Abstract
Background: Although the prevalence of posttraumatic stress disorder (PTSD) is twice as high in women as it is in men, the role of estrogen in the risk for PTSD is not well understood. Deficits in fear inhibition and impaired safety signal learning may be biomarkers for PTSD. We examined menstrual cycle phase and serum estradiol levels in naturally cycling women while they were undergoing a novel conditioned inhibition procedure that measured their ability to discriminate between cues representing danger versus safety and to inhibit fear in the presence of safety cues.
Methods: Sample 1 included healthy participants in whom we compared inhibition of fear-potentiated startle during the follicular (lower estrogen) and luteal (higher estrogen) phases of the menstrual cycle. We used the same paradigm in a traumatized clinical population (sample 2) in whom we compared low versus high estradiol levels.
Results: In both samples, we found that lower estrogen in cycling women was associated with impaired fear inhibition.
Limitations: In the clinical sample, the low estradiol group was on average older than the high estradiol group owing to the random recruitment approach; we did not exclude participants based on hormonal status or menopause.
Conclusion: Our results suggest that the lower estrogen state during normal menstrual cycling may contribute to risk for anxiety disorders through dysregulated fear responses.
Introduction
Posttraumatic stress disorder (PTSD) is a severe anxiety disorder that may develop after exposure to a traumatic event. Women have a 2-fold greater risk for PTSD than men,1–4 leading to substantial health disparities.5 However, little is known about the influence of female gonadal hormones on PTSD etiology. There is evidence to suggest that estrogen may have a protective function in anxiety regulation.6 Studies using rodent models of anxiety have shown that females in the proestrous phase of their cycle (marked by high estrogen levels) show fewer fear- and anxiety-related behaviours than females in the metestrous or diestrous phases (lower estrogen levels).7–10 Women are more likely to report symptoms of depression and anxiety during premenstrual, postpartum and perimenopausal periods, when estrogen levels are low.11–16 Moreover, neuroimaging studies show greater activation of neural networks involved in fear when women are scanned during the early follicular phase of their menstrual cycle (low estrogen levels) than when they are scanned midcycle (high estrogen levels).17,18 Hence, natural fluctuations of estrogen across the reproductive cycle may factor into the disproportionate incidence of PTSD in women. However, few studies have examined the influence of menstrual cycle phase and estrogen levels in laboratory-based models that explicitly probe for PTSD phenotypes. The objective of the present study was to examine the effects of estrogen on a biomarker of PTSD using translational methods.
There is a growing literature showing that the inability to inhibit conditioned fear is a biomarker of PTSD.19–22 In the Pavlovian fear extinction model of fear inhibition, a conditioned stimulus (CS) that was previously paired with an aversive unconditioned stimulus (US) is repeatedly presented in the absence of the US, which leads to a reduction of conditioned fear responding.21 Previous research has shown that healthy women with low estrogen levels have deficits in fear extinction recall.23 We recently demonstrated in women with PTSD that low estrogen, but not high estrogen, is associated with fear extinction deficits.24 Understanding the influence of estrogen on fear inhibition could shed light on mechanisms underlying sex differences in psychopathology.
Our laboratory developed a conditioned inhibition paradigm that allows for the independent analysis of fear expression and fear inhibition, and can be used in clinical populations to determine which of the 2 processes is dysregulated.25 The procedure, referred to as conditional discrimination (abbreviated AX+/BX−), involves reinforcing stimulus X conditional upon the presence of either stimulus A or B. Stimulus A increases fear with training as the participant learns that A and X presented together predict the aversive unconditioned stimulus (US). Stimulus B becomes inhibitory in that B presented with X predicts the absence of the US (i.e., B is a safety signal). The presentation of A and B together (AB) results in a reduced fear response to A because B transfers its inhibitory property to A. Thus, the AB trials are referred to as conditioned inhibition test trials and indicate the ability to transfer safety to a danger cue. Consistent with these predictions, we have found greater fear expression in the presence of AX versus BX and in the presence of AX versus AB in healthy individuals.25 However, participants with high levels of PTSD symptoms do not show fear inhibition in this paradigm (i.e., they are unable to suppress fear responses during AB trials).26,27
The purpose of the present study was to use this conditioned inhibition paradigm to investigate the influence of natural estrogen fluctuations on fear inhibition. To this end, we used the same experimental paradigm in 2 participant samples, applying a translational approach from preclinical to clinical populations. The first sample included healthy participants tested at different points in their menstrual cycles, whereas the second sample included women recruited from an inner-city population shown to have high rates of trauma exposure and PTSD incidence,28 divided into high and low estrogen groups based on serum samples. We hypothesized that women with low estrogen levels would show deficits in fear inhibition relative to those with high estrogen levels.
