Abstract
Background: Using biological evidence to define subtypes within the heterogeneous population with obsessive–compulsive disorder (OCD) is important for improving treatment response. Based on age at onset, OCD can be clustered into 2 groups, each of which is more homogeneous with respect to clinical and cognitive phenotype. However, the neural bases for these phenotypic differences need to be established to construct evidence-based homogeneous groups.
Methods: We compared brain volumes, clinical symptoms, and neurocognitive function for 49 people with early-onset OCD and 52 with late-onset OCD (participants in both groups were unmedicated or drug-naïve), and 103 healthy controls. We performed regression analyses to examine group × volume interaction effects on clinical outcomes or neurocognitive function in people with OCD.
Results: We observed larger volumes in the precentral, orbitofrontal, middle frontal, and middle temporal gyri in people with early-onset OCD compared to those with late-onset OCD. Poorer visuospatial construction in early-onset OCD was correlated with a larger left middle frontal gyrus volume. Impaired visuospatial memory in people with early-onset OCD and cognitive inflexibility in people with late-onset OCD were correlated with increased and decreased volume in the left middle frontal gyrus, respectively. We found group × volume interactions for obsessive–compulsive symptom scores in the left middle temporal gyrus of people with OCD.
Limitations Although we divided the subtypes using the commonly adopted criterion of age at onset, this criterion is still somewhat controversial.
Conclusion: We provided the neural bases for clinical and neurocognitive differences to demonstrate that biological evidence underlies the distinctions between early- and late-onset OCD. This study suggests that different treatment options should be considered for the OCD subtypes, because their neurobiology differs and is related to distinct phenotypic profiles.
Introduction
Unsatisfactory treatment outcomes have been reported in a substantial proportion of people with psychiatric disorders, including obsessive–compulsive disorder (OCD), because of heterogeneity in the patient populations.1 The Research Domain of Criteria project addresses the need to subdivide psychiatric disorders into more homogeneous groups using biological evidence to find better treatment options.2,3 People with OCD can be categorized based on age at onset: the early-onset group has less of a response to serotonin reuptake inhibitors than the late-onset group, although both groups show similar outcomes from cognitive behavioural therapy.4–6 However, the basis for the neuropharmacological difference between these subgroups is still controversial,7 and more neurobiological evidence related to early- and late-onset OCD is needed.
The incidence of OCD has a bimodal distribution: an early peak occurs before puberty, and another occurs around young adulthood.8 Differences in genetic load and comorbidities suggest a distinct pathophysiology for early- and late-onset OCD.9 These 2 subgroups are thought to be different in their clinical symptoms and neurocognitive function, but these differences are also controversial. For example, some studies have found that people with early-onset OCD are characterized by greater severity of obsessive–compulsive symptoms than people with late-onset OCD,4,5,10,11 but others have shown comparable severity between the 2 groups.12–15 Neurocognitive function is also different between groups for visuospatial construction and memory, verbal fluency and executive function.12,16,17 In particular, poorer visual recall is apparent in people with early-onset OCD,17 while people with late-onset OCD have worse verbal fluency and executive function, especially cognitive flexibility.12,16 These phenotypic differences suggest relative homogeneity within the 2 groups, but biological evidence, which is useful for finding better treatment options, has not been provided. Thus, the neurobiological mechanisms underlying the phenotypic differences between early- and late-onset OCD remain to be investigated.
Neuroimaging studies have found neurobiological relevance for differences related to the age at onset of OCD.9 In particular, people with early-onset OCD have larger volumes in their medial frontal and temporoparietal cortices.18,19 Exner and colleagues19 suggested that the structural alterations in people with early-onset OCD are a result of neuroplastic changes that occur with the constant struggle to suppress unwanted intrusive thoughts over a longer duration. In contrast, a mega-analysis reported no difference in cortical thickness between people with early- and late-onset OCD, but this finding was in patients who were medicated.20 This point needs to be further studied to obtain reliable results, because medication can have confounding effects. However, if brain differences are present, they can be used to find neural bases for the phenotypic differences between early- and late-onset OCD.
