Abstract
Background: Auditory verbal hallucinations (AVHs) are a core symptom of schizophrenia. Previous reports on neural activity patterns associated with AVHs are inconsistent, arguably owing to the lack of an adequate control group (i.e., patients with similar characteristics but without AVHs) and neglect of the potential confounding effects of medication.
Methods: The current study was conducted in a homogeneous group of patients with schizophrenia to assess whether the presence or absence of AVHs was associated with differential regional cerebral glucose metabolic patterns. We investigated differences between patients with commenting AVHs and patients without AVHs among a group of dextral antipsychotic-naive inpatients with acute first-episode schizophrenia examined with [18F]fluorodeoxyglucose positron emission tomography (FDG-PET) at rest. Univariate and multivariate approaches were used to establish between-group differences.
Results: We included 9 patients with AVHs and 7 patients without AVHs in this study. Patients experiencing AVHs during FDG uptake had significantly higher metabolic rates in the left superior and middle temporal cortices, bilateral superior medial frontal cortex and left caudate nucleus (cluster level p < 0.005, family wise error–corrected, and bootstrap ratio > 3.3, respectively). Additionally, the multivariate method identified hippocampal–parahippocampal, cerebellar and parietal relative hypoactivity during AVHs in both hemispheres (bootstrap ratio < −3.3).
Limitations: The FDG-PET imaging technique does not provide information regarding the temporal course of neural activity. The limited sample size may have increased the risk of false-negative findings.
Conclusion: Our results indicate that AVHs in patients with schizophrenia may be mediated by an alteration of neural pathways responsible for normal language function. Our findings also point to the potential role of the dominant caudate nucleus and the parahippocampal gyri in the pathophysiology of AVHs. We discuss the relevance of phenomenology-based grouping in the study of AVHs.
Introduction
Auditory verbal hallucinations (AVHs) are defined as perceptions of verbal material in the auditory modality in the absence of corresponding external stimuli.1 They are a core symptom of schizophrenia, reported in 60%–80% of patients, often resulting in notable distress and potentially harmful behaviour to the patient and others. Despite their prevalence, the neural correlates of AVHs are not yet fully understood. Elucidating the pathophysiologic basis of AVHs is important for the understanding of neurobiologic processes underlying schizophrenia and for the development of more effective treatment strategies for patients who are unresponsive to conventional therapy.
Over the last few decades, neuroimaging research has attempted to distinguish the neural mechanism involved in the development of AVHs in patients with schizophrenia. Volumetric studies have consistently shown that severity of AVHs is associated with reduced relative volume in the left auditory cortex, left amygdala, left insula and prefrontal cortex.2–8 In parallel, functional neuroimaging studies have uncovered an extensive network of areas associated with the hallucinatory state itself. Despite considerable variability among studies, the general picture that hallucinating patients display relative hyperactivity in bilateral frontotemporal, cerebellar and thalamostriatal networks has emerged.9 Importantly, most of these regions have also been implicated in language perception, processing and production, both of inner and overt speech. One potential pathway to disrupted language function, and possibly AVHs, is proposed to result from alterations in frontotemporal white matter tracts. Favouring this hypothesis, diffusion tensor imaging research in auditory hallucinators has confirmed such anomalies in some of the most important frontotemporal connections.10,11
Relevant to the study of hallucinations, AVHs are highly heterogeneous from a linguistic point of view. Although it is assumed that different subtypes of AVHs would share a common final pathway,12 neural substrates involved in AVHs may actually be a function of their phenomenologic variants,13,14 given that normal brain activation patterns vary with linguistic complexity.15,16 Cluster analyses of the characteristics of AVHs have given support to this notion by showing that high linguistic complexity may share neural commonality with hearing multiple voices and hallucinations with systematized content.13 We propose that studying a specific subgroup of hallucinators, based on phenomenologic dimensions of AVH, may be valuable in reducing the underlying neural variability associated with this symptom and resolving some inconsistencies attributable to heterogeneous sampling in previous research. In the present study we only included hallucinating patients who had prominent and frequent AVHs with common phenomenologic features (i.e., complex commenting hallucinatory voices).
