Abstract
Background: Depression and stress are major risk factors for suicidal behaviour, and some studies show abnormalities of proinflammatory cytokines in the serum and cerebrospinal fluid (CSF) of depressed and suicidal patients. However, it is not clear if similar abnormalities of cytokines are present in the brain of suicidal and depressed patients.
Methods: We therefore determined the mRNA (using real-time polymerase chain reaction) and protein (using enzyme-linked immunosorbent assay and Western Blot) expression levels of interleukin (IL)-1β, IL-6, tumour necrosis factor (TNF)-α, lymphotoxin A, lymphotoxin B, IL-8, IL-10 and IL-13 in the prefrontal cortex (PFC) obtained from 24 depressed individuals who died by suicide and 24 nonpsychiatric controls.
Results: We observed that the mRNA and protein levels of IL-1β, IL-6, TNF-α, and lymphotoxin A were significantly increased, and levels of anti-inflammatory cytokine IL-10, and of IL-1 receptor antagonist (IL-1RA) were significantly decreased in the PFC of depressed individuals who died by suicide compared with controls. There were no significant differences in the protein and mRNA levels of IL-8 and IL-13 in the PFC.
Limitations: The main limitation of this study is that some of the suicide group had been taking antidepressant medication at the time of death.
Conclusion: Our results suggest that alterations of cytokines may be associated with the pathophysiology of depressed suicide and there may be an imbalance between pro- and anti-inflammatory cytokines in people who die by suicide. The causes of these increases in the brain of people who die by suicide, therefore, need to be investigated further.
Introduction
Suicide is a major public health concern, as 44 000 individuals die by suicide annually in the United States.1 Depression and stress are major risk factors for suicide. Abnormalities of the immune system have been suggested to contribute to the pathogenesis of depression based on a growing body of evidence. Several studies suggest that altered immune states may be present in patients with depression (for reviews, see Howren and colleagues2 and Valkanova and colleagues3).
Cytokines comprise a heterogeneous group of messenger molecules produced by immunocompetent cells, such as lymphocytes and macrophages. Several studies suggest that the administration of cytokines to animals4 or humans5,6 causes a set of behavioural changes, known as “sickness behaviour,” that is similar to depression.
Evidence that cytokines might play an important role in inducing depression has also been provided by the observation that the administration of cytokine interferon (IFN)-α to patients with cancer or melanoma causes sickness behaviour (for a review, see Dantzer and colleagues7). However, the main evidence suggesting that abnormalities of proinflammatory cytokines may be associated with the pathophysiology of depression is based on observations by many investigators that the levels of proinflammatory cytokines and their soluble receptors appear to be increased in the serum of depressed patients.8–10 Stressful events have also been shown to cause changes in immune function. 11 Several studies indicate that stress increases infection and autoimmune disease due to stress-related immunosuppression, and exposure to stressful life events causes impairment in various aspects of cellular immune function (for reviews, see Garcia-Bueno and colleagues11 and Goshen and Yirmia12). Stress-induced alterations in the hypothalamic–pituitary–adrenal (HPA) axis function cause changes in cytokine levels and function, 12 providing evidence for stress-related changes in cytokines.
Both depression and stress are major risk factors for suicidal behaviour; therefore, it is not surprising that abnormal immune function has also been implicated in suicidal behaviour (for a review, see Black and Miller13). Some recent studies suggest that cytokines may be abnormal in individuals who die by suicide or in suicidal patients. For example, Lindqvist and colleagues14 found increased levels of interleukin (IL)-6 in the cerebrospinal fluid (CSF) of suicide attempters compared with controls, and Janelidze and colleagues15 observed elevated levels of IL-6 and tumour necrosis factor (TNF)-α with decreased levels of IL-2 in suicide attempters. The available evidence thus suggests that there are abnormalities in proinflammatory cytokines in people who die by suicide and in suicide attempters. However, it is not clear if there are alterations of cytokines in the brain of depressed individuals who die by suicide. Shelton and colleagues16 studied the expression of inflammatory genes in postmortem brain samples (Brodmann area [BA] 10) of depressed patients. They found that several genes, including IL1A and IL2, were upregulated in the prefrontal cortex (PFC) of depressed patients. In an earlier study, we determined the protein and mRNA levels of proinflammatory cytokines, IL-1β, IL-6 and TNF-α, in the PFC of teenagers who died by suicide and controls.17 We found that both the protein and mRNA levels of these 3 cytokines were significantly increased in the teenagers who died by suicide.