Using a relatively large number of people with OCD who were unmedicated or drug-naïve, we proposed to explore the neural bases for the phenotypic differences between early- and late-onset OCD. In particular, we aimed to determine whether certain brain structures are associated with clinical and neurocognitive differences between the groups. We hypothesized that people with early-onset OCD would exhibit larger medial frontal, parietal and temporal cortical volumes than people with late-onset OCD. We also hypothesized that these regional volumes would have distinct associations with clinical symptoms and neurocognitive function in people with early- and late-onset OCD. We expected that uncovering these neural bases would add biological background to attempts to construct evidence-based homogeneous subgroups in the population with OCD.
Methods:
Participants
We recruited 101 people with OCD via the OCD clinic in the Department of Psychiatry at Seoul National University Hospital. Diagnosis of OCD was made by experienced psychiatrists according to criteria from the Diagnostic and Statistical Manual of Mental Disorders, fourth edition (DSM-IV).21 Patients had been unmedicated for at least 1 month or were drug-naïve (60 unmedicated, 41 drug-naïve). We administered the Yale–Brown Obsessive Compulsive Scale (Y-BOCS),22 the Hamilton Depression Rating Scale (HAM-D)23 and the Hamilton Anxiety Rating Scale (HAM-A)24 to assess the severity of obsessive–compulsive symptoms and accompanying depression and anxiety. We divided patients into 2 groups: early-onset OCD (age at onset < 17 yr; n = 49) and late-onset OCD (age at onset ≥ 17 yr; n = 52).25 We also constructed a subset of patient groups based on duration of illness rather than age. We recruited 103 healthy controls through Internet advertisements. Healthy controls were matched for age, sex, handedness and intelligence quotient (IQ), and screened using the Structured Clinical Interview for DSM-IV Axis-I Disorders, Non-patient Edition.26 Participants with a lifetime history of psychotic disorders; neurologic disorders, including a clinically important history of head trauma; substance use disorder; or mental retardation were excluded from the study. We obtained written informed consent from participants after providing a complete description of the study. This study was approved by the institutional review board of Seoul National University Hospital.
Neuropsychological tests
We assessed neurocognitive function using a battery of neuropsychological tests that covered visuospatial ability, verbal fluency, executive function and decision-making skills. We measured visuospatial ability using the Rey-Osterrieth Complex Figure Test (RCFT) subsets: the copy condition to assess constructional ability, and the immediate recall and delayed recall conditions to assess memory.27 We assessed verbal fluency using the Korean version of the Controlled Oral Word Association Test (COWAT).28 We evaluated organization ability and cognitive flexibility to determine executive function. We assessed organization using the picture arrangement and block design subtests of the Korean version of the Wechsler Adult Intelligence Scale (WAIS)29 and the RCFT organization subtest. We assessed cognitive flexibility using the Intra-Extra Dimensional Set Shift (IED) task.30 Because cognitive flexibility is associated with decision-making quality,31 we also conducted exploratory tests of decision-making and risk-taking behaviours using the Cambridge Gambling Task.32
Image acquisition and processing
We acquired high-resolution T1-weighted images from all participants using a 3 T Trio MRI scanner (Siemens MAGNETOM Trio) at Seoul National University Hospital. We used a 12-channel head coil and a magnetization-prepared rapid gradient echo sequence with the following imaging parameters: echo time 1.89 ms, repetition time 1670 ms, flip angle 9°, voxel size 1.0 × 0.98 × 0.98 mm3, 208 slices.
We preprocessed the structural T1 images according to the voxel-based morphometry (VBM) pipeline33 in the Statistical Parametric Mapping toolbox version 12 (SPM12; http://www.fil.ion.ucl.ac.uk/spm/). We segmented the images into grey matter, white matter and cerebrospinal fluid using the unified segmentation algorithm. The images were normalized to a study-specific template using a high-dimensional Dartel algorithm that applied a tissue segmentation map; then, the normalized images were resliced into the Montreal Neurological Institute space using a flow field and affine transformation matrix.34 Modulation and smoothing with a full width at half maximum Gaussian kernel of 8 mm were applied to the grey matter images to increase the signal-to-noise ratio.