Methods
Sample 1: healthy community sample
Participants
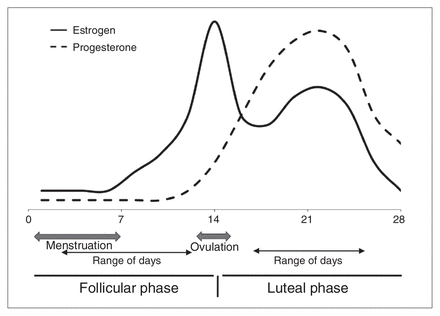
All women in this sample were healthy volunteers from the Emory and Atlanta VA Medical Center community who responded to a research flyer. The women were scheduled to participate relative to their current menstrual cycle (i.e., they were asked to call the research study on the first day of their cycle and were scheduled either for days 2–12 or days 17–25 of their cycle (Fig. 1). All women provided informed consent, and the Emory University Institutional Review Board and the Atlanta VAMC approved the study protocol.
Typical estrogen and progesterone fluctuations across the 28-day human menstrual cycle. The solid line represents estrogen levels, which peak at ovulation and remain relatively elevated during the luteal phase. The dashed line represents progesterone level, which peaks after ovulation and is high during the luteal phase. The data for this study were collected 2–12 days since the last menstrual period for the follicular group, and 17–25 days since the last menstrual period for the luteal group.
To be included, women had to be between 18 and 55 years old and have regular menstrual cycles. We excluded those with a history of Axis I disorders diagnosed using the Structured Clinical Interview for DSM-IV,29 head injury or neurologic disorder. We also excluded women currently using oral contraceptives or other hormonal therapy, women who were in menopause or pregnant and those who had a positive urine drug screen. We screened for hearing impairment using an audiometer (Grason-Stadler, Model GS1710) with a threshold of 30 dB[A]SPL at frequencies ranging from 250 to 4000 Hz.
Experimental design
Fear-potentiated startle and inhibition of fear-potentiated startle were assessed using the AX+/BX− conditional discrimination paradigm.25 Each CS was a compound of 2 lights presented serially. The AX+ compound served as the reinforced stimulus to produce fear (CS+), and the BX− compound served as the nonreinforced stimulus to produce safety signal learning (CS−). Each compound CS had 1 distinct cue (A or B) and 1 common cue (X). The fear inhibition test stimulus was a compound of the previously conditioned A and B cues to determine transfer of inhibition (by B) to the fear response to A.30 The A, B and X stimuli were green, purple or blue light panels 5 cm × 10 cm in size (counterbalanced colour assignment across participants) mounted on the wall 1 m from the participant’s seat. The aversive US was a 250 ms airblast with an intensity of 140 psi directed to the larynx. The startle probe was a 108 dB[A]SPL, 40 ms burst of broadband noise with near instantaneous rise time, delivered through headphones.
The test session began with a habituation phase consisting of 6 noise alone (NA) startle probes to reduce initial startle reactivity. Immediately after habituation, participants underwent the conditioning phase that included 6 NA trials, 6 AX+ trials paired with the US, and 6 nonreinforced BX− trials. The testing phase seamlessly followed the conditioning phase and consisted of 3 NA trials and 3 AB trials. In all phases of the experiment, intertrial intervals were of randomized duration, ranging from 9 to 22 seconds.