The neurobiology of teenage suicide may differ somewhat from adult suicide,18–21 therefore, in the present study we sought to determine the protein and mRNA expression levels of inflammatory cytokines TNF-α, TNF-β, lymphotoxin A, lymphotoxin B, IL-1β, IL-6, IL-1 receptor antagonist (IL-1RA), IL-8, IL-10 and IL-13 in the PFC of depressed adults who died by suicide and matched controls. We selected these cytokines based on the following rationale. Levels of proinflammatory cytokines, such as IL-1β, IL-6 and TNF-α, have been shown by our group17,22 and by other investigators (for reviews, see Black and Miller13 and Dowlati and colleagues8) to be higher in both serum and postmortem brain of depressed individuals who died by suicide. In order to examine if there is an imbalance between the pro- and anti-inflammatory cytokines, we studied the protein and mRNA expression of the anti-inflammatory cytokines IL-10 and IL-13. We studied IL-8 because it has been shown by some investigators to be lower in the CSF of suicidal patients.23 Lymphotoxin A and lymphotoxin B are members of the TNF superfamily and are important in brain function, and we have recently shown these to be altered in the postmortem brain of individuals with schizophrenia.24 Since TNF-α has been found to be increased in both serum and postmortem brain of depressed patients, it was of interest to examine whether the other members of the TNF superfamily are also abnormally expressed in the postmortem brain of people who died by suicide.
Based on our own studies and those reported in the literature, we hypothesized that the brain levels of proinflammatory cytokines IL-1β, IL-6 and TNF-α would be higher and that levels of IL-8, IL-10 and IL-13 would be lower in the postmortem brain of depressed individuals who died by suicide than in controls. We also hypothesized that levels of the other members of the TNF superfamily, that is lymphotoxin A and lymphotoxin B, would be higher, similar to TNF-α, and that there would be an imbalance between the proinflammatory cytokines (i.e., IL-1β, IL-6 and TNF-α) and the anti-inflammatory cytokines (i.e., IL-10 and IL-13).
Methods
Sample and diagnoses
The study was performed on the PFC (BA9) of 24 depressed individuals who died by suicide and 24 nonpsychiatric controls. Brain tissues were obtained from the Maryland Brain Collection at the Maryland Psychiatric Research Center, Baltimore. Tissues were collected only after a family member gave informed consent. All tissue from controls and depressed individuals was grossly examined by experienced neuropathologists. Toxicology data were obtained by analysis of urine and blood samples. All procedures were approved by the University of Maryland Institutional Review Board (IRB) and University of Illinois IRB.
The nonpsychiatric control group included accident victims with no history of psychiatric disorders or substance abuse and/or family history of psychiatric disorders. They could be male or female, and from any ethnic or racial background. We excluded those who were drivers in car accidents, those who died due to fire, and those who died after prolonged hospital admission. We also excluded those with major medical or neurologic disorders, those who were HIV-positive and those with a history of psychiatric disorders.
Diagnostic method
Diagnosis was based on the Structured Clinical Interview for DSM-IV (SCID I).25 At least 1 family member and/or a friend, after giving written informed consent, underwent an interview. Diagnoses were made by consensus between 2 psychiatrists based on the data obtained in the interview, medical records from the case, and records obtained from the Medical Examiner’s office. We confirmed that controls lacked mental illnesses using these consensus diagnostic procedures.
Determination of mRNA levels
We extracted total RNA from 100 mg of tissue using TRIZOL reagent (Invitrogen, Thermo Fisher Scientific Inc.), which was followed with DNase treatment according to the manufacturer’s instructions. The RNA concentration and purity was determined by measuring the optical density with NanoDrop®ND-1000 (NanoDrop Technologies, LLC) and using the 260/280 nm ratio with expected values between 1.8 and 2.0. The RNA quality was assessed using an Agilent Bio-analyzer 2100 (Agilent Technologies) to determine the RNA integrity number (RIN). All samples had 28S/18S ratios greater than 1.2 and RINs above 7.0.
The expression levels of mRNA were determined using a 2-step real-time polymerase chain reaction (qPCR) method. Here, 1 μg of total RNA was reverse transcribed using 50 ng of random hexamers, 2 mM dNTP mix, 10 units of ribonuclease inhibitor and 200 units of MMLV-reverse transcriptase enzyme in a final reaction volume of 20 μl.