Statistical analyses
We compared demographic and neurocognitive variables among the early-onset OCD, late-onset OCD and control groups using the χ2 test or 1-way analysis of variance (ANOVA), followed by post hoc Bonferroni tests, in SPSS version 23 (IBM). We compared clinical outcomes between early- and late-onset OCD groups using 2-tailed independent samples t tests.
We performed VBM analysis for the T1 images. Using a generalized linear model in SPM12, we conducted ANOVA with Bonferroni post hoc tests on regional grey matter volumes for the early-onset OCD, late-onset OCD and control groups. Because potential confounding variables were matched between groups, we conducted the VBM analysis without nuisance regressors.35 We conducted multiple comparison corrections using AlphaSim implemented in AFNI (afni.nimh.nih.gov), which employs Monte Carlo simulations to control the false-positive rate.36,37 We maintained a cluster-level-corrected false-positive detection rate of p < 0.05 using a voxel-level threshold of p < 0.001 with a cluster extent (k) empirically determined by Monte Carlo simulation (n = 10 000 iterations). Based on the Anatomic Automatic Labelling system, 38 we labelled the brain regions that survived correction.
For the grey matter regions that were significantly different between the early- and late-onset OCD groups, we conducted Pearson and partial correlations of regional volumes, respectively, with clinical outcomes and neurocognitive function within each group. We controlled for IQ score for neurocognitive correlations. We also performed linear regression analyses with group, regional volume and group × volume interaction as predictors of the dependent variable of symptom severity in people with OCD. We performed regression analyses with IQ as an additional predictor of the dependent variable of neurocognitive function in people with OCD. We also examined correlations between illness duration and symptom severity within each subtype of OCD.
Results:
Demographic and clinical characteristics
Demographic and clinical variables are presented in Table 1 and Appendix 1, Table S1, available at jpn.ca/190028-a1. We found no significant demographic differences between groups according to the results of the ANOVA, χ2 and post hoc tests. The early-onset OCD group was significantly younger at onset (t87 = −13.98, p < 0.001) and had a longer duration of illness (t74 = 6.65, p < 0.001) than the late-onset OCD group. We found no significant differences in the severity of obsessive–compulsive symptoms, depression or anxiety. The Y-BOCS total (r = 0.28, p < 0.05) and obsessive scores (r = 0.32, p < 0.05) were positively correlated with duration of illness in the early-onset OCD group but not in the late-onset OCD group (total: r = 0.14, p = 0.34; obsession subscale: r = 0.003, p = 0.99).
Demographic and clinical characteristics
Neurocognitive function
The ANOVA results showed significant group effects for the RCFT copy and immediate recall measures (F2,181 = 3.76, p < 0.05; F2,181 = 3.40, p < 0.05); COWAT letter (F2,180 = 3.87, p < 0.05); WAIS block design (F2,200 = 3.35, p < 0.05); and IED errors (F2,160 = 5.98, p < 0.01). In the post hoc results, the early-onset OCD group had a significantly lower score on the RCFT copy measure (mean ± standard deviation 17.63 ± 2.87) than the late-onset OCD group (18.52 ± 1.15), but this difference was only a trend compared to the control group (18.39 ± 1.17). Compared with controls, the 2 OCD subgroups performed significantly worse in the following neurocognitive function tests: RCFT immediate recall and IED in the early-onset OCD group; COWAT letter, WAIS block design and IED in the late-onset OCD group (Table 2). The results of the subset analysis are presented in Appendix 1, Table S2 and results.
Comparison of neurocognitive function between groups
Whole-brain morphometry
The results of 1-way ANOVA showed significant group effects for the left precentral gyrus, left middle frontal gyrus (MFG), right medial and anterior orbitofrontal gyrus (OFG), left posterior OFG, right superior temporal gyrus, left middle temporal gyrus (MTG), right thalamus, right lingual gyrus, left cerebellar lobule III and right cerebellar lobule VIII (see details in Appendix 1, Table S3).