Startle response measurements
We measured the acoustic startle response (eyeblink component) via electromyography (EMG) of the right orbicularis oculi muscle. Two 5 mm Ag/AgCl electrodes were positioned about 1 cm under the pupil and 1 cm below the lateral canthus. All resistances were less than 6 kg-ohms. Electromyograph activity was amplified and digitized using a startle response monitoring system (SR-LAB; San Diego Instruments). The EMG signal was band-pass filtered at frequencies of 30 and 1000 Hz. We scored peak startle amplitude 20–120 ms after the startle probe onset.
Contingency awareness measurement
Similar to previously published methods,31 we used a 3-button response keypad (SuperLab; Cedrus Corporation) in the startle sessions in coordination with the San Diego Instruments startle response system to collect trial-by-trial ratings of US expectancy. Participants were instructed to respond on each trial by pressing 1 of 3 buttons: the “+” key when they expected a CS to be followed by the airblast, the “−“ key when they did not expect the airblast and the “0” key when they were uncertain of what to expect.
Data analysis
The group variables in the analyses were the menstrual cycle phase during testing: follicular and luteal. We assessed startle reactivity for the NA trials by averaging the startle response to the probe in the absence of a CS. We assessed startle potentiation by comparing startle magnitude on the NA trials and the AX+ trials using a 2-way repeated-measures analysis of variance (RM-ANOVA) with group (2 levels: follicular, luteal) as the between-group factor and trial type (2 levels: NA, AX+) as the within-subjects factor.
To index fear-potentiated startle, we calculated percent potentiation for each CS type to account for individual differences in startle magnitude and startle habituation. This value was derived as follows: percent startle potentiation = 100 × (startle magnitude during CS trials – NA startle) ÷ (NA startle). We used an RM-ANOVA with trial type as a within-subjects factor (3 levels: AX+, BX−, AB) and group as a between-group factor. We included 2 contrasts in the model. First, differential conditioning compared the percent startle potentiation to AX+ and BX− during the last block of conditioning. Second, fear inhibition compared percent startle potentiation during the last block of AX+ and the test block of AB. Contingency awareness was assessed using the same RM-ANOVA, but with US expectancy ratings as the dependent variable. In all RM-ANOVAs we used the Greenhouse-Geisser statistic with an α level of 0.05. Significant between-group differences were followed up by post hoc Tukey tests. We performed all analyses in SPSS version 17.0 for Windows.
Sample 2: traumatized clinical sample
Participants
The sample included women recruited from primary care clinics at Grady Memorial Hospital in Atlanta, Ga., which serves a primarily African-American, low socioeconomic, inner-city population.32,33 Women were excluded if they had active psychosis, bipolar disorder or any major medical illnesses, as assessed by health and physical examinations conducted by study clinicians. We also excluded women who were pregnant or hearing-impaired. Medication use was not an exclusion criterion. All participants were screened for hearing impairment using an audiometer, as described previously. Prior to their participation, all participants provided written informed consent, and the Emory University Institutional Review Board and Grady Memorial Hospital approved the study protocol.
Clinical assessment
The modified PTSD Symptom Scale (mPSS) is a psychometrically valid 17-item self-report scale assessing PTSD symptomatology over the 2 weeks before rating.34–36 It has been shown to have high internal consistency (α = 0.85).35 The categorical definition of PTSD was determined based on DSM-IV criteria.
The Traumatic Events Inventory (TEI)36 assesses lifetime history of trauma exposure and is a measure of trauma resulting from child abuse and other causes. Previously shown to have high internal consistency (α = 0.94), the TEI assesses experience and frequency of 13 types of traumatic events and has been validated in this population.32,33,36
Startle response measurements
Our startle response data acquisition equipment was upgraded between the experiments involving sample 1 and those involving sample 2. For sample 2, we measured acoustic startle responses using EMG recordings sampled at 1000 Hz and amplified using the EMG module of the BIOPAC MP150 system (Biopac) for Windows. The acquired data were filtered, rectified and smoothed in MindWare software (MindWare Technologies) and exported for statistical analyses. The response keypad was connected to the Biopac system to assess contingency awareness in a manner similar to that of sample 1.
