Abstract
Background: Mindfulness-based stress reduction (MBSR) alleviates depression and anxiety in adults with autism spectrum disorder (ASD); however, underlying therapeutic neural mechanisms and mindfulness-specific effects have yet to be elucidated.
Methods: We randomly assigned adults with ASD to MBSR or social support/education (SE). They completed questionnaires that assessed depression, anxiety, mindfulness traits, autistic traits and executive functioning abilities as well as a self-reflection functional MRI task. We used repeated-measures analysis of covariance (ANCOVA) to evaluate behavioural changes. To identify task-specific connectivity changes, we performed a generalized psychophysiological interactions (gPPI) functional connectivity (FC) analysis on regions of interest (ROIs; insula, amygdala, cingulum and prefrontal cortex [PFC]). We used Pearson correlations to explore brain–behaviour relationships.
Results: Our final sample included 78 adults with ASD — 39 who received MBSR and 39 who received SE. Mindfulness-based stress reduction uniquely improved executive functioning abilities and increased mindfulness traits, whereas both MBSR and SE groups showed reductions in depression, anxiety and autistic traits. Decreases specific to MBSR in insula–thalamus FC were associated with anxiety reduction and increased mindfulness traits, including the trait “nonjudgment;” MBSR-specific decreases in PFC–posterior cingulate connectivity correlated with improved working memory. Both groups showed decreased amygdala–sensorimotor and medial–lateral PFC connectivity, which corresponded with reduced depression.
Limitations: Larger sample sizes and neuropsychological evaluations are needed to replicate and extend these findings.
Conclusion: Together, our findings suggest that MBSR and SE are similarly efficacious for depression, anxiety and autistic traits, whereas MBSR produced additional salutary effects related to executive functioning and mindfulness traits. Findings from gPPI identified shared and distinct therapeutic neural mechanisms, implicating the default mode and salience networks. Our results mark an early step toward the development of personalized medicine for psychiatric symptoms in ASD and offer novel neural targets for future neurostimulation research.
Clinical Trial Registration: ClinicalTrials.gov identifier NCT04017793.
Introduction
Autism spectrum disorder (ASD) is one of the most pervasive neurodevelopmental conditions, affecting 1 in 54 children,1 who are at greater risk for developing co-occurring neuropsychiatric disorders.2 Estimates put the risk of depression around quadruple that of the general population,3 with an anxiety disorder occurrence of about 40 %4 in individuals with autism. Consequences can be devastating, including a sevenfold greater likelihood of suicide.5 Moreover, deficits in executive functioning — critical to managing mood and anxiety6 — remain impaired across the lifespan of individuals with ASD.7 Together, these challenges compound pre-existing ASD-related difficulties with maintaining long-term independence and employment.8 Although current pharmacotherapies are largely unsuccessful because of poor medication compliance, lack of therapeutic effectiveness and adverse effects,9 mindfulness-based therapies (MBTs) show promise in mitigating depression and anxiety symptoms in adults with ASD10 and may improve executive functioning, as shown in other populations.11
Mindfulness-based stress reduction (MBSR) is an intensive 8-week meditation intervention aimed at directing attention toward present-moment awareness with a nonjudgmental and nonreactive attitude. In 2013, Spek and colleagues12,13 adapted the original MBSR protocol for adults with ASD and reported reductions in depression and anxiety relative to wait-list controls that persisted 9 weeks after the intervention. Antidepressant and anxiolytic effects of MBSR have since been replicated in randomized controlled trials (RCTs) using cognitive behavioural therapy (CBT)14 and social support/relaxation education (SE)15 comparison groups. Other improvements in quality of life, emotion regulation, autistic traits, stress and rumination have also been reported.12,16,17 Interestingly, social support is embedded in MBTs, and the lack thereof has been linked to loneliness and depression in adults with autism.18 Together, early research with small samples suggests MBTs have broad therapeutic efficacy in adults with ASD; however, it remains unclear if these effects are specific to mindfulness. In addition, the magnitude of therapeutic improvements in ASD has yet to be thoroughly characterized using rigorous clinical designs with active comparison interventions.
Little is known about the therapeutic neural mechanisms of MBSR in adults with ASD, with the exception of a single pilot study implicating the middle cingulate cortex (or dorsal anterior cingulate cortex [dACC]) in MBSR-elicited alleviation of depression symptomatology.15 In major depressive disorder (MDD), MBTs are theorized to exert therapeutic effects by normalizing maladaptive patterns of negative self-beliefs and attentional biases and rumination via cortical midlines structures.19 In adults with ASD, self-focused attention predicts depressive symptoms, supporting this idea.20 Mindfulness-based therapies enhance decentring, the capacity to take a detached perspective of one’s thoughts and emotions, which is associated with less depression21 and offers a potential mechanism of action in ASD. Accordingly, functional MRI (fMRI) tasks tapping into self-referential cognition activate overlapping brain regions as those modulated by meditation, including the insula, prefrontal cortex (PFC), cingulum and amygdala.22–24 A 2016 systemic review cited these brain regions as key targets of MBSR, irrespective of task and neuroimaging modality.25 Elucidating therapeutic neural mechanisms of MBSR may provide novel insights into the cause of psychiatric comorbidities in ASD, assist in developing personalized-medicine approaches and inform novel targets for neurostimulation research.
