Abstract
The peptide neurotensin has been studied for more than 30 years. Although it is widely distributed in the central and peripheral nervous systems, neurotensin has been more intensely studied with regard to its interactions with the central dopamine system. A number of claims have been made regarding its possible implication in many diseases of the central nervous system, including schizophrenia. In this review, we describe briefly the basic biology of this neuropeptide, and then we consider the strengths and the weaknesses of the data that suggest a role for neurotensin in schizophrenia, drug abuse, Parkinson’s disease, pain, central control of blood pressure, eating disorders, cancer, neurodegenerative disorders and inflammation.
Introduction
Neurotensin (NT) is a 13-amino-acid peptide originally isolated in 1973 from bovine hypothalami.1 This neuropeptide is found in the central nervous system (CNS), as well as in the gastrointestinal tract. However, this review will focus on the CNS effects of NT. Like many other neuropeptides, NT acts as a neuromodulator in the nervous system where the close association between NT and dopamine (DA) systems has been well documented (for review, see Binder et al2). The neuromodulation of DA transmission by NT raises the possibility that the neurotensinergic system could be implicated in diseases where the common determinant is a deregulation of DAergic transmission, such as schizophrenia, drug abuse and Parkinson’s disease.2 In this review, we will attempt to provide a summary of current knowledge about NT with special emphasis on the evidence suggesting its implication in CNS diseases.
Biosynthesis, maturation, release and degradation of NT
Like all neuropeptides, NT is synthesized as part of a larger inactive precursor that also contains neuromedin N (NN), a 6-amino-acid NT-related peptide.3,4 In the rat, the NT/NN precursor consists of 169 amino acid residues4 containing 4 pairs of dibasic residues (Lys-Arg). The endoproteolytic processing of this precursor at pairs of basic residues is mediated by members of the prohormone convertase (PC) family.5 The major PC involved in this process in the brain is likely to be PC2, because its expression colocalizes with all regions immunoreactive for NT.6,7 Although expression of PC1 and PC5A in the brain is more limited, these endoproteolytic enzymes also have the capacity to process the NT/NN precursor6,8,9 and could contribute to the regional differences observed in the relative proportions of NT and NN among brain structures.10
Once processed as an active peptide, NT is stored in dense-core vesicles11,12 and released in a calcium-dependent manner.13–15 NT transmission is terminated primarily by cleavage of NT in the C-terminal hexapeptide sequence that accounts for the biological activity of the peptide.16 Several endopeptidases belonging to the family of metallopeptidases have also been shown to contribute to the physiological inactivation of NT, such as neutral endopeptidase 24.11 (EC3.4.24.11),17 angiotensin-converting enzyme,18 endopeptidase 24.15 (EC3.4.24.15)19 and endopeptidase 24.16 (EC3.4.24.16).20 However, endopeptidase 24.16 is considered the main endopeptidase contributor to the inactivation of NT, because it is expressed ubiquitously.21–23
NT localization in the CNS
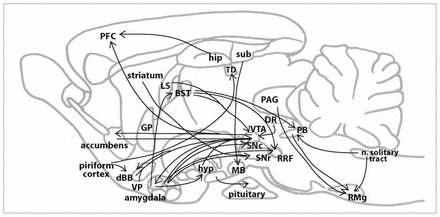
NT-producing neurons and their projections are widely distributed in the CNS, which explains the wide range of effects of this peptide. In the rat brain, a number of NT-containing neural circuits have been suggested (Fig. 1): (1) from cells in the central nucleus of the amygdala to the bed of the stria terminalis,24 to the substantia nigra pars compacta,25 to the substantia nigra pars reticulata,26 to the ventromedial nucleus of the hypothalamus; 27 (2) from the hippocampus projecting through the cingulate cortex to the frontal cortex;28 (3) from the arcuate and paraventricular nuclei of the hypothalamus to the neurointermediate lobe of the pituitary gland;29 (4) from cells in the ventral tegmental area (VTA) to the nucleus accumbens, the diagonal band of Broca, the prefrontal cortex and the amygdala; 30,31 (5) from cells in the endopyriform nucleus and prepyriform cortex to the anterior olfactory nucleus, the nucleus of the diagonal band of Broca and the dorsomedial thalamic nucleus; 32,33 (6) from cells in the periaqueductal grey matter (PAG), the parabrachial nucleus and the nucleus of the solitary tract to the nucleus raphe magnus;34 (7) from cells in the preoptic region and the rostral part of the lateral hypothalamus, the rostral part of the lateral septum, the bed nucleus of the stria terminalis, the ventromedial ventral pallidum, the dorsal raphe nucleus and the diagonal band of Broca to the VTA;35 (8) from the subiculum to the mammillary bodies;36 (9) from the dorsal lateral portion of the bed nucleus of the stria terminalis to the parabrachial nucleus; 37 (10) from the striatum to the substantia nigra pars reticulata; 38 and (11) from the central nucleus of the amygdala and the bed nucleus of the stria terminalis to the retrorubral field.39 In addition to neurotensinergic pathways to and from the VTA, high concentrations of NT have also been observed in regions associated with DAergic projections, such as the caudate nucleus, globus pallidus and putamen40,41 (for a review, see Binder et al2). At the spinal cord level, NT-containing cells have been found in the substantia gelatinosa.41
Sagittal representation of neurotensinergic pathways in the rodent brain. BST = bed nucleus of the stria terminalis; dBB = diagonal band of Broca; DR = dorsal raphe nucleus; GP = globus pallidus; hip = hippocampus; hyp = hypothalamus; LS = lateral septum; MB = mamillary bodies; n. solitary tract = nucleus of the solitary tract; PAG = periacqueductal grey; PB = parabrachial nucleus; PFC = prefrontal cortex; RMg = raphe magnus nucleus; RRF = retrorubral field; SNc = substantia nigra pars compacta; SNr substantia nigra pars reticulata; sub = subiculum; TD = dorsal thalamic nucleus; VP = ventral pallidum; VTA = ventral tegmental area.
