corresponds to the pulvinar nucleus, which is an association nucleus with reciprocal projections with the prefrontal, superior temporal and parietooccipital cortices.55,56 It participates in modulating cortical signal processing, specifically, visual and auditory attention.57 In this regard, pulvinocortical disconnections have been proposed to support visual attentional deficits in schizophrenia.58 Volume reductions and metabolic alterations of the pulvinar nucleus have also been reported in patients with schizophrenia,59–61 particularly in those with more severe disease.62,63 Thus the pulvinar is a key thalamic region for the pathophysiology of schizophrenia, and an aging effect on its microstructure, as suggested by this study, may contribute to compromising cortico–pulvinar–cortical connections in patients with schizophrenia.64,65
The adhesio interthalamica is a thin midline structure connecting the 2 thalami and containing several nuclei involved in attention. It has been suggested that the absence of this structure is a marker of early neurodevelopmental changes in schizophrenia. In our study, it was absent in about 3% of participants in the control group and in about 9% of patients; however, this difference was not statistically significant. At least 6 prior MRI studies evaluated the prevalence of absent adhesio interthalamica in schizophrenia patients, reporting both positive66–68 and negative findings.28,69,70 In particular, absence of adhesio interthalamica was more common in firstepisode patients when compared with the control group.68 Another study observed the lack of adhesio interthalamica in almost 11% of participants in the control group and in 18% of schizophrenia patients69 without, however, finding any statistically significant group differences. Further, a recent twin study did not observe any abnormal frequency of the adhesio interthalamica in schizophrenia.28 Our findings are in line with prior negative investigations showing that the prevalence of absent adhesio interthalamica, although often more frequent in patients with schizophrenia, did not significantly differ in comparison with the control group. Therefore, the literature only in part sustains the role of the absence of adhesio interthalamica for the pathophysiology of the disease, which may be relevant for the neurodevelopment of particular subgroups, such as female schizophrenia patients67 or patients with more severe negative symptoms.70
Limitations
This study has some limitations that should be considered. First, our sample largely comprised patients treated over the long-term. Thus whether thalamic disruption preceded the onset of the illness or appeared subsequently as a result of illness course or psychotropic treatment cannot be elucidated. However, clinical variables, including length of illness and antipsychotic lifetime administration, did not significantly affect ADC values, suggesting that disruption of thalamic integrity is independent of illness severity, chronicity or medication. Also, we recruited a larger number of patients who were well matched to participants in the control group, providing adequate power. Second, part of our control group was selected from persons undergoing MR scanning for dizziness, which may represent a methodological limitation. However, they were fully recovered at the time of imaging and had no evidence of central nervous system abnormalities on their scans; auditory evoked potentials were not investigated in these individuals. Third, no specific thalamic nuclei were investigated; therefore, we cannot exclude the possibility that volumes of particular subnuclei may actually differ between patients and the control group. Finally, the ROI positioning for detecting ADC values was based on a geometric anatomical division of the thalamus that only partially corresponds to thalamic nuclei. Finally, as a possible strength, it should be noted that our DWI protocol was short and clinically feasible in the real world of psychiatric practice. It could potentially be added to any conventional scan session as a complementary method for the evaluation of microstructure organization not detectable by sMRI. This would represent a clinical and scientific advantage, according to the evidence for impaired thalamic and white matter communication in patients with schizophrenia.71–73
In conclusion, this study found widespread disrupted thalamic cytoarchitecture in the presence of preserved volumes in a large sample of patients with schizophrenia who were representative of those living in the geographically defined catchment area of South Verona (i.e., 100 000 inhabitants). Abnormalities of thalamic microstructure in schizophrenia may represent bizarre organization at the cellular level, resulting in perturbation of corticothalamic connectivity and being relevant for the cognitive disturbances reported in patients with this disorder. Future diffusion imaging studies should further explore thalamic coherence in patients at high risk for schizophrenia and in first-degree relatives.
Acknowledgements
Preliminary findings of this work were presented at the 60th Society of Biological Psychiatry Meeting; 2004 April 29–May 1; New York (NY). This work was partly supported by grants from the American Psychiatric Institute for Research and Education (APIRE Young Minds in Psychiatry Award), the Italian Ministry for Education, University and Research (PRIN n. 2005068874), the Veneto StartCup 2007 to Dr. Brambilla and a grant from Regione Veneto, Italy, (159/03, DGRV n. 4087).
Footnotes
Medical subject headings: psychotic disorders; magnetic resonance; neurosciences.
Competing interests: None declared.
Contributors: Drs. Agarwal, Pozzi Mucelli, Tansella and Brambilla designed the study. Mr. Rambaldelli and Drs. Perlini, Bellani, Cerini, Isola, Versace and Gasparini acquired the data. Drs. Dusi, Kitis, Balestrieri and Brambilla analyzed the data. Drs. Agarwal and Brambilla wrote the article. Mr. Rambaldelli and Drs. Perlini, Dusi, Kitis, Bellani, Cerini, Isola, Versace, Balestrieri, Gasparini, Pozzi Mucelli, Tansella and Brambilla reviewed the article. All authors gave final approval for publication.
- Received July 2, 2007.
- Revision received November 2, 2007.
- Revision received January 10, 2008.
- Accepted January 11, 2008.