We conducted a double-blind RCT to compare the clinical efficacy of an 8-week MBSR intervention with an SE intervention for depression, anxiety, autism symptoms, executive functioning and mindfulness traits in an adequately powered sample of adults with ASD. We also sought to identify shared and distinct alterations to functional self-reflection neurocircuitry in the insula, cingulum, PFC and amygdala.25 We used a task-dependent fMRI functional connectivity analysis known as generalized psychophysiological interactions (gPPI) to identify mindfulness-induced changes to self-reflection neurocircuitry in these regions of interest (ROIs). We then evaluated these connectivity metrics for associations with therapeutic improvement. We hypothesized that MBSR and SE would elicit reductions in depression, anxiety and autistic traits based on previous literature on MBSR and SE in ASD;26,27 that MBSR would have additional efficacy for executive functioning and mindfulness traits, as found in other populations;11 that the MBSR group alone would show alterations to insular, ACC and lateral PFC (lPFC) functional connectivity, given previous findings for MBSR neuroimaging pertaining to interoception, salience detection and executive functioning;28–30 and that both groups would show alterations to amygdala, posterior cingulate cortex (PCC) and medial PFC (mPFC) functional connectivity patterns, based on previous literature showing inverse relationships between social support and depression and anxiety in ASD,26 and salutary effects of social support on amygdala reactivity and default mode network (DMN) dynamics.31,32
Methods
Participants and intervention groups
Our study protocol was approved by Arizona State University’s Institutional Review Board, and all participants provided written, informed consent, in accordance with the Declaration of Helsinki.
Adults with ASD who met the following inclusion criteria were recruited: an ASD diagnosis confirmed using the Autism Diagnostic Observation Schedule-2 (ADOS-2),33 administered by an experienced research-reliable member of the Southwest Autism Resource and Diagnostic Center, and DSM-534 criteria, as assessed by an experimental psychologist with 20 years of experience in ASD; and an IQ score greater than 70 on the Kaufman Brief Intelligence Test, Second Edition (KBIT-2).35 We excluded participants based on active suicidality, history of traumatic brain injury and substance abuse, but not on the basis of sex, age or general comorbidities (e.g., single cases of epilepsy, Tourette syndrome, bipolar disorder and hearing loss). No handedness restrictions were imposed. Power analyses for repeated measures analysis of variance (ANOVA) based on MBSR depression findings in ASD (medium effect size, ηp2 = 0.35) have suggested a sample size of 82 was sufficient to detect medium effects between groups with power set at 0.80.14
Participants were randomly assigned to an 8-week intervention of MBSR or SE by a team member unassociated with the delivery of treatment, data collection or analyses, using a block design “ABBA” sequence. The standard MBSR protocol was minimally adapted for adults with ASD as described previously.15
Self-reported behavioural measures
Pre- and postintervention questionnaires were administered by team members blinded to participant allocation (E.H., D.O., L.M. and S.D.) at the fMRI appointment within 1 month before and after the intervention to assess depression (Beck Depression Inventory-II [BDI-II]); state and trait anxiety symptoms (State–Trait Anxiety Inventory [STAI]-1 and -2); autism traits (Social Responsiveness Scale, second edition [SRS-2]); mindfulness traits (Five Facets Mindfulness Questionnaire [FFMQ], and its 5 subscales, “Describe,” “Observe,” “Act,” “Nonreactivity” and “Nonjudgment”); and executive functioning abilities (Behavior Rating Inventory of Executive Function [BRIEF] and its 8 subscales: “Working Memory,” “Shift,” “Monitor,” “Inhibit,” “Initiate,” “Plan,” “Organize” and “Emotional Control”). An intention-to-treat approach was implemented, which allowed participants with complete pre/post data sets to be included in the analysis, regardless of extent of participation in the intervention.
Acquisition of fMRI
Imaging data were collected on a 3 T Philips Ingenia scanner with a maximum gradient strength of 45 mT/m at the Barrow Neurological Institute (Phoenix, Arizona). T2*-weighted images were acquired with a gradient echo, echoplanar pulse sequence to elicit blood oxygen level–dependent (BOLD) contrast. The scanning parameters were slice thickness 3 mm, echo time (TE) 30 ms, repetition time (TR) 2000 ms, flip angle 90°, in-plane resolution 64 × 64, field of view 240 mm, and voxel size 3 mm × 3 mm. Images from the first 3 TRs were discarded to allow for stabilization. 3D magnetization–prepared rapid acquisition gradient echo T1 images were used for preprocessing (170 sagittal slices, slice thickness 1.2 mm, in-plane resolution 256 × 256, field of view 240 mm).