Pharmacology of NT receptors
Both central and peripheral actions of NT depend on recognition of the peptide by specific receptors at the plasma membrane of target cells. Three different NT receptors, referred to as NTS1, NTS2 and NTS3/sortilin, have been cloned so far42 (for review, see Hermans and Maloteaux43 and Mazella44). NTS1 and NTS2 belong to the G-protein-coupled receptor superfamily45–48 and were initially distinguished pharmacologically on the basis of their affinities for NT and their differential sensitivity to the histamine antagonist levocabastine.49
Indeed, the type 1 NT receptor is often referred to as the high-affinity binding site (Kd = 0.1–0.3 nmol/L) and is insensitive to levocabastine.49 In rat brain, NTS1 transcripts are located primarily in neurons from the septum, substantia nigra, VTA, zona incerta, suprachiasmatic nucleus, as well as prefrontal, entorhinal and retrosplenial cortices.50,51 NTS1-containing nerve terminals were seen by immunohistochemical studies in the caudate putamen, bed nucleus of the stria terminalis, olfactory tubercle, as well as the lateral septum, amygdala, lateral habenula and nucleus accumbens.52,53
NTS1 is functionally coupled to the phospholipase C and the inositol phosphate (IP) signalling cascade (for review, see Vincent et al42 and Hermans and Maloteaux43). Other possible signalling could occur through activation of cyclic guanosine monophosphate (cGMP), cyclic adenosine monophosphate (cAMP), arachidonic acid production,54,55 mitogen-activated protein (MAP) kinase phosphorylation56–58 and inhibition of Akt activity.59 NTS1 undergoes agonist-dependent internalization when exposed to its natural agonist NT. This phenomenon is well documented in neurons and in NTS1-expressing cell lines (for review, see Beaudet et al,60 and also Hermans et al,61 Vandenbulcke et al62 and Nguyen et al63). The effects mediated by NTS1 are usually blocked by its selective nonpeptide antagonist SR48692,64 or by the broad-spectrum antagonist SR142948A65 that recognizes both NTS1 and NTS2. The generation of mice lacking NTS1 receptors66,67 has allowed the attribution to NTS1 of the hypothermic, impaired motor coordination and gastrointestinal motility effects of NT. A role for NTS1 in analgesia has also been suggested,68,69 but this is still controversial.66
The NT type 2 receptor (NTS2) has a lower affinity for NT (Kd = 3–10 nmol/L) than NTS1 and is sensitive to levocabastine. Unlike its high-affinity counterpart, NTS2 expression is more diffusely distributed throughout the brain. In the rat brain, high densities of NTS2 mRNA and protein were detected in many regions documented to receive a dense neurotensinergic innervation, such as the bed nucleus of the stria terminalis, olfactory bulb, substantia nigra, VTA and PAG.70–72 However, some of the highest concentrations of NTS2 receptors were found in areas devoid of neurotensinergic inputs such as the cerebral cortex, hippocampus and cerebellum,72 suggesting that endogenous ligands other than NT may activate this receptor.73 At the cellular level, NTS2 expression was initially detected in both neurons70,74 and astrocytes,70,75,76 although recent immunostaining experiments have reported signal only in neurons throughout the brain,72 a puzzling observation.
The pharmacological and signalling properties of NTS2 are still controversial. In particular, it is unclear whether NT acts as an agonist, inverse agonist or antagonist at this site. Depending upon the species from which the NTS2 was isolated47 and the cell system used to evaluate signalling (oocytes, Chinese hamster ovary cells, COS-7 cells and HEK293 cells), different results have been reported. The only common result is the agonist action of SR48692,74,77–80 which was originally described as an antagonist with a higher affinity for NTS1 than NTS2 receptors.64, In addition, both NT and levocabastine exert a species-dependent agonist (mouse)47 or antagonist (human) profile.78,79 Indeed, in cell lines expressing human NTS2, SR48692 activated Ca2+ mobilization or IP formation, whereas both NT and levocabastine antagonized this response. The species-dependent pharmacological properties of NTS2 could be linked to differences in the constitutive activity of this ghrelin-family receptor. 79,81 The most convincing data supporting an agonist role of NT at rodent and human NTS2 receptors is the ability of NT to produce ligand-induced internalization of receptor–ligand complexes.80,82,83 This internalization of NTS2 is apparently associated with activation of extracellular signal–regulated kinases 1/2 (ERK1/2) but not Ca2+mobilization.83
The type 3 NT receptor (NTS3), which was originally identified as the intracellular sorting protein sortilin, is unique among neuropeptide receptors in that it is a single transmembrane domain receptor of the type I family.84 NTS3/sortilin, like NTS1, binds NT with high affinity once converted to its mature form upon cleavage by furin.85 This receptor is found in neurons, glia and adipocytes.76,86,87 In the rat brain, regions expressing both high levels of NTS3/sortilin mRNA and protein include the piriform cortex, hippocampus, islands of Calleja, medial and lateral septal nuclei, amygdaloid nuclei, thalamic nuclei, supraoptic nucleus, substantia nigra and Purkinje cell layer of the cerebellar cortex.88 NTS3/sortilin has the ability to bind a variety of other ligands in addition to NT, such as the receptor-associated protein (RAP)89,90 and lipoprotein lipase (LpL).91 It is thus possible that NTS3/sortilin also subserves non-NT-related functions in the mammalian CNS. To date, very little is known about the physiological role of the NTS3/sortilin receptor. The strongest evidence that NTS3 can behave as a true NT receptor lies in the recent demonstration that this protein may be involved in the NT-induced migration of human microglial cells via the stimulation of both MAP and phosphatidylinositol (PI)3-kinase-dependent pathways. 92 NTS3/sortilin may also be involved in the growth of certain cancer cell lines93 and in the modulation of NT signalling via mediation of NT uptake and degradation.86,94,95
In closing, let us mention that a candidate fourth NT receptor has been proposed, although little is currently known about its physiological relevance. This receptor, named SorLA/LR11, is, like NTS3, a single transmembrane domain receptor of the type 1 family.96 In the CNS this receptor is expressed in neurons,97 but its role in the effects of NT remains to be determined. It has been suggested that it regulates the processing of the amyloid precursor protein.98
Major physiological roles of NT in the CNS
In this review, we focus our attention on the roles of NT in the CNS. However, it is important to point out that this peptide is also highly expressed peripherally where it acts as a modulator of the gastrointestinal and cardiovascular systems.99–102
Action of NT on the DA system
Neurotensinergic innervation of DAergic neurons
There is strong anatomical evidence for the interaction between NT and the DAergic system. Midbrain DAergic neurons located in the VTA, substantia nigra and retrorubral field are densely innervated by NT-containing axons.35,41 The origin of the NT plexus that innervates DA neurons is not fully established, but some of the neurotensinergic input to the VTA originates from the lateral preoptic area and the rostral part of the lateral hypothalamus.35 The origins of the neurotensinergic fibres innervating the substantia nigra and retrorubral field remain controversial.
