Abstract
Objective: In patients with major depressive disorder (MDD), enhanced activation of the rostral anterior cingulate cortex (rACC) during conflict resolution has been demonstrated with the use of functional magnetic resonance imaging (fMRI), which suggests dysregulation of the affective compartment of the ACC associated with error monitoring and cognitive control. Moreover, several previous studies have reported disrupted structural integrity in limbic brain areas and the orbitofrontal cortex in MDD. However, the relation between structural and functional alterations remains unclear. Therefore, the present study sought to investigate whether structural brain aberrations in terms of grey matter decreases directly in the medial frontal regions or in anatomically closely connected areas might be related to our previously reported functional alterations.
Methods: A sample of 16 female, drug-free patients with an acute episode of MDD and 16 healthy control subjects matched for age, sex and education were examined with structural high-resolution T1-weighted MRI; fMRI images were obtained in the same session.
Results: Voxel-based morphometry (VBM) revealed grey matter decreases in the orbitofrontal and subgenual cortex, in the hippocampus-amygdala complex and in the middle frontal gyrus. The relative hyperactivation of the rACC in terms of inability to deactivate this region during the Stroop Color-Word Test showed an inverse correlation with grey matter reduction in the orbitofrontal cortex.
Conclusion: The present study provides strong evidence for an association between structural alterations in the orbitofrontal cortex and disturbed functional activation in the emotional compartment of the ACC in patients with MDD during cognitive control.
Introduction
Aside from depressed mood and anhedonia, patients with the diagnosis of major depressive disorder (MDD) experience worries and negative thoughts about themselves and their future that they perceive as being automatic, persistent and unintended. This leads to a cycle of depression producing even more rumination and accompanying emotional arousal. This subjectively observed inability to suppress negative thoughts has been supported by objective evidence from the neuropsychological perspective. Patients with MDD have exhibited not only an attentional bias toward negative emotional stimuli1 but also a more general deficit in cognitive control processes and decision making.2,3 Especially in cognitive paradigms with conflicting response alternatives, patients with depression revealed a lack of inhibitory control of prepotent stimulus-response contingencies and showed enhanced interference sensitivity.4
Converging lines of evidence from cognitive neuroscience indicate that lateral prefrontal regions, that is, the ventrolateral prefrontal cortex (VLPFC; Brodmann’s area [BA] 44/45) and dorsolateral PFC (DLFPC; BA 9/46) and the anterior cingulate cortex (ACC; BA 24/32), play a crucial role in flexible goal-directed behaviour involving cognitive functions such as the selection and activation of appropriate responses (VLPFC), the monitoring of conflicts and performance outcome (ACC) and the cognitive inhibition of task-irrelevant responses (DLPFC).5
Neuroimaging studies have provided strong evidence for dysfunctions in these areas in patients with MDD. Attenuated resting-state blood flow and glucose metabolism in prefrontal and anterior cingulate brain areas were reported in restingstate positron emission tomography studies.6,7 Further, functional magnetic resonance imaging (fMRI) studies with cognitive paradigms taxing executive functions reported attenuated activations in the PFC and ACC when patients showed an impaired task performance in comparison with matched healthy control subjects8 and a relative hyperactivity in patients when they were matched for task performance.9
In our previous study with the Stroop Color-Word Test (SCWT),10 an established neuropsychological test taxing cognitive inhibition processes, we directly tested the hypothesis of disrupted prefrontal and anterior cingulate functions.11 Our main finding was that patients showed a behavioural performance and dorsal ACC (dACC) activity comparable to healthy control subjects but a relative hyperactivity in the rostral ACC (rACC) and left DLPFC. The rACC activity correlated positively with the Stroop interference (more time spent to inhibit the word reading during the incongruent v. the congruent condition) and with left DLPFC activity. We interpreted these results as an inability of patients with MDD to inhibit cognitive interferences from the emotional state, which they probably compensate for with stronger cognitive control exerted by the DLPFC to produce normal behavioural performance.
