Abstract
Background: Autism spectrum disorders (ASDs) are associated with auditory hyper- or hyposensitivity; atypicalities in central auditory processes, such as speech-processing and selective auditory attention; and neural connectivity deficits. We sought to investigate whether the low-level integrative processes underlying sound localization and spatial discrimination are affected in ASDs.
Methods: We performed 3 behavioural experiments to probe different connecting neural pathways: 1) horizontal and vertical localization of auditory stimuli in a noisy background, 2) vertical localization of repetitive frequency sweeps and 3) discrimination of horizontally separated sound stimuli with a short onset difference (precedence effect).
Results: Ten adult participants with ASDs and 10 healthy control listeners participated in experiments 1 and 3; sample sizes for experiment 2 were 18 adults with ASDs and 19 controls. Horizontal localization was unaffected, but vertical localization performance was significantly worse in participants with ASDs. The temporal window for the precedence effect was shorter in participants with ASDs than in controls.
Limitations: The study was performed with adult participants and hence does not provide insight into the developmental aspects of auditory processing in individuals with ASDs.
Conclusion: Changes in low-level auditory processing could underlie degraded performance in vertical localization, which would be in agreement with recently reported changes in the neuroanatomy of the auditory brainstem in individuals with ASDs. The results are further discussed in the context of theories about abnormal brain connectivity in individuals with ASDs.
Introduction
People with autism spectrum disorders (ASDs) display impaired social interaction and communication skills and a pattern of rigid and repetitive behaviour, according to the DSM-IV criteria.1 In addition, ASDs are associated with both hyposensitivity and hypersensitivity to sensory stimuli.2–4 These are not considered primary features of ASDs in the DSM-IV, but they are included as criteria in the DSM-5 (www.dsm5.org). Given the impaired verbal communication skills of people with ASDs, the questions of whether and how auditory processing is affected and of how this relates to the underlying neural substrate are highly relevant. This is also evidenced by a rapidly increasing interest in auditory and speech perception in individuals with ASDs.5–7 Findings indicate that children with ASDs have enhanced pitch perception, difficulty understanding speech in noisy environments and a reduced likelihood of orienting toward auditory social stimuli.8–13
Auditory brainstem responses have longer and less consistent latencies in individuals with ASDs.14 The primary auditory cortex in people with ASDs and auditory hypersensitivity show a stronger response to pure tone pip stimuli.15 Studies investigating speech-in-noise perception have shown that people with ASDs have more difficulty than controls understanding speech in noise with (spectro)temporal dips.10 This effect, a weaker comodulation masking release16 in people with ASDs, was also found in a later study, suggesting that controls were better able to integrate information over temporally separated intervals than participants with ASDs.11 Spatial attention has been shown to be affected and accompanied by abnormal event-related brain potentials in people with ASDs in a spatial hearing experiment.17 Recent postmortem investigations of the brainstem olivary complex (part of the auditory pathway) in decedents who had ASDs revealed a greatly reduced size of the medial superior olive (MSO), with lower numbers of stellate and fusiform neurons and, to a lesser degree, reduced size of the other nuclei of the superior olivary complex (SOC).18,19 These are, to the best of our knowledge, the only histological studies investigating these structures in decedents who had ASDs, and unfortunately patient and control brains were not perfectly matched for sex and level of functioning. Nevertheless, they provide an interesting perspective on auditory processing in brains affected by ASDs.
