Abstract
Background: Accumulating evidence indicates that schizophrenia is associated with brain immune activation. While a number of reports suggest increased cytokine levels in patients with schizophrenia, many of these studies have been limited by their focus on peripheral cytokines or confounded by various antipsychotic treatments. Here, well-characterized patients with schizophrenia, all receiving olanzapine treatment, and healthy volunteers were analyzed with regard to cerebrospinal fluid (CSF) levels of cytokines. We correlated the CSF cytokine levels to previously analyzed metabolites of the kynurenine (KYN) pathway.
Methods: We analyzed the CSF from patients and controls using electrochemiluminescence detection with regard to cytokines. Cell culture media from human cortical astrocytes were analyzed for KYN and kynurenic acid (KYNA) using high-pressure liquid chromatography or liquid chromatography/mass spectrometry.
Results: We included 23 patients and 37 controls in our study. Patients with schizophrenia had increased CSF levels of interleukin (IL)-6 compared with healthy volunteers. In patients, we also observed a positive correlation between IL-6 and the tryptophan:KYNA ratio, indicating that IL-6 activates the KYN pathway. In line with this, application of IL-6 to cultured human astrocytes increased cell medium concentration of KYNA.
Limitations: The CSF samples had been frozen and thawed twice before analysis of cytokines. Median age differed between patients and controls. When appropriate, all present analyses were adjusted for age.
Conclusion: We have shown that IL-6, KYN and KYNA are elevated in patients with chronic schizophrenia, strengthening the idea of brain immune activation in patients with this disease. Our concurrent cell culture and clinical findings suggest that IL-6 induces the KYN pathway, leading to increased production of the N-methyl-d-aspartate receptor antagonist KYNA in patients with schizophrenia.
Introduction
The role of immune activation in schizophrenia has received increasing interest in recent years. Epidemiological studies have shown that infections and autoimmune diseases are clinically important risk factors for the development of schizophrenia.1–3 Furthermore, a recent study integrating results from a meta-analysis of genome-wide association studies in schizophrenia found that the most significant changes were observed in genetic loci related to the immune system.4 Also, patients with schizophrenia show microglial activation, as revealed by positron emission tomography.5,6 Further, postmortem studies using messenger RNA (mRNA) expression or immunohistochemical detection have shown increased levels of immune-related compounds.7–9 A number of investigations have focused on cytokines, but these studies have not yielded a unanimous picture, possibly because of a number of confounding factors, such as smoking and dietary habits, body mass index (BMI), type and duration of antipsychotic treatment and drug abuse — all factors potentially affecting the immune system.10 However, according to recent meta-analyses,11,12 some cytokines are significantly associated with schizophrenia (e.g., interleukin [IL]-6). Still, in the majority of these studies, only peripheral cytokines were investigated, and reports of brain cytokine levels in patients with schizophrenia are relatively sparse and limited by the lack of a control group of healthy volunteers or by a low sensitivity of the assay. Recently though, cerebrospinal fluid (CSF) IL-1β was shown to be markedly elevated in first-episode patients compared with healthy individuals.13 A few studies have also shown that CSF IL-6 is elevated in patients with chronic schizophrenia.14,15 Although evidence of immune activation in schizophrenia is emerging, its functional relationship to aberrant brain neurotransmission and behaviour has been unclear. Activation of the kynurenine (KYN) pathway has been suggested as a mechanism serving to transfer information from the immune system of the brain to neurons.16–18 Tryptophan degradation along this pathway occurs in various cells, including monocyte-derived macrophages and microglia, and produces several neuroactive compounds, such as quinolinic acid, an N-methyl-d-aspartate (NMDA) receptor agonist and kynurenic acid (KYNA), a blocker of the NMDA receptor and the α7 nicotinic receptor.18 Indeed, in patients with schizophrenia and those with bipolar disorder with psychosis, elevated levels of KYNA and its precursor KYN in the CSF or in the postmortem brain have been consistently reported.19–26
The aim of the present study, using a well-characterized cohort of olanzapine-treated patients with chronic schizophrenia, was to investigate CSF levels of cytokines and correlate those to previously measured levels of tryptophan metabolites of the KYN pathway in the same patients. Further, since an association between CSF IL-6 and the production of KYNA has been found in patients, as reflected by the tryptophan:KYNA ratio, we investigated a putative interplay between this cytokine and the production of KYNA in human astrocyte cultures.
