Abstract
Background Glutamate transmission is implicated in drug-induced behavioural sensitization and the associated long-lasting increases in mesolimbic output. Metabotropic glutamate type 5 (mGlu5) receptors might be particularly important, but most details are poorly understood.
Methods We first assessed in mice (n = 51, all male) the effects of repeated dextroamphetamine administration (2.0 mg/kg, i.p.) on locomotor activity and binding of the mGlu5 ligand [3H]ABP688. In a parallel study, in 19 stimulant-drug-naïve healthy human volunteers (14 female) we administered 3 doses of dextroamphetamine (0.3 mg/kg, p.o.) or placebo, followed by a fourth dose 2 weeks later. We measured [11C]ABP688 binding using positron emission tomography before and after the induction phase. We assessed psychomotor and behavioural sensitization using speech rate, eye blink rate and self-report. We measured the localization of mGlu5 relative to synaptic markers in mouse striatum using immunofluorescence.
Results We observed amphetamine-induced psychomotor sensitization in mice and humans. We did not see group differences in mGlu5 availability following 3 pre-challenge amphetamine doses, but group differences did develop in mice administered 5 doses. In mice and humans, individual differences in mGlu5 binding after repeated amphetamine administration were negatively correlated with the extent of behavioural sensitization. In drug-naïve mice, mGlu5 was expressed at 67% of excitatory synapses on dendrites of striatal medium spiny neurons.
Limitations Correlational results should be interpreted as suggestive because of the limited sample size. We did not assess sex differences.
Conclusion Together, these results suggest that changes in mGlu5 availability are not part of the earliest neural adaptations in stimulant-induced behavioural sensitization, but low mGlu5 binding might identify a higher propensity for sensitization.
Introduction
Altered glutamate neurotransmission has been proposed to contribute to the pathophysiology of psychiatric disorders, including addictions and schizophrenia.1,2 Repeated exposure to addictive drugs and emotionally relevant stimuli such as stress can elicit responses that become progressively larger, a process called sensitization.3–5 Sensitized psychomotor responses are commonly used to index incentive sensitization, the strengthening of the appetitive motivational properties of a stimulus or event.6 These effects are considered to be central to the aberrant salience hypotheses of addiction6,7 and psychosis.8,9 One hypothesized locus for these drug-induced changes is at cortical glutamatergic projections onto striatal medium spiny neurons that modulate responses to addictive drugs and emotionally relevant environmental stimuli.10,11
The metabotropic glutamate type 5 (mGlu5) receptor makes important contributions to the development of sensitization in male laboratory rodents. Mice lacking mGlu5 (mGlu5 knockout) do not develop behavioural sensitization to cocaine,12 and mGlu5 negative allosteric modulators diminish the development and expression of sensitization to cocaine and dextroamphetamine (Amph) in rats.13–15 Signalling through mGlu5 can also modulate drug and non-drug associative learning.16,17 In the context of drug reward, mGlu5 negative allosteric modulators also reduce self-administration of Amph and cocaine, extinction of cocaine seeking, and drug and cue-induced reinstatement of drug-seeking behaviours following extinction. 18–20 Conversely, exposure to relatively extensive cocaine administration regimens decreased mGlu5 levels in the nucleus accumbens.18,21,22 In humans, positron emission tomography (PET) studies with the specific mGlu5 allosteric ligand [11C]ABP688 identified reduced mGlu5 availability in male and female tobacco smokers23 and in cocaine-dependent men and women following a brief period (days or weeks) of abstinence. 24,25 These findings raise the possibility that mGlu5 could contribute to the altered responses to drugs and environmental stimuli that develop following a repeated drug regimen.
Based on these observations in both animal models and clinical populations, allosteric modulators of mGlu5 are currently being investigated for the treatment of addiction.26–28 Determining if and how mGlu5 availability changes during a sensitization-inducing regimen can inform our understanding of disease etiology and aid in future medication development. The first objective of this study was to test in both mice and humans whether the long-lasting sensitization of behaviourally activating effects produced by repeated exposures to the psychostimulant Amph was related to changes in mGlu5 availability. The second objective was to use immunofluorescence to localize mGlu5 at the level of the synapses of striatal medium spiny neurons. We predicted that Amph treatment would induce changes in mGlu5 availability in the striatum and prefrontal cortex of humans and mice, and that these changes would be associated with greater psychomotor activating responses to Amph.