Self-reflection task using fMRI
Functional MRI scans were obtained during a self-reflection task before and after the intervention. Participants responded yes or no to trait adjectives for 2 conditions: self-condition (participants decided whether they possessed the trait adjective) and word-condition (participants decided whether the adjective was positively valenced) (see Pagni and colleagues15 for a detailed task description).
Preprocessing and modelling of fMRI
Preprocessing of fMRI data was completed using SPM12 (Wellcome Trust Centre for Neuroimaging, http://www.fil.ion.ucl.ac.uk/spm), including slice timing correction, realignment to the mean image, coregistration to the skull-stripped T1, normalization to the Montreal Neurological Institute (MNI) space using the Diffeomorphic Anatomical Registration Through Exponentiated Lie Algebra (DARTEL) method,36 and 8 mm full-width at half-maximum Gaussian kernel spatial smoothing. Scrubbing parameters for scans exceeding 5 mm relative displacement were generated using the Artifact Detection Tools toolbox in CONN (https://www.nitrc.org/projects/conn). The CONN-fMRI toolbox uses anatomical component correction (aCompCor) to remove noise and movement confounds on a voxel-to-voxel basis. Denoising parameters consisted of white matter (6P), cerebrospinal fluid (CSF; 6P), realignment (6P) and scrubbing (117P); no derivatives or polynomial expansion were applied. We included parameters for the effects of pre-word (2P), pre-self ( 2P), post-word ( 2P) and post-self (2P) with first-order derivatives. Data were bandpass filtered (0.008–0.09 Hz), despiked after regression and linear detrended. We used gPPI, a more recent version of PPI with improved sensitivity, to identify task-modulated functional connectivity. The gPPI was computed using bivariate regression in CONN with the following regressors: regressor for self-conditions, regressor for word-condition, time-series regressor for the seed region, PPI term for self-condition, and PPI term for word-condition.
Functional connectivity analyses
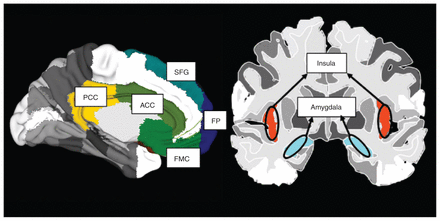
All behavioural and neuroimaging analyses were performed by researchers blinded to participant allocation (B.P. and B.B.B.). We selected neuroanatomical ROIs based on a systematic review of MBSR-mediated neural changes implicating the insula, amygdala, cingulum, dlPFC and mPFC,25 with dlPFC defined as the bilateral superior frontal gyrus (SFG) and mPFC defined as the bilateral frontal medial cortex (FMC) and frontal pole (FP). At the second level, we performed seed-to-voxel analyses using the Harvard–Oxford atlas in CONN at hypothesized ROIs for behavioural group × time interactions (MBSR > SE) and main effects of time (MBSR + SE) for post-self > pre-self and post-word = pre-word contrasts, with the number of classes as a covariate. We set α as a false discovery rate (FDR)–corrected p value less than 0.05 for target clusters. Significant ROIs were Bonferroni-corrected for left or right hemisphere for the insula, amygdala, SFG, FMC and FP (pFDR = 0.025); anterior and posterior divisions of the cingulum were corrected similarly (ACC and PCC, respectively, pFDR = 0.025). Owing to stringent FDR and Bonferroni correction, we accepted 1- and 2-sided/tailed (positive and negative contrast) tests. Regions of interest are shown in Figure 1.
Regions of interest (ROIs) for seed-based gPPI functional connectivity analysis. ACC = anterior cingulate cortex; FMC = frontal medial cortex; FP = frontal pole; PCC = posterior cingulate cortex; SFG = superior frontal gyrus. Image generated using the Harvard–Oxford human brain atlas tool.
Behavioural analyses
We performed repeated-measures analyses of covariance (ANCOVAs) using the Statistical Package for the Social Sciences, version 26 (SPSS-26), to examine behavioural group × time interactions and main effects of time, with the number of classes attended entered as a covariate. Our primary measures of interest were BDI-II, STAI-1, STAI-2, SRS-2, FFMQ (total score) and BRIEF (total score); we performed exploratory analyses for FFMQ and BRIEF subscales without multiple comparison correction. For measures that showed significant behavioural change, we performed 2-sided Pearson correlations with age and IQ.
Exploratory brain–behaviour relationships
We calculated pre- to post-change scores for significant behavioural group × time interactions and main effects of time. Next, significant functional connectivity changes that survived FDR and Bonferroni correction from the gPPI group by time and main effect of time analyses were extracted and entered into a 1-tailed Pearson correlation analysis in SPSS-26 with α set at p less than 0.05, with the connectivity changes hypothesized to be in a therapeutic direction.