Estrogen assays
Experienced nurses obtained fasting whole blood specimens by venipuncture between 8:00 am and 9:00 am at the Clinical Interactions Network within the Atlanta Clinical and Translational Science Institute. To obtain serum for estradiol measurement, blood was kept at room temperature for 30 minutes to allow clotting, centrifuged and then transferred to a −80º C freezer for storage until analysis. Estradiol assays were completed by the Yerkes Biomarkers Core Laboratory at Emory University using a commercially available radioimmunoassay kit (product #KE2D1; Siemens Healthcare Diagnostics). Samples whose coefficient of variation (CV) between the replicates exceeded 20% were repeated and the values averaged. The interassay CV% was 11.3% at 176.13 pg/mL and 14.56% at 1308.09 pg/mL (n = 55). The intra-assay CV% was 17.61% at 31.40 pg/mL (n = 1).
Data analysis
The group variables in the analyses were the high and low estrogen groups derived from the median split of serum estradiol (E2) levels. All RM-ANOVAs and dependent variables were identical to those in sample 1, except for the group (high E2, low E2).
Results
Sample 1: healthy community sample
Demographics
This sample included 28 women: 14 tested during the follicular phase and 14 tested during the luteal phase of their cycle. The group variables in the analyses were the menstrual cycle phase during testing: follicular and luteal. The 2 groups did not differ in age (mean 32.9, standard error of the mean [SEM] 3.0 yr in the follicular phase group v. mean 29.3, SEM 1.8 yr in the luteal phase group; F1,27 = 1.07, p = 0.31) or racial composition, (35.7% black, 50.0% white and 14.3% Asian in the follicular phase group v. 28.6% black, 71.4% white and 0% Asian in the luteal phase group; χ2 = 2.64, p = 0.27).
Fear-potentiated startle
We assessed fear-potentiated startle by comparing startle magnitude on the NA trials and the AX+ trials using a 2-way RM-ANOVA with group as the between-group factor and trial type as the within-subjects factor. This analysis revealed a significant main effect of trial type (F1,26 = 27.03, p < 0.001), but no main or interaction effect of group. Startle magnitude was significantly higher during the AX+ trials than the NA trials in the follicular phase (mean 267.29 [SEM 54.48] μV for the AX+ trials v. mean 152.00 [SEM 31.78] μV for the NA trials; F1,13 = 10.71, p = 0.006) and for the luteal phase (mean 257.12 [SEM 54.48] μV for the AX+ trials v. mean 108.02 [SEM 31.78] μV for the NA trials; F1,13 = 20.38, p = 0.001).
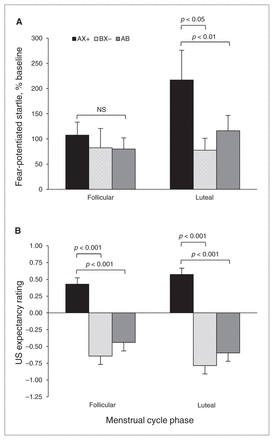
For differential conditioning, we compared AX+ and BX− trials during the last block of conditioning, and for fear inhibition we compared AX+ and AB test trials. We again used an RM-ANOVA with group as the between-group factor and trial type as the within-subjects factor. Figure 2A shows the percent startle potentiation to the different trial types across groups. The analysis showed a significant main effect of trial type (F2,52 = 5.10, p = 0.009), but no main effect of group (F1,26 = 1.44, p = 0.24) or interaction effect (F2,52 = 2.27, p = 0.11). Within-group contrasts showed that the women in the luteal phase showed significant discrimination between AX+ and BX− trials (F1,13 = 6.73, p = 0.022) and significant inhibition on AB compared with AX+ trials (F1,13 = 9.62, p = 0.008). On the other hand, women in the follicular phase showed neither discrimination nor inhibition (both F < 1.00).