Methods
Study populations
Mice
We used male C57BL6 mice (2 to 6 months old) for all experiments in rodents. We used tissue from 3 wild-type mice, 1 mGlu5 knockout mouse (mGlu5-KO; Jackson Laboratory; Grm5 Null/Knockout #003558), and 1 transgenic B6.Cg-Tg (Drd1a-tdTomato)6Calak/J mouse for immunofluorescence experiments. Animal housing, breeding and care were performed in accordance with the Canadian Council on Animal Care guidelines (http://ccac.ca/en_/standards/guidelines) and approved by the Facility Animal Care Committee of the Douglas Research Centre (protocols 2008–5643 and 2014–7479).
Humans
Nineteen healthy volunteers completed the study (14 women, 5 men). Participants were selected for high novelty-seeking traits, defined as scores ≥ 20 on the Tridimensional Personality Questionnaire novelty-seeking scale,29 given a population mean of 15 ± 5.30 Novelty-seeking is associated with an increased tendency to develop sensitization in rodents and humans,3,31 and this criterion was expected to increase the likelihood of observing sensitization while using a limited drug regimen. Detailed inclusion/exclusion criteria and screening procedures are described in Appendix 1, available at jpn.ca/190162-a1.
Assessment of behaviour
Amph sensitization in mice
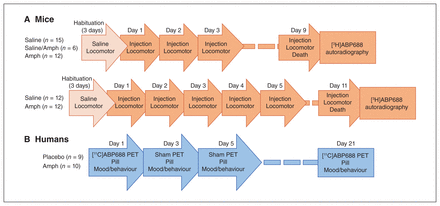
We used 2 Amph sensitization regimens in mice (Fig. 1A): a 3-dose pretreatment regimen intended to reflect the relatively mild sensitization effects expected in humans; and a 5-dose pretreatment regimen more similar to those found in the existing literature.4 We treated the mice with 2 mg/kg Amph or saline (i.p.) daily for 3 days (n = 15 for saline, n = 12 for Amph) or 5 days (n = 12 per group), as previously described. 11 A 6-day latency followed the induction phase. On day 7 post-induction, mice were given the same treatment they had received previously (i.e., saline or 2 mg/kg Amph). A fifth group of mice (n = 6) received 3 injections of saline pretreatment and a challenge dose of Amph. All procedures were carried out during the light cycle, and we measured locomotor activity after each dose (Appendix 1). Animals were killed immediately after the final behavioural session, between 1400 and 1600 h. Their brains were collected, frozen in 2-methylbutane (−30°C) and stored at −80°C.
Testing schedule for dextroamphetamine (Amph) sensitization in mice and humans. Each subject/participant received the treatment described on the left in every session, with the exception of the Saline/Amph mouse group, which received saline pretreatment (3 doses) and Amph challenge on day 9. See Appendix 1 for detailed descriptions of the experimental sessions.
Amph sensitization in humans
In a randomized, double-blind study, we administered a sensitizing regimen of Amph (Dexedrine) or visually identical placebo to healthy volunteers according to the schedule in Fig. 1B. In 3 consecutive sessions 48 hours apart (days 1, 3, and 5), participants received Amph or placebo. We measured subjective responses, eye-blink rate, speech rate,32 motor response (Actiwatch AW-16, Philips Respironics) and physiologic responses to the drug for 2.5 hours after administration (Appendix 1). Three weeks after the first dose, participants received a final challenge dose or placebo (day 21) — the same treatment they received in the initiation phase. This drug treatment regimen has been reported by our laboratory and others to induce sensitization of striatal dopamine release and behaviour in humans.3,33 Sensitization was operationally defined as a greater response to the drug on day 21 compared to day 1. Based on previous work, we expected to observe sensitization in motor measures and on self-reported measures of activation and energy levels (visual analogue scales [VAS] measuring alertness, mind racing and energy) but not on measures of euphoria/high (VAS measuring excitement, rush, high, euphoria; Addiction Research Center Inventory [ARCI] Amph subscale) or conscious motivation (VAS measuring drug liking, wanting to take again).3,7 Protocol deviations (removed researcher blind, n = 2 placebo; altered follow-up behaviour schedule, n = 1 Amph) are detailed in Appendix 1. The study was conducted in accordance with the Declaration of Helsinki and was approved by the Research Ethics Board of the Montreal Neurological Institute at McGill University. All participants provided written informed consent.