Results
Participant demographics and behavioural effects
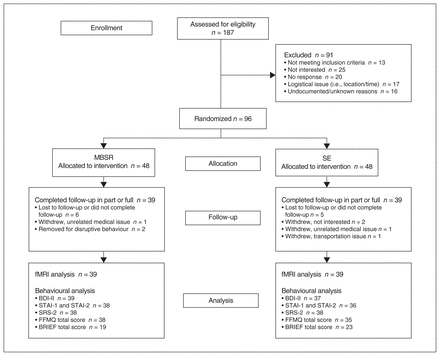
Ninety-six adults with ASD met our inclusion criteria; however, an attrition rate of 18.75% led to a final sample of 78 participants with partial or full pre- and postintervention data sets (39 who received MBRS and 39 who received SE; Figure 2). Group demographic characteristics are shown in Table 1. The groups did not differ with respect to sex distribution, age, IQ, autism severity or number of classes attended.
Participant flow chart detailing eligibility and treatment groups as well as participant retention and data attrition. BDI-II = Beck Depression Inventory-II; BRIEF = Behavior Rating Inventory of Executive Function; FFMQ = Five Facets Mindfulness Questionnaire; MBSR = mindfulness-based stress reduction; SE = social support/education; SRS-2 = Social Responsiveness Scale, second edition; STAI = State–Trait Anxiety Inventory.
Participant demographic characteristics and self-report means, standard deviations, and ranges
Behavioural measures were not complete for all participants. We added the BRIEF questionnaire for the second and third cohorts; therefore, this measure had fewer data points (n = 51) available for analysis. In addition, the following measures had missing questionnaires from either before or after the intervention or had less than 20% of the questions completed: BDI-II, n = 2; STAI-1 and STAI-2, n = 4; SRS-2, n = 2; FFMQ, n = 5; and BRIEF, n = 10 (Figure 2).
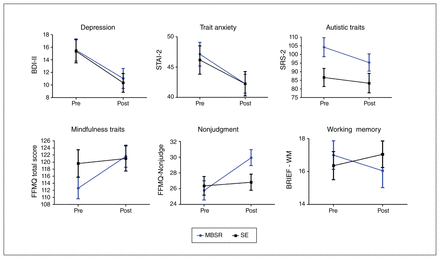
Main effects of time (collapsed by group, MBSR + SE) were detected on the BDI-II, STAI-2 and SRS-2, indicating decreases in depression, trait anxiety and autistic traits across participants; no state anxiety changes (STAI-1) were detected (Table 2). We detected a group × time interaction for FFMQ total score; our exploratory analysis showed an MBSR-specific increase on the FFMQ Nonjudgment subscale (Table 2 and Figure 3B). No other FFMQ subscales showed significant effects. There was no significant group × time interaction for the BRIEF total score; however, our exploratory analyses showed a group × time interaction on the BRIEF Working Memory sub-scale, with an MBSR-specific improvement (Table 2 and Figure 3B). No other BRIEF subscales showed significant effects. Baseline IQ did not correlate with change scores on the BDI-II (r68 = −0.021, p = 0.0868), STAI-2 (r67 = 0.033, p = 0.791), SRS-2 (r72 = 0.002, p = 0.990), FFMQ Nonjudgment subscale (r68 = −0.001, p = 0.996) or BRIEF Working Memory subscale (r37 = 0.001, p = 0.995). Age also did not correlate with change scores on the BDI-II (r74 = 0.021, p = 0.861), STAI-2 (r73 = 0.126, p = 0.290), SRS-2 (r78 = 0.077, p = 0.504), FFMQ Nonjudgment subscale (r74 = −0.029, p = 0.808) or BRIEF Working Memory subscale (r42 = 0.111, p = 0.486).
Behavioural change from preintervention to postintervention for mindfulness-based stress reduction (MBSR) and social support/ education (SE) groups. Error bars indicate standard error of the mean. There was a significant main effect of time shown on Beck Depression Inventory-II (BDI-II), State–Trait Anxiety Inventory (STAI)-2, and Social Responsiveness Scale, second edition (SRS-2) measures, indicating decreased depression, anxiety and autistic traits postintervention for both groups. There was a significant group × time interaction shown on Five Facets Mindfulness Questionnaire (FFMQ) total score, the FFMQ Nonjudgment subscale, and on the Behavior Rating Inventory of Executive Function (BRIEF) Working Memory subscale, indicating MBSR-specific increases in overall mindfulness and nonjudgment and decreases in executive functioning challenges.