This view is supported by studies examining the functional heterogeneity of ACC regions.12,13 Whereas the dorsal (“cognitive”) part, through its strong interconnections with parietal, prefrontal and supplementary motor areas, is mainly activated in cognitively demanding tasks, the rostral (“affective”) part is strongly connected to the orbitofrontal cortex (OFC), amygdala, hippocampus and periaqueductal grey matter and has been found to be mainly activated in tasks requiring processing and integration of affect-related information. Moreover, the affective part has additionally been shown to correlate with autonomic responses during execution of cognitive tasks (e.g., in the Stroop task).14
An interesting aspect is the reported dynamic interplay between these subdivisions in healthy subjects, which shows a relative deactivation in the dACC during emotional paradigms, and in the rACC during cognitive tasks, in comparison with baseline, possibly to allocate resources for effective cognitive processing.15 Moreover, there is strong evidence for a so-called “default mode” of the brain,16,17 a physiological baseline involving mainly cortical midline areas such as the dorsomedial PFC (DMPFC) and ventromedial PFC (VMPFC) as well as the posterior cingulate cortex (PCC), which reveal a high resting-state activity (e.g., during passive viewing of simple visual stimuli) but an attenuated activity during goal-directed behaviour. Although the meaning of this physiological baseline is not fully understood, there are suggestions that this activity in the midline cortical areas can be interpreted in terms of continuous self-referential processing.18
One prominent area within the default mode brain network is the VMPFC including the rACC; in our previous fMRI study,11 activity relative to baseline was elevated in patients and decreased in healthy control subjects during the Stroop task.
In the present study, our main purpose was to investigate why patients suffering from depression are unable to inhibit conflicting affective responses in terms of deactivation of the rostral, or affective, part of the ACC. One possible explanation for this observation is that the investigated patients had structural abnormalities directly in this region or in brain areas interconnected with the rACC, such as the subgenual cortex, OFC, amygdala or hippocampus.
Structural imaging studies have reported a relatively consistent volume reduction in the hippocampus of patients with MDD.19 In their positron emission tomography study on patients with familial MDD, Drevets and colleagues20 observed reduced glucose metabolism and cerebral blood flow in the subgenual PFC in association with a volume reduction in the subgenual cortex that could also be demonstrated in a postmortem neuropathological study.21 Further, Bremner and colleagues, 22 in a volumetric MRI study, as well as Rajkowska and colleagues,23 in a postmortem study, were able to demonstrate reduced volume of the OFC in patients with depression that was due to decreases in glial cells but also to decreases in neuronal cell density. Additionally, Rajkowska and colleagues23 reported a similar reduction in cell density for the DLPFC. Despite this evidence of structural alterations in frontolimbic areas in patients with depression, the relation of these structural changes to functional aberrations remains unclear.
In light of these findings, to reveal potential structural underpinnings of the reported elevated rACC activity, we performed a voxel-based morphometry (VBM) analysis with T1-weighted images of the same sample of patients and control subjects as were investigated with the Stroop task in our previous study. VBM represents a sophisticated and objective whole brain technique to assess subtle regional grey matter changes.
On the basis of previous study findings and anatomical considerations, we hypothesized that the abnormal activity of the rACC in patients with MDD might be directly related to grey matter reduction occurring mostly in the medial PFC, such as the subgenual area or OFC.
In an exploratory analysis, we examined the relation between these regional structural findings and clinical variables (illness duration, the number of previous depressive episodes and symptom severity). Further, we investigated the relation between Stroop performance and grey matter density in patients with depression.
Additionally, from the fMRI perspective, we explicitly investigated the hypothesis of the abnormal default mode of brain activity in patients with MDD by concentrating on deactivations, relative to baseline, within the frontocingulate network during Stroop task performance.
Methods
Subjects
Sixteen female patients who met the criteria for MDD according to the Structured Clinical Interview for DSM-IV Axis I Disorders (SCID)24 were recruited from the inpatient and outpatient service of a psychiatric university hospital. Detailed demographic and clinical characteristics are outlined in Table 1. None of the patients had a history of lithium administration or electroconvulsive therapy. All study patients underwent a washout period of at least double the half-life of their preexisting medications and were free of psychotropic medication for a week, on average, at the time of the fMRI measurement.
Demographic and clinical characteristics of the sample
Sixteen healthy female control subjects matched for age and education (Table 1) were recruited through local newspaper advertisements and screened for psychiatric or neurologic diseases. The sample composition and the inclusion and exclusion criteria were identical to those in our previous paper. One patient was excluded from structural analyses because of movement artifacts during the structural MRI scanning procedure.
The study protocol was approved by the Ethics Committee of the University of Jena.