Effect of NT on DAergic neuron firing rate and DA release
NT increases the firing rate of mesencephalic DAergic neurons and has a depolarizing effect, at least in part via NTS1 activation.103–105 This increase in firing rate is Ca2+-dependent and requires IP3 receptor activation.106 Because NT modulates the activity of DAergic neurons, it is thus not surprising that NT also modulates DA release. NT has been shown to modulate DA release from striatal, nucleus accumbens and retinal slices.107–110 Depending on the experimental model, and the brain area where it is injected, NT can induce an increase or a decrease in DA release. Using electrochemical detection and a carbon fibre electrode, NT, when applied intracerebroventricularly, was found to increase DA levels in the nucleus accumbens. 111 However, using microdialysis, perfusion of NT in the nucleus accumbens was found to lead to a local decrease in DA release by a γ-aminobutyric acid-A (GABAA)-receptor-dependent mechanism.112 Moreover, microdialysis of antibodies against NT in the cerebral ventricles leads to an increase in DA release in the nucleus accumbens.113 NT could also act to facilitate DA release in the accumbens by decreasing the ability of the dopamine D2 receptor (D2R) to mediate presynaptic inhibition at DAergic axon terminals.114 Finally, NT injected in the VTA causes an increase in DA release in the nucleus accumbens.115 This is likely to be explained by the ability of NT to increase the firing rate of DAergic neurons. However, the possible contribution of a decrease in somatodendritic autoreceptor function remains to be evaluated.
Regulation of DA receptor function
In addition to its action on the excitability of DAergic neurons, NT can also act to regulate the function of DA receptors. Indeed, there is biochemical and electrophysiological evidence suggesting an interaction with D2R. First, binding experiments showed that NT induces a modest decrease in the affinity of D2R for its agonists.116–119 In-vivo and in-vitro studies showed that NT increases the dissociation constant (Kd) of D2R without affecting Bmax. These results suggest that the function of D2R receptors is affected by NT, but the number of functional receptors remains unchanged.120–124 The interaction between NT and D2R was also measured by evaluating DA release in the striatum. Perfusion of NT in this region decreases the ability of DA agonists to inhibit basal DA levels, an observation that is compatible with the existence of some functional interaction between NT and D2R. However, whether this interaction occurs directly on DAergic axon terminals is unclear.125 Modification of D2R function by NT was also evaluated electrophysiologically. Indeed, patch clamp recordings suggest that NT and D2R may regulate in an opposing fashion a similar potassium conductance in DAergic neurons, thus providing a possible explanation for an NT–D2R interaction:126 NT would increase cellular excitability by inhibiting potassium channels, whereas a D2 agonist would decrease excitability by activating the same channels. According to such a model, NT would thus simply prevent the D2 agonist from activating the potassium channels by independently inhibiting them. However, extracellular recordings of the firing of midbrain DAergic neurons showed that NT is able to reduce the inhibitory effect of the D2R agonist, even at concentrations of NT that do not increase firing. 127–129 This observation argues against the idea that the interaction depends on the regulation of a common potassium channel by the 2 receptors.
Action of NT on other neurotransmitter systems
In addition to its widely studied interaction with the DAergic system, NT also affects other neurotransmitter systems.
Evidence for the effect of NT on cholinergic transmission
In the CNS, cholinergic neurons are found in the midbrain, striatum and magnocellular complex of the basal forebrain. 130,131 There is evidence for the expression of NT receptors by cholinergic neurons in the basal forebrain and also for neurotensinergic innervation of this structure, suggesting a direct modulation of these neurons by NT.132–134 Anatomical data also suggest that some cholinergic neurons may be modulated indirectly by NT. For example, an indirect modulation of cholinergic neurons is mediated by NT receptors located on DAergic neurons innervating the striatum.135 The effect of NT on acetylcholine (ACh) release was also studied, and it was shown that NT increases K+-evoked ACh release in striatal brain slices.136 However, a contradictory result has been obtained by in-vivo microdialysis in the striatum, where no effect of NT on ACh release was detected.137 The effect of NT on ACh release in the cortex also seems to be complex. It was reported that NT depresses ACh release in frontal cortex slices, has no effect in the occipital cortex and has a potentiating effect in the parietal cortex.138 Electrophysiological experiments showed that in cholinergic neurons of the basal forebrain magnocellular complex, NT produces depolarization and rhythmic bursting.139–141 The same result has been obtained in cholinergic neurons of the medial septum and vertical limb of the diagonal band nuclei.142 NT-induced excitation has been associated in nucleus basalis of Meynert cholinergic neurons with a reduction in inwardly rectifying K+ conductances.143
Effect of NT on serotonergic neurotransmission
There are anatomical data suggesting an interaction between NT and serotonergic neurons. Indeed, neurons of the rostral part of the raphe synthesize NT, whereas NT receptors are widely expressed in most of the raphe.72,144–146 Much of the raphe nuclei also receives significant neurotensinergic innervation. 147,148 There is physiological evidence for the presence of NTS1 on serotonergic neurons. Indeed, a subpopulation of serotonergic neurons located in the nucleus raphe magnus and in the dorsal raphe respond to NT by an increase in their firing rate, an effect that is blocked by SR48692.149,150 The functional role of NT in the raphe remains to be determined, but it may participate in the modulation of some of the known functions of the serotonergic system including nociception,151 sleep–wakefulness cycle regulation152 and stress-related responses.153
Effect of NT on GABAergic neurotransmission
The activity of GABAergic neurons evaluated either by measuring firing rate or by sampling extracellular GABA can be increased by NT in the hippocampus, striatum, prefrontal cortex and globus pallidus.137,154–156 Other studies have shown that NT decreases GABA release both in the substantia nigra and the ventral thalamus by a D2R-dependent mechanism.157 These effects were counteracted by SR48692, suggesting that they were mediated by NTS1. Such an increase in GABA release could result in a number of indirect effects. For example, NT-mediated GABA release in the striatopallidal pathway inhibits pallidal GABAergic neurons projecting to the subthalamic nucleus, resulting ultimately in an increase in glutamate release.158