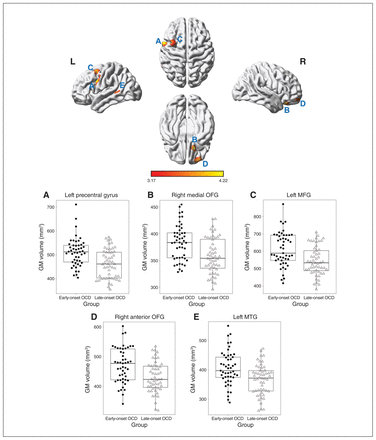
As shown in Figure 1, the early-onset OCD group had larger volumes in the left precentral gyrus (peak at −58, 8, 33; T = 4.21; corrected p < 0.001; cluster size = 326); right medial OFG (peak at 18, 28, −26; T = 4.10; corrected p < 0.01; cluster size = 244), right anterior OFG (peak at 32, 56, −18; T = 3.88; corrected p < 0.01; cluster size = 287), left MFG (peak at −38, 9, 56; T = 3.90; corrected p < 0.001; cluster size = 358), and left MTG (peak at −52, −42, 2; T = 3.69; corrected p < 0.05; cluster size = 197) than the late-onset OCD group. Other post hoc and subset VBM results are presented in Appendix 1, Tables S3 and S4.
Structural differences in grey matter volumes between patients with early- and late-onset obsessive–compulsive disorder (OCD). Top: Brain regions that were significantly larger in early-onset OCD than in late-onset OCD; these regions included (A) the left precentral gyrus, (B) the right medial orbitofrontal gyrus (OFG); (C) the left middle frontal gyrus (MFG); (D) the right anterior OFG; and (E) the left middle temporal gyrus (MTG). Bottom: plots showing significantly larger regional grey matter (GM) volumes in early-onset OCD than in late-onset OCD.
Association between regional volumes and clinical or neurocognitive variables
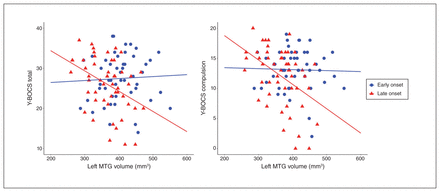
The late-onset OCD group showed negative correlations between left MTG volume and Y-BOCS total (r = −0.40, p < 0.01) and compulsion scores (r = −0.44, p < 0.01), in addition to HAM-D score (r = −0.31, p < 0.05). The early-onset OCD group showed negative correlations between left MTG volume and HAM-A score (r = −0.28, p < 0.05). The regression model predicted Y-BOCS total (F3,97 = 3.74, p < 0.05, adjusted R2 = 0.08) or compulsion score (F3,97 = 5.46, p < 0.01, adjusted R2 = 0.12) in the left MTG of people with OCD. The Y-BOCS total score was predicted by the effects of group (β = 1.52, p < 0.05) and group × volume interaction (β = −1.66, p < 0.05); the interaction indicated that patients with a smaller left MTG volume and late-onset OCD would have a higher Y-BOCS total score. The Y-BOCS compulsion score was also predicted by the effects of group (β = 2.34, p < 0.05) and group × volume interaction (β = −2.69, p < 0.01), indicating that patients with a smaller left MTG volume and late-onset OCD would have a higher Y-BOCS compulsion score. The group × volume interaction effects for Y-BOCS scores in people with OCD are presented in Figure 2.
Group × volume interaction effect of the left middle temporal gyrus (MTG) on the severity of obsessive–compulsive symptoms across patients with early- and late-onset obsessive-compulsive disorder. Patients with a later onset and a smaller left MTG volume had higher total (left) and compulsion (right) scores on the Yale–Brown Obsessive Compulsive Scale (Y-BOCS).
The early-onset OCD group showed negative correlations between left MFG volume and performance in the RCFT subsets of copy (r = −0.30, p < 0.05) and immediate recall (r = −0.36, p < 0.05). In the late-onset OCD group, a smaller left MFG volume was significantly correlated with a greater number of errors on the IED (r = −0.30; p < 0.05). However, we found no group × volume interaction effects for neurocognitive function in people with OCD.