Cognitive paradigm
The Stroop task was presented in an event-related design and consisted of a congruent and an incongruent condition. In the congruent condition, colour words were presented in the colour denoted by the corresponding word (e.g., the word “red” shown in red); in the incongruent condition, colour words were displayed in one of 3 colors not denoted by the word (e.g., the word “green” shown in red). This target stimulus was presented in the centre of the display screen. Two possible answers (colour words in black type) were presented below it (in the lower visual field) to minimize contextual memory demand. By pressing 1 of 2 buttons (with the index or middle finger), the subjects had to indicate the Word Type colour that corresponded spatially to both possible answers. Stroop stimuli were presented for 1500 milliseconds, with an interstimulus interval of 10.5 seconds, and were pseudorandomly jittered over the repetition time (TR). Further details can be found in our previous paper.
Functional and structural MRI scanning procedure
The functional and structural data were collected at the same time on a 1.5 T Siemens Magnetom Vision whole-body system (Siemens, Erlangen, Germany) equipped with a head volume coil. Head immobilization was established by head pads within the head coil. A series of 440 T2-weighted images were obtained with the use of a gradient-echo planar sequence (TR = 2000 ms, echo time [TE] = 60 ms, flip angle = 90°, field of view = 240 mm) with 19 contiguous transversal slices of 5-mm thickness, in 2 sessions (220 scans in each session). Matrix size was 64 × 64 pixels with in-plane resolution of 3.75 × 3.75 mm.
High-resolution structural T1-weighted volume scans were obtained in sagittal orientation (TR = 15 ms, TE = 5 ms, flip angle = 30°, field of view of 256 mm) with a slice thickness of 1 mm and in-plane resolution of 1 × 1 mm.
Functional data analysis
For functional image processing and statistical analyses, we used the Statistical Parametric Mapping-2 (SPM2) software (Wellcome Department of Imaging Neuroscience, London, UK, 2003). We described the several analysis steps in detail in our previous paper.11
The functional data were corrected for differences in time acquisition by sinc interpolation, realigned at the first image of every session, normalized to the Montreal Neurological Institute (MNI) reference brain and smoothed with a Gaussian kernel (10 mm, full width at half maximum [FWHM]).
The data were subsequently analyzed voxel-wise within the general linear model to calculate statistical parametric maps of t statistics for condition-specific effects. Prior to this analysis, the data were high-pass filtered with a cut-off period of 128 seconds and corrected for serial correlations choosing the AR(1) variable.
We performed a fixed-effect model at a single-subject level to create images of parameter estimates; they were then entered into a second-level analysis (random-effects analysis) using the subjects’ individual 1-sample t tests.
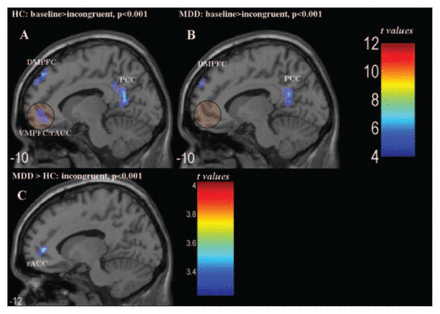
To test the default-mode hypothesis, a 1-sample sample t test was computed within groups for the contrast baseline greater than incongruent condition. A between-group comparison in regard to the incongruent condition, which was depicted in our previous paper,11 is displayed in Figure 1C and Table 2. According to our previous paper, predefined image masks of frontocingulate areas were applied as derived from existing literature on the Stroop task. Regions of interest were defined according to the WFU_Pickatlas (www.fmri.wfubmc.edu) and included the following regions: the cingulate gyrus, inferior frontal gyrus, medial frontal gyrus, middle frontal gyrus and superior frontal gyrus.
Baseline greater than incongruent Stroop condition (p < 0.001, cluster size > 19). (A) HC subjects showed a relative deactivation in the PCC and in the VMPFC/rACC and DMPFC. (B) MDD patients revealed relative decreases in BOLD signal in the PCC and DMPFC. (C) The direct comparison between patients and control subjects is displayed, revealing mainly a relative hyperactivity in the rACC in patients during the incongruent Stroop condition. BOLD = blood oxygen level dependent; DMPFC = dorsomedial prefrontal cortex; HC = healthy control; MDD = major depressive disorder; PCC = posterior cingulate cortex; rACC = rostral anterior cingulate cortex; VMPFC = ventromedial prefrontal cortex.