Effects of menstrual cycle phase on conditioned discrimination in a healthy community sample. (A) Percent startle potentiation to the different trial types across groups. The women in the luteal phase showed significant discrimination between AX+ and BX− (p < 0.05) and significant inhibition on AB compared with AX+ trials (p < 0.01). On the other hand, women in the follicular phase showed neither discrimination nor inhibition. (B) Aversive unconditioned stimulus (US) expectancy. Women in the luteal phase showed robust discrimination between AX+ and BX− (p < 0.001) and significant inhibition on AB compared with AX+ trials. However, unlike their startle data, results of women in the follicular phase also demonstrated significant discrimination and significantly less US expectancy on the AB trials compared with AX+. AX+ > BX− = discrimination; AX+ > AB = inhibition.
Contingency awareness
To test awareness of reinforcement contingencies for the different trial types, we used the same RM-ANOVA with group as the between-group factor, trial type as the within-subjects factor and with US expectancy ratings as the dependent variable. Figure 2B shows the US expectancy ratings to the different trial types across groups. The analysis showed a significant main effect of trial type (F2,52 = 64.104, p < 0.001), but no main effect of group or interaction effect. Consistent with the startle data, the women in the luteal phase showed robust discrimination between AX+ and BX− trials (F1,13 = 131.58, p < 0.001) and significant inhibition on AB compared with AX+ trials (F1,13 = 94.87, p < 0.001). However, unlike their startle data, women in the follicular phase also demonstrated significant discrimination (F1,13 = 27.51, p < 0.001) and significantly less US expectancy on the AB trials than on the AX+ trials (F1,13 = 25.73, p < 0.001).
Sample 2: traumatized clinical sample
Demographics and clinical description
This sample included 44 women: 22 with low E2 levels (mean 6.2 [SD 2.68] pg/mL) and 22 with high E2 levels (mean 95.4 [SD 106.24] pg/mL). The low E2 group was on average older than the high E2 group (mean 48.4 [SD 10.4] yr v. mean 34.2 [SD 9.7] yr; F1,41 = 20.87, p < 0.01). However, racial composition did not differ between groups (81% black and 19% white in the low E2 group v. 85% black and 15% white in the high E2 group; χ2 = 0.17, p = 0.68). Table 1 shows the trauma history and PTSD symptoms in the high E2 and low E2 groups. Although both groups experienced high levels of trauma, with more than 70% of participants reporting multiple traumas and interpersonal trauma, only one-third of participants in each group met the criteria for PTSD. The 2 groups did not differ on these variables.
Clinical characteristics of the traumatized study sample, by estradiol levels
Fear-potentiated startle
We assessed fear-potentiated startle by comparing startle magnitude on the NA trials and the AX+ trials using a 2-way RM-ANOVA with group (low E2, high E2) as the between-group factor and trial type as the within-subjects factor. This analysis revealed a significant main effect of trial type (F1,43 = 17.97, p < 0.001), but no main or interaction effect of group. Startle magnitude was significantly higher during the AX+ trials than the NA trials in both groups (mean 80.61 [SD 17.17] μV for the AX+ trials v. mean 43.06 [SD 8.36] μV for the NA trials; F1,21 = 13.05, p = 0.002 in the low E2 group and mean 56.27 [SD 11.87] μV in the AX+ trials v. mean 35.98 [SD 7.97] μV in the NA trials; F1,21 = 5.38, p = 0.030 in the high E2 group).
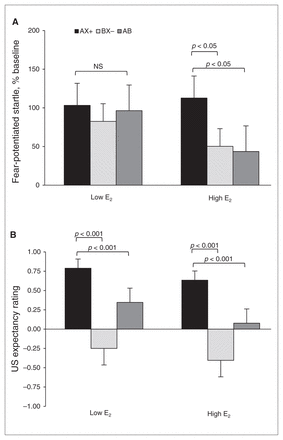
For differential conditioning, we compared AX+ and BX− during the last block of conditioning, and for fear inhibition we compared AX+ and AB test trials. We again used an RM-ANOVA with group as the between-group factor and trial type as the within-subjects factor. Figure 3A shows the percent startle potentiation for the different trial types across groups. The analysis showed a trend toward a main effect of trial type (F2,84 = 2.83, p = 0.07), but no main effect of group (F1,42 = 0.57, p = 0.46) or interaction effect (F2,84 = 1.24, p = 0.27). The contrasts revealed that women in the high E2 group showed significant discrimination between AX+ and BX− (F1,21 = 4.93, p = 0.038) and significant inhibition on AB compared with AX+ trials (F1,21 = 4.34, p = 0.050). On the other hand, women in the low E2 group showed neither discrimination (F1,21 = 1.81, p = 0.19) nor inhibition (F < 1.00).