Real-time PCR was performed using Pre-designed Taqman gene expression assays (Applied Biosystems) for all target and housekeeping genes on a MX3005p sequence detection system (Agilent Technologies). The TaqMan assay IDs are listed in Table 1. To determine the stability and optimal number of housekeeping genes, we used geNORM version 3.4 (PrimerDesign Ltd) according to the manufacturer’s instructions26 and tested 12 commonly used reference genes from different functional classes in 10 samples from each test group.26 The average gene-stability measure ranked β-actin and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) as the most stable genes in our samples. The PCR efficiency was tested over 5-log dilution series and confirmed that all target genes and housekeeping genes had similar amplification efficiencies. For each primer/probe set, the PCR was performed using 10 μL of cDNA diluted 1:10-fold. Each qPCR plate included “no reverse transcriptase” and “no template” controls to eliminate nonspecific amplification. One sample from each target gene was run on a gel to confirm specificity, and all samples were run in triplicate. Target gene qPCR data are normalized to the geometric mean of β-actin and GAPDH and then expressed relative to the control samples using the 2−(ΔΔCt) method, where ΔΔCt = (Ct target – Ct internal control) subject – (Ct target – Ct internal control) control. The Ct internal control is the geometric mean of β-actin and GAPDH Ct values. Outliers were excluded if the normalized (ΔCt) values were greater than 2 standard deviations from the group mean. The relative expression levels are reported as the fold change, and ΔCt values are used for further statistical analysis (Applied Biosystems).
TaqMan primers/probes used for real-time polymerase chain reaction analysis
Determination of brain protein levels
The levels of IL-1β IL-6 and TNF-α were determined in brain homogenates (100 mL) with an enzyme-linked immunosorbent assay (ELISA) using commercially available Quantakine kits (R & D Systems, Inc.) for human IL-1β, human IL-6 and human TNF-α, according to the manufacturer’s instructions.
Immunolabelling of other cytokines (i.e., lymphotoxin A, lymphotoxin B, IL-1RA, IL-8, IL-10 and IL-13) was determined using the Western blot method in membrane fractions, as described by our group in an earlier study.27 Briefly, equal volumes of membrane fractions isolated by this procedure (30 μg of protein in 20 μL) were loaded onto 4%–12% (weight per volume) Bis-Tris gel and electrophoresed. The blots were initially developed using the polyclonal primary antibodies (1:1000 dilution) and subsequently using horseradish-peroxidase-linked secondary anti-rabbit antibody at a dilution of 1:3000. The membranes were stripped using stripping solution (Chemicon International) and probed with β-actin monoclonal primary (1:5000 dilution; Sigma Chemical Co.) and anti-mouse secondary antibody (1:5000 dilution). The levels of inflammatory cytokine proteins were calculated as a ratio of the optical density of the primary antibody to the optical density of β-actin antibody. The following polyclonal antibodies were purchased from Santa Cruz Biotechnology, Inc.: IL-8 (molecular weight 8 kDa), IL-10 (molecular weight 20 kDa), IL-13 (molecular weight 13 kDa) and IL-1RA (molecular weight 25 kDa). Lymphotoxin A polyclonal antibody (molecular weight 19 kDa) was purchased from Abnova, and lymphotoxin B (molecular weight 25 kDa) was purchased from Abcam. Secondary antibodies were purchased from GE Healthcare (Life Sciences).
Statistical analysis
We performed the data analyses using the SAS 9.2 statistical software package. A generalized linear model was fitted to compare the effects of 2 groups — depressed individuals who died by suicide and nonpsychiatric controls — on inflammatory cytokine expression by adjusting the effects of age, sex, postmortem interval (PMI) and brain pH. To examine whether the protein and mRNA expression of cytokines was affected by the PMI, age, sex, or brain pH, we included these variables as covariates in the model.
As we performed several tests, we also provided the significant results using the false discovery rate (FDR) controlling procedure for multiple testing. In addition, each outcome measure was tested for normality using the Kolmogorov–Smirnov test. The non-normal outcome measure was log-transformed and then used for the analysis. Protein levels of IL-1β, IL-6, IL-8 and TNF were not normal according to Kolmogorov–Smirnov testing; however, after log-transformation they were normally distributed (IL-1β: p = 0.06; IL-6: p = 0.06; IL-8: p = 0.15; and TNF-α: p = 0.25). A similar observation was made in mRNA levels for IL-13 and IL-10.