Activation of regions showing significant* blood oxygenation level dependent signal increase in patients compared with healthy control subjects in the incongruent Stroop condition
All resulting statistics were subject to a significance level of p < 0.001, with a spatial extent threshold of 20 contiguous voxels corresponding to the expected number of voxels per cluster. The reported coordinates were transformed into Talairach space.
Structural data analysis
We applied an optimized VBM protocol as developed and described by Good and colleagues,25 using the VBM-toolbox (http://dbm.neuro.uni-jena.de/) implemented in the SPM2 package. First, from the images of all patients and control subjects, we created a study-specific template set consisting of a mean T1-weighted image and a mean grey matter image. For this purpose, we applied a 12-parameter affine and nonlinear spatial transformation to spatially normalize images to the SPM2 T1-weighted template (MNI, Montréal). Then we segmented them into grey matter, white matter and cerebrospinal fluid compartments and smoothed them with a Gaussian kernel of 8 mm FWHM. Last, we averaged the images to provide study-specific templates with reduced scanner- and population-specific bias.
Next, we processed the original images of all patients and control subjects, applying the following operations: First, we segmented all images, using the study-specific T1 template. Subsequently, we normalized the extracted grey matter images to the customized grey matter template, applying 12-parameter affine and nonlinear spatial transformation, which we then reapplied to the original structural images. Using a modified mixture model cluster analysis, we corrected these normalized images for nonuniformities in signal intensity and partitioned them into grey and white matter and cerebrospinal fluid. To improve the segmentation algorithm, we applied the hidden Markov random field model,26 which models spatial dependencies at the tissue-class membership level and which is implemented in the VBM toolbox. Finally, the resulting images were smoothed with a Gaussian kernel of 12 mm FWHM.
We performed voxel-by-voxel 1-way analysis of variance with these grey matter images to test for differences in regional grey matter concentration between control subejcts and patients. To avoid possible edge effects around the border between grey and white matter, only voxels with absolute grey matter values above 0.15 were entered into the analysis. All statistical images were thresholded at p < 0.001 (uncorrected for multiple comparisons), and only clusters with a minimum of 193 voxels are reported according to expected voxels per cluster threshold (kE). To investigate an association between demographic and clinical data, Stroop performance data and altered grey matter density, we performed a correlational analysis in the patients, using SPSS 13.1 (SPSS Inc., Chicago, Ill.).
Combined fMRI-VBM analysis
To test the effect of structural brain changes on the blood oxygen level dependent (BOLD) signal in the rACC/VMPFC, we extracted the first eigenvariate (main component of the PCA) from the resulting clusters of differences in grey matter density and correlated these according to Pearson’s correlation coefficient with parameter estimates from the fMRI analysis, using SPSS 13.1. To confirm the significance of this correlation, we used SPM2 to additionally perform a correlational analysis between the contrast incongruent condition greater than fixation baseline and the first eigenvariate of the grey matter values from the OFC cluster in patients. We masked this correlation with the contrast patients greater than control subjects with regard to the incongruent condition (unmasked, p < 0.001) to correlate only voxels that were statistically different between groups.
Moreover, we tested the hypothesis of decreased grey matter density directly in the region of the functional activation difference, that is, in the rACC. For this purpose, we created a region-of-interest image of the functional rACC activation difference in patients and used this image as a mask image in the VBM analysis, performing a small volume correction.
In light of structural differences between patients with depression and control subjects, we tested the significance and robustness of the relative rACC hyperactivity in patients for the contrast incongruent greater than fixation baseline while controlling for the grey matter values from the OFC cluster by means of analysis of covariance in SPM2.
Results
Differences in grey matter
With regard to global grey matter alterations (in litres), there were no significant differences between patients (mean 0.666, standard deviation [SD] 0.062 L) and healthy control subjects (mean 0.673, SD 0.058 L).