Controls showed neurocognitive correlations between left precentral gyral volume and the RCFT subsets of copy (r = −0.27, p < 0.05) and immediate recall (r = −0.24, p < 0.05).
Discussion
To provide a biological basis for the phenotypic distinctions between OCD subgroups, we investigated the neural mechanisms behind the clinical and neurocognitive differences between people with early- and late-onset OCD who were drug naïve or unmedicated. We found 2 structural brain markers for differences in clinical symptoms and neurocognitive function. People with OCD who had a smaller MTG volume and later onset of disease had more severe obsessive–compulsive symptoms. In addition, poorer visuospatial construction in people with early-onset OCD was related to larger MFG volume. The enlarged MFG volume in people with early-onset OCD was also related to impaired visuospatial memory; the decreased volume in people with late-onset OCD was associated with cognitive inflexibility. These results point to several neural bases for the distinct phenotypes of people with early- and late-onset OCD.
Because people with early-onset OCD experience a more aggressive clinical course of the disease that manifests with a greater number of obsessive–compulsive symptoms,11 we expected that they would have more severe symptoms than people with late-onset OCD. However, we found that the 2 OCD subgroups were comparable in terms of symptom severity, in line with some previous findings.12–15 According to Stewart and colleagues,39 people with early-onset OCD have poorer clinical outcomes with a longer duration of illness. People with early-onset OCD in studies reporting higher symptom severity had symptoms for more than 17 years,4,5,10 and patients with less severe symptoms had them for less than 13 years.12–15 Similarly, people with early-onset OCD in the present study had a relatively short illness duration. Their symptoms became more aggravated as the duration of illness increased, a finding that we did not observe in the late-onset OCD group. These findings demonstrate that the severity of obsessive–compulsive symptoms might have been comparable between the 2 subgroups if people with early-onset OCD had not been ill for longer to develop fully deteriorated symptoms.
With respect to phenotypic differences, researchers have also investigated neurocognitive function in early- and late-onset OCD. Owing to a lack of understanding of the neural mechanisms underlying each group, most neurocognitive functions related to OCD symptomatology have been targeted. 40 However, findings have been inconsistently reported, which can be attributed to small sample sizes, confounding medication effects and differences in sample characteristics.8,12,17 In the present study, we observed worse visuospatial construction performance in people with early-onset OCD. These patients also showed relatively inefficient encoding of visuospatial information compared to controls. In contrast, Hwang and colleagues12 reported worse visuospatial memory in people with late-onset OCD. However, that finding could be attributed to the lower overall level of general intelligence.12,41 In addition, this inconsistent finding could be the result of different criteria for age at onset. However, we believe the present study has several strengths, because we used a larger sample and controlled for confounding variables, such as level of intelligence and medication effects.41,42 Our results were in line with the theoretical notion that memory deficits in OCD arise from deficits in the construction of visuospatial information rather than in the storage and retrieval of memory.43 We also found that people with late-onset OCD performed worse when it came to cognitive shifting, organizational ability and verbal fluency, which has been shown previously.12,16 Using a relatively large sample of unmedicated or drug-naïve patients, our results demonstrate that visuospatial impairments (especially construction and encoding) and executive dysfunction are important features of early- and late-onset OCD, respectively.