We did not observe any significant effect of confounding variables, such as age, race, sex, PMI or brain pH, on protein expression for the main outcome measures between the depressed individuals who died by suicide and the nonpsychiatric control groups. However, there was a significant effect of age (TNF-α), PMI (IL-1β, lymphotoxin A) and brain pH (IL-10) on mRNA expression.
Results
Demographic and clinical characteristics of the sample
The demographic and clinical characteristics of the depressed individuals who died by suicide and the nonpsychiatric controls are described in Table 2. There was no significant difference in the mean age and PMI between the 2 groups. Also shown are the presence and absence of psychoactive medications and the cause of death.
Characteristics of nonpsychiatric controls (n = 24) and depressed individuals who died by suicide (n = 24) included in the present study
mRNA expression levels of cytokines in the PFC
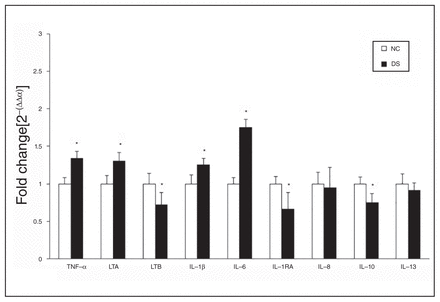
We determined the mRNA levels of TNF superfamily members, consisting of TNF-α, lymphotoxin A (also known as TNF-β) and lymphotoxin B, and the mRNA levels of IL-1β, IL-6, IL-1RA, IL-8, IL-10 and IL-13 in the PFC of depressed individuals who died by suicide and controls (Fig. 1). These results are also shown as dot plots in Appendix 1, Figure S7, available at jpn.ca/170192-a1. Our results showed that overall there were differences between the suicide and control groups in mRNA levels of TNF-α (F5,43 = 3.51, p = 0.01) while adjusted for confounding variables. A post hoc t test showed that the mRNA levels of TNF-α (t = −3.30, p = 0.002) and lymphotoxin A (t = −2.30, p = 0.03) were significantly higher in the PFC of depressed individuals who died by suicide than in controls (Fig. 1). However, mRNA levels of lymphotoxin B (t = 2.18, p = 0.03) were significantly lower in depressed individuals who died by suicide than in controls (Fig. 1).
Mean mRNA expression levels of inflammatory cytokines, including tumour necrosis factor (TNF)-α, lymphotoxin A (LTA), lymphotoxin B (LTB), interleukin (IL)-1β, IL-6, IL-1 receptor antagonist (IL-1RA), IL-8, IL-10, and IL-13, in the prefrontal cortex (PFC) of depressed individuals who died by suicide (DS) and nonpsychiatric controls (NC). The data are shown as the fold change in mRNA levels ± standard error of the mean. *p < 0.05.
A comparison of mRNA levels of IL-1β, IL-6 and IL-1RA between the 2 groups revealed overall significant differences in the mRNA levels of IL-6 (F5,44 = 8.48, p < 0.001). Post hoc analysis showed significant differences in the mRNA levels of both IL-1β (t = −2.11, p = 0.04) and IL-6 (t = −5.87, p < 0.001) and that mRNA levels were significantly higher in depressed individuals who died by suicide than in controls. However, mRNA levels of IL-1RA (t = 2.28, p = 0.03) were significantly lower in the suicide than in the control group (Fig. 1).
The mRNA levels of IL-8, IL-10 and IL-13 in depressed individuals who died by suicide and nonpsychiatric controls are shown in Figure 1. A post hoc t test showed that the levels of IL-10 were significantly lower (t = 3.12, p = 0.003) in the suicide than in the control group. However, the mRNA levels of IL-8 and IL-13 did not differ significantly.
Using the FDR controlling procedure with q = 0.05, we found that the mRNA levels of IL-6, TNF-α, IL-10, IL-1RA, lymphotoxin B and IL-1β were significantly different in depressed individuals who died by suicide compared with controls.