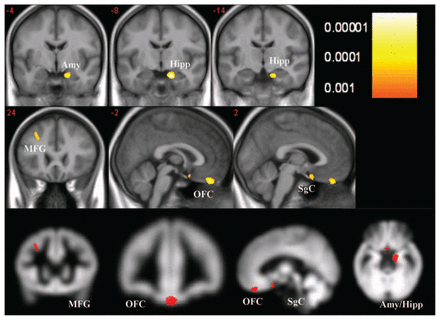
When we tested differences in regional grey matter density between patients and healthy control subjects (p < 0.001), we found 4 clusters of regional decreases in grey matter in patients with MDD, as displayed in Figure 2 and outlined in Table 3. The largest cluster comprised voxels from the right amygdala and right hippocampus formation (subiculum). The second cluster consisted of voxels from the medial OFC, including the left and right portion of the gyrus rectus (BA 11). Additionally, we found a reduction of grey matter volume in the subgenual cortex (BA 25) and in the left middle frontal gyrus (BA 8/9). Testing the hypothesis of decreased grey matter density directly in the rACC, we found no significant difference between patients and healthy control subjects (small volume correction).
Regions of significant grey matter reduction in patients with major depressive disorder compared with healthy control subjects (p < 0.001, cluster size > 192). Amy = amygdala; Hipp = hippocampal formation; MFG = medial frontal gyrus; OFC = orbitofrontal cortex; SgC = subgenual cortex.
Activation of regions showing significant* grey matter reduction in patients with unipolar depression compared with healthy control subjects
Relation to fMRI activation data
The detailed results of the functional MRI analysis are presented in detail elsewhere.11
In Figure 1A and 1B, 1-sample t tests of the contrast fixation baseline greater than incongruent Stroop condition are illustrated separately for patients and control subjects to display brain areas of relative deactivation during Stroop performance. Control subjects revealed a relative deactivation during the incongruent condition in the PCC (x = 4, y = −54, z = 19; p < 0.001; k = 870), in the dorsomedial PFC (x = −14, y = 55, z = 32; p < 0.001; k = 486) and in the ventromedial PFC (x = −8, y = 48, z = −6; p < 0.001; k = 276) including the rACC. Patients showed a relative deactivation during the incongruent condition only in the PCC (x = −10, y = −51, z = 22; p < 0.001; k = 296) and in the dorsomedial PFC (x = −12, y = −59, z = 30; p < 0.001; k = 105). In agreement with the results of these 1-sample t tests, the direct between-groups comparison of the incongruent condition (Fig. 1C) revealed as a main finding a significant relative hyperactivity in the rACC, as displayed in Table 2 and reported in our previous study.11
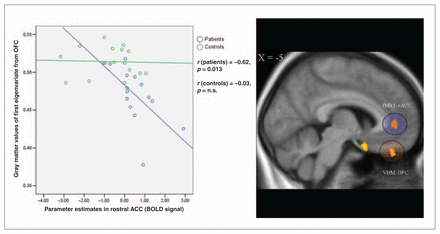
Testing our main hypothesis of a relation between this inability of patients with MDD to deactivate the rACC during the Stroop task and structural brain alterations, we found a significant negative Pearson’s correlation between parameter estimates in the rACC from the fMRI analysis and grey matter values in the medial OFC (r = −0.62, p = 0.013) in patients and not in healthy control subjects (r = −0.03, p = nonsignificant), as illustrated in Figure 3. Both correlation coefficients were significantly different (z = −1.74, p < 0.05). No significant correlations resulted between rACC parameter estimates and other brain areas of decreased grey matter density in patients.
The scatterplot depicts the correlation of the parameter estimates extracted from maximum activation in the rACC and the grey matter values of first eigenvariate from the OFC in patients with depression and control subjects. On the T1 overlay, we mapped the cluster of decreased grey matter density in the OFC in patients and the functional rACC cluster of relative hyperactivity in patients for the direct group comparison (incongruent Stroop condition greater than fixation baseline). For better visualization, we used a threshold of p < 0.005 for the functional overlay. ACC = anterior cingulate cortex; BOLD = blood oxygen level dependent; fMRI = functional magnetic resonance imaging; OFC = orbitofrontal cortex; rACC = rostral ACC; VBM = voxel-based morphometry.
Using SPM2 for the correlation between OFC grey matter values and the contrast incongruent Stroop condition greater than fixation baseline in patients, we observed, in accordance with the SPSS analysis, a significant negative correlation only in the rACC (x = −12, y = 45, z = 3; p < 0.05, familywise error corrected).
Controlling for the effect of decreased grey matter density in the OFC, we still observed relative hyperactivity in the rACC in patients with depression compared with healthy control subjects with regard to the incongruent Stroop condition (x = −14, y = 52, z = −6; p < 0.001).