Methods
Patients
We collected CSF from patients with DSM-IV-verified schizophrenia or schizoaffective disorder from January 2005 through December 2007. All patients were outpatients with no history of drug abuse and were medicated with olanzapine (2.5–25 mg/d) as the only antipsychotic drug. Full details of the study design and patient characteristics, including serum and CSF concentrations of olanzapine (analyzed by liquid chromatography and tandem mass spectrometry, as described prevously27) have been published elsewhere.28 All patients included in the study were somatically healthy according to routine laboratory analyses and physical examination. Patients who received medical treatment for infectious diseases, cancer, diabetes type 1 or 2 with a blood glucose level higher than 15 mmol/L, a history of myocardial infarction, hypertension (systolic blood pressure > 160 mm Hg, diastolic blood pressure > 110 mm Hg) or medicated with drugs affecting platelet function were not included in the study. The only concomitant drugs accepted were zopiclone and lithium. Smoking was permitted. Symptom severity was rated in all patients using the Brief Psychiatric Rating Scale (BPRS) and Global Assessment of Functioning (GAF) scale. Plasma glucose, insulin, triglycerides and cholesterol where analyzed at the Department of Clinical Chemistry, Linköping University Hospital, Sweden, as part of routine clinical analyses.
Healthy volunteers
Healthy controls were mainly recruited among medical students, hospital staff members and their relatives from January 2005 through December 2008. They were all in good physical health, as confirmed by a medical check-up, including routine laboratory tests and a physical examination. All volunteers were free from medication for at least 1 month and free from any form of substance abuse, except for smoking. Volunteers were subjected to a semi-structured interview, using the Structured Clinical Interview of DSM-IV disorders29 and a questionnaire for personality disorders,30 or interviewed by a psychiatrist to exclude mental illness. None of them had a family history of major psychosis or suicide in first- or second-degree relatives, and they were all found to be free from current signs of psychiatric morbidity or difficulties in social adjustment at the time of sampling.
CSF sampling
Lumbar puncture was performed after a minimum of 8 hours of fasting (coffee not allowed). There were no restrictions on posture or rest. At about 8 am, a disposable needle (BD Whitacre Needle 0.7 × 90 mm) was inserted at the L4–5 level with the participant in the right decubitus position. The CSF was allowed to drip into a plastic test tube. Two 6 mL fractions of CSF were collected and protected from light and centrifuged at 3500 rpm for 10 minutes within 30 minutes of the puncture. Each 6 mL sample was divided into 2 plastic tubes and stored at −70°C until analysis. All CSF samples had been frozen and thawed twice before cytokine analysis.
The CSF levels of tryptophan, KYN and KYNA from a subsample of patients (n = 11, all men) and controls (n = 18, all men) have previously been published.25
Policy and ethics
The work described in the present study was carried out in accordance with the Declaration of Helsinki. The Ethics Committee of the Medical Faculty of Linköping University, Sweden; the Swedish Medical Products Agency; and the Swedish Data Inspection Board approved the studies involving patients and healthy volunteers, respectively. All patients and healthy volunteers received verbal and written information about the study and provided written informed consent. At this time patients were stable owing to medication, they showed no acute psychosis, and they had a relatively high level of functioning. The same senior psychiatrist (E.S., K.L.) followed the patients for the duration of the study and was convinced that the patients understood the information and accepted to participate.
Human astrocyte culture
Human embryonic primary cortical astrocytes were purchased from ScienCell Research Laboratories and cultured according to manufacturer’s recommendations. All experiments were performed on cells in passage 4. Cells were serum starved (0.02% fetal bovine serum and 0.01% growth supplement mix) for 24 hours before the experiments and were then stimulated with IL-6 (10 ng/mL) for 24, 48 and 72 hours. Cell culture supernatants were collected and immediately frozen on dry ice and kept at −80°C until analysis. Prior to analysis, residual protein was precipitated using the following procedure. Samples were centrifuged at 14000 rpm for 5 minutes, and an equal volume of perchloric acid (0.4 M) was added to the supernatants before additional centrifugation. Following addition of 70% perchloric acid, samples were centrifuged twice. All cell culture experiments were performed in triplicate and repeated twice.