These and other abnormalities reported across a broad range of processes in the auditory domain6,7 suggest that the pathophysiology of ASDs may not be exclusively confined to higher-level (top–down) cognitive processing, but may already be present at the level of primary sensory (bottom–up) processing. The concept of more pervasive neural deficits is closely related to the hypothesis of abnormal connectivity throughout the brain in people with ASDs.20–23 Two specific theories of deficient connectivity focus on deficits in temporal binding21 (reduced γ-band synchronization between local brain networks) and the ratio of excitation to inhibition in the brain.23 Functional and structural imaging studies have also suggested abnormal subcortical connectivity in people with ASDs.24–26 Recent investigations seem to point toward a combination of short-range overconnectivity and long-range underconnectivity in people with autism.27 This “connectivity hypothesis” of ASDs claims that the social and cognitive abnormalities may be explained by poor long-range connectivity between distant regions of the brain and excessive short-range connectivity within and between nearby regions.22 This hypothesis has recently been called into question, as motion-related artifacts may have contributed to findings in functional connectivity studies.28–30
The auditory pathway in the human brain is relatively well understood31 and, as such, offers a good opportunity to obtain better understanding of ASDs at the neural level. This is particularly promising for the neural mechanisms underlying sound localization, as the roles and neural connectivities of the different auditory brainstem nuclei in localization are to a large degree described and understood.32,33 We reasoned that global connectivity deficits and abnormal brainstem development could profoundly and predictably affect sound localization. We performed 3 behavioural experiments, which are outlined in the sections that follow; an overview is given in the Appendix (Table S2, available at cma.ca/jpn). While we cannot unambiguously map behavioural measurements to these neural mechanisms, any observed atypicalities would provide a new perspective on auditory processing in individuals with ASDs and could be a starting point for future research.
Horizontal and vertical localization of noise stimuli within background noise
Horizontal localization relies predominantly on the detection of interaural time differences (ITDs), originating from the different distances of the ears to the sound source, and interaural level differences (ILDs), caused by frequency-dependent masking by the head.32 For low frequencies, ITD detection is the primary mechanism. It relies on coincidence detection of spikes travelling along the ipsilateral and contralateral auditory nerve fibres. Hence, ITD detection critically depends on the accurate timing of inputs at the ears.34–36 For higher frequencies, starting from about 2 kHz, ILD detection by excitatory–inhibitory (EI) and inhibitory–excitatory (IE) cells becomes the primary mechanism.37 These cells are sensitive to the ILD at their best frequency. As ITD and ILD detection integrate binaural information, they rely on intact long-range neural connectivity between the brainstem nuclei of the left and right pathways.
Sound-source location in the vertical plane is determined from the spectral shape properties of the acoustic input that arise from the complex geometry and associated direction-dependent filtering of the pinna.32 Vertical localization at lateral locations38 does not rely on integrating binaural information, but is primarily a “within-stream” monaural process that is thought to rely on short-range connectivity within auditory nuclei (i.e., within tonotopic maps).34,35 It has been shown that abnormalities in this processing stream can be sensitively demonstrated by adding competing background noise to increase the difficulty of the localization task.39
We designed an experiment to investigate whether horizontal and vertical localization performance are affected in people with ASDs and whether difficulties in signal/noise separation add to this.
Vertical localization of sweep stimuli
To investigate impaired temporal integration in participants with ASDs, we performed a localization experiment with repetitive frequency-modulated sweeps of different repetition periods. The instantaneous spectral properties of sweeps are narrowband, but as the moving centre frequency covers all audible frequencies, a fast sweep can be temporally integrated by the brain to provide sufficient spectral information for vertical sound localization.40 The sweep experiment allowed us to systematically vary the temporal integration window across sweeps by varying their speeds and thus allowed us to test our hypothesis that healthy control participants can integrate spectral information over a longer window than participants with ASDs.
Precedence effect
When the same sound originates from 2 locations with a short temporal delay, there is a temporal window within which the brain will fuse the sounds into a single source; this phenomenon is known as the precedence effect.41,42 In most cases the fused sound will appear to originate from a point between the actual sound locations and weighted by the stimulus delay. The time difference up to which this effect occurs (about 5–50 ms) is at least an order of magnitude larger than the maximum ITD caused by the distance between the ears (about 650 μs). The precedence effect can be interpreted as an aspect of temporal integration sensitivity. We hypothesized that a shorter temporal integration window in participants with ASDs would result in a shorter time window for the precedence effect in this group than in healthy controls.
Methods
Participants
Measurements were performed in adults with ASDs and healthy controls. We obtained written informed consent from all participants. The study was approved by the local ethical committee (CMO regio Arnhem-Nijmegen).