Consistent with our hypothesis and previous findings,18,19 we demonstrated that people with early-onset OCD had larger grey matter volumes in the frontal and temporal cortices than people with late-onset OCD. These regions are involved in the cortico–striato–thalamo–cortical (CSTC) circuit, a major pathophysiological model of OCD.44,45 In this circuit, cortical regions are connected to the striatum and thalamus, which play a key role in the control of thoughts and behaviours. 46 In addition to cortical differences, the 2 patient subgroups have demonstrated different brain mechanisms in the striatal and thalamic regions.47,48 For example, lower serotonin transporter availability in the striatum has been observed only in people with late-onset OCD.47 As well, thalamic function is different between groups.48 We suggest that the differences in the striatum and thalamus between people with early- and late-onset OCD may be further linked to the abovementioned differences in cortical structures. In the present study, we found a larger caudate volume in people with early-onset OCD than in controls; however, we found no difference in the striatum when we compared the 2 patient groups (Appendix 1, Table S3). This could have occurred because structural neuroimaging is not as sensitive as functional neuroimaging when capturing the different serotonergic systems of the subcortical regions between people with early- and late-onset OCD. From another perspective, increased MTG volume could be a compensatory change. Although it has been reported that executive function is substantially impaired in people with OCD,40 the early-onset group in the present study had relatively preserved executive function. Because the temporal cortex is involved in executive function,49 our findings showing increased temporal cortical volume may allow executive function in people with early-onset OCD to be spared to some degree. Our findings showing the distinct biological backgrounds of early- and late-onset OCD further support the bimodal distribution model of illness onset,10 and not the neurodegenerative/progressive disease model.
In contrast, Koprivova and colleagues18 demonstrated lower occipital grey matter density in patients with early-onset OCD. However, those patients were on medication, which may have contributed to occipital cortical thinning.18,45 People with early-onset OCD are usually exposed to psychiatric medications for a longer duration.10 Thus, we considered higher medication exposure in patients with early-onset OCD to be a confounder. In addition, smaller dorsomedial prefrontal cortex/anterior cingulate cortex volume, a frequently reported brain abnormality in OCD,44,50 was not replicated in the present study. Piras and colleagues50 suggested that the generalization of neuroimaging findings could be limited by the heterogeneous profiles of symptom dimensions. For example, patients with predominant aggressive/checking, contamination/cleaning or sexual/religious symptoms have smaller dorsomedial prefrontal cortex/anterior cingulate cortex volumes.51–53 Compared with previous meta/mega-analyses,44,45 these symptom types were less predominant in our patients (Table 1). This inconsistency could be attributed to clinical heterogeneity.
We first matched the 2 OCD subgroups according to age rather than duration of illness, because people with early-onset OCD generally experience a longer period of illness.5,8,12,47 However, this classification raises the question of which effects are related to age at onset and which are related to duration of illness. We performed an alternative analysis using a subset of samples matched according to duration of illness and obtained similar results (Appendix 1, Tables S1, S2 and S4). However, the late-onset group in this subset also showed poorer organization ability and smaller right precentral gyrus volume than the early-onset group. We considered these differences to be affected by age at onset, and the unreplicated findings (e.g., visuospatial impairments and larger MTG volume in the early-onset group) might be more attributable to duration of illness.
More than finding phenotypic differences between people with early- and late-onset OCD, the primary goal of the present study was to identify the neural bases for the different phenotypes. Interestingly, we found that the MTG was the neural basis for the different clinical symptom profiles. Smaller MTG volume in the late-onset OCD group represented more severe obsessive–compulsive symptoms, despite the fact that overall the 2 patient groups had comparable symptom severity. In particular, patients with later disease onset and a smaller left MTG volume were more likely to have more severe obsessive–compulsive symptoms. Because the MTG is connected to the striatal regions, the complex dysfunction of the CSTC circuit (including the MTG) in late-onset OCD may be related to symptom manifestation.45,46 Indeed, previous studies have reported that a structural decrease in MTG volume is involved in OCD pathophysiology and is associated with levels of symptom severity.45,54 In contrast to the correlations we observed for symptoms in people with late-onset OCD, none of the brain abnormalities in people with early-onset OCD were associated with obsessive–compulsive symptoms, even though these patients had enlarged grey matter volumes (Appendix 1, Table S3). Structural alterations that appeared to be irrelevant for clinical symptoms could be a result of neuroplastic changes made to suppress unwanted intrusive thoughts in people with early-onset OCD.19 These findings supported our hypothesis that there are different neural bases underlying the clinical symptoms in the 2 patient groups, and they suggest that the MTG is a key region related to obsessive–compulsive symptoms in people with late-onset OCD.