Protein expression levels of cytokines in the PFC
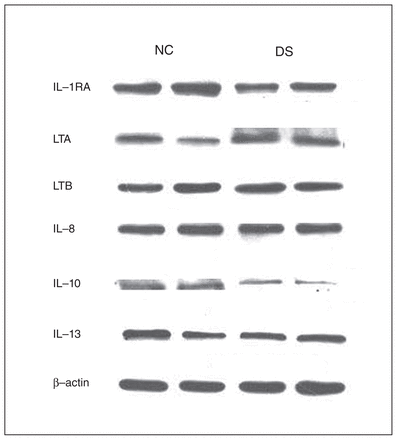
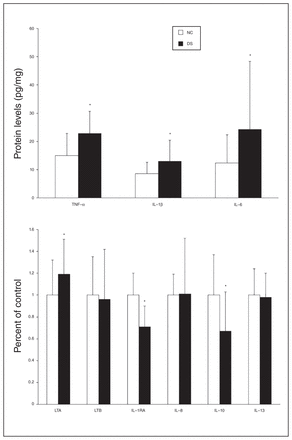
Because we observed differences in the mRNA levels of some cytokines in the depressed individuals who died by suicide compared with nonpsychiatric controls, we examined whether there were comparable differences in the protein levels of these cytokines in depressed individuals who died by suicide. We determined the protein expression levels of these cytokines in the PFC in both groups. Representative immunoblots showing the protein expression of lymphotoxin A, lymphotoxin B, IL-1RA, IL-8, IL-10 and IL-13 in 2 controls and in 2 individuals who died by suicide are shown in Figure 2. The detailed blots for these cytokines are shown in Appendix 1, Figures S1–S6. The protein expression levels are shown in Figure 3A and 3B. The protein expression results are shown as dot plots in Appendix 1, Figure S8–S9. Analysis of variance results showed that there were significant differences between the 2 groups in the protein levels of TNF-α (F5,46 = 4.60, p = 0.002), IL-1β (F5,46 = 5.93, p < 0.001), IL-1RA (F5,42 = 5.30, p < 0.001), IL-6 (F5,46 = 2.72, p = 0.03), IL-10 (F5,42 = 2.47, p = 0.04) and lymphotoxin A (F5,42 = 3.74, p = 0.07). The post hoc t test showed that the protein levels of TNF-α were significantly increased in depressed individuals who died by suicide (t = 4.50, p = 0.006), compared with controls (Fig. 3A). The protein levels of lymphotoxin A were also significantly increased in the suicide (t = 4.36, p < 0.001) compared with the control group (Fig. 3B). However, the protein levels of lymphotoxin B did not differ significantly between the groups (Fig. 3B).
Representative Western blots showing the immunolabelling of interleukin (IL)-1 receptor antagonist (IL-1RA), lymphotoxin A (LTA), lymphotoxin B (LTB), IL-8, IL-10, IL-13 and β-actin in the prefrontal cortex membrane fraction of 2 nonpsychiatric controls (NC) and 2 depressed individuals who died by suicide (DS).
(A) Mean protein expression levels (pg/mg tissue) of tumour necrosis factor (TNF)-α, interleukin (IL)-1β and IL-6 and (B) percent of control of lymphotoxin A (LTA), lymphotoxin B (LTB), IL-1 receptor antagonist (IL-1RA), IL-8, IL-10 and IL-13 in the prefrontal cortex of depressed individuals who died by suicide (DS) and nonpsychiatric controls (NC). Values are given as the mean ± standard deviation. *p < 0.05.
The IL-1β (t = 2.65, p = 0.01) and IL-6 (t = 2.35, p = 0.03) protein levels were significantly higher in the suicide than in the control group (Fig. 3A), whereas the protein levels of IL-1RA were significantly decreased in the suicide group (t = −5.27, p < 0.001; Fig. 3B). The protein levels of IL-8 and IL-13 were not significantly different between the groups. The protein levels of IL-10 were significantly lower in the suicide than in the control group (t = −3.16, p = 0.003).
Using the FDR controlling procedure with q = 0.05, we found that the protein levels of IL-6, TNF-α, IL-10, IL-1RA, lymphotoxin A and IL-1β differed significantly between depressed individuals who died by suicide and controls.
Effect of antidepressants and the mode of death
We examined whether the observed changes in the protein and mRNA expression of cytokines in the PFC of the sample we studied were associated with previous treatment and/or presence of antidepressant drugs or with the mode of death. In the suicide group, 11 of 24 individuals had been taking antidepressants at the time of death. When we compared the mRNA and protein expression levels of the cytokines between suicide completers who were on antidepressants and those who were free of antidepressants, we did not find any significant differences in any of the cytokines we studied.