We also sought to address several other problems in the limited literature assessing the hallucinatory state itself. To our knowledge, no previous studies on AVH have solely employed a sample with never-treated first-episode schizophrenia, and as a result the studies were unable to rule out the potential effects of medication. Additionally, a within-group (before–after) design comparing presence and remission of AVHs has been the most widely used method in positron emission tomography (PET) studies and has been previously used by our group.17 This approach, however, does not allow for distinguishing between generation and remission mechanisms of symptoms and cannot ascribe findings to AVHs, considering the parallel progression of other psychotic symptoms and the confounding effects of medication. Linked to this prevalent design is perhaps the biggest limitation of previous research: the general lack of a matched comparison group of patients with schizophrenia but without AVHs. Therefore, the motivation behind this study was to provide a direct between-group comparison of patients with acute schizophrenia with and without AVHs as a more suitable approach in overcoming the aforementioned confounds and more accurately characterizing the neural correlates of AVHs.
Taking these considerations into account, we aimed to investigate the regional differences in neural activity between auditory hallucinators and nonhallucinators among never-medicated first-episode patients in an attempt to elucidate the pathophysiology specific to patients with schizophrenia with AVHs.
Methods
Patients
We recruited antipsychotic-naive inpatients with first-episode schizophrenia, with and without AVHs, at the Hospital Clinic of Barcelona. Diagnoses were established using the Structured Clinical Interview (SCID) for DSM-IV Axis I Disorders.18 The patients with AVHs were part of a within-group study reported elsewhere.17 We also included a group of age- and sex-matched healthy controls. All participants were screened with medical history, physical examination, structural T1- and T2-weighted magnetic resonance imaging (MRI) and laboratory testing. Exclusion criteria were history of head trauma, neurologic disorders, DSM-IV criteria for alcohol dependence and a positive urine test for illicit drugs on the day of admission to our hospital. Hallucinating patients were included if they reported experiencing prominent and frequent AVHs and were able to describe these experiences. All participants in the AVH group were interviewed again after scan acquisition to confirm the presence of AVHs during fluorodeoxyglucose (FDG) uptake. Based on the clinical judgment of at least 2 experienced psychiatrists (E.P., E.F.-E.), we considered patients who had never reported experiencing perceptive alterations in the auditory modality or manifested any hallucinatory behaviour, such as soliloquy or unmotivated laughter, to be nonhallucinators. Clinical assessment was based on several comprehensive clinical interviews and complete anamnesis recounted by patients and their relatives, along with behavioural observation upon admission to the hospital. A negative pregnancy test was obtained from all women before PET acquisition. After individual appraisal of the capacity to consent, all patients provided written informed consent to participate in the study, which was approved by the ethics committee of our hospital (Comité Ético de Investigación Clínica, Hospital Clínic de Barcelona).
Psychopathology scales
Clinical assessment was performed with the Positive and Negative Syndrome Scale (PANSS),19 the Clinical Global Impression Scale (CGI)20 and the Comprehensive Assessment of Symptoms and History (CASH).21 We asked all hallucinating patients about characteristics of AVHs they had experienced during the 30 minutes after FDG injection. Frequency and formal features of AVHs were rated immediately before data acquisition using the psychotic symptom rating scale (PSYRATS).22,23 No patients were tagged as outliers after an individual assessment of scale scores.