Effect of NT on glutamate neurons
NT increases glutamate release in some brain regions, such as the striatum, globus pallidus, frontal cortex and the substantia nigra.157–160 Considering that an excess of glutamate can induce excitotoxicity within the context of conditions such as stroke, Alzheimer’s disease and Parkinson’s disease, it is interesting to note that NT can enhance glutamate excitotoxicity in both mesencephalic and cortical neurons.161,162 In addition to enhancing glutamate release, NT can modify the function of glutamate receptors. Indeed, NT amplifies N-methyl-d-aspartate (NMDA)-mediated signals by a protein kinase C (PKC)-dependent mechanism. 162 It is well known that stimulation of the NMDA receptor mediates glutamate-induced excitotocixity, leading to apoptotic cell death or necrosis.163,164 NT can increase the number of apoptotic cells induced by glutamate exposure.162
Neuroendocrine effects of NT
One of the major central functions of NT is to regulate neuroendocrine systems (for review, see Rostene and Alexander165). A number of essential nuclei implicated in hormone secretion and regulation, such as the arcuate nucleus, the parvocellular division of the paraventricular nucleus and the periventricular nucleus of the hypothalamus, as well as the pituitary, receive neurotensinergic projections and express NT binding sites.29,166 NT influences neurons that synthesize and secrete gonadotropin-releasing hormone, somatostatin and corticotrophin-releasing factor (CRF).167 In addition to neurotensinergic projections arriving from other regions, NT is synthesized in some hypothalamic secretory cells located for the most part in the arcuate and paraventricular nuclei. 168,169 It appears that NT produced locally contributes to the regulation of the release of some neuromodulators and hormones such as CRF, galanin, enkephalin, cholecystokinin and growth hormone–releasing hormone170–172 and could thus be implicated in stress-related functions. Compatible with this, long-term delivery of SR48692 to the paraventricular nucleus of the hypothalamus attenuated stress-induced elevations of the release of corticosterone and adrenocorticotropic hormone (ACTH).173 In addition, there is a mutual regulation between NT and neuroendocrine systems, because androgen, estrogen, glucocorticoid and thyroid hormone affect NT synthesis in the hypothalamus and anterior pituitary.174–177
Action on non-neuronal cells
In addition to its action on neurons, NT has been reported to affect glial cells such as astrocytes and microglia. Evidence for the effects of NT on astrocytes is provided by the fact that these cells express NT-binding sites in many brain areas such as the midbrain, cortex, striatum, brain stem and spinal cord.178 The subtype of NT receptor expressed by these cells is presently unclear. A functional study of cultured VTA astrocytes showed that the calcium increase induced by NT was partly sensitive to SR48692 and was not mimicked by the NTS2 agonist levocabastine.179 This result suggests that astrocytes express NTS1 but also that another undefined receptor could be present on these cells. Other studies, using embryonic rat midbrain and postnatal cerebral cortex cell cultures, showed that NTS2 was expressed by a subpopulation of glial cells and that NTS2 expression was increased in reactive astrocytes. 75,76,174 Compatible with the idea that NT receptors are expressed by a subpopulation of astrocytes, it was reported that whereas NT-induced intracellular Ca2+ mobilization can be detected in about 30% of primary mesencephalic astrocytes, this response is absent from purified adherent astrocytes from the same region.106 Single-cell reverse transcription-polymerase chain reaction (RT-PCR) experiments from acutely dissociated astrocytes, as well as double in situ hybridization experiments at different developmental time points, would be useful to clarify the expression pattern of NT receptors in astrocyte subpopulations. The physiological role of NT receptors in astrocytes remains largely unknown. However, because there are increasing data suggesting the involvement of astrocyte-derived messengers in regulating neuronal activity and synaptic transmission,180,181 future work is required to directly investigate the hypothesis that NT induces the release of chemical messengers from astrocytes, thus indirectly regulating neurons in the mesencephalon or other structures. Interestingly, it was shown recently that although NT-induced Ca2+ mobilization in astrocytes is not necessary for the excitatory effect of NT on midbrain DAergic neurons in culture, this excitation is enhanced when neurons are grown together with NT-responsive astrocytes.106
Finally, NT also affects the activity of microglial cells. More specifically, NT elicits the migration of a microglial cell line by an NTS3-dependent process. It was also shown that ERK1/2 and PI3-kinase intracellular pathways are implicated in this effect.92 Such results suggest that NT plays a role in cellular responses to inflammation or brain lesion, because microglia motility is very important for these processes.
Role of NT in schizophrenia?
On the basis of a growing literature suggesting the existence of anatomical and physiological interactions between NT and the mesolimbic DA system, Charles Nemeroff published an influential paper 25 years ago entitled “Neurotensin: Perchance an endogenous neuroleptic?”182 This much-cited paper introduced the bold suggestion that NT could act to inhibit DA neurotransmission, in much the same way as antipsychotic (or neuroleptic) drugs. Nemeroff also proposed that perhaps schizophrenia symptoms result in part from a deficit in brain NT, leading to excessive DA neurotransmission. Here we will briefly reconsider this hypothesis and ask whether there are sufficient data to support it.
First, what is the evidence supporting an intimate relation between DA and NT? As stated previously, NT is indeed found to be abundantly expressed in the ventral midbrain in an intricate network of thin fibres. In addition, NT-binding sites and NT receptors are expressed in high numbers by DA neurons. These basic observations certainly are in agreement with the idea that NT is a key regulator of the DA system. However, it is important to remember that NT is also abundant in a number of additional nuclei in the brain and, unless a deficit in NT was restricted to the ventral midbrain or to the regions receiving the densest DA projections, a disease implicating a decrease in NT would be expected to be accompanied by a large variety of symptoms not restricted to those found in schizophrenia.