Group behavioural effects
Functional connectivity specific to MBSR
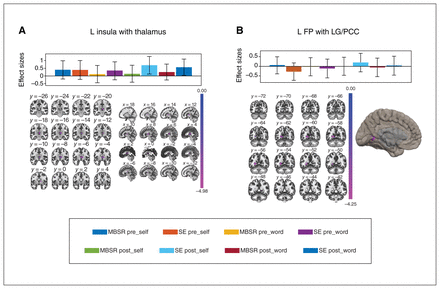
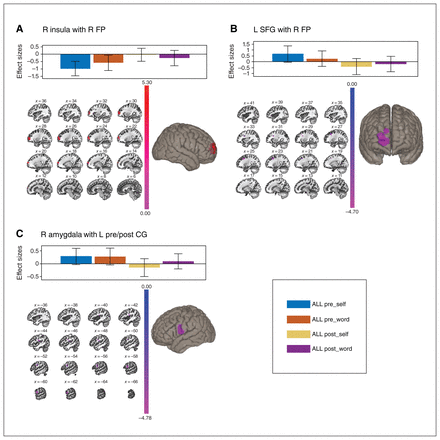
The left insula showed an MBSR-specific decrease in connectivity with the bilateral thalamus (Table 3 and Figure 4A). In addition, the left FP showed an MBSR-specific decrease in connectivity with the lingual gyrus (LG) and PCC (Table 3 and Figure 4B). No other ROIs showed group-specific effects after FDR and Bonferroni correction.
Functional connectivity changes: group × time interactions (pFDR < 0.025). Top: bar graphs adjusted for covariate of classes; bottom left: coronal slice view; bottom right: sagittal slice view or inflated brain of target cluster. (A) Decreased connectivity of the left insula with the bilateral thalamus. (B) Decreased connectivity of the left frontal pole (FP) with lingual gyrus (LG) and posterior cingulate cortex (PCC). FDR = false discovery rate; L = left; MBSR = mindfulness-based stress reduction; R = right; SE = social support/education.
Significant group × time interactions and main effects of time at ROIs
Nonspecific functional connectivity
We evaluated the main effects of time to identify nonspecific intervention effects. We found that the right insula showed an attenuated anticorrelation with the right FP following the intervention (Table 3 and Figure 5A). For the dlPFC, the left SFG transitioned from positive correlation (pretreatment) to anticorrelation (post-treatment) with the right FP (Table 3 and Figure 5B). The left amygdala transitioned from a positive (pretreatment) to a negative correlation (post-treatment) with the left pre- and postcentral gyrus and central operculum (Table 3 and Figure 5C). No other ROIs showed significant effects after FDR and Bonferroni correction.
Functional connectivity changes: main effects of time (across all participants; pFDR < 0.025). Top: bar graphs adjusted for covariate of classes; bottom left: sagittal slice view; bottom right: inflated brain of target clusters. (A) Increased connectivity of the right insula with the right frontal pole (FP). (B) Decreased connectivity of the left superior frontal gyrus (SFG) with the right frontal pole (FP). (C) Decreased connectivity of the right amygdala with the left pre- and post-central gyrus (CG). FDR = false discovery rate; L = left; R = right.
Brain–behaviour relationships
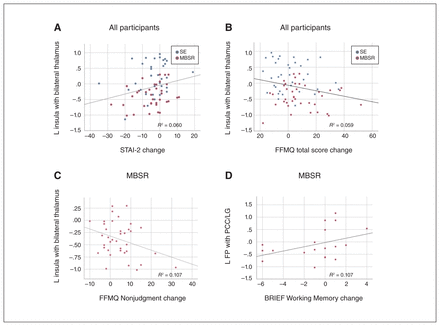
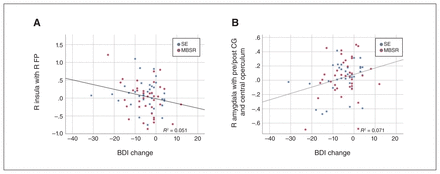
For MBSR-specific connectivity changes, decreased insula–thalamus connectivity was associated with decreased trait anxiety across all participants (r73 = 0.246, p = 0.018), increased FFMQ total scores across all participants (r73 = −0.244, p = 0.019) and increased FFMQ Nonjudgement sucscale scores across all participants (r72 = −0.336, p = 0.002) and in the MBSR group alone (r37 = −0.327, p = 0.024; Figure 6A–C). In addition, decreased FP–PCC connectivity correlated with working memory improvements (defined as decreases in BRIEF Working Memory subscale scores) across all participants (r41 = 0.296, p = 0.030) and in the MBSR group alone (r19 = 0.399, p = 0.045; Figure 6D). For group-nonspecific connectivity changes, decreased amygdala–pre-/ postcentral gyrus connectivity was associated with reduced depression across all participants (r73 = 0.267, p = 0.011) and in the SE group alone (r36 = 0.330, p = 0.025; Figure 7A). Increased insula–FP connectivity also correlated with decreased depression across all participants (r73 = −0.226, p = 0.028) and in the MBSR group alone (r37 = −0.318, p = 0.028; Figure 7B). No other correlations were significant for the other behaviour or connectivity measures.