Cytokine analyses
We analyzed IL-1β, IL-2, IL-4, IL-6, IL-8, IL-10, IL-18, tumour necrosis factor (TNF)-α, interferon (IFN)-α-2a and IFN-γ in CSF using a customized Human Ultra-Sensitive 10-Plex Kit (MesoScale Discovery) in 2011. The assays were analyzed as per the manufacturers protocol (www.mesoscale.com), including a long primary incubation time (overnight at 4°C). The sample volume was 50 μL. The intra-assay coefficient of variation was below 20% for all analytes presented. In analogy with the study by Maier and colleagues,31 particularly reliable detections of IL-6 and IL-8 was achieved. The limits of detection in our analysis were as follows: IL-1β (0.06 pg/mL), IL-2 (0.23 pg/mL), IL-4 (0.20 pg/mL), IL-6 (0.26 pg/mL), IL-8 (0.12 pg/mL), IL-10 (0.23 pg/mL), IL-18 (1.57 pg/mL), TNF-α, (0.18 pg/mL), IFN-α-2a (0.74 pg/mL) and IFN-γ (0.24 pg/mL).
Analysis of tryptophan metabolites
We analyzed tryptophan, KYN and KYNA using a reversed phase high-performance liquid chromatography (HPLC) system, as previously described;23,25 50 μL samples were manually injected.
Analysis of KYN in astrocyte media
Standard curves were prepared in the range of 0.0004 to 10 μmol/L of KYN dissolved in Dulbecco phosphate-buffered saline (PBS) aliquoted and stored below −70°C. Astrocyte media and standard samples (50 μL) were prepared using solid phase extraction (Oasis MAX). We used D4-KYN as the internal standard and added it to each standard and media sample to a final concentration of 0.5 μmol/L. We purchased PBS from Gibco and KYN from Buchem.
After solid phase extraction the sample eluate was evaporated with nitrogen at 50°C and redissolved with 100 μL 0.1% formic acid in MilliQ water. We injected 7.5 μL of the filtrate into an Acquity HPLC system (Waters Corporation) equipped with a HSST3 2.1 × 100 mm, 1.8 μm particle column. The detection was performed using a Waters Xevo TQ-S triple quadrupole mass spectrometer operating in positive ionization MS/MS configuration. The mobile phase was run at a flow rate of 300 μL/min and consisted of 0.1% formic acid in MilliQ water (A phase) and 95% acetonitrile 0.1% formic acid (B phase) starting with 10% B for 2.5 minutes following gradient elution, with a total run time of 10 minutes. The formic acid and acetonitrile were purchased as MS-grade from Sigma-Aldrich. The mass spectrometer was tuned for KYN and set at capillary voltage of 2.0 kV, cone voltage of 4 V, source temperature of 150°C, desolvation temperature of 600°C, desolvation gas flow of 1000 L/hr and collision energy of 16 eV. Mass spectral transition was m/z 209 > 146 for KYN and 213 > 150 for the internal standard.
Calibration was performed using standards covering the range of the media concentration. Eleven concentration points were used to establish a linear calibration curve and plotted using the ratio of analyte peak area over internal standard peak area after integration by Masslynx 4.1 software (Waters Corporation). The retention time for KYN was 1.8 minutes.
Statistical analysis
Analyses of potential confounders were performed using a Mann–Whitney U test or Fisher exact test. Comparisons of CSF IL-6, IL-8, tryptophan, KYN, KYNA, tryptophan:KYN ratio, KYN:KYNA ratio and tryptophan:KYNA ratio between patients and controls were first analyzed using the Mann–Whitney U test. These analyses were then also performed using logistic regression analyses with age as a covariate and group (controls v. patients) as the dependent variable. We performed all correlation analyses using a Spearman rank correlation analysis. All values are reported as medians and interquartile ranges (IQR). We analyzed cell culture data using 1-way analysis of variance (ANOVA) with Bonferroni correction for comparisons within each time point. Differences in IL-6 between freeze/thaw cycles were analyzed using the Mann–Whitney U test. All values are reported as means and standard errors of the mean. We checked the assumptions of each test that we used. All reported p values are 2-sided. We performed our analyses using SPSS Statistics version 20.0 (IBM Inc.) or Prism version 6.0 (GraphPad Software Inc.).