In the suicide group, we also examined the effect of the mode of death (i.e., violent suicide v. nonviolent suicide). We did not find a significant difference between the violent (n = 12) and the nonviolent (n = 12) suicide subgroups on the protein and mRNA expression on any of the cytokines we studied.
Discussion
In this study, we found that the protein and mRNA levels of proinflammatory cytokines TNF-α, lymphotoxin A, IL-1β, and IL-6 were significantly increased and those of anti-inflammatory cytokines IL-10 and IL-1RA were significantly decreased in the PFC of depressed individuals who died by suicide compared with nonpsychiatric controls. There were no significant differences in the protein and mRNA levels of IL-8 and IL-13 in the PFC of the suicide compared with the control group. These observations suggested an upregulation of proinflammatory cytokines and downregulation of 1 of the anti-inflammatory cytokines (i.e., IL-10) in depressed individuals who died by suicide.
Some studies have suggested that antidepressant treatment may decrease the levels of proinflammatory cytokines in depressed patients. For example, Kim and colleagues28 did not find significant changes in IL-23 and IL-17 after 6 weeks of antidepressant treatment. However, some studies have reported decreased levels of proinflammatory cytokines after antidepressant treatment.29,30 We did not find an effect of antidepressant treatment on the cytokines in the suicide group in the present study. Since antidepressant treatment decreases cytokines and we found increased cytokine levels in the suicide group, it is unlikely that this increase was due to prior treatment with antidepressants.
Several studies have suggested abnormalities of proinflammatory cytokines in suicidal behaviour (for reviews, see Black and Miller,13 Ducasse and colleagues,31 and Serafini and colleagues32). However, these studies were primarily performed in the serum or CSF of suicidal and nonsuicidal patients and controls. Additionally, these studies primarily determined the protein expression levels of cytokines. Some studies that were conducted in the CSF of suicidal patients14 suggested abnormalities of CNS cytokines in suicidal patients, but such cytokine abnormalities are not clearly shown to be present in the brain.
Abnormalities of cytokines have been implicated in the pathophysiology of major depressive disorder, based on both human and animal studies (for reviews, see Miller and colleagues33 and Schiepers and colleagues10). Most, but not all of these studies indicate abnormalities of the proinflammatory cytokines IL-1β, IL-6 and TNF-α both in the serum and the postmortem brain samples. The studies of serum and CSF in depression have been reviewed by Dowlati and colleagues.8 They analyzed the studies for several cytokines, such as IL-1β, IL-6, TNF-α, IL-4, IL-2, IL-8 and IL-10 in depression. Their meta-analysis found that the levels of IL-6 and TNF-α were significantly increased in depressed patients. Some studies also found significant effects of antipsychotic treatment on the levels of cytokines. Although IL-1β was not found by Dowlati and colleagues8 to be significantly increased in depressed patients, IL-1β has been generally found to be increased by some investigators.
Although there are several studies of cytokines in patients with major depressive and bipolar disorders, there are only a few studies of cytokines in suicidal patients with mood disorders. There have also been several meta-analyses of cytokines in suicidal patients, including the one by Black and Miller.13 Since we studied cytokines in depressed individuals who died by suicide, we discuss some of the findings of cytokines in suicidal patients, particularly those with major depressive disorder. Janelidze and colleagues15 determined the plasma levels of IL-2 and TNF-α in suicide attempters, depressed nonsuicidal patients and nonpsychiatric controls. They found increased levels of IL-6 and TNF-α as well as decreased levels of IL-2 in suicide attempters compared with depressed non-suicidal patients and controls.
Lindqvist and colleagues14 determined the protein levels of IL-1β, IL-6, IL-8 and TNF-α in the CSF and plasma of suicide attempters and nonpsychiatric controls. They found that the protein levels of IL-6 in the CSF were significantly higher in suicide attempters than in controls. They also found that patients who died by violent suicide had higher IL-6 levels than controls. In addition, they found that the levels of IL-1β, IL-8 and TNF-α did not differ between the suicide attempters and controls.