Brain–behaviour scatterplots for connectivity changes specific to mindfulness-based stress reduction (MBSR; p < 0.05). Associations between (A) insula–thalamus connectivity and State–Trait Anxiety Inventory-2 (STAI-2) scores across all participants, (B) insula–thalamus connectivity and Five Facets Mindfulness Questionnaire (FFMQ) total scores across all participants, (C) insula–thalamus connectivity and FFMQ Nonjudgment subscale scores for participants who received the MBSR intervention, and (D) left frontal pole (FP)–posterior cingulate cortex (PCC)/lingual gyrus (LG) connectivity and Behavior Rating Inventory of Executive Function (BRIEF) Working Memory subscale scores for participants who received the MBSR intervention. L = left; SE = social support/education.
Brain–behaviour scatterplots for nonspecific intervention connectivity changes (p < 0.05). Associations between (A) insula–frontal pole (FP) connectivity and Beck Depression Inventory-II (BDI-II) scores across all participants and (B) amygdala–pre/postcentral gyrus (CG) and BDI-II scores across all participants. MBSR = mindfulness-based stress reduction; R = right; SE = social support/education.
Discussion
As hypothesized, both the MBSR and SE groups showed reductions in depression, anxiety and autistic traits, which suggests that social and educational support are adequate in diminishing psychological symptoms and improving mental health. Our findings align with those of Sizoo and Kuiper,14 who found no group differences in antidepressant and anxiolytic effects between MBSR and cognitive behavioural therapy (CBT). Such findings advocate for the use of active control groups rather than wait-list control groups to discern the “active ingredients” of mindfulness therapy.10,12 Importantly, future work including wait-list control groups will help characterize changes associated with time and test/retest effects to better understand the unique therapeutic elements of MBSR and SE. In addition, brain-based investigations are critical to distinguishing whether therapeutic mechanisms are nonoverlapping or overlapping. If nonoverlapping, neural mechanisms can be targeted in an additive manner to maximize efficacy and promote long-term outcomes; if overlapping, SE is sufficient, and potentially preferred because it may be more affordably delivered by community leaders and accessible via online social platforms. However, if mindfulness traits and working memory capacity are the outcomes of interest, MBSR may be preferred.
Mindfulness training uniquely enhanced nonjudgment and improved working memory abilities, but did not broadly affect mindfulness traits and executive functioning as hypothesized. Alleviation of psychological symptoms based on MBSR may be mediated by its emphasis on cultivating a nonjudgmental and nonreactive awareness.37 Lack of changes in mindfulness facets of acting, observing and describing are in line with a study that used a CBT comparison group38 but not a wait-list control comparison showing increases in all 5 traits.39 Our results suggest mindfulness training targets nonjudgment, but it remains unclear whether this effect is population specific or a product of active control group selection.39 Other studies have shown that pretreatment levels of trait mindfulness and autism severity predict the magnitude of MBSR outcomes.40,41 In addition, total practice hours mediate mindfulness trait changes, which in turn mediate mental health outcomes,38 supporting the notion that mindfulness abilities are key dose–response–dependent therapeutic mechanisms. These factors should be considered in future studies aimed at developing personalized medicine approaches for adults with ASD.17
Although findings about the influence of MBSR on broad executive functioning have been mixed, improvements in memory have been found consistently in adolescents and adults.42,43 Corroborating these relationships, the extent of meditation training is associated with working memory and short- and long-term memory performance, which also suggests dose–response effects.44 Our hypothesis of broad executive functioning improvements was only partially supported, with MBSR-specific improvements being restricted to working memory. Follow-up studies using objective executive functioning measures are required to elaborate these relationships in adults with ASD.
Changes induced by MBSR in insular activity and functional connectivity are well-established and attributed to alterations in interoception, exteroception and emotional information processing.25,28,29 In support of our hypothesis, the present study extends this body of work in adults with ASD by showing MBSR-specific decreases in insula–thalamus connectivity using a gPPI approach. Mindfulness-dependent changes in insula–thalamus connectivity were also reported by Farb and colleagues,45 who suggested that mindfulness training facilitates the transition from narrative self-focus, governed by the DMN, to present moment awareness, governed by the insula, thalamus and sensorimotor cortices. Furthermore, differences in insula–thalamus connectivity have been reported in meditators compared with nonmeditators, and after psychopharmacological administration of lysergic acid diethylamide, which possesses robust anti-depressant and anxiolytic properties.46,47 Together, these studies suggest that insula–thalamus connectivity changes are not task- or population-specific and may be a common therapeutic correlate of meditation and psychedelic-assisted psychotherapy.