A first argument in favour of the role of NT as an endogenous antipsychotic-like compound is that animal studies have shown quite convincingly that injections of NT in the VTA or in projection areas such as the nucleus accumbens cause effects that are similar to those of single administration of antipsychotic drugs. For example, when injected in the VTA, both NT and antipsychotic drugs increase the firing rate of DA neurons.103,183 Similarly, both NT (indirectly) and antipsychotics (directly) inhibit D2R function when injected in the nucleus accumbens. 114,116–119,121,122,124,125 At the behavioural level, both agents cause sedative-like effects and muscle relaxation, as well as hypothermia and vacuous chewing movements (for review, see Dobner et al184 and Kinkead and Nemeroff185). Dissimilarities have also been reported, so it would be fair to conclude that the match is not perfect. However, some of the reported differences could be dose related. For example, at low doses NT may decrease D2 receptor function, thus leading to a partial disinhibition of DA signalling. However, at higher doses, NT apparently has additional effects that are unrelated to a decrease in D2 receptor function. For example, it directly depolarizes DA neurons by activating cationic channels, an effect that antipsychotic drugs do not have.186
A second and arguably the most direct argument for a role of NT in schizophrenia is that a number of published studies, all of them emanating from Nemeroff and collaborators, report that cerebrospinal fluid (CSF) NT levels are lower in a subgroup of subjects with schizophrenia than in controls.187–193 What in fact has been observed is that CSF NT levels are lower in a subgroup of nonmedicated patients with schizophrenia who are most severely affected, with an inverse correlation between symptom severity and NT levels. In addition, in some of the studies, a partial normalization of CSF NT levels was noted in response to antipsychotic drug treatment, although there was no correlation between recovery of NT levels and extent of improvement of symptoms.189 It should also be noted that in one postmortem study, an increase in cortical NT levels was detected in patients with schizophrenia.194 Although the finding of reduced CSF NT levels appears to be a reproducible observation, no reports replicating this work have yet to come from other laboratories. The results have to be interpreted with caution. First, all the data come from CSF measurements, and the relation between CSF and brain NT levels is unclear, in part because part of the signal could reflect spinal cord levels of NT. Second, the finding of reduced CSF NT levels is limited to a subgroup of patients; group comparisons have failed to find an overall decrease in CSF NT levels when all patients were considered. Finally, although the hypothesis of a link between compromised brain NT and schizophrenia is attractive, an alternative hypothesis is that NT levels are lower in some nonspecific manner in the most severely affected nonmedicated patients because of a generalized decrease in cognitive activity. If this is true, then perhaps the CSF and brain levels of a number of markers of neuronal activity will be found to be lower in the most severely affected patients. Antipsychotic drug treatment would then act indirectly to increase NT levels by promoting partial recovery of cognitive activity by reducing positive symptoms.
But then again, the possibility that antipsychotic drugs act at least in part by restoring brain NT levels is not unreasonable, because most antipsychotic drugs have been reported to increase the expression of NT mRNA in the ventral and dorsal striatum195–198 (but see Mijnster et al199). If this hypothesis is correct, then one would expect that NT receptor antagonists could actually have negative effects on the symptoms of patients with schizophrenia. Although NT receptor antagonists have been explored for many years by pharmaceutical companies, to date, the results of only 1 clinical trial reporting the effect of an NT receptor antagonist (SR48692) on schizophrenia have been published. In that report, no significant effect of the antagonist was found.200 On the basis of the original hypothesis of an NT deficit in schizophrenia, some have proposed that NT receptor agonists hold more promise as potential antipsychotic drugs.201–204 A limited number of such agonists have been developed, and some are reportedly able to cross the blood–brain barrier.205–209 No clinical trial using such agonists in the treatment of schizophrenia has yet been reported, but animal studies have shown that NT or some of these systemically active agonists can display an antipsychotic-like profile.201–203,205,209–214 For example, a number of animal studies, many of which have studied the pre-pulse inhibition (PPI) of the startle reflex as a model of sensorimotor gating deficits in schizophrenia, have confirmed possible links between NT and DA in such schizophrenia-relevant paradigms. Injection of NT in the nucleus accumbens, but not in the VTA, reduces the ability of amphetamine to perturb PPI in the rat55,202,215 (but see Feifel et al216). This observation is compatible with the idea stated above that NT acts in the DA target structures to reduce DA signalling. In addition, a centrally active NT agonist administered subcutaneously produced a similar effect55,217 (but see Rompre218 for a psychotimulant-like effect of centrally administered NT, suggesting that at certain doses NT could also act to enhance DA-dependent behaviours). Although not tested in a PPI paradigm, a recently developed NTS1 ligand, KH28, was reported to reduce amphetamine-induced hyperlocomotion.214 Compatible with the idea that a deficit in NT could influence PPI, the NT receptor antagonist SR142948A prevented the ability of the antipsychotic haloperidol to reduce isolation-rearing-induced perturbation of PPI.219 Finally, PPI is disrupted in NT-knockout mice.220 Such results, though far from proving a link between NT and schizophrenia, nonetheless can be taken as arguing in favour of the biological plausibility of this hypothesis.
Beyond clinical trials with NT agonists, it will not be easy to pursue further in human patients the hypothesis of a link between NT and schizophrenia. There is currently no approach to measuring brain NT levels in living humans, and no association between schizophrenia and mutations or polymorphisms in NT or NT receptor genes has been reported.221
Implication of NT in drug abuse?
A possible implication of NT in drug abuse has been studied for close to 25 years.222 The primary drive for this hypothesis is based on the similarities between some of the effects of NT and of psychostimulant drugs. This work has been somewhat controversial, in large part because the effect of NT depends on many parameters such as the dose used, the injection procedure and localization, as well as the experimental model or species used. As mentioned in previous sections, injection of NT in the nucleus accumbens tends to reduce some behavioural effects of psychostimulant drugs. However, the psychostimulant-like actions of NT occur when the peptide is injected in the VTA.223 For example, intra-VTA injections of NT in the rat increase locomotor activity and elevate DA release in the nucleus accumbens.223–227 This is compatible with the localization of excitatory NT receptors on VTA DA neurons that give rise to the mesocorticolimbic pathway,228 as mentioned previously. NT could thus produce psychostimulant-like effects when injected in the VTA simply by enhancing the firing of DA neurons, leading to elevations of extracellular DA in the nucleus accumbens. NT can also increase the firing of substantia nigra DA neurons104 and cause an elevation of glutamate levels in this structure.128,157
Moreover, NT presents other psychostimulant characteristics. When injected in the rat VTA, NT acts as a primary reinforcer in the conditioned place preference paradigm229 and can be self-administered.230 In rats, NT potentiates brain-stimulation reward.231–234 When injected intracerebroventricularly, NT also sensitizes rats to the locomotor-activating effects of amphetamine235 and increases the sensitivity of mice to ethanol.236,237 Moreover, systemic administration of the NT receptor antagonists SR48692 and SR142948A reduces sensitization to amphetamine, suggesting that endogenous NT could play some role in this drug-induced long-term plasticity. 238–240 Whether NT specifically in the VTA is critically involved is less clear. In fact, psychostimulants such as cocaine and amphetamine cause an increase in NT mRNA expression in the striatum and the nucleus accumbens,241–244 areas where NT has been shown to decrease the effects of psychostimulants. Interestingly, an increase in NT immunoreactivity after methamphetamine or cocaine administration has also been reported in the substantia nigra,245,246 but no data are available for the VTA. Amphetamine and metamphetamine also increase NT levels in the medial prefrontal cortex.247 Finally, it should be noted that rhesus monkeys can reportedly not be trained to self-administer the centrally active NT receptor agonist NT69L intracerebroventricularly.248 In addition, NT reduces bar pressing for food rewards.249 Together, these results nonetheless suggest that NT may play a role in the effects of psychostimulants, but the mechanism involved is unclear. If the major cellular mechanism of the psychostimulant effect of NT is increased DA neuron firing, an open question is the specific mechanism involved. As stated previously, NT can activate cationic channels and inhibit potassium channels, thus directly depolarizing DA neurons. This could perhaps be sufficient to explain the increase in firing rate. However, inhibition of somatodendritic D2 autoreceptors by NT could also be involved. Experiments directly addressing this issue are required.