Isung and colleagues23 evaluated the vascular endothelial growth factor and IL-8 levels in the CSF of medication-free suicide attempters and healthy male controls. They found that suicide attempters had lower vascular endothelial growth factor and IL-8 levels in the CSF than controls. In a meta-analysis, Black and Miller13 combined all suicidal and nonsuicidal patients and controls and found that the levels of IL-β were significantly increased in patients with suicidal behaviour compared with patients with nonsuicidal behaviour; no other markers were statistically significant. However, they found significant differences in the effect size estimates for IL-6, IL-10 and TNF-α. This meta-analysis also compared the blood levels of cytokines in suicidal patients and controls. They concluded that the blood levels of IL-6, IL-10, and C-reactive protein were significantly increased in patients with suicidal behaviour compared with controls. In addition, there were no significant differences for IL-1β and TNF-α between suicidal patients and controls. Ducasse and colleagues,31 in their meta-analysis of cytokine studies in suicidal and nonsuicidal patients and controls, found no differences in the IL-6, TNF-α, and IL-1β levels between suicidal and nonsuicidal patients. Therefore, the meta-analysis results of cytokine studies in suicidal and nonsuicidal patients appear to be inconsistent, although some studies suggest differences in IL-1β, IL-6, and TNF-α between suicidal and nonsuicidal patients and controls.
Although there are several studies of cytokines in the serum of depressed and suicidal individuals, the number of cytokine studies in depression, bipolar disorder or schizophrenia is limited. Shelton and colleagues34 studied the expression of inflammatory cytokines in the postmortem brain of depressed and suicidal individuals and found that several genes, such as IL1A and IL2, were upregulated in the PFC of depressed individuals.34 Dean and colleagues35 observed transmembrane TNF-α in the postmortem brain samples from individuals with bipolar disorder and schizophrenia. They found that there was a significant increase in transmembrane TNF-α in BA24, but not BA46, in those with bipolar disorder. Protein levels were not altered in those with schizophrenia. The authors also found that the levels of soluble TNF-α were not altered in any of the brain areas in bipolar disorder or schizophrenia. They also found that the levels of TNFR1 were increased in BA24 and BA46 of those with major depressive and bipolar disorders. In an earlier study of postmortem brain samples, Dean and colleagues36 found that transmembrane TNF-α was significantly increased in BA46, but not in BA24, in individuals with major depressive disorder, and there was no change in soluble TNF in either of the CNS regions they studied.
Tonelli and colleagues37 studied cytokines in the postmortem brain of suicide completers and found sex-related changes in TNF-α and IL-1β levels in the PFC. We have previously found increased levels of IL-1β, IL-6 and TNF-α in BA10 of teenagers who died by suicide.17 In a recent study, Clark and colleagues38 found, contrary to their expectations and previous study, that the mRNA levels of IL-2, IL-33, interferon (IFN)-γ, TNF-α and CCL2 were lower in the ventrolateral PFC of depressed individuals than in nonpsychiatric controls.
Wang and colleagues39 observed HPA-associated genes and a number of factors that stimulate or inhibit corticotropin-releasing factor in the hypothalamus of depressed individuals and matched controls. Whereas they found alterations in corticotropin-releasing factor and CRFR1 mRNA levels in depressed patients, they did not find changes in cytokine genes TNF-α or IL-1β in depressed individuals compared with controls. The present study, in combination with other studies discussed here, may suggest that cytokine changes in depressed postmortem brain may be region-specific.
Steiner and colleagues40 found increased microgliosis in the postmortem brain of suicidal individuals with depression and schizophrenia, suggesting immune function abnormalities in suicide completers. Torres-Platas and colleagues41 found astrocyte hypertrophy in anterior cingulate white matter of depressed individuals who died by suicide (for a review, see Mechawar and Savitz42). These studies indicate abnormal immune function in postmortem brain of depressed individuals who died by suicide. Torres-Platas and colleagues43,44 also found that the ratio of primed over ramified microglia was significantly increased in the dorsal anterior cingulate cortex in depressed individuals who died by suicide. However, there were no significant differences in overall microglial densities between individuals who died by suicide and controls.
The source of cytokines in the postmortem brain and mechanism of their increase in individuals who die by suicide is unclear. Cytokines have been shown to be present in the human brain; they can also be synthesized in the human brain (for a review, see Vidrih and colleagues45). On the other hand, cytokines can cross the blood–brain barrier under certain conditions (for reviews, see Schiepers and colleagues10 and Vidrih and colleagues45). Thus, increased proinflammatory cytokines in the postmortem brain of individuals who die by suicide may be related to both processes.