Results
Participants
We included 23 patients (15 men, median age 37 [range 23–49] yr; 8 women, median age 35 [range 26–46] yr), 2 of whom had schizoaffective disorder, and 37 controls (23 men, median age 24 [range 21–51] yr; 14 women, median age 23 [range 19–32] yr) in the study. The median age was significantly higher in patients than controls (median 35.0, IQR 32.0–41.0 v. median 23.0, IQR 22.0–25.5, p < 0.001). Sex distribution, the percentage of smokers and BMI did not differ significantly between the groups (Table 1). In addition to olanzapine, 2 patients were taking zopiclone and the 2 patients with schizoaffective disorder were taking lithium. Six patients and 3 controls were smokers.
Demographic characteristics of patients with schizophrenia and controls
Analysis of potential confounders
No association was found between the CSF cytokines (IL-6 or IL-8) and age, plasma glucose, cholesterol, triglycerides, serum insulin or CSF/serum olanzapine. We found that CSF IL-8, but not IL-6, correlated with BMI in patients after age correction (Table 2).
Clinical characteristics and their association with IL-6 and IL-8
Detection of cytokines in CSF
We detected IL-6 in the CSF in all 37 controls and in 21 patients, and IL-8 was detected in the CSF of 36 controls and 22 patients. The other cytokines analyzed were undetectable (IFN-γ, IL-1β and IL-4) or were found to be very close to the detection limit of the assay, hereby only detected in a limited number of samples (IL-2, IL-10, IL-18, TNF-α, and IFN-α-2a).
CSF IL-6 in patients versus controls
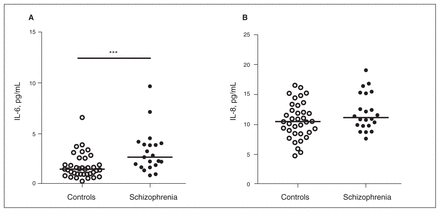
The CSF IL-6 concentration was elevated in patients with chronic schizophrenia compared with controls (2.68 pg/mL, IQR 1.79–3.99 pg/mL, n = 21 v. 1.50 pg/mL, IQR 1.01–2.25 pg/mL, n = 37, p = 0.001; Fig. 1A). When stratifying the analysis by sex, IL-6 was significantly increased both in men (patients: 2.68 pg/mL, IQR 1.67–3.88 pg/mL, n = 15; controls: 1.12 pg/mL, IQR 0.85–1.68 pg/mL, n = 23, p = 0.006) and women (patients: 3.25 pg/mL, IQR 1.92–5.83 pg/mL, n = 6; controls: 1.76 pg/mL, IQR 1.41–2.59 pg/mL, n = 14, p = 0.026). No correlation between age and CSF IL-6 was observed in either patients (r = 0.062, p = 0.79) or controls (r = 0.018, p = 0.92). In line with this, IL-6 was significantly higher in patients than controls after adjusting for age (p = 0.002).
(A) Interleukin (IL)-6 and (B) IL-8 in the cerebrospinal fluid (CSF) of controls and patients with schizophrenia. Each point represents the concentration of a single CSF sample, and the horizontal lines represent the median for each group. ***p < 0.001 (Mann–Whitney U test).
CSF IL-8 in patients versus controls
The CSF IL-8 levels did not differ between patients with schizophrenia and controls (11.15 pg/mL, IQR 9.80–15.24 pg/mL, n = 22 v. 10.5 pg/mL, IQR 8.55–12.99 pg/mL, n = 36, p = 0.16, and p = 0.74, adjusted for age and BMI; Fig. 1B).
Correlations between IL-6 and KYN and KYNA
The CSF levels of KYN were increased in patients with schizophrenia compared with controls (patients: 57.9 nM, IQR 48.0–71.3, n = 23; controls: 32.2 nM, IQR 26.2–49.0, n = 37, p < 0.001 and p = 0.007, adjusted for age). The CSF levels of KYNA were also increased in patients compared with controls (patients: 1.87 nM, IQR 1.63–2.29, n = 23; controls: 1.50 nM, IQR 1.14–1.92, n = 37, p = 0.006 and p = 0.040, adjusted for age). There were no significant differences in CSF tryptophan between patients with schizophrenia and controls (1.71 nM, IQR 1.61–1.87, n = 23 v. 1.77 nM, IQR 1.47–1.94, n = 37, p = 0.83 and p = 0.72, adjusted for age).