Relation to clinical variables and Stroop test performance
Controlling for the effects of age, we found no significant correlations between illness duration, number of prior episodes of depression and grey matter values in the regions of significantly reduced grey matter in patients with MDD.
As we already stated in our previous paper,11 we found no significant association between symptom severity as assessed by the Hamilton Depression Rating Scale (HAMD)27 and BOLD signal in the rACC.
Testing the relation between symptom severity and structural data, we found a significant negative correlation only between decreased grey matter in the left middle frontal gyrus and HAMD total score in patients (r = −0.66, p = 0.008, adjusted with Bonferroni correction).
Controlling for the effect of BOLD signal in the rACC, we found no significant correlations between regions of decreased grey matter density and Stroop interference score (adjusted with Bonferroni correction).
Discussion
The present study aimed to combine hemodynamic and morphometric measurements to investigate whether potential structural abnormalities in patients with MDD might be related to specific functional abnormalities in a cinguloprefrontal network.11 The main result of the previous fMRI study was that medication-free female patients with acute depression revealed an absence of activity decrease in the rACC (resulting in hyperactivation relative to baseline and to healthy control subjects) that correlated positively with the interference score in the Stroop task.
As we further investigated in the present paper, healthy control subjects deactivated this region (in terms of significantly lower BOLD signal during the incongruent condition compared with fixation baseline) as well as the PCC and DMPFC. These regions were previously shown to be part of the so-called default-mode network and were consistently reported to reveal a decreased blood flow or negative BOLD signal during various cognitive tasks.16
Hence, the observed activation difference between patients and healthy control subjects is not a simple difference in the amount of activation above fixation baseline but represents, rather, completely different activation directions. Patients with depression were not able to suppress task-irrelevant activity in the rACC, as was seen in healthy control subjects, and therefore might have a disruption in the default-mode network.
Thus the question is, Why are patients unable to suppress the interfering rACC/VMPFC activity? One potential explanation might be structural changes (e.g., in grey matter in the region of functional activation difference or in regions closely connected with the rACC/VMPFC).
As a main finding of the current structural VBM analysis, we observed significantly reduced regional grey matter density in several regions in patients with MDD. In addition to extensive volume reductions in the left amygdala/hippocampus formation, predominantly in the subiculum, left middle frontal gyrus (BA 8/9) and subgenual cortex (BA 25), we found a significant grey matter decrease bilaterally in the gyrus rectus of the medial OFC (BA 11). We did not find a significant between-groups difference in grey matter density in the rACC/VMPFC area.
Most important, the combination of fMRI and VBM analyses revealed the hypothesized relation between structural and functional abnormalities in MDD patients in terms of a significantly negative correlation only between decreased grey matter values in the medial OFC and elevated BOLD signal in the VMPFC/rACC during the Stroop task.
The morphometric findings of our study, which is to our knowledge the first combined fMRI–VBM study in patients with MDD, correspond to findings in several previous volumetric and postmortem studies: in a postmortem histopathological analysis of the density and size of prefrontal neurons, significant reductions in DLPFC and OFC regions were detected in MDD patients when compared with healthy subjects.23
With regard to grey matter decreases in the OFC, an MRI study by Bremner and colleagues22 revealed a 32% volume reduction of the OFC (gyrus rectus) in patients with MDD. Similar findings have been reported by Lacerda and colleagues. 28 Findings show that lesions in the OFC, which is known to be critically involved in emotional processing (predominantly its medial parts15), are associated with abnormalities in a wide range of affective behaviours, such as depressed mood, aggression, affective instability, anxiety and anhedonia. This indicates that volume reductions in this region might be etiologically related to the disorder of major depression.29,30
Likewise, volume reductions in the subgenual ACC have been associated with MDD in an MRI volumetric study20 and in postmortem studies that revealed glial reduction in the corresponding grey matter.21 A recent multimodal study by Pezawas and colleagues31 also reported volume reductions in the subgenual ACC in healthy subjects with a genetically increased risk of depression. Alternatively, Pizzagalli and colleagues32 did not find any morphometric differences in the subgenual ACC in melancholic as well as in nonmelancholic depression subjects relative to healthy control subjects, but did find a reduced resting-state metabolism and increased delta activity only in patients with melancholic depression. Whereas some studies on amygdala volume in MDD patients revealed glial cell and volume reductions,33,34 Frodl and colleagues35 demonstrated an increased amygdala volume in patients with a first depressive episode in comparison with patients with recurrent MDD and healthy control subjects, as well as no differences between the latter 2 groups.