Because clinical associations are not enough to fully explain OCD pathophysiology, neurocognitive models are suggested as alternatives for expanding our understanding.55 While the MTG region was associated with clinical phenotypes, we found the MFG region to be the neural basis for neurocognitive differences. A larger MFG volume in people with early-onset OCD was associated with poor visuospatial ability, especially construction and encoding. In addition, the structural decrease in MFG volume in people with late-onset OCD was related to cognitive inflexibility. This region is involved in multifaceted cognitive functions, because the dorsolateral prefrontal cortex, an analogous region to the MFG, forms connections with the striatal and thalamic regions in the CSTC circuit and controls executive function and memory processing. 55 For example, dysfunction in the dorsolateral prefrontal cortex contributes to difficulties in visuospatial construction, memory processing and set-shifting in psychiatric and neurologic disorders.56,57 Our findings suggest that different disruptions of the MFG in this circuit might contribute to poor visuospatial construction and memory in early-onset OCD but cognitive inflexibility in late-onset OCD.
Limitations
The present study had several limitations. First, there has been no strong consensus related to the criterion of age at onset for differentiating between OCD subtypes. People with early-onset OCD are thought to have subclinical symptom onset before puberty, and obsessive–compulsive symptoms appear after puberty in people with late-onset OCD.11 Using this information, we designated the cutoff for age at onset to be 17 years, classifying the 2 patient subgroups approximately at puberty. A similar criterion has been applied in previous studies.8,12–15,47 Second, although all patients in the study were either unmedicated for at least 1 month or drug-naïve, previous exposure to medication could still have affected structural brain changes in the unmedicated group. We further examined volumetric differences between the 41 drug-naïve and 60 unmedicated OCD patients and found no volume differences between these groups, so it is unlikely that previous medication exposure affected structural brain changes in unmedicated patients (Appendix 1, Table S5). Third, a nonsignificantly larger total intracranial volume in people with early-onset OCD could have influenced the VBM results. When we controlled for total intracranial volume, we observed a pattern of findings similar to those reported in the literature, but the results did not survive correction for multiple comparisons (Appendix 1, Table S6). However, the additional adjustments in our already matched samples could have produced biased and overcorrected results. 35,58 Moreover, the unadjusted results themselves were meaningful, because the regional differences between the OCD subtypes showed large effect sizes (0.72 < d < 0.82) when total intracranial volume difference had a small to medium effect size (d = 0.36). Finally, participants in the present study were mostly young adults, and this could have produced biased results. However, including relatively young participants in the study may have the advantage of excluding age-related decreases in brain volume and cognitive performance.59
Conclusion:
In the present study, we identified the neural bases for the different profiles of clinical symptoms and neurocognitive function for people with early- and late-onset OCD. These neural bases demonstrate that there is biological evidence for the phenotypic differences between the 2 patient groups. These findings may serve as a neurobiological background for constructing evidence-based homogeneous subgroups of OCD and helping to find better treatment options for early- and late-onset OCD.
Acknowledgments
The authors thank the families who participated in the study for their availability, as well as the MRI technician at Seoul National University Hospital and the research coordinators at the Clinical Cognitive Neuroscience Centre for their help in recruiting participants and arranging schedules for the study. This research was supported by the Basic Science Research Program and the Basic Research Laboratory Program through the National Research Foundation of Korea (NRF; grant no. 2019R1A2B5B03100844, 2018R1A4A1025891).
Footnotes
Competing interests: None declared.
Contributors: T. Kim, S. Kwak, J.-W. Hur, T.Y. Lee and J.S. Kwon designed the study. T. Kim, S. Kwak and W.-G. Shin acquired the data, which T. Kim, J.-W. Hur, J. Lee, W.-G. Shin, M. Kim and J.S. Kwon analyzed. T. Kim wrote the article, which all authors reviewed. All authors approved the final version to be published and can certify that no other individuals not listed as authors have made substantial contributions to the paper.
- Received February 10, 2019.
- Revision received June 9, 2019.
- Accepted July 23, 2019.