We recruited high-functioning participants with ASDs from referrals to the department of psychiatry at the Radboud University Nijmegen Medical Centre and from participants in a previous study.43 People with ASDs were included if they had a clinical diagnosis of autistic disorder or Asperger syndrome according to DSM-IV1 criteria and if they had no comorbid Axis I disorders. Clinical diagnoses were established by experienced clinicians on the basis of a careful developmental history and psychiatric evaluation. Clinical diagnoses were confirmed by administering a structured interview, the Autism Diagnostic Interview-Revised (ADI-R),44 among the parents or caretakers of participants. We recruited healthy control participants from the Donders Institute database. We tested the nonverbal intelligence of participants using Raven’s Advanced Progressive Matrices (APM). Participants were included if they were between 18 and 35 years old. We excluded individuals who reported hearing impairment, severe neurologic impairment or severe psychiatric comorbidity.
The sweep localization experiment was performed in the groups described above; participants were asked to come back for the noise localization and precedence experiments. Since not all participants were still available, new healthy control participants were included in these experiments.
Experimental setup
Experiments were performed in a dark 3 × 3 × 3 m room with reflection-dampening walls, preventing echoes above 500 Hz. Stimuli were presented from 1 of 58 identical speakers (Visaton SC 5.9) mounted on a motorized hoop measuring 2.5 m in diameter that could rotate around the earth-vertical axis at a precision better than 0.1°. Speakers were mounted on the hoop at 5° increments, from −55° to 85° on the front half, and from −52.5° to 87.5° on the back half, thus allowing for a 2.5° resolution in the elevation direction. The participant was seated comfortably in a modified chair, with his or her head in the centre of the hoop. Two fixed background speakers were positioned at a height of 1.5 m on both sides of the frontal wall (see the Appendix, Fig. S1). Stimuli were generated offline using MATLAB (Mathworks) at a sampling rate of 48.828 kHz, had 5 ms sinusoidal onset and offset envelopes, and were presented from a real-time processor (RP2.1, Tucker-Davis Technologies, System 3). Custom-made amplifiers that allowed for a per-trial attenuation drove the speakers on the hoop; an off-the-shelf amplifier (Philips FA569) drove the background speakers. The experiment was con-trolled by custom-written software running on a standard personal computer (Dell).
Two sets of single-turn magnetic-field coils attached along the edges of the side walls (horizontal) and floor and ceiling (vertical) generated the oscillating magnetic fields for the search-coil method at 60 kHz and 80 kHz, respectively. The participant wore a lightweight spectacle frame with a pickup coil attached on its nose bridge, allowing precise measurement of the head orientation at a resolution better than 0.1°. A laser diode attached to the spectacle frame in the centre of the pickup coil projected onto a small (1 cm2) plate fixed at about 30 cm in front of the head, providing the participant with a convenient head-fixed visual pointer. To convert measurements on the horizontal and vertical channels into azimuth and elevation head orientation angles, we used a visual calibration procedure in which the participant pointed at target light-emitting diodes (LEDs) throughout the relevant region of measurement within the frontal hemifield. Head orientation was expressed in azimuth and elevation coordinates, defined as the angles with the medial and horizontal planes, respectively.45
Noise localization experiment
In each trial, we presented a white noise background (0.2–20 kHz) with a sound pressure level (SPL) of of 62 dB from the fixed speakers for a duration of 2500 ms. After a random delay of 450–700 ms from white noise onset, 1 of the speakers on the hoop played a broadband target stimulus consisting of quasi–white noise (0.2–20 kHz) looped with a 20 ms period (sounding like a buzzer) for a total duration of 150 ms including 50 ms on/offset envelopes. Target locations were selected pseudorandomly from a flat distribution within −75° and 75° azimuth and −45° and 60° elevation. Intensity varied between −20 and 0 dB in steps of 5 dB with respect to the background stimulus. In addition, we included control localization trials without the background noise and at the maximum target intensity.
We instructed participants to aim the visual pointer at a fixation LED (middle of the frontal wall) at the start of the trial and then to quickly aim it in the direction of the target sound stimulus and to maintain fixation for about a second. We included 29 trials per sound level, yielding a total of 174 trials.