Quantification of mGlu5
Autoradiography (mice)
Fresh frozen brains from mice that underwent behavioural tests and 1 mGlu5-KO mouse were sliced at 12 mm on a Cryostat, slide-mounted and stored at −80°C. We performed autoradiography with [3H]ABP688 for as previously described, 34 with minor modifications (Appendix 1).
PET scans (humans)
Participants underwent PET scans between 1000 and 1330 h on days 1 and 21, immediately followed by pill administration. On a separate day, we performed MRI scans for anatomic coregistration. To minimize the effects of different levels of procedural stress on behavioural measures, we performed sham PET scans before the drug session on days 3 and 5. A venous catheter was inserted, and the participant lay in the scanner for 66 minutes without data acquisition.
We performed [11C]ABP688 synthesis as previously described. 25 Following a 6-minute transmission scan with 137Cs to correct for tissue attenuation, we initiated a 60-minute dynamic scan concomitant with a 1-minute injection of 369 ± 29.1 MBq [11C]ABP688 (92 ± 3.8% [E]-isomer) via a catheter placed in the antecubital vein. We collected list-mode data and reconstructed them as previously described.25 We determined mean regional binding potential (BPND) values from 10 grey matter regions of interest (Appendix 1) using the simplified reference tissue model,35 with cerebellar grey matter as a reference. We compared voxel-wise BPND values between days 1 and 21 within each treatment group using SPM12 (Wellcome Functional Imaging Laboratory).
Immunofluorescence
To better understand the role of mGlu5 in the striatum, we performed immunofluorescence experiments on unfixed, frozen coronal sections of mouse brain to localize this receptor relative to synaptic markers. Detailed procedures are presented in Appendix 1. Brain sections taken at the level of the striatum from wild-type or mGlu5-KO mice were incubated with mGlu5 mouse antiserum (1:700; EMD Millipore MABN139) in the presence or absence of VGLUT1 guinea pig antiserum (1:2500; EMD Millipore AB5905) and PSD95 rabbit antiserum (1:2000; Abcam ab18258). To visualize mGlu5 on a subset of medium spiny neurons, we performed immunofluorescence experiments on sections from a Drd1a-tdTomato mouse, which expressed a modified DsRed fluorescent protein under control of the mouse Drd1a promoter. We acquired triple-label and Drd1a-tdTomato mouse images on a laser scanning confocal microscope (Zeiss LSM 880 with airyscan detector; Carl Zeiss). We used Imaris software version 8.0.2 (Bitplane AG) to render fluorescent labelling as surfaces and to perform colocalization analysis. We performed distance analysis using the DiAna plugin36 for ImageJ software (National Institutes of Health).
Results
Behavioural sensitization to Amph in mice and humans
Locomotor response to Amph in mice
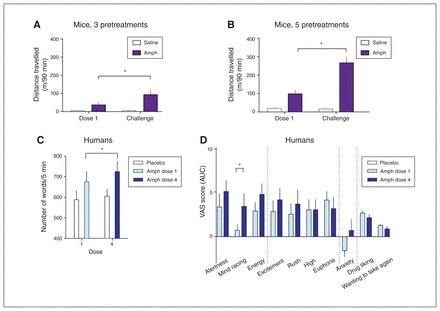
We first explored whether mGlu5 was altered following behavioural sensitization to Amph in mice. Locomotor activity responses to the last dose of Amph were significantly greater than responses to dose 1 in animals treated with either 3 prechallenge doses (treatment × session interaction F1,25 = 13, p = 0.001; challenge versus dose 1, Amph t11 = −3.2, p = 0.008, saline t14 = −0.22, p = 0.83) or 5 pre-challenge doses (treatment × session interaction F1,21 = 30, p < 0.001; challenge versus dose 1 Amph t10 = −5.2, p < 0.001, saline t11 = 0.97, p = 0.36; Fig. 2A and B; Appendix 1, Fig. S1 and Table S1).