A final and obviously critical question is whether NT is involved in direct drug effects or drug addiction mechanisms in humans. Currently, very few data have been published. In one study, no association was found between a polymorphism in the proneurotensin gene and alcohol dependence in a Finnish population.250 In summary, although accumulating animal research argues in favour of the implication of NT in drug abuse mechanisms, essentially no data are available in humans. The possibility of using NT receptor ligands as possible pharmacological tools to treat drug addiction205 can thus be considered promising but perhaps a bit premature.
Implication of NT in Parkinson’s disease?
Parkinson’s disease (PD) results from a progressive loss of nigrostriatal DAergic neurons. The decrease in striatal DAergic innervation due to this loss is responsible for motor disturbances characteristic of the disease such as akinesia, muscular rigidity and tremor. The close relation between NT and the DAergic system suggests that NT could be associated with PD.
Numerous studies have tried to determine whether PD is associated with changes in the neurotensinergic system. Biochemical and histological studies of brain tissues from patients who died from PD showed a decrease in the number of NT-binding sites in the substantia nigra, VTA, caudate nucleus, putamen and globus pallidus compared with healthy subjects.251–254 Using in situ hybridization, it was also possible to show more specifically that NTS1 mRNA was decreased in the substantia nigra of subjects with parkinsonism.255 PD can be partly mimicked in animal models by destroying the nigrostriatal pathway using toxins such as 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) or 6-hydroxydopamine (6-OHDA). In these PD animal models, there is a decrease in the number of NT-binding sites in the striatum and substantia nigra.228,256–258 In the substantia nigra, the decrease in the number of NT-binding sites is surely the result of the loss of DAergic neurons that express NT receptors. However, the interpretation of the decrease in NT-binding sites in the striatum is not that straightforward. Indeed, contradictory results concerning the exact localization (presynaptic or postsynaptic) of NT receptors have been obtained in the striatum. In some studies, it was reported that almost all NT receptors within the striatum were located on DAergic terminals,259,260 whereas others reported that NT receptors were mostly expressed by striatal intrinsic neurons.29,256,261 Another study showed that the decrease in NT receptors in the monkey striatum treated with MPTP is smaller than the decrease in DA concentration, suggesting only a partial localization of NT receptors on nigrostriatal projections.257 The same result was obtained comparing the presence of NT receptors and the DA content in postmortem brain tissues from patients with PD.251,254 The decrease in striatal NT-binding sites may not be only due to the loss of NT receptors on DAergic neurons but could also be the result of an effect on striatal neurons themselves. More anatomical studies should be done to determine whether NT receptors expressed by striatal medium spiny neurons are specifically decreased in PD.
In addition to the quantification of NT-binding sites, the concentration of tissue NT has also been measured in order to determine changes induced by PD on the neurotensinergic system. It has been shown that plasma NT concentration is higher in patients with parkinsonism compared with control patients and also higher in untreated patients compared with levodopa-treated patients.262 Studies of postmortem brains further showed that NT levels remain unchanged in PD compared with healthy subjects in the caudate nucleus, putamen and globus pallidus, while they were higher in the substantia nigra compacta and reticulata263,264 and decreased in the hippocampus. 264 Whether the increase in NT concentration in the substantia nigra is causally related to PD or whether it results indirectly from the loss of DA neurons or from drug therapy is not easy to determine. Indeed, because most patients with PD are subjected to drug therapy, it is difficult to evaluate plasma NT concentration in nonmedicated patients. However, it is interesting to compare PD to incidental Lewy body disease. The presence of Lewy bodies in the brain is correlated with substantia nigra cell death, even if the decrease is smaller than in PD,265,266 and is considered to represent the PD presymptomatic phase.267 These patients do not show any pathological symptoms and so do not receive medication. Interestingly, there is an increase in NT content in the postmortem brain of patients with Lewy body disease, even though the effect is not as large as in PD.268 At the very least, this finding suggests that the increase in NT in PD is probably not secondary to pharmacotherapy.
NT levels were also measured in PD animal models and, surprisingly, the opposite results have been obtained. In 6-OHDA-lesioned rats, NT-immunoreactivity is increased in the striatum and globus pallidus, whereas no change occurs in the substantia nigra.269,270 In MPTP-lesioned monkeys, no change in NT concentration was found in the striatum.271 In this same model, a previous study did not show any change in NT in the substantia nigra and striatum.272
Although the relation between NT and PD has not been fully elucidated, one can speculate that the increase in NT levels observed in the human brain with PD is the result of an adaptive mechanism in reaction to the loss of NT receptors and DAergic neurons. This increase in NT concentration could represent an attempt of the system to increase DAergic drive by stimulating the remaining NT receptors on DAergic neurons. Within this context, an obvious question is whether NT agonists could be useful as adjunctive treatment of PD. Some studies have indeed shown that exogenous NT can display antiparkinsonian properties in 6-OHDA-lesioned animals. In fact, intracerebroventricular administration of NT or some of its analogues results in attenuation of muscular rigidity and tremors observed in these animals.273,274 In addition, in 6-OHDA-lesioned rats, the NT agonist NT-69L has an antiparkinson-like effect, because it blocks rotating behaviour induced by d-amphetamine and apomorphine.275 The exact mechanism of this NT-induced antiparkinson-like effect has not yet been determined. Based on the previously mentioned ability of NT to decrease D2R receptor function in the striatum, the use of NT receptor antagonists in the treatment of PD has also been considered. Administration of the NTS1 antagonist SR48692 has been shown to enhance striatal DAergic transmission.125 However, a clinical trial with SR48692 in patients with PD showed no improvement of symptoms.276
In conclusion, although no data are currently available to support a direct, causal role of NT in PD, some interesting correlative evidence has been obtained and some promising recent studies on animal models support the potential role of NT agonists in the symptomatic treatment of PD.