Acquisition and reconstruction of PET data
All patients fasted for at least 4 hours before intravenous injection of [18F]-FDG (4.7 MBq/kg of body weight). At the moment of injection, they all had a blood glucose concentration below 130 mg/dL. For the AVH group, FDG was administered when the patient demonstrated hallucinatory behaviour. From at least 10 minutes before FDG administration to 30 minutes after, all patients were silent and rested in a dark room to avoid visual stimulation. Image acquisition began 35 minutes after FDG injection using a PET scanner (Advance NXi; GE Healthcare). A 1-minute emission scan was performed to verify the correct brain position in the field of view (14.5 cm). A 20-minute emission scan and a 7-minute transmission data scan were then acquired using a 2-dimensional mode and 283 × 336 matrix. The slice thickness was 5 mm. Image reconstruction was performed on a SUN workstation (SUN Microsystems Mountain View) using a 128 × 128 × 35 matrix, with a voxel size of 4.29 × 4.29 × 4.25 mm3. We used the ordered-subsets expectation maximization algorithm with 28 subsets and 2 iterations for the reconstruction. Attenuation correction was performed using the acquired transmission data.
Data processing and statistical analysis
Univariate analysis — parametric inference
Preprocessing and statistical analysis of PET images were performed using SPM5 (Statistical Parametric Mapping 5, Well-come Trust Centre for Neuroimaging; www.fil.ion.ucl.ac.uk/spm). Preprocessing included spatial normalization into Montreal Neurological Institute (MNI) space and smoothing with a Gaussian kernel (12 mm full-width at half-maximum). The primary aim of the study was to establish differences in relative glucose metabolic rate (rGMR) between the AVH and non-AVH groups using a voxel-wise 2-sample t test for independent groups. We assumed unequal variance of measurements. Proportional scaling was applied to fit the mean global activity of grey matter to 50 mL/dL/minute for each scan. We used a relative proportional threshold masking of 0.8. This analysis was also repeated using global activity as a covariate in a general linear model analysis of covariance (ANCOVA), without scaling the images, to assess the potential impact of global normalization on results.24,25 The resulting t statistic image was thresholded at t14 > 3.787 (p < 0.001, uncorrected) at the voxel level, with a minimum cluster size of 20 adjacent voxels. Only clusters significant at p < 0.05 (family wise error–corrected) according to Gaussian random field theory are reported.26 A multiple regression model with 3 dummy regressors (representing the AVH, non-AVH and healthy control groups) and an intercept was also estimated. An AVH > healthy controls comparison was created and used to mask the results of the AVH > non-AVH comparison. Because of the increase in degrees of freedom in this model, results were thresholded using a more stringent false discovery rate (FDR) q at 0.05. We performed whole-brain and region-of-interest (ROI) analyses to correlate rGMR with severity of hallucinations in the AVH group using simple linear regression and the PSYRATS total score as a variable of interest. The bilateral superior temporal and inferior frontal cortices, regions previously associated with severity of AVHs,9 were selected a priori as separate ROIs for this purpose. We performed ROI analyses using WFU PickAtlas Tool, version 2.4.27,28
Multivariate analysis — nonparametric inference
In addition to the univariate approach, we sought to cross-validate our results with a partial least squares (PLS) analysis. Partial least squares analysis is similar to principal components analysis; the difference is that the PLS solutions are constrained to a part of the data related to experimental manipulations or effects of interest (i.e., the covariance matrix).29 The analysis decomposes this covariance matrix into latent variables (LVs), each of which is a linear combination of the observed data (with specific weights given to the elements —voxels — comprising the data) that explains a certain percentage of the covariance.30 Images were preprocessed as described above and analyzed using PLS software (www.rotman-baycrest.on.ca/pls, version 5.1003291). We used a mean-centering approach to assess between-group differences in rGMR. Statistical significance of LVs was assessed by means of a permutation test without replacement using 1000 permutations, and the reliability of the contribution of each voxel salience (weight for a given LV) was determined using bootstrap estimates of the salience standard errors by obtaining 1000 bootstrap samples.31 The bootstrap ratio (BSR) is the ratio of the voxel salience to its estimated standard error. We report LVs with a significance of p < 0.05 based on permutation testing and voxels surviving a set threshold of BSR ± 3.3, which is roughly equivalent to a 99.9% confidence interval (CI).