Sensitization of behavioural response to dextroamphetamine (Amph) in mice and humans. A and B: mice. Locomotor activity after dose 1 and challenge in animals given 3 or 5 pre-challenge doses of 2 mg/kg Amph (i.p.). Locomotor activation is presented as the sum of the distance travelled in 90 minutes after amphetamine injection (mean ± standard error of the mean [SEM]). C and D: humans. C: speech rate in placebo (n = 9) and Amph (n = 10) groups following the first and fourth dose, mean ± SEM. D: self-reported drug effects (visual analogue scales [VAS]) in Amph-treated participants (n = 10). Values represent mean ± SEM area under the curve (AUC) of the scores on each subscale during the drug session. Alertness, mind racing and energy were considered activating effects; excitement, rush, high and euphoria were considered euphoric/high effects; drug liking and wanting to take again were considered conscious motivational effects. We observed a significant treatment × subscale effect (p = 0.021) on activating effects only. *p < 0.05 in planned pair-wise comparisons of ratings on day 1 versus day 2.
Psychoactivating effects of Amph in healthy humans
Groups were similar in terms of age, sex, smoking status and stimulant drug history, although participants randomized to the placebo group had slightly higher novelty-seeking scores (Table 1).
Participant and scan characteristics
A significant treatment × session interaction emerged for speech rate (F3,48 = 4.8, p = 0.005). This reflected a significant increase in speech rate after the fourth dose compared to the first dose of Amph (t8 = −4.0, p = 0.003) but not of placebo (t9 = −0.61, p = 0.56; Fig. 2C). Drug treatment did not affect eye-blink rate or motor activity as measured by Actiwatch counts (data not shown).
We observed significant main effects of treatment on activating responses (alertness, mind racing and energy VAS; F1,17 = 9.7, p = 0.006) and feelings of euphoria/high (excitement, rush, high and euphoria; F1,17 = 14, p = 0.001), but not on anxiety (F1,17 = 0.023, p = 0.88) or conscious motivation (drug liking and wanting to take again; F1,17 = 0.19, p = 0.67; Appendix 1, Table S1). An absence of some drug effects was not surprising given the relatively low doses and our population of healthy drug non-users. For the activating effects, a subscale × treatment interaction was also present (F2,34 = 4.3, p = 0.021). We performed planned comparisons within each group based on a priori hypotheses (Fig. 2D). Among the activating effects subscales, ratings for mind racing were significantly greater on day 21 (fourth dose of Amph) than on day 1 (t9 = −2.5, p = 0.035), although these findings did not survive correction for multiple comparisons. Activating VAS summary scores (t9 = −2.0, p = 0.08) and ARCI Amph scores (Z = −1.8, p = 0.08) showed trend-level effects reflecting higher scores on day 21. We observed no changes for these measures in the placebo group (drug liking t8 = 2.1, p = 0.07; other subscales p > 0.18; Appendix 1, Fig. S2).
Plasma Amph concentration increased, and blood pressure, heart rate and serum cortisol levels were significantly elevated following each drug administration (p < 0.05), but these did not differ across sessions (p > 0.25; Appendix 1).
Changes in mGlu5 with Amph sensitization
Binding at mGlu5 in mice
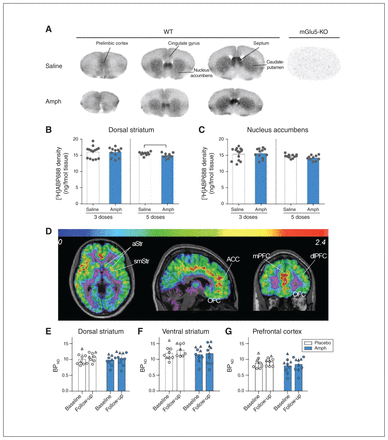
We found that [3H]ABP688 binding sites were not present in brain slices from the mGlu5-KO mouse or in the presence of 10 μM MPEP, a selective mGlu5 antagonist. This finding indicated that the ligand bound specifically to mGlu5. We then measured [3H]ABP688 binding sites on coronal sections from the mice that underwent behavioural tests (Fig. 3A). The regional distribution of mGlu5 was similar in mice and humans. We found that [3H]ABP688 binding sites were highly abundant in the septum, dense in the striatum and relatively abundant in the cingulate and prelimbic cortex in mice (Fig. 3A), consistent with previous work.37 In the salinetreated animals, binding was significantly different across the 3 regions quantified (F25,1 = 47, p < 0.001), reflecting higher binding in the striatum and nucleus accumbens than in the prelimbic cortex (p < 0.001). We found no difference in mGlu5 binding in the dorsal striatum or nucleus accumbens in animals pretreated with 3 Amph doses compared to saline-treated controls (p > 0.50; Fig. 3B and C and Appendix 1, Table S2). We found that mGlu5 was significantly reduced in the dorsal striatum in animals pretreated with 5 Amph doses compared to saline-treated controls (t19 = 2.2, p = 0.038). We observed a nonsignificant trend toward a decline in the nucleus accumbens of Amph-treated mice (t19 = 1.8, p = 0.086; Fig. 3B and C). We found no difference in binding between saline-treated controls and mice that received saline pretreatment followed by a single challenge dose of Amph (t9 = 0.81, p = 0.44; Appendix 1, Fig. S3) and no change in binding in the prelimbic cortex (3 pretreatment doses t25 = 0.17, p = 0.50; 5 pretreatment doses t19 = 0.17, p = 0.86).