Sweep experiment
Stimuli consisted of repeated full-range (0.2–24.4 kHz) frequency sweeps, generated using the voltage-controlled oscillator function in the signal processing toolbox in MATLAB. Sweeps had durations of 1, 2, 4, 8, 16 and 32 ms and were repeated to produce stimuli that all had a total duration of 150 ms. All stimuli were presented at 0 azimuth, with elevations between −45° and 60° and with no background sound. Participants received the same visual pointing instructions as they did for the noise localization experiment. There were 132 trials.
Precedence experiment
In the precedence experiment, we presented a 100 ms white noise stimulus at an SPL of 62 dB from both background speakers (located at −45° and 45° azimuth and 0° elevation) with a delay of 0–40 ms in either the left or right channel. Participants were instructed to push a button if they heard 2 sounds (“2 onsets”). There were 150 trials.
Statistical analysis
Prior to further analysis, we extracted head movement parameters (representing pointer position) for the localization and sweep experiments from the measured data. A custom developed toolbox46 automatically detected responses using velocity and acceleration criteria. Some detected intervals needed manual adjustment. The head orientation at the end of the participant’s response was the response angle used for further analysis.
We parameterized participant performance in the localization experiments with 2 measures: the response gain, which is the slope of a linear fit of response angles to stimulus angles, and the Pearson linear correlation coefficient between stimulus and response data. We substituted trials without a response by setting the azimuth and elevation coordinates to zero. In the correlation computation, we only included trials where the participant made a response. If the participant responded in fewer than 3 trials for a given condition, the correlation was set to zero. This only occurred for the lowest intensity category in the noise localization experiment. We assessed group level effects using repeated-measures analysis of variance (ANOVA) with a significance threshold of α = 0.05. We calculated statistics using SPSS version 19 (IBM Corporation).
In the precedence experiment, we applied logistic regression to model the responses per participant. From the regression coefficients, we computed the point where 2 sounds were reported in 50% of the cases as the threshold. We assessed group differences using a 2-tailed, 2-sample t test with a significance threshold of α = 0.05.
Results
The characteristics of the included participants are shown in Table 1. Ten participants with ASDs and 6 controls participated in all experiments. One patient with an ASD was excluded owing to neurologic abnormalities and 1 was excluded owing to hearing impairment. Two controls and 1 patient with an ASD were excluded because they had not performed the task correctly. For 9 participants it was not possible to obtain ADI-R data because no parent or caretaker was available. One participant had a score below the cut-off on the social interaction scale, 2 scored below the cut-off for stereotypical behaviour and 4 did not meet the onset cut-off. The clinical picture, however, was very typical in all of these cases. All other ADI-R scores were above the cut-off in all domains.
Demographic and clinical characteristics of participants with autism spectrum disorders and healthy controls
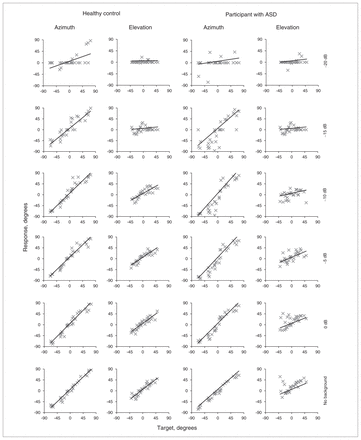
Figure 1 shows all responses from the noise localization experiment of an example participant from either group, sorted by different signal-to-noise ratios (SNRs). Note that azimuth performance in both participants is very reliable up to the point starting below −15 dB, where the participants had great difficulty hearing the stimulus. In contrast, elevation performance decreased more gradually for all SNRs. This pattern is consistent with earlier findings obtained from healthy controls.39
Localization responses for different signal-to-noise ratios (SNRs) and without noise background (control condition) in an example control (C14) and autism spectrum disorder (ASD; A21) participant. Scatter plots show individual trials, and lines show linear regression.