Results
Participants
We recruited 18 right-handed antipsychotic-naive inpatients with first-episode schizophrenia. Of these, 11 reported prominent AVHs and 7 had a clear absence of past or current AVHs. Of the 11 participants in the AVH group, 2 were excluded from further analyses owing to a failure to hallucinate during the scanning process. The 16 patients included in the analyses had a mean age of 26.62 (standard deviation [SD] 5.50) years and 43.75% were women. We included 8 age- and sex-matched healthy controls in the study (mean age 28.14 [SD 4.63] yr, and 50% were women).
There were no significant differences between the AVH and non-AVH groups in sociodemographic characteristics. The percentage of bilingual participants was 66.66% in the AVH group and 71.42% in the non-AVH group. Across all bilingual participants, 81.81% were proficient in Spanish and Catalan, whereas the remaining 2 were proficient in Spanish and either Portuguese or Arabic, respectively. No statistically significant clinical differences other than scores in the hallucinatory behaviour item on the PANSS were found between the AVH and non-AVH groups. The PANSS scores for all patients are reported in Table 1. After a minimum 1-year follow-up period in our outpatient clinic, the diagnosis of schizophrenia was confirmed in all patients.
Sociodemographic and clinical characteristics of the study sample
Clinical features of the AVH group
Table 2 reports the PSYRATS mean scores for each domain for the AVH group. The CASH interview showed that 88.88% of the patients reported marked (“clear evidence of voices that occur frequently”) to severe (“clear evidence of voices that occur almost daily”) auditory hallucinations. All of the patients reported commenting hallucinatory voices, and some of them (55.55%) perceived conversing voices as well. All presented with hallucinations of critical/derogatory content, which alternated with complimenting and commanding voices in some cases. All patients reported that their hallucinations had a substantial impact on their actions. The proportion of patients who attributed hallucinatory voices to an external source was similar to that of patients who believed the voices originated internally. None reported types of hallucinations other than auditory ones during FDG uptake. At follow-up, there was complete remission of hallucinations following treatment with risperidone (4–9 mg/d) in all patients.
Differences in rGMR between AVH and non-AVH groups
Univariate analysis — parametric inference
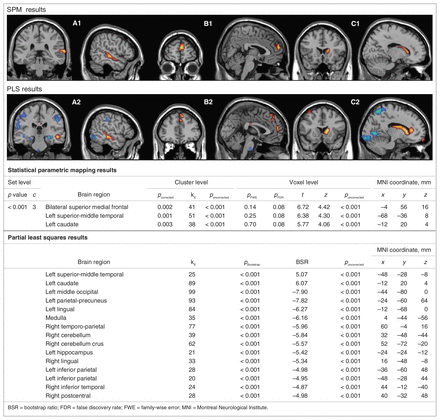
Patients with AVHs showed significantly higher rGMR in the left superior middle temporal cortices, bilateral superior medial frontal cortex and left caudate nucleus than those without AVHs (Fig. 1). There were no regions of significantly increased rGMR in the non-AVH compared with the AVH group at the set threshold. We observed an additional cluster of higher rGMR in the left anterior cingulate cortex in patients with AVHs when we reduced the voxel-level threshold to p < 0.01 on an exploratory basis (cluster-level p = 0.001, corrected for multiple comparisons, kE = 189; Tmax = 5.24, Zmax = 3.84, local MNI coordinates x, y, z = −20, −24, −32 mm). No regions of increased rGMR were observed at this level for the non-AVH > AVH comparison. Results from the multiple regression model including healthy controls ([AVH > non-AVH] contrast masked by [AVH > healthy controls]) confirmed that the observed hyperactivity in the left temporal cortex, medial frontal cortex and caudate in the AVH group represents deviation from normality (Tmax = 6.35, Tmax = 6.03, Tmax = 5.75, respectively). All 3 regions were significant at p < 0.028 at the voxel level, FDR-corrected; local MNI coordinates were the same as those reported in Figure 1 for the AVH > non-AVH comparison. There were no differences in global rGMR between the AVH and non-AVH groups (p = 0.52). Furthermore, results from the ANCOVA model yielded the same between-group effects (p < 0.034, cluster-level corrected) as those detected using proportional scaling, thus ruling out a potential normalization bias on results.