Implication of NT in pain mechanisms?
The implication of NT in analgesia has been studied for many years.277 A high density of NT receptors is present throughout the PAG and the rostral ventromedial medulla, 2 structures implicated in descending nociceptive circuits. 52,151,278,279 NT has analgesic effects that are naloxone independent and consequently not dependent on opioids.280 The antinociceptive effect of NT has been reported after injection of the peptide in many brain areas, as well as intracisternally, 281 intrathecally,282 intracerebroventricularly283 or directly in areas rich in NT innervation such as the amygdala, medial preoptic area, thalamus, PAG and nucleus raphe magnus.284 The location of the injection site can modify the intensity of the response to NT. Indeed, when injected in the central part of the amygdala, NT induces analgesia and increases hotplate response latency more effectively than if injected intracerebroventricularly. 285 Compatible with the possible implication of endogenous NT in pain suppression mechanisms, a microdialysis study showed that NT is endogenously released in the PAG in mice after unilateral hindpaw inflammation using Complete Freund’s Adjuvant.286
Analgesic effects of NT have been widely reported, but in most of these studies relatively high concentrations of NT were used. Studies performed using lower, perhaps more physiological, concentrations suggest that NT may actually induce hyperalgesia instead of analgesia. Such a phenomenon is observed when picomolar range concentrations (0.03–0.3 pmol/L) of NT are injected in the rostral ventromedial medulla, although injections at higher doses in the same nucleus induce analgesia.284,287–289 In addition, infusion of SR48692 in the rostral ventromedial medulla or intraperitoneal injection of this compound facilitates analgesia, therefore suggesting a hyperalgesia function for endogenous NT.287,289,290
Both NTS1 and NTS2 appear to be implicated in NT-induced analgesia. The first studies of NT receptor subtypes mediating analgesia showed that the analgesic effect of NT was mediated by a subtype of receptor that is pharmacologically distinct from the previously characterized high-affinity levocabastine-insensitive receptor and low-affinity levocabastine-sensitive receptors.291,292 Different results were obtained when suitable antagonists for the different receptors became available. Indeed, SR48692, a relatively selective antagonist of NTS1 when used at low doses, had no effect on NT-induced analgesia, which suggested that another NT receptor subtype, presumably NTS2, was responsible for the phenomenon.293 A subsequent study showed that levocabastine, an NTS2 agonist, displays an analgesic effect.294 However, another study showed that SR48692 inhibits NT-induced analgesia with complex multiphasic dose–response characteristics,289 suggesting the implication of more than one NT receptor subtype. A recent study also argues for the implication of NTS1, in addition to NTS2, in the analgesic effect of NT.69 Indeed, it was shown using the tail-flick test that, in addition to NT, the 2 NTS2 agonists levocabastine and JMV-431 both induce an antinociceptive response. However, the antinociceptive effect of NT was partly abolished by co-administration of SR48692, suggesting that, in addition to NTS2, NTS1 also plays a modest role. Using NTS1-knockout mice, it was also possible to show that this receptor indeed plays a role in at least some kinds of painful stimuli, because these mice present a defect in NT-induced analgesia in the hot-plate test,68 whereas no modification occurs in another nociceptive test, such as the phenyl-p-benzoquinone (PBQ)-induced writhings.66 Concerning hyperalgesia, it has been shown that endogenous NT facilitates visceral pain response, because in NT-knockout mice and in SR48692-treated rats there is attenuation of visceral nociception.290 The fact that both NTS1 and NTS2 are implicated in the central role of NT in pain control is compatible with the fact that both receptors are expressed in the PAG and raphe nuclei.72,151
Taken together, the published data are thus compatible with a role of NT in pain mechanisms. Further evaluation of the possible use of NT agonists or antagonists as analgesic compounds thus seems warranted. Compatible with this, the NT analogue NT69L has been reported to cause a rapid and persistent antinociceptive effect as determined by the hot-plate test.295
Possible implication of NT in other diseases
NT is widely distributed in the brain and is known to affect many neurotransmitter systems. Growing evidence suggests that NT is associated with different pathologies in addition to those directly related to the DAergic system discussed up to now.
Implication of NT in the central control of blood pressure
Hypotension was the first reported physiological effect of NT.1 A possible role of this peptide in hypertension would thus not be surprising. In anesthetized rats, NT injected intracerebroventricularly induces hypotension, whereas a smaller decrease in blood pressure is observed in nonanesthetized rats.296,297 The NT receptor antagonist SR142948A is more effective than SR48692 in blocking this effect.102 The nucleus of the solitary tract plays an important role in the central control of arterial pressure, because it receives signals from baroreceptors and chemoreceptors of the cardiovascular system. NT injected into the nucleus of the solitary tract causes a decrease in blood pressure.298,299 NT69, an NT receptor agonist that crosses the blood–brain barrier, induces hypotension. 248 The opposite results were reported with conscious rats, in which an intracerebroventricular injection of NT induces hypertension.300–302 The results obtained in these different studies are thus conflicting, and the cause of this discrepancy remains unknown. However, we can speculate that differences in the site of injection or in anesthesia may account for this. Nonetheless, the results presented here are clearly compatible with some role for NT in central regulation of blood pressure. In addition to these results obtained using NT injections, it has been shown that in spontaneously hypertensive rats, there is a decrease in NT concentrations in many brain areas implicated in blood pressure regulation, such as the hypothalamus, medulla oblongata, pons, pituitary and spinal cord.303 Interestingly, suprachiasmatic nucleus NT contents are greatly decreased in hypertensive patients compared with controls.304 The use of NT receptor knockout mice would perhaps help to clarify the implication of NT in arterial pressure, but changes in blood pressure have apparently not yet been evaluated in such animals.