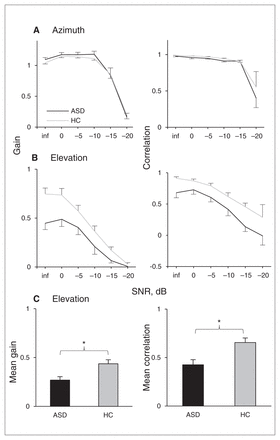
The group analyses show that azimuth localization had very good trial-to-trial reproducibility for SNRs of −10 dB and higher in both the ASD and control groups, with both gain and correlation decreasing for the low SNRs (Fig. 2A). Differences between the ASD and control groups and interactions with SNRs were not significant (all p > 0.10). For the elevation response components, both gain and correlation were significantly lower for the ASD than the control group (Fig. 2B, C; gain: F1,18 = 6.45, p = 0.021, partial η2 = 0.26; correlation: F1,18 = 8.28, p = 0.010, partial η2 = 0.32). The group × SNR interaction after Greenhouse–Geisser (GG) correction was significant for gain (F5,90 = 4.00, pGG = 0.019, partial η2 = 0.18), but not for correlation (pGG = 0.637). We used GG correction, as both ANOVAs violated the sphericity assumption for repeated-measures ANOVA (Mauchly test of sphericity p < 0.001 in both tests).
Mean performance (regression and correlation coefficients per participant per stimulus level) in the noise localization experiment for participants with autism spectrum disorders (ASDs) versus healthy controls (HC). Error bars represent standard errors of the mean. Note that the gain is slightly higher than 1 for signal-to-noise ratios (SNRs) of −10 dB and higher. *p < 0.05.
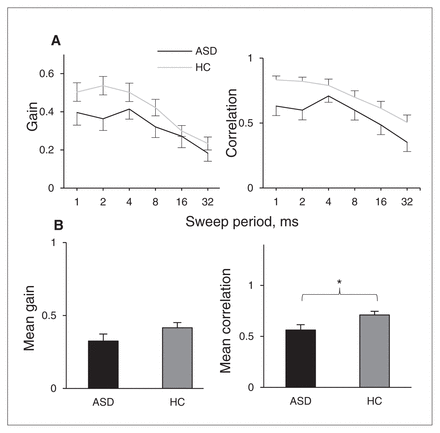
Figure 3A summarizes the results for the sweep localization experiments. The data show the response gains and correlations in the elevation direction as a function of sweep duration of 1–32 ms. Participants with ASDs showed significantly lower correlation scores than controls (Fig. 3B, F1,36 = 5.70, p = 0.022, partial η2 = 0.14; Mauchly test of sphericity p = 0.05). The difference in gain was not significant (p = 0.13). Group × period interactions were not significant for either gain or correlation (both p > 0.10).
Group results for localization of sweeps. Error bars represent standard error of the mean. *p < 0.05. ASD = autism spectrum disorder; HC = healthy control.
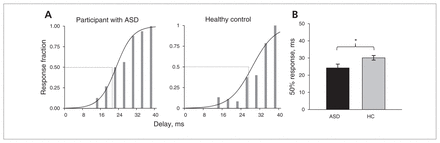
Figure 4A shows the measured psychometric curve from an example participant in the ASD and control groups during the precedence experiment, in which the relative fraction of perceived double stimuli is plotted against speaker delay. For short delays, the participant reported hearing a single sound in all trials, whereas 2 sounds were reported for the longest delays. In the group results (Fig. 4B), the average 50% response point was 30.1 ± standard deviation (SD) 4.2 ms for healthy control listeners and 24.2 ± 7.1 ms for listeners with ASDs; this difference was significant (t14.7 = −2.26, p = 0.039, Cohen d = 1.0; equal variances not assumed).
Precedence results. A) Example response curve from a participant with an autism spectrum disorder (ASD; A02) and a control (HC; C11). Bars = binned responses, 4 ms bin width. Line = logistic regression. B) Group means of 50% response point. Error bars are standard errors of mean. *p < 0.05.