Effects of estradiol (E2) levels on conditioned discrimination in a traumatized clinical sample. (A) Percent startle potentiation to the different trial types across groups. The women in the high E2 group showed significant discrimination between AX+ and BX− (p < 0.05) and significant inhibition on AB compared with AX+ trials. On the other hand, women in the low E2 group showed neither discrimination nor inhibition. (B) Aversive unconditioned stimulus (US) expectancy. Women in the high E2 group showed robust discrimination between AX+ and BX− (p < 0.01) and significant inhibition on AB compared with AX+ trials. However, unlike their startle data, women in the low E2 phase also demonstrated significant discrimination and inhibition (p < 0.001). AX+ > BX− = discrimination; AX+ > AB = inhibition.
Given that PTSD is associated with impaired discrimination and fear inhibition, we repeated the RM-ANOVA only in participants who did not meet criteria for PTSD (low E2n = 15, high E2n = 16). This analysis also demonstrated significant discrimination (F1,15 = 4.35, p = 0.054) and significant inhibition (F1,15 = 6.84, p = 0.020) in the high E2 group. Once those with PTSD were removed from the analysis, the low E2 group also showed discrimination between AX+ and BX− (F1,14 = 5.38, p = 0.036). However, this group still did not show inhibition of fear (F1,14 = 1.16, p = 0.30).
To control for the group difference in age, given that the low E2 group likely included menopausal women (since they were not excluded from the study), we also repeated the RM-ANOVA only in women younger than 50 years (low E2n = 7, high E2n = 21). By making this selection, the groups no longer differed in age (mean 38.3 [SD 12.4] yr in the low E2 group v. mean 34.2 [SD 9.7] yr in the high E2 group; F1,27 < 1.0). In this analysis, we again found that only women with high E2 showed significant discrimination (F1,20 = 4.70, p = 0.042) and significant inhibition (F1,20 = 4.45, p = 0.048).
Contingency awareness
To test awareness of reinforcement contingencies for the different trial types, we used the same RM-ANOVA with group as the between-group factor, trial type as the within-subjects factor and US expectancy ratings as the dependent variable. Figure 3B shows the US expectancy ratings for the different trial types across groups. The analysis showed a significant main effect of trial type (F2,76 = 23.73, p < 0.001), but no main effect of group or interaction effect. Contrasts indicated that women in the high E2 group showed robust discrimination between AX+ and BX− (F1,19 = 23.16, p < 0.001) and significant inhibition on AB compared with AX+ trials (F1,19 = 17.95, p < 0.001). However, unlike their startle data, the results of women in the low E2 group also demonstrated significant discrimination (F1,19 = 12.84, p = 0.002) and inhibition (F1,19 = 9.38, p = 0.006).
Discussion
The present study tested the ability of women with hormonally distinct menstrual cycle phases to discriminate between danger and safety cues and to inhibit fear in the presence of safety cues. Our findings showed that women in the luteal phase of their cycle demonstrated significant discrimination between AX+ and BX− trials and significant inhibition on AB compared with AX+ trials. On the other hand, women in the follicular phase showed neither discrimination nor inhibition (Fig. 2A). To address the uncertainty of self-reported menstrual cycle phases, we measured blood serum estradiol levels in a separate sample of women. In a similar pattern of results, but now with comparable levels of fear-potentiated startle in the 2 groups, women in the high estradiol group showed significant discrimination and fear inhibition, whereas women in the low estradiol group did not (Fig. 3A). These deficits were not due to unawareness of stimulus contingencies, because response pad results indicated that all experimental groups showed significantly less US expectancy on BX− and AB trials than on AX+ trials despite group differences in startle physiology (Figs. 2B and 3B).31 Our previous investigations have also revealed a discrepancy between response pad data (i.e., cognitive learning) and startle responses (i.e., psychophysiological learning),26 suggesting that the impaired inhibition of fear is not due to a learning deficit, but rather a deficit in top–down fear regulation by cortical structures.