NT and eating disorders
Food intake is controlled in part centrally by the hypothalamus, which is the site of integration of multiple signals such as hormones, neurotransmitters and peptides. Among them, NT is known to reduce food intake. Indeed, NT injected in the VTA or in the ventromedial part of the hypothamalus induces a decrease in food intake.305,306 There are also changes in NT expression in animals with eating disorders. Indeed, NT expression is increased in the hypothalamus of anorexic animals. 307 NT interacts with other food intake regulators such as leptin, a hormone produced by adipose tissues that reduces food intake. Administration of leptin increases hypothalamic NT expression in healthy rats308 as well as in hypothalamic cell lines in vitro.309 In addition, the NTS1 antagonist SR48692 appears to reverse leptin’s action on satiety.310 The expression of NT is also downregulated in animal models lacking leptin.311 Other evidence for a role of NT in food intake comes from results obtained with NTS1-deficient mice (NTS1−/−), which show a small increase in body weight compared with control animals.66 Pharmacological studies also support a role of NT in body weight homeostasis. The NT analogue NT69L reduces body weight and food intake in healthy and obese rats.312 These results are promising with regard to the possible use of NT ligands as therapeutic drugs in eating disorders.
NT and cancer
NT expression arises early in embryonic development, and it has been suggested that it could thus play a role in cell cycle regulation.313,314 Compatible with this hypothesis, NT has been associated with the progression and differentiation of tumours in the periphery and central nervous systems.315 The implication of NT in cancer has been studied extensively in the periphery, where it has been shown that the size of some colon or lung tumours increases in the presence of NT316,317 and becomes smaller in the presence of the NTS1 antagonist SR48692.317,318 The presence of NTS1 has been reported in human tumours of the ovary, pancreas and prostate and Ewing’s sarcomas, as well as centrally in meningiomas and astrocytomas. 315,319–321 The transcription of the NTS1 gene is also apparently increased in human colonic adenomas.322 In dividing human neuroblastoma (CHP 212) and murine neuroblastoma (N1E-115) cells, it has been reported that during prolonged NT agonist stimulation, NTS1 receptors are recycled to the cell surface, thus allowing constant cell sensitization and long-term activation of mitogen-activated protein kinases p42 and p44,323 which is compatible with a potential role in oncogenic regulation.
A possible implication of NTS3 in cancer has also been explored. Indeed, NTS3 and NTS1 are present in almost all cancer cells from the prostate, pancreas and colon.58,93 However, no data have yet shown the presence of NTS3 in central tumours. Little is known about the signalling pathways activated by NTS3, but the receptor is present inside the cell in close proximity to the nucleus.95 This characteristic gives NTS3 a good profile for being a candidate in mediating NT-induced proliferative signals. Considering the strong evidence in favour of a role of NT in peripheral tumours, it would seem that additional efforts should be directed toward a better comprehension of the role of NT in cancers of the CNS.
NT and neurodegenerative disorders and stroke
There is currently little evidence for a role of endogenous NT in neurodegenerative disorders or stroke. A possible implication of NT in neurodegenerative disorders is nonetheless suggested by the finding that in the suprachiasmatic nucleus of patients with Alzheimer’s disease, there is a significant decrease in NT-containing neurons.324 Because of the lack of conclusive evidence, it is nonetheless difficult to know whether NT can directly contribute to neurodegenerative diseases. However, NT enhances glutamate-induced excitotoxicity through NTS1 activation, as suggested by the blockade of this effect with SR48692.161 In sharp contrast, others have suggested that NT reduces neuronal death and infarct volume in experimental ischemia models.325,326 This protective effect of NT could be partly explained by the ability of this peptide to induce hypothermia, a condition that is well known to be neuroprotective against ischemia-induced central infarcts.327
NT and inflammation
Findings both in and outside the nervous systems have long supported a role of NT in inflammation, acting as a proinflammatory cytokine. It was first described that peripheral NT elicits inflammatory symptoms such as vasodilatation, enhanced vascular permeability, mast cell degranulation, and enhancement of directional migration and phagocytosis of neutrophils.328 NT was later shown to enhance the production of interleukin-1 (IL-1) by activated alveolar macrophages.329 In the gastrointestinal tract, a tissue rich in NTS1, NT has been involved in the pathophysiology of colonic inflammation.330 NT is also known to stimulate IL-8 secretion and IL-8 gene expression in human colonocyte cells.331 Although NT is usually thought to be proinflammatory, an opposite effect has been recently described in chronic inflammatory damage induced by dextran sodium sulfate in mice. In this model, NTS1 activation has been shown to stimulate intestinal wound healing through a cyclooxygenase (COX)-2 dependent pathway.332
The first evidence for the involvement of NT in immune responses in the CNS was provided by the finding that the endotoxin lipopolysaccharide is able to induce the expression of NT mRNA in corticotropin-releasing hormone neurons of the paraventricular nucleus of the hypothalamus.333 On the basis of this work, it has been speculated that NT may be released in the pituitary portal blood to trigger pituitary responses associated with mobilization of the immune system. More recently, it has been shown that NT also induces expression of several cytokines/chemokines including IL-1 and macrophage inflammatory protein (MIP)-2 in microglia cells, which are otherwise known to express only the NTS3 receptor, 334 thus supporting the involvement of NTS3 in CNS inflammation. Finally, it is interesting to note that descending inhibition and facilitation during peripheral inflammation are due in part to inflammation-induced changes in the rostral ventromedial medulla that involve NT and glutamate receptors. 335 In spite of the scarce evidence for a role for NT in CNS inflammation, speculation has arisen about the possibility that an NT proinflammatory stimulus acting through microglia cells could contribute to brain inflammation in degenerative diseases such as PD and Alzheimer’s diseases.
Future challenges and conclusions
Together, the results presented in this review demonstrate that although endogenous NT has not been shown conclusively to be causally involved in any one CNS disorder, there is a trail of evidence arguing for its implication in many. Among those, drug dependence, pain and cancer appear to be the most promising. Future work using genetically modified animals, gene downregulation strategies and new centrally active pharmacological ligands should continue to advance our understanding of the role of NT in health and disease.
Footnotes
Medical subject headings: central nervous system; dopamine; neurotensin; Parkinson disease; pathophysiology; schizophrenia.
Competing interests: None declared.
Contributors: All authors contributed to the conception and design of the review, analyzed the data, wrote and reviewed the article, and gave final approval for publication.
- Received December 1, 2005.
- Revision received January 26, 2006.
- Revision received January 30, 2006.
- Accepted January 30, 2006.