Brain regions showing differences in relative glucose metabolic rate (rGMR) in patients with schizophrenia with auditory verbal hallucinations (AVHs; n = 9) compared with patients with schizophrenia without AVHs (n = 7). Groups of 2 slices in coronal and sagittal planes show clusters significantly hyperactive (hot colours) or hypoactive (cold colours) in the AVH group. The upper panel shows results from univariate analysis using statistical parametric mapping (SPM; thresholded at p ≤ 0.001, uncorrected, cluster size ≥ 20 voxels). The lower panel shows results from multivariate analysis using partial least squares (PLS; abridged for cluster size ≥ 20 voxels, BSR > [3.3]). From left to right, the images display higher rGMR along the left superior and middle temporal cortices (A1 and A2), bilateral superior medial frontal cortex (B1 and B2) and left caudate nucleus (C1 and C2) in hallucinating patients. Bilateral hippocampal hypoactivation is also shown in the coronal view of A2. The table contains p values and statistics regarding the observed intergroup differences for all clusters (abridged results for clusters larger than 20 adjacent voxels; PLS significant results for the superior medial frontal cortex cluster are not included owing to small cluster size). Images are overlaid on a canonical single-subject T1 template. All coordinates are reported in MNI space. Images are presented in accordance with radiologic convention (left is right).
Multivariate analysis — nonparametric inference
Mean-centering PLS identified 2 LVs, 1 of which was statistically significant (p = 0.009) and accounted for 100% of the crossblock covariance. Whereas clusters positively weighted on this LV fully coincided with the findings of the univariate analysis (Figs. 1 and 2), an additional group of negatively weighted clusters on this LV were also observed (BSR ± 3.3). Regions showing relative hypoactivity in the AVH group included the bilateral cerebellar cortices, parietal cortex, lingual gyri, left precuneus, bilateral hippocampi and parahippocampal gyri.
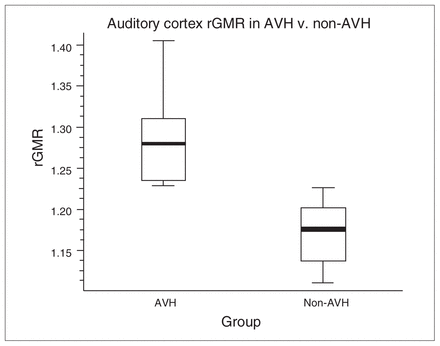
Differences in auditory cortex relative glucose metabolic rate (rGMR) in patients with schizophrenia with auditory verbal hallucinations (AVHs) compared with patients with schizophrenia without AVHs. The graph represents rGMR in each group (average and range) and demonstrates significantly different (p < 0.001) and nonoverlapping values between the 2 groups at a voxel located in the left middle temporal gyrus (Montreal Neurological Institute coordinates x, y, z = −68, −40, 8 mm).
Correlations between rGMR and PSYRATS scores
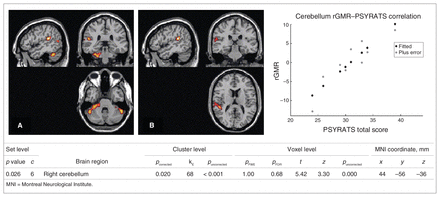
Whole-brain correlation analysis with the PSYRATS total score in patients with AVHs did not yield any significant results at the set voxel-level threshold of p < 0.001, uncorrected. However, when we reduced this threshold to p < 0.01 on an exploratory basis, we observed a positive correlation for a cluster of voxels located in the right cerebellar hemisphere (cluster-level p = 0.020, family wise error–corrected; Fig. 3). The ROI-based simple linear regression analysis at the threshold of p < 0.01 additionally showed that rGMR in the right superior temporal cortex was correlated with the PSYRATS total score (cluster-level p = 0.041, corrected for multiple comparisons; kE = 28, Tmax = 8.07, Zmax = 3.93, local MNI coordinates x, y, z = 48, −40, 16 mm), whereas there was no significant correlation with the inferior frontal cortex. All results remained significant (cluster-level p < 0.05, corrected for multiple comparisons) after controlling for individual scores on the conceptual disorganization item on the PANSS, which differed at a trend level between patients with and without AVHs (Table 1). Visual inspection of the scatter plots did not reveal any outliers.