The 2 samples of women in the present study differed in several key aspects. The first sample included only healthy volunteers who were not taking oral contraceptives and had normal menstrual cycles. The second sample was recruited from a highly traumatized urban population and included women who may not have been cycling due to menopause or use of oral contraceptives to examine the effects of estrogen in a true clinical population. We used the estrogen levels rather than the cycle information as the determining factor in this clinical sample. As mentioned previously, in both cases, low estrogen was associated with deficits in fear inhibition on the startle measure. Although in both samples US expectancy was significantly lower on AB trials than on AX+ trials, the traumatized sample showed more variability and higher levels of uncertainty in their responses (see Fig. 3B). This is likely owing to the increased age range and potentially more difficulty in the traumatized sample in attending to the experimental stimuli.
The present study confirmed that hormonal status selectively influences fear inhibition, but not fear potentiation. This is consistent with the findings of Milad and colleagues,23,37 who found that menstrual cycle phase and higher estrogen were associated with increased extinction recall, which requires inhibition of fear in the presence of a cue that was previously paired with a US and subsequently extinguished. Therefore, both extinction recall and conditioned discrimination involve learning to suppress fear in the presence of safety cues. Together, these studies support the idea of a protective function of estrogen in anxiety regulation, and emphasize the need for more research examining the influence of hormonal status on fear inhibitory processes.
While the present findings point to an influence of cycling levels of estrogen on fear inhibition in women, fluctuations in progesterone levels might also contribute to these observations (Fig. 1). Milad and colleagues23 did not find a significant effect of blood progesterone levels on extinction recall in women, although they did find progesterone effects in female rats.37 Given that progesterone has also been implicated in anxiety regulation38 and emotion processing,39 further research is needed to elucidate the precise roles of progesterone and estrogen in modulating fear inhibition in women. However, a recent study found that administration of estrogen in a rodent model rescued fear extinction deficits observed both in rodents and humans with low estrogen due to hormonal contraceptives,40 lending more support to estrogen as the hormone of action. It is important to note that cycling hormones may have very different effects on behaviour and emotion than constant levels, as might be seen during contraceptive use or hormone replacement. For example, our study found that the dynamic changes in estrogen implicated an association between low estrogen and impairments in fear inhibition; a rodent study of gonadectomized females with estrogen replacement found that estrogen resulted in less fear inhibition.41 A difficulty in investigating the effects of menstrual cycle in humans has been that women’s retrospective reports with regard to onset of cycle tend to be unreliable. Furthermore, the exact days included in the cycle definitions vary across laboratories. If late follicular phase is included, estrogen levels will be high due to ovulation, whereas the late luteal phase can include estrogen withdrawal before the next cycle. Therefore, measuring actual hormone levels in addition to cycle phase provides more information on the effects of gonadal hormones.
Given the greater prevalence of PTSD in women than men, it may seem contradictory that low, rather than high estrogen was associated with impaired fear inhibition, which has been linked to PTSD.22 Interestingly, studies comparing fear extinction in men with either high- or low-estrogen women have found that men respond similarly to women with high levels of estrogen,23 indicating that testosterone may also affect fear inhibition either directly or through aromatization to estrogen. Moreover, the association between stress and gonadal hormones in modulating risk is not well understood. Studies have found sexually dimorphic effects of cortisol on fear conditioning and extinction,42 as well as sex and menstrual cycle effects on limbic neurocircuitry.17 Despite the observed sex differences in the prevalence of PTSD, very few studies have examined the association between hormonal status in women and risk for PTSD. A notable recent study by Bryant and colleagues43 investigated whether menstrual cycle phase at the time of trauma exposure influenced symptom levels. The study found that women in the midluteal phase during trauma exposure were more likely than women at any other phase of the menstrual cycle to have intrusive memories of the event. This effect was associated specifically with flashback severity rather than PTSD symptoms in general, suggesting that cycle phase may affect memory formation, rather than general vulnerability for the disorder. As this study did not assess hormone levels, it is difficult to conclude whether flashbacks were associated with lower or higher estrogen levels. These studies underscore the complexity of the interactions between trauma and sex-specific dimorphisms in neural and endocrine function and the need for further investigation of these interactions with regard to risk for anxiety disorders.