Brain–behaviour correlations showed that those with the greatest decreases in insula–thalamus connectivity had greater reductions in anxiety (across all participants) and greater increases in nonjudgment toward experience (driven by MBSR). This association mirrors findings reported by Parkinson and colleagues,48 who found that insula connectivity generally correlated with mindfulness traits, and Gorman and colleagues49 who proposed a “fear network” that implicated the insula and thalamus in the context of anxiety disorders. Moreover, individuals with high levels of worry also showed insula hyperactivation during emotional processing;50 however, to our knowledge, no studies to date have referenced insula–thalamus connectivity in the context of MBSR-induced effects or anxiety; therefore, our study provides a novel therapeutic neural mechanism specific to mindfulness meditation in adults with ASD.
Frontal pole–PCC connectivity changes specific to MBSR align with results reported by Kral and colleagues,51 who found MBSR-specific alterations in this pathway. However, their finding was in the opposite direction, likely arising from resting-state versus task-based design or the use of a neurotypical population.51 Aberrant FP–PCC connectivity profiles during self-reflection have been identified in ASD and associated with age, suggesting that mindfulness training may intervene on an ASD-affected circuit.52 Posterior cingulate cortex activity is a common feature of novice but not expert meditators, which is suspected to represent the transition from effortful toward effortless attention to present moment experience;53 therefore, our findings of frontal regulation of the DMN match what is expected in novice practitioners. The identified PCC cluster also encompassed the LG — a frequently reported region in the MBSR literature — which contributes to episodic memory.54 In line with this, FP–LG/PCC connectivity changes in our study correlated with improvements in working memory and might reflect decoupling of the anterior–posterior DMN axis, a circuit that displays pronounced atypicality in ASD underpinning deficits in self-awareness.55
Both the MBSR and SE groups showed a loss of pre-existing FP–insula anticorrelations, suggesting common social factors rather than mindfulness training elicited DMN–salience network (SN) connectivity alterations. While we predicted FP connectivity would be nonspecific to the intervention group, we expected insula connectivity to be specific to MBSR. Thus, our hypothesis was partially supported. Increases in cortical thickness in the right FP and in PFC–insula connectivity induced by MBSR have been reported;56 however, to our knowledge, this study is the first to report FP–insula connectivity changes. Whether these connectivity changes are unique to ASD is unclear; however, they may be clinically relevant. Cognitive behavioural therapy has been shown to alter PFC–insula connectivity in patients with depression.57 The negative correlation we observed between FP–insula connectivity and depression symptomatology adds further support to the potential clinical significance of DMN–SN interactions.
Our hypothesis of MBSR-specific alterations to SFG connectivity was not supported. Instead, both the MBSR and SE groups showed a transition from correlated to anticor-related SFG and FP activity. In line with the nonspecificity aspect, CBT and MBSR were compared and both altered lateral and medial PFC activity in patients with social anxiety.58 Previous work has established coactivation of the SFG and mPFC during self-reflection in ASD, as well as hyperconnectivity accompanying self-centrism and excessive self-evaluation in patients with MDD.19 Our findings suggest both MBSR and SE alter lateral–medial PFC connectivity during self-referential neural processing and warrant follow-up to evaluate clinical relevance.
The absence of cingulum (ACC and PCC) and medial frontal cortex (MFC) connectivity changes were unexpected, as the DMN and cortical midline structures are commonly reported therapeutic neural substrates of MBSR.51 In a pilot study, we identified middle cingulate connectivity changes with the sensorimotor network that were not replicated in this larger sample.15 Although we did observe MBSR-specific and MBSR-nonspecific changes in the FP, the lack of effects in major DMN hubs — namely, the MFC and cingulum — may reflect a unique property of mindfulness training or interventions more generally for adults with ASD. For example, aberrations in within- and between-DMN connectivity in ASD55 may have affected how the interventions were received and interacted with brain function. No behavioural intervention studies examining the DMN have been published in ASD, making conjecture difficult.
Our hypothesis of intervention nonspecific alterations to amygdala connectivity was supported; however, we anticipated amygdala–PFC connectivity changes, as reported in the context of emotion regulation.25 Instead, both MBSR and SE showed amygdala–pre-/postcentral gyrus decoupling. We surmise that social engagement akin to both interventions over the 8-week period reduced feelings of isolation and loneliness, an effect seen with other socially oriented interventions,18 and diminished amygdala input to the sensorimotor system. Attenuation of amygdala response induced by MBSR is often interpreted as top–down inhibition, expressed neurobiologically as increased PFC–amygdala connectivity (see Gotink and colleagues25 for a review). While activation studies have found decreased signal in the amygdala and increased signal in sensorimotor regions, to our knowledge, this is the first study to identify connectivity changes in this limbic–sensorimotor circuit.