Correlations between relative glucose metabolism rate (rGMR) and severity of hallucinations as indicated by Psychotic Symptom Rating Scale22,23 (PSYRATS) scores in patients with schizophrenia with auditory verbal hallucinations (AVHs). (A) Whole-brain correlations with PSYRATS scores. Only significant correlations at p < 0.05, cluster level–corrected are shown in the table (exploratory set threshold p = 0.01, voxel level–uncorrected). The scatter plot shows a positive correlation between rGMR in the right cerebellum and PSYRATS total score at local maxima (t = 5.42, cluster-level p = 0.020). (B) Results from region of interest–based analysis. There was a significant positive correlation between rGMR in the right superior temporal cortex and PSYRATS scores. Images are overlaid on a canonical single-subject T1 template. All coordinates are reported in MNI space. Images are presented in accordance with radiologic convention (left is right).
Discussion
The results of the present study indicate a functional involvement of the frontotemporal cortex and dominant caudate nucleus in patients with first-episode schizophrenia with complex commenting AVHs. The left superior and middle temporal region, bilateral superior medial frontal cortex and left caudate were found to be relatively hyperactive in right-handed patients with schizophrenia with AVHs compared with those without AVHs. Relative hypoactivity in the hippocampal complex, cerebellum and parietal cortex was also observed in patients with AVHs. Importantly, these findings are unlikely related to the effects of medication or to clinical symptoms other than AVHs.
Relative hyperactivity in the left superior and middle temporal cortices, including the primary auditory cortex,32 is consistently observed in studies of AVH.9 Interestingly, this temporal region, which is engaged during the perception of hallucinatory voices, contains a voice-selective subregion activated in healthy individuals during passive listening.33 A critical role of the temporal cortex in AVH is directly supported by transcranial magnetic stimulation studies, which show that targeting the left temporoparietal region is most effective in reducing AVHs.34,35
In a follow-up exploratory analysis, we also showed that severity of hallucinations was associated with rGMR in the right superior temporal cortex and cerebellum. These results are consistent with the superior temporal region’s putative role in the recognition of prosody and emotional characteristics of perceived speech.36,37 Considering that the PSYRATS directly assesses negative (emotional) content of hallucinations (Table 2), hyperactivity in this region, as associated with higher severity scores, could indicate more unpleasant/negative AVHs in some patients.
Our findings further point to a functional involvement of the superior medial frontal cortex in AVHs. Several lines of research suggest that this area is involved in the construction of internal self-representations and personally related associations,38,39 enabling higher order personal interpretations of external stimuli. In the context of language processing, increasing frontal activation spanning the superior and medial frontal cortex has been shown with increasing complexity of perceived speech.16,40–42 The superior medial frontal activity seen in our study during AVHs could be related to the high linguistic complexity and the personal relevance of the commenting hallucinations. This is in line with the notion that certain neural substrates of AVHs are linked to their phenomenology.13,14 For example, Sommer and colleagues43 recently showed that AVHs mainly activate the right homologues of language areas in treated chronic hallucinators mostly experiencing insulting/commanding AVHs characterized by low linguistic complexity. Differences in patient characteristics and phenomenology of AVHs could partly account for the diverging results seen between our studies.