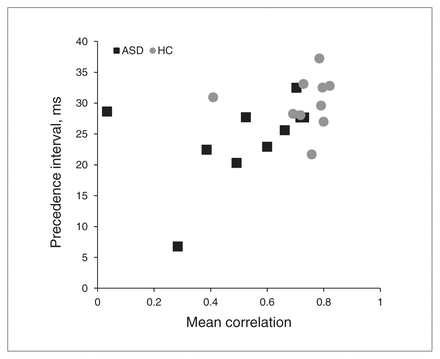
To investigate whether performance in the noise localization and precedence experiments were related, Figure 5 shows both measures per participant. Performance in the 2 experiments significantly correlated across all participants (r = 0.47, p = 0.037).
Scatter plot of vertical localization performance in the localization task versus 50%-point in the precedence experiment. Correlation: r = 0.47, p = 0.037. The −20 dB category was excluded in the computation of the performance average, as the estimate of the correlation coefficient for this category was unreliable owing to the number of trials that the participants could not hear at this intensity level. ASD = autism spectrum disorder; HC = healthy control.
See the Appendix for an exploration of age, which was not included in the analyses, as an explanatory variable.
Discussion
Horizontal and vertical localization of noise stimuli within background noise
In the noise localization experiment, participants with ASDs and healthy controls performed equally well in horizontal plane localization for all employed SNRs. The similarity in performance between the groups confirms that both healthy controls and participants with ASDs had few problems hearing the stimuli for SNRs above −20 dB and were attentively performing the task.
Interestingly, participants with ASDs performed markedly worse at vertical localization in this experiment. In contrast to horizontal localization,33 vertical localization is predominantly a monaural process, especially for lateral targets.38 A possible explanation for the impairment in performance is abnormal connectivity within the cochlear nuclei and the inferior colliculi, which are believed to be involved in elevation detection.47,48
A sharp reduction in the number of fusiform neurons in the MSO in patients with ASDs has been reported in the literature.18,19 It has been suggested that similar neurons in the cochlear nucleus play a role in elevation processing47 and orienting toward elevated sources.49,50 The efferent fibres from these neurons project to the inferior colliculus and the medial geniculate body.51 A reduction in the number of fusiform neurons in the cochlear nucleus could, therefore, impair downstream relaying of elevation information from the cochlear nucleus.
We observed a significant group × SNR interaction for the vertical response gain in this experiment. The curves in Figure 2B suggest that the between-group effect of gain could be multiplicative rather than additive, which could potentially explain the interaction. Although the results are compatible with impaired vertical localization at a low SNR in participants with ASDs (a shift of the curve to the left), we cannot, therefore, unambiguously attribute the significant interaction to such an effect.
Vertical localization of sweep stimuli
Like in the noise localization experiment, we also observed that participants with ASDs performed worse on vertical localization in the sweep localization experiment. The difference between groups appeared largest for the fastest sweeps, although the group × period interaction was not significant. It has been shown previously that the fast sweeps contain adequate spectral information and should therefore be the easiest stimuli to localize in the vertical plane.40 The apparent lack of a between-group difference that is specific to the slower sweeps suggests that there is no difference in the length of the spectrotemporal integration window, but rather that fundamental elevation processing is affected in people with ASDs (as in the noise localization experiment).
Precedence effect
Participants with ASDs showed a weaker precedence effect than controls. Previously reported thresholds for the precedence effect vary greatly with stimulus type and response task. A threshold of 22 ms has been reported for 100 ms noise bursts,42,52 although in that experiment the criterion was “equal loudness of lead and lag,” instead of the “double onset” criterion that we used in the present study. Larger values (30–50 ms) have been reported for speech.42 In light of these previous findings, the thresholds reported in the present study seem plausible. The shorter threshold in participants with ASDs could hint at a reduced ability to integrate information over time and thus appears to be consistent with the temporal binding hypothesis.21 Moreover, the time scale of the precedence effect falls exactly in the hypothesized time-scale of temporal binding deficit21 (i.e., in the 10–40 ms range of the γ-band frequencies).