Decreased connectivity between the amygdala and sensori-motor system correlated with alleviation of depressive symptoms across all participants, offering a shared neural correlate of MBSR and SE. Amygdala changes induced by MBSR are suspected to be a key factor in improved emotional regulation,25 with amygdala dysfunction underlying psychopathology;59 pre-/postcentral gyrus connectivity changes have also been reported in adults with ASD following MBSR.15 Our findings suggest that limbic–sensorimotor decoupling may be a nonspecific metric of depression reduction in adults with ASD. Future work should seek to replicate and extend these findings using ROI-to-ROI and causal approaches.
Limitations
The greatest limitation to the present analysis is the modest sample size, which may have been underpowered to detect small to medium effects. Nevertheless, this study is the largest of its kind in adults with ASD and was powered to detect medium to large effects. Results yielded significant group × time interactions and main effects of time on behavioural measures and seed-to-voxel correlations at ROIs. Another potential limitation is the susceptibility of fMRI analyses to type 1 errors. To reduce these concerns, we harnessed α inflation by using rigorous statistical methods, including FDR and Bonferroni correction. We also used a generalized psychophysiological interactions approach to elucidate task-specific modulation, a method that, compared to standard approaches, achieves improved model fit, specificity and sensitivity.60
Other limitations pertain to generalizability, including the recruitment of intellectually abled adults, which limits conclusions related to children and adolescents, and adults who experience intellectual challenges (IQ < 70). To increase generalizability, we enrolled adults within a wide age range (age 18–80 yr), of all sex/gender orientations and handedness, regardless of other comorbidities common to ASD (e.g., epilepsy, Tourette syndrome, hearing loss). Although we did not directly evaluate the influence of IQ, autism severity, age or sex on outcomes, a correlation analysis found no effect of IQ and age within the study sample. Additionally, self-report measures are limited, and future integration of objective cognitive and clinical measures in MBSR studies of adults with ASD are warranted. Future research should seek to characterize the complex interplay of demographic factors on clinical outcomes, use clinician and performance-based assessments to confirm and extend these findings, and use mediation/moderation analyses to elucidate psychological and neural mediators of long-term therapeutic improvements.17
Conclusion
The current study contributes to a growing endeavour to treat mental health comorbidities and identify therapeutic biomarkers in ASD and, to our knowledge, marks the first comprehensive investigation into the therapeutic neural correlates of MBSR in this population. We identified MBSR-specific and intervention-nonspecific effects in behaviour and brain connectivity patterns. Superior therapeutic outcomes were observed with MBSR than with SE in the domains of working memory and mindfulness abilities. However, both MBSR and SE led to significant reductions in depression, anxiety and autistic traits. These clinically meaningful changes suggest social support is similarly efficacious and sufficient to reduce psychiatric symptoms in adults with ASD. Additionally, this study elucidated mindfulness-specific and -nonspecific neural mechanisms. Connectivity changes driven by MBSR were found in the insula and thalamus, corresponding with decreased trait anxiety and increased mindfulness; and in anterior–posterior DMN axis connectivity, corresponding with improved executive functioning. Nonspecific connectivity changes were found across all participants in SN–DMN connectivity, corresponding with reductions in depression; in lateral–medial PFC axis connectivity; and in the limbic–sensorimotor system, corresponding with reductions in depression. Overall, results suggest overlapping and nonoverlapping neural substrates of MBSR and SE in adults with ASD. Novel connectivity metrics provide critical information for the development of personalized medicine, causal connectivity analyses and neurostimulation research.
Acknowledgements
The authors are grateful to the participants who made this study possible and to Sharmeen Maze for her dedication to pristine MRI data collection.
Footnotes
Competing interests: B. Pagni received support for article processing charges from the ASU Graduate Program Student Association’s Publication Grant. M. Walsh receives funding from the National Institute of Mental Health ( NIMH F31MH122107). M. Dixon received payment for execution of the program as part of this study. L. Ballard declares having received honoraria for facilitation of guided mind-fulness medications and educational presentations on mindfulness meditation.
Contributors: B. Pagni, M. Dixon and B. Braden designed the study. B. Pagni, E. Hill, M. Walsh, S. Delaney, D. Ogbeama, L. Monahan, N. Guerithault, M. Dixon, L. Ballard and B. Braden acquired the data, which B. Pagni, E. Hill, M. Walsh, J. Cook, M. Dixon and B. Braden analyzed. B. Pagni, M. Dixon and B. Braden wrote the article, which all authors revised. All authors gave final approval of the version to be published and agreed to be accountable for all aspects of the work.
Funding: This work was supported by Arizona State University (start-up funding) and the National Center for Complementary and Integrative Health (F31AT01097). The funding sources were not involved in the research or preparation of the manuscript.
Data sharing: Data are available upon reasonable request to the corresponding author.
- Received August 31, 2022.
- Revision received November 16, 2022.
- Revision received December 5, 2022.
- Accepted December 9, 2022.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/