Among frontal regions presumably involved in AVHs, the Broca area has received much attention because of its established role in the generation of inner (and overt) speech.44 However, we failed to observe a differential involvement of this area in AVHs compared with its engagement in psychotic symptoms other than AVHs. Preferential differences between the acute hallucinatory state and an antipsychotic-induced remission in frontal metabolism, as previously reported by our group,17 could partly represent a medication effect, since some frontal regions (including the Broca area) are known to be susceptible to metabolic changes following antipsychotic treatment.45–47 However, this interpretation remains to be tested in future studies directly comparing patients with schizophrenia with and without AVHs before and after treatment.
The caudate nucleus also exhibited relative hyperactivity in the AVH group. The dominant caudate nucleus has a central role in cognitive and motor functions, including certain linguistic processes,48 and has been implicated in regulating the language switch in proficiently bilingual individuals.49,50 Interestingly, most of our patients with AVHs were proficient in both Spanish and Catalan, and a considerable proportion of them reported alternating hallucinatory voices in more than 1 language. Considering that the left caudate is the primary recipient of corticostriatal projections from association regions in the language-dominant hemisphere,49 caudate dysfunction itself could be a key factor in the development of AVHs.
Activation of the cerebellum, more specifically the right cerebellar hemisphere, during speech generation has been shown in language neuroimaging research.51 Our findings of abnormal cerebellar activity suggest involvement of this structure in AVHs. Purkinje cells in the cerebellum have an important role in filtering neocortical input through modulation of their inhibitory output to the deep cerebellar nuclei. This circuit appears to be dysfunctional in patients with schizophrenia, which has been suggested to result in misinterpretation of inputs.52
Hypoactivity associated with AVHs was seen in the hippocampi–parahippocampal gyri. Deactivation of the parahippocampal gyrus preceding the hallucinatory experience was recently reported in a high temporal resolution functional magnetic resonance imaging (fMRI) study,53 a finding that is also in partial agreement with previous research on AVHs.54,55 Findings from postmortem and structural imaging studies56 along with functional abnormalities of verbal and nonverbal memory processes57–59 provide evidence for a potential role of the hippocampal complex in schizophrenia. The relation between parahippocampal hypoactivity and engagement of language networks, particularly the auditory cortex, could be critical in the generation of AVHs and needs to be considered in future studies.
A number of strengths to this study should be considered. As far as we know, no previous functional neuroimaging studies of AVH have been conducted in a sample of antipsychotic-naive patients with first-episode schizophrenia using a non-AVH patient control group comparable on clinical and sociodemographic characteristics. Only 1 other study employed a control group of nonhallucinating patients with acute schizophrenia; however, not all of the participants were antipsychotic-naive.60 Our cross-sectional design also offers better control than before–after designs over treatment-related confounds. With a few exceptions, studies on AVH have not controlled for the potential impact of variations in AVH phenomenology. Here, we evaluated a clinically homogeneous group of hallucinating patients, all of whom experienced prominent, complex commenting hallucinatory voices. Lastly, some advantages of our neuroimaging modality in the study of AVH warrant mention. Most of the studies on AVH conducted in the last few years used fMRI. Unless special sampling techniques are used, the intense noise generated by the MRI scanner can saturate the hemodynamic response in the auditory cortex such that any auditory activation due to AVHs could be underestimated.61,62 Despite its low temporal resolution, FDG-PET has the important advantage of being a silent technique and, compared with fMRI, it can serve as a better tool for capturing accumulated neural activity over several minutes.17,63–65 The specific strengths and weaknesses associated with PET and fMRI, along with their distinct measurements of neural activity, render these techniques complementary in the study of AVH.
In combining results of this study, we speculate that a failure to deactivate the temporal cortex may be fundamental to the generation of AVHs. Spontaneous increased activity in this region may generate auditory perceptions without external stimuli, such as those reported by some patients with epilepsy during ictal activity.66 An enduring failure of cerebellar–subcortical modulatory circuits in schizophrenia can interfere with the ability to filter out this aberrant input, leading to misinterpretation of both its source and meaning. Depending on specific characteristics of the percept, such as its linguistic complexity or the personal relevance of its content, certain components of associative circuits in the frontal cortex might secondarily be engaged, similar to the processing of actual external perceptions.