General discussion
We will briefly consider how the results we discussed could relate to the theory of abnormal connectivity in people with ASDs.22,27 With respect to vertical localization, connectivity theories of ASDs would suggest local overconnectivity,20 and this seems plausible, as either under- or overconnectivity would likely have a detrimental effect on performance. Findings of reduced sizes of the relevant nuclei in people with ASDs and reduced sizes of their neuronal populations, however, could point to reduced connectivity.18,19 As binaural processing is fundamental for the precedence effect, factors contributing to the shorter temporal window may include reduced long-range connectivity between the left and right pathways. Hence, the combination of the localization and precedence experiments is compatible with this part of the connectivity hypothesis. The significant correlation between performance in the noise localization and precedence experiments (Fig. 5) suggests that both effects may indeed originate from the same root cause at the neuronal level.
The hypothesis of reduced temporal binding in people with ASDs21 may provide an explanation for a weaker precedence effect in this population. Note, however, that there was no indication of deficient coincidence detection in azimuth processing in the first experiment. This indicates that temporal binding may indeed be affected, but that this may be more appropriately viewed as having consequences at longer time scales, perhaps in more complex neural systems, rather than a direct problem in neural timing.
Fusiform cells in the auditory brainstem have been described as pyramidal cells as well, and this may hint at a relation between our findings and reported abnormalities in development of other brain areas. Pyramidal cells of smaller size have been observed in layers III, V and VI of Brodmann areas 44 and 45.53 These neurons are involved in longer-range cortical and subcortical projections. In the same layers of the fusiform gyrus, neurons were found to be smaller and fewer in number.54 In this context it should also be noted that, as it seems likely that neuronal atypicalities are present throughout the brain, changes in cortical processing could provide complementary or alternative explanations for impaired task performance.
It has been speculated19 that the reduced number of fusiform and stellate cells may be caused by neuroblasts failing to migrate to the SOC or that they may not survive after failing to form connections to the cochlear nuclei. A potentially interesting aspect of these data, although the authors did not mention this, is that in people with ASDs, the number of MSO neurons seems to increase with age until adulthood, whereas in controls, the number seems constant over the age range covered. A delayed development trajectory of the brain is one of the main features of theories on ASDs;55 a potential developmental aspect in the neural structure of the auditory pathway suggests that investigating sound localization in children may prove worthwhile.
Limitations
With the broadband stimuli that we used in the present study, it is not possible to disentangle the ITD and ILD detection mechanisms that contribute to horizontal localization. Follow-up studies could use stimuli with only low- or high-frequency content to distinguish between the 2 mechanisms.
Group sizes were relatively small in the experiments investigating localization in noise and in the precedence experiment. For this reason it was not possible to perform analyses on performance differences among participants in the ASD group. It may be fruitful to investigate subgroups and correlations of localization task performance with other diagnostic measures in future research.
Conclusion
We observed 2 abnormalities in auditory processing in people with ASDs: participants with ASDs performed worse than controls on vertical sound localization, and the temporal window for the precedence effect was shorter in participants with ASDs than controls. The functional abnormality in vertical localization may be a reflection of a reduction in structural connectivity that has been reported in the literature18,19 and is consistent with the hypothesis of reduced long-range connectivity.20,27 The temporal binding hypothesis of ASD21 could explain the shorter temporal window in which the precedence effect occurs.
Footnotes
Competing interests: C. Kan is on the ADHD Advisory Board at Eli Lilly and has received royalties for 2 Dutch books on autism. J.K. Buitelaar declares an internal grant from the University Medical Centre, Radboud University. No other competing interests declared.
Contributors: E. Visser, M. Zwiers, A.J. van Opstal and J.K. Buitelaar designed the study. E. Visser, C.C. Kam and L. Hoekstra acquired the data, which E. Visser, M. Zwiers, A.J. van Opstal and J.K. Buitelaar analyzed. E. Visser, M. Zwiers and A.J. van Opstal wrote the article, which all authors reviewed and approved for publication.
- Received September 5, 2012.
- Revision received January 14, 2013.
- Revision received March 20, 2013.
- Accepted March 20, 2013.