The tryptophan:KYN and the tryptophan:KYNA ratios were also significantly lower in patients than controls (patients: p < 0.001 and p = 0.005, adjusted for age; controls: p = 0.034 and p = 0.023, adjusted for age), whereas the KYN:KYNA ratio was close to significantly decreased in patients (p = 0.05 and p = 0.10, adjusted for age). We detected a correlation between IL-6 and the production of KYNA in patients with schizophrenia (r = −0.49; p = 0.024), as reflected by the tryptophan:KYNA ratio.
The effects of IL-6 on KYN and KYNA formation in human cortical astrocytes cells
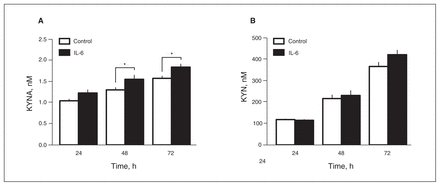
To investigate a putative association between the elevated levels of IL-6 and KYNA observed in patients, we exposed cultured fetal human cortical astrocytes to recombinant human IL-6 (10 ng/mL). Increased levels of KYNA were detected in the cell medium after 48 hours (1.55 ± 0.097 nM v. 1.29 ± 0.054 nM, p = 0.038) and 72 hours (1.83 ± 0.070 nM v. 1.56 ± 0.053 nM, p = 0.045) of IL-6 stimulation (Fig. 2A). The KYN levels were not affected by IL-6 treatment at any time point (Fig. 2B).
(A) Kynurenic acid (KYNA) and (B) kynurenine (KYN) in human cortical astrocytes exposed to interleukin (IL)-6 for 24, 48 and 72 hours. Bars represent means ± standard errors of the mean *p < 0.05 versus control (1-way analysis of variance).
Effects of repeated thawing/freezing cycles on the stability of IL-6
No significant difference in IL-6 concentrations was observed between CSF samples that were thawed once from −70°C (1.03 ± 0.29 pg/mL, n = 5) and samples after 2 thawing/freezing cycles from −70°C (1.01 ± 0.28 pg/mL, n = 5, p = 0.84).
Discussion
In the present study, CSF inflammatory markers were analyzed in a well-characterized cohort of olanzapine-treated outpatients with chronic schizophrenia. Among the 10 different cytokines analyzed, we found increased levels of IL-6 in patients compared with controls. Levels of other cytokines were unchanged (IL-8) or were found to be under the detection limit of the assay. Elevated levels of CSF IL-6 were not associated with age, sex, BMI or smoking habits or with any of the metabolic parameters measured, such as plasma levels of glucose, insulin, cholesterol and triglycerides. Furthermore, in line with previous studies,32,33 treatment with olanzapine appeared not to influence IL-6, as CSF or serum levels of olanzapine did not correlate with CSF IL-6.
Levels of CSF IL-6 have previously been studied in other psychiatric conditions. In major depression, CSF IL-6 was reported not to differ from controls.34,35 Further, in euthymic patients with bipolar disorder or in suicide attempters with depression this cytokine is reduced or elevated, respectively.36,37 The presently observed increase in CSF IL-6 in patients with chronic schizophrenia is in agreement with results of previous studies analyzing this cytokine in plasma or CSF.11,12,14,15,38 However, in a recent study from our laboratory, CSF IL-6 levels were not altered in patients with first-episode schizophrenia compared with healthy volunteers;13 rather, in those patients CSF IL-1β was increased. In the present study, IL-1β was undetectable both in patients and controls. The discrepancy between the results of our previous study and the present results may be related to the use of different platforms for the analysis of cytokines (Luminex vs. MesoScale) or to the fact that all CSF samples in the present study had been stored for 3–6 years and frozen and thawed twice. Both storage time and multiple freezing/thawing cycles are reported to affect the concentration of cytokines, including IL-1β, in human samples.39 However, both of our investigations and another study39 suggest that IL-6 may be an exception in this regard. The reason for the discrepancy with regard to CSF IL-6 levels in patients with first-episode versus chronic schizophrenia is unclear, but may be related to the chronic progress of the disease.