MRI studies of hippocampal volumes in MDD also lack consistency. Thus, while several studies found no differences between patients and healthy control subjects in grey matter hippocampal volume,36,37 several other studies revealed significant reductions in grey matter volume in the hippocampus formation in patients with MDD.38,39 A meta-analysis by Videbech and colleagues19 reported an average 8% volume reduction of the left hippocampus and an average 10% reduction of the right hippocampus in patients with MDD. Major causes for the heterogeneity of these volumetric findings are probably differences in scanning protocols (e.g., slice thickness), in tracing procedures and in sample composition (e.g., sex and age of subjects).19
The lack of association between grey matter volume and clinical variables such as illness duration is not surprising given inconsistencies in earlier studies that investigated the relation between illness duration or number of previous episodes and the volume of a region of interest. MacQueen and colleagues40 found a significant logarithmic association and Sheline and colleagues41 a significant linear association between hippocampus volume and total illness duration. However, other studies35,38,42 did not find any association between clinical variables and decreased volume in the hippocampus or amygdala. For the OFC, in agreement with our study, Bremner and colleagues22 and Lacerda and colleagues28 reported no significant association between OFC volume decrease and clinical variables. Further, the lack of correlation in our study might be attributable to the comparatively smaller sample size, which might not be sensitive enough to detect this kind of association, and a very small variance regarding past depressive episodes (range 0–3). The significant negative correlation between HAMD total score and decreased grey matter density in the left middle frontal gyrus should be interpreted with caution. It is difficult to relate state-dependent variables such as acute symptoms of depression to relatively stable, long-term structural changes.
With regard to our focus of investigation, namely, the integration of structural and functional abnormalities, the correlation between VMPFC/rACC BOLD signal (which was significantly increased in patients owing to their inability to deactivate this brain area during the incongruent Stroop condition) and structural brain alterations yielded a significantly negative result between VMPFC/rACC BOLD signal and OFC grey matter values. Thus patients with a strong grey matter volume reduction revealed a high BOLD signal in the VMPFC/rACC, whereas patients with less distinct structural abnormalities seemed to be better able to deactivate the VMPFC/rACC during cognitive performance.
Given the strong anatomical interconnections between the rostral part of the ACC and the medial OFC,43 this finding suggests that volumetric abnormalities in the medial OFC might be a major factor influencing the patients’ reduced ability to deactivate the VMPFC/rACC and might be associated with a potentially chronically increased signal in the rACC.
We concluded from our results that local grey matter differences directly in the rACC do not lead to differences in BOLD signal between healthy control subjects and patients depression. Additionally, after controlling for the effect of decreased grey matter in the OFC, we still observed relative rACC hyperactivity in patients with depression, compared with healthy control subjects, with regard to the incongruent Stroop condition, which provides compelling evidence for functional abnormality in the rACC.
We therefore assume that, for the Stroop task, irrelevant functional hyperactivity in the rACC of patients with depression might be explained by potential abnormal functional activity in the OFC. Our fMRI sequence was not optimized to acquire echo planar images with sufficient signal in basal structures predominantly in the OFC, owing to local susceptibility gradients near the air/tissue interface. Therefore, we could not test this hypothesis of disrupted functional connectivity and were not able to relate these potential functional changes in MDD patients to observed local grey matter decreases. We can only speculate that grey matter decrease in the OFC potentially leads to abnormal functional activity in this region. This in turn may influence rACC activity. It would be promising to test this hypothesis and to relate grey matter decreases in these regions to functional activations with an appropriate paradigm and optimized echo planar imaging sequence.
The rACC bordering on the VMPFC constitutes the “affective” part of the ACC and has frequently been found to show increased activity in healthy subjects during processing of pain and affective stimuli (e.g., during experimentally induced sadness).44
Moreover, the rACC/VMPFC interface constitutes part of the so-called default network; it has previously been demonstrated to decrease in activity in healthy subjects during cognitive processing and to be abnormally activated in patients with MDD during rest.17,31,45,46 Our findings strongly suggest that structural abnormalities might be at least partly responsible for the inability of MDD patients to flexibly up- and downregulate activation in this symptomatically relevant area. Other parts of the default-mode network within the frontocingulate areas (i.e., the DMPFC and PCC) decreased their activity similarly in patients and healthy control subjects during the incongruent trial, relative to fixation baseline, indicating a normal functioning of these areas in MDD.