Limitations
There are several important limitations to the current study. The limited sample size could make our analyses susceptible to a type-II error and potentially underestimate some actual rGMR differences between the study groups. As is inherent to research on AVH, hallucinations are transient phenomena and their identification relies on patients’ subjective reports. A multimodal approach with additional intrasession assessment of AVHs using higher temporal resolution techniques could provide valuable information on the temporal course of AVHs. Linked to the nondemanding nature of our paradigm is the lack of a reliable estimation of the total time spent hallucinating in relation to tracer uptake time. Therefore, our findings, although likely reflecting processes that occur during hallucinations, could also represent metabolic patterns underlying acute AVH liability or perihallucinatory neural activity. Lastly, we selected a clinically homogeneous group of hallucinating patients to reduce the within-group variability. This strategy, however, makes it difficult to assess neural correlates of different subtypes of AVHs and impedes a finer insight into AVH phenomenology itself.
Conclusion
We found differing brain metabolic patterns contingent on the presence or absence of AVHs in antipsychotic-naive patients with first-episode schizophrenia. Relative hyperactivity of the left superior and middle temporal cortices, bilateral superior medial frontal cortex and left caudate nucleus was seen in patients experiencing linguistically complex commenting hallucinations. A multivariate analysis additionally revealed parallel prominent cerebellar, parietal and hippocampal–parahippocampal hypoactivity. These distinct brain activation patterns are unlikely related to clinical symptoms other than AVHs. Taken together, results indicate that AVHs in patients with schizophrenia may be mediated by an alteration of neural pathways responsible for normal language function, specifically those implicated in the perception and processing of external language.
Acknowledgements
This work was supported by the Fundació Marató of TV3 (registered number 012410), Janssen-Cilag; and Ministerio de Ciencia y Tecnología (grant number SAF-2002–04270-C02–02), the Fondo de Investigación Sanitaria (grant numbers G03/185, C03/06), DIUE (Government of Catalonia, Comissionat per Universitats I Recerca del Departament d’Innovació, Universitats I Empresa, grant number 2009SGR1295) and the Instituto de Salud Carlos III, Centro de Investigación Biomédica en Red de Salud Mental, CIBERSAM. The authors thank the Fundación Española de Psiquiatría y Salud Mental for providing funding for research training.
Footnotes
Competing interests: Dr. Horga declares having received grant and travel support from the Alicia Koplowitz Foundation. Dr. Parellada declares having received grant support and lecture fees from Janssen-Cilag and GlaxoSmithKline and grant support from Lilly. Dr. Lomeña declares having received lecture fees from GE Healthcare. Dr. Font declares having received grant support from Maraton TV3 and Janssen-Cilag and travel support from Janssen-Cilag. Dr. Bernardo declares having received grant support from Bristol-Myers Squibb, CIBERSAM, Eli Lilly, FIS-Institute of Health Carlos III, the Government of Catalonia, Janssen-Cilag, Marató TV3, Organon and Pfizer, and declared board membership with and lecture fees from Bristol-Myers Squibb, Eli Lilly, Janssen-Cilag, Mylan and Pfizer. None declared for Drs. Fernández-Egea, Mané, Falcón, Konova, Pavia and Ros.
Contributors: Dr. Horga designed the study, performed the analyses, interpreted the results and drafted the manuscript. Drs. Parellada, Lomeña, Fernández-Egea, Font and Bernardo designed the study protocol. Drs. Konova, Mané and Falcón assisted with data analyses, provided methodological advice and helped editing the manuscript. Drs. Pavia, Ros and Lomeña supervised the imaging protocol and data collection. All authors contributed to, revised and approved the final manuscript.
- Received May 23, 2010.
- Revision received August 11, 2010.
- Accepted October 15, 2010.