In the present study we confirm that CSF levels of KYN and KYNA are increased in patients with schizophrenia. We also discovered a positive correlation between CSF IL-6 and the tryptophan:KYNA ratio. Proinflammatory cytokines, such as IFN-γ, TNF-α or IL-1β are known to induce tryptophan 2,3-dioxygenase (TDO) and/or indolamine 2,3-dioxygenese (IDO), rate-limiting enzymes of the KYN pathway.40,41 Further, injection of IL-6 into the rat hippocampus has been found to be associated with the induction of IDO.42 A decreased tryptophan:KYNA ratio, tentatively reflecting the activity of IDO/TDO, would explain the elevation in CSF KYN and KYNA consistently found in patients with schizophrenia. Indeed, increased TDO activity in the postmortem brains of patients with schizophrenia has been reported.22,43 In line with this, the present study shows that fetal human cortical astrocytes respond to application of IL-6 with an increased KYNA synthesis, suggesting an intimate interplay between IL-6 and the KYN pathway.
Increased levels of brain KYNA may be the link between immune activation and aberrant neurotransmission and behaviour in patients with schizophrenia. Thus, experimental studies demonstrate that elevated KYNA affects brain glutamatergic/dopaminergic neurotransmission, hereby implicating activation of the KYN pathway in established models of schizophrenia.18,43,44 Moreover, elevated KYNA induces schizophrenia-like behaviour, such as disrupted prepulse inhibition45 and auditory sensory gating46 as well as impaired contextual discriminations,47 spatial working memory48,49 and attentional set-shifting,50 in rodents. Notably, a specific inhibitor of KYN aminotransferase II, which reduces brain KYNA levels, prevents ketamine-induced working memory impairments and tends to attenuate hallucinatory-like behaviours in primates.51 Together, these findings strongly support the hypothesis that increased brain KYNA constitutes a major trigger for cognitive and psychotic symptoms and should encourage the development of high-quality biomarkers and novel treatment approaches based on the KYN pathway.
Limitations
The major limitation of the present study was the use of CSF that had been frozen and thawed twice. Although KYNA is a stable compound and not affected by repeated freezing and thawing,52 some cytokines are considerably more sensitive in this regard. Also, the use of a multiplex assay to quantify cytokines may yield less sensitivity than a singleplex procedure. The median age differed significantly between patients and controls; however, we controlled for this confounder in the statistical analysis. Finally, given the circadian rhythmicity of CSF IL-6,53 lumbar puncture was performed only once in each individual.
Conclusion
Our data support previous findings that brain IL-6, KYN and KYNA are elevated in patients with chronic schizophrenia and strengthen the idea of brain immune activation in people with the disease. The increased production of KYNA in fetal human astrocytes following exposure of IL-6 shows that this cytokine is able to induce the activity of the KYN pathway. No correlations between CSF IL-6 and psychiatric symptom scores (GAF and BPRS) were found, possibly owing to the fact that all patients were well controlled, stable and under chronic treatment with olanzapine. Future studies may reveal whether elevation in brain IL-6 contributes to psychiatric symptoms in patients with schizophrenia.
Acknowledgements
This work was supported by grants from the Swedish Medical Research Council awarded to G. Engberg (2009–3068, 2011–4789), S. Erhardt (2009–7053, 2013–2838), C. Svensson (2009–3068) and M.-L. Dahl (2008–2578, 2013–2592); the Swedish Brain foundation, Petrus och Augusta Hedlunds Stiftelse, Östergötland County Council; the AstraZeneca-Karolinska Institutet Joint Research Program in Translational Science, the Karolinska Institutet (KID). The authors thank the patients and healthy volunteers for their participation and the health professionals who facilitated the work.
Footnotes
↵* These authors contributed equally to this work.
Competing interests and funding: None declared. The clinical research study was sponsored by Linköping University Hospital with an in-kind contribution to the sample analysis from AstraZeneca. AstraZeneca neither influenced nor sponsored the clinical research performed at Linköping University. These data arise from a sample collection, not from a prospective trial and are therefore not recorded in any clinical trial registry.
Contributors: L. Schwieler, M. Larsson, E. Skogh, C. Svensson, S. Erhardt and G. Engberg designed the study. L. Schwieler, M. Larsson, E. Skogh, M. Kegel, S. Abdelmoaty, M. Bhat, M. Samuelsson, M.-L. Dahl and S. Erhardt acquired the data, which L. Schwieler, M. Larsson, F. Orhan, A. Finn, C. Sellgren, I. Shuppe-Koistinen, C. Svensson, S. Erhardt and G. Engberg analyzed. L. Schwieler, M. Larsson, S. Erhardt and G. Engberg wrote the article, which all authors reviewed and approved for publication.
- Received May 15, 2014.
- Revision received August 13, 2014.
- Accepted September 3, 2014.