An imbalance between proinflammatory cytokines, such as IL-1β, IL-6, and anti-inflammatory cytokines, such as IL-4 and IL-10, has been found in depressed patients.46,47 In our study, we not only observed an increase in proinflammatory cytokines, but also a decrease in anti-inflammatory cytokine IL-10. This may even exaggerate the abnormality of proinflammatory cytokines in individuals who die by suicide.
The mechanism by which cytokines can induce depression or suicidal behaviour is not clear. However, cytokines influence the neurotransmitters system, particularly the serotonergic system (for a review, see Miller and colleagues48), as well as the HPA axis (for reviews, see Turnbull and Rivier49 and Zunszain and colleagues50), both of which have been implicated in the pathophysiology of depression and suicide. For example, proinflammatory cytokines cause changes in 5-hydroxytryptamine (5HT) transmission in the brain areas that are implicated in the pathophysiology of depression and suicide, such as the PFC, hippocampus and amygdala.45 Cytokines decrease activity of presynaptic neurons and cause changes in 5HT uptake as well as changes in the synthesis of 5HT.45 Cytokines also reduce tryptophan availability by activating the enzyme indoleamine 2,3-deoxygenase.7 They affect reuptake activity through the activation of signalling pathways, such as MAP kinase.51 TNF and IL-1 increase the expression of the serotonin transporter.52–54 Proinflammatory cytokines activate the enzyme indoleamine 2,3-deoxygenase leading to the synthesis of kynurenine from tryptophan, the precursor of serotonin, and thus affecting the synthesis and levels of serotonin by its depletion. This depletion in serotonin levels may induce or cause depressive symptoms in some individuals.
Cytokines can also cause changes in the HPA axis system (for reviews, see Turnbull and Rivier49 and Zunszainet and colleagues50), which again has been implicated in both depression and suicidal behaviour. A number of cytokines, such as IL-1β and TNF-α, are potent activators of the HPA axis50 and they can cause the expression and the release of corticotropin-releasing factor and the pituitary release of adrenocorticotropic hormone (for a review, see Turnbull and Rivier49). The HPA axis feedback mechanism can also be altered by cytokines. Acute cytokine administration to humans was shown to cause both elevated plasma levels of adrenocorticotropic hormone and cortisol and an increase in the corticotropin-releasing factor release.55,56 Thus, cytokines may affect 2 major systems, the serotonergic mechanism and the HPA axis mechanism, which have been shown to be involved in the pathophysiology of depression and suicidal behaviour.
Limitations
This study has some limitations. We do not have body mass index information for the individuals included, and it is not certain if any of the observed changes in cytokines are related, at least in part, to the body mass index. Some of the individuals in the suicide group had been taking antidepressants at the time of death. Although we did not observe differences in cytokine levels in depressed individuals treated with antidepressants and those free of antidepressants, this is one of the limitations of a postmortem brain study. The number of postmortem samples available for these studies was limited. Although we had 24 pairs of depressed individuals who die by suicide and matched nonpsychiatric controls for this study, our small sample size may still be a limitation.
Conclusion
These studies of cytokines in the PFC of depressed individuals who died by suicide suggest abnormalities of both proinflammatory cytokines, such as IL-1β, IL-6 and TNF-α, and anti-inflammatory cytokines, such as IL-10, in suicidal and depressed individuals. These studies also suggested an imbalance between pro- and anti-inflammatory cytokines based on the increase in proinflammatory cytokines and decrease of anti-inflammatory cytokines observed in depressed individuals who died by suicide. This study also suggests that abnormalities of cytokines that have been observed in the serum or CSF of suicidal patients are also present in the postmortem brain of depressed and suicidal individuals.
Footnotes
Funding: This research was supported by grants RO1MH098554 and RO1MH106565 (Dr. Pandey) from the National Institute of Mental Health, Rockville, MD. The funding source had no role in the study design; collection, analysis and interpretation of data; or writing of the manuscript.
Competing interests: None declared.
Contributors: G. Pandey designed the study. H. Rizavi, H. Zhang and X. Ren acquired the data, which G. Pandey and R. Bhaumik analyzed. G. Pandey wrote the article, which all authors reviewed. All authors approved the final version to be published and can certify that no other individuals not listed as authors have made substantial contributions to the paper.
- Received September 24, 2017.
- Revision received January 10, 2018.
- Accepted January 22, 2018.