Limitations
One the limitations of the study is the lack of availability of estrogen data in the first sample. This study was conducted as part of a larger study in healthy volunteers focusing only on fear-potentiated startle responses and did not involve the collection of blood samples. Although retrospective self-report of the menstrual cycle is highly unreliable, we minimized this issue by scheduling women prospectively relative to the first day of their cycle. To understand the influence of menstrual cycle position on vulnerability for PTSD, we also examined fear inhibition in a clinical sample whose population was previously shown to have high rates of trauma exposure and PTSD.33 Because fear inhibition has been shown to be impaired in individuals with PTSD,26 it is possible that PTSD symptoms were associated with fear inhibition deficits in the present study; however, the estradiol groups did not differ in terms of trauma history or PTSD symptoms. Furthermore, we did a separate analysis only in participants who did not meet the criteria for PTSD and found the same pattern of results in the high E2 group. In the low E2 group, deficits were again found in fear inhibition, but not in discrimination. Thus, it is unlikely that PTSD was a confounding factor in the fear inhibition deficits seen in the low E2 group. However, it is possible that having PTSD may have contributed to discrimination deficits, but only in the low E2 group. These results suggest that low levels of estrogen may be a vulnerability factor in women that is exacerbated by trauma-related psychopathology.24 Another limitation of the clinical sample was that the low E2 group was older than the high E2 group. This was due to the random recruitment approach in which we did not exclude participants based on hormonal status or menopause. However, we controlled for age differences and menopause by repeating our analysis only in women younger than 50 years — effectively eliminating group differences in age — and again replicated our original results.
Conclusion
Our findings implicate a role of estrogen on neural processes that regulate fear inhibition and suggest that low estrogen may be associated with increased risk for anxiety disorders through dysregulated fear responses. These findings may have important implications for psychiatric treatments that are sensitive to the menstrual cycle. Future studies aimed at understanding sex differences in PTSD pathogenesis should pay particular attention to the hormonal status of women.
Acknowledgements
This research was supported by the Mental Health Service, Atlanta DVA Medical Center; the STC Program Center for Behavioral Neuroscience of the National Science Foundation under Agreement No. IBN-9876754 (venture grant, PI, E. Duncan); the American Psychiatric Association/GlaxoSmithKline (PI, E. Duncan); National Institute of Mental Health Grants MH47840 (PI, M. Davis), MH071537 (PI, K.J. Ressler) and MH070129 (PI, T. Jovanovic); IRACDA grant number K12-GM000680 (PI, E.M. Glover); Howard Hughes Medical Institute (PI, K.J. Ressler); the Department of Defense/Congressionally Directed Medical Research Program Award # W81XWH-08-2-0170 (PI, S.D. Norrholm); the Atlanta Clinical Translational Science Institute; the NIH National Centers for Research Resources (M01 RR00039); and the Emory University General Clinical Research Center at Grady Hospital. We thank Allen Graham, Angelo Brown and Nineequa Blanding for their assistance with participant recruitment and data collection.
Footnotes
Competing interests: None declared for K.B. Mercer. As above for E.M. Glover, S.D. Norrholm, M. Davis, E. Duncan, B. Bradley, K.J. Ressler and T. Jovanovic.
Contributors: All authors contributed to study design, reviewed the article and approved its publication. E.M. Glover and T. Jovanovic acquired the data and wrote the article. They analyzed the data with S.D. Norrholm.
- Received July 6, 2012.
- Revision received December 20, 2012.
- Revision received February 15, 2013.
- Accepted February 20, 2013.