The VMPFC has been detected to play a relevant role in the context of self-referential processing (e.g., when subjects have to make judgements about their own abilities and traits).18 Thus our finding of a significantly increased signal in the VMPFC (or the reduced ability to deactivate the VMPFC) in patients with MDD seems plausible when it is taken into consideration that increased (and predominantly negative) self-referenced processing (in the form of automatic and unstoppable pondering about one’s own “inabilities” and “defective” character traits) is a characteristic symptom of the disorder.
Consequently, our data strongly indicate that the reduced ability of MDD patients to deactivate this medial perigenual area, which encompasses the rACC and ventromedial prefrontal regions, might play a relevant role in the psychopathology of the disorder. Further, the data suggest that this functional disturbance might be closely related to structural abnormalities in more inferior regions that are anatomically closely connected to this region. Our results, moreover, corroborate previous studies reporting structural abnormalities in several limbic and cortical regions. However, the question of whether these abnormalities are a cause or consequence of the disorder remains to be elucidated and requires further study.
Recapitulating our data and considering the substantial anatomical connection of the medial OFC and VMPFC to limbic structures such as the amygdala, subiculum and entorhinal cortex,47 the present fMRI VBM study revealed structural and functional aberrations within this reciprocal network that is mainly engaged in processing and integrating affective information. Cognitive aspects of depression, such as rumination and deficient cognitive control, as well as another accompanying core symptom of depression, such as anhedonia (as a sign of disturbance in the reward-processing network), may be directly related to disturbed connectivity within the orbitofrontal-limbic network. Thus the inability to inhibit the activity of the rACC together with the positive relation between the level of this activity and the amount of interference in the Stroop task offer strong evidence for dysbalance of the orbitofrontal-limbic network in controlling maladaptive affective and vegetative responses during cognitive processing that is strongly related to structural lesions within this network.
Limitations
Some potential limitations of the present study should be mentioned. We studied only female patients, so our results cannot be generalized to the whole population of patients with depression. The rationale for this selection was to reduce the potential variance in BOLD signal due to sex effects. Because male and female patients clearly differ with regard to their illness characteristics, including illness severity and concurrent symptoms (for a review, see Marcus and colleagues48), there is reason to assume that differences between male and female patients also exist on the cerebral level. However, potential sex differences in brain metabolism have not to date been studied in patients with MDD. Because the lifetime prevalence rates of depression are about 2 to 3 times higher for women than men, the results of our study can be generalized to most patients with depression.
Further, the limitations of the VBM method should be mentioned. VBM strongly depends on correct segmentation and classification of brain tissue types. Moreover, to statistically compare the grey matter images between groups, they have to be registered to a common anatomical space. Thus either systematic tissue classification differences between groups or registration error may produce between-groups effects that are not attributable to real differences in grey or white matter.49
Conversely, VBM represents an unbiased, objective and comprehensive method for testing differences in local composition of brain tissue after global shape differences are discounted. The VBM method is an established tool in morphometry and was successfully applied in various studies with neurologic or psychiatric patients.31,50,51 The validity of the methods can be shown (e.g., Keller and colleagues’52 comparison of hippocampus volume with VBM and manual segmentation).
Despite these limitations, our results provide strong evidence for an association between functional and structural abnormalities in patients with MDD. Further studies with a larger number of patients are needed to further elucidate the nature of this relation.
Acknowledgements
We thank all the patients and control subjects who participated in this study. This work was supported by an IZKF program grant of the Friedrich-Schiller-University of Jena (TMWFK B30701-015/-016) and the Bundesministerium für Bildung und Forschung BMBF (FKZ01ZZ0105).
Footnotes
Medical subject headings: magnetic resonance imaging; depressive disorder; depression.
Competing interests: None delcared.
Contributors: Drs. Wagner, Sauer and Schlösser designed the study. Dr. Wagner and Ms. Schachtzabel acquired the data, which Drs. Wagner, Koch, Reichenbach and Schlösser analyzed. Drs. Wagner, Koch and Schlösser wrote the article, and Drs. Koch, Reichenbach, Sauer and Schlösser and Ms. Schachtzabel revised it. All authors gave final approval for the article to be published.
- Received May 21, 2007.
- Revision received September 4, 2007.
- Accepted October 16, 2007.