Binding availability of mGlu5 following sensitization. A: mGlu5 binding density in the mouse brain measured by [3H]ABP688 autoradiography. B and C: in mice, mGlu5 density quantified with [3H]ABP688 was reduced following sensitization only in the group treated with 5 prechallenge doses. D: representative [11C]ABP688 BPND distribution in the brain of 1 human participant. E to G: no change in BPND from baseline to follow-up in placebo- or Amph-treated participants. ACC = anterior cingulate cortex; Amph = dextroamphetamine; aStr = associative striatum; BPND = binding potential, non-displaceable; dlPFC = dorsolateral prefrontal cortex; mGlu5 = metabotropic glutamate type 5 receptor; mGlu5-KO = mGlu5-knockout; mPFC = medial prefrontal cortex; OFC = orbitofrontal cortex; smStr = sensorimotor striatum; WT = wild-type.
Availability of mGlu5 in humans
We used PET with [11C]ABP688 to assess mGlu5 binding availability across the brain before and after stimulant sensitization (Table 2 and Fig. 3D to G). As we observed previously in a larger sample,38 the overall BPND was higher in men than women (t15 = −2.7, p = 0.015) by an average of 26% across regions (Appendix 1, Figure S4). A single day 1 scan in each of the Amph and placebo groups was excluded because of low injected activity and high injected mass, respectively (Appendix 1), resulting in a final sample size for primary PET comparisons of 9 Amph-treated participants and 8 placebo participants. Detailed test–retest analyses of variability in the placebo group have been reported previously.39
Regional [11C]ABP688 BPND values in the Amph and placebo groups
We observed no significant treatment or session effects, and no interaction in the striatal, cortical or limbic regions (Table 2 and Appendix 1, Table S2). We observed no change in BPND from day 1 to 21 in either group in paired comparisons (Table 2 and Fig. 3E to G). Results were similar when sex was included as a covariate. Voxel-wise comparisons found no clusters of significant increase or decrease in either group (threshold p = 0.001, corrected for false discovery rate).
Correlation analyses of the relationship between mGlu5 and behavioural sensitization
Post-Amph mGlu5 availability and locomotor sensitization in mice
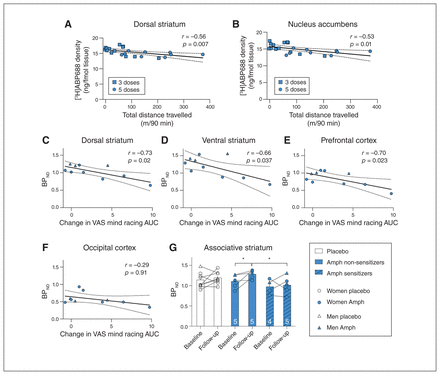
Across all Amph-treated mice, mGlu5 binding in the nucleus accumbens and dorsal striatum following amphetamine regimens were negatively correlated with initial drug response (dorsal striatum r = −0.45, p = 0.038; nucleus accumbens r = −0.42, p = 0.050) and with extent of locomotor sensitization (dorsal striatum r = −0.56, p= 0.007; nucleus accumbens r = −0.53, p = 0.01; Fig. 4A and B, and Appendix 1, Table S3). We did not observe this in saline-treated animals (p > 0.54), suggesting that this finding was not because of a relationship between mGlu5 levels and locomotor activity independent of drug treatment.
Low metabotropic glutamate type 5 receptor (mGlu5) binding is associated with greater sensitization. A and B: in mice, lower mGlu5 density after a challenge dose was associated with greater sensitization (total distance travelled in 90 minutes post-dextroamphetamine (Amph) at challenge minus day 1) in the groups receiving 3 pre-challenge doses (squares) and 5 pre-challenge doses (circles). C to E: [11C]ABP688 BPND in the striatum and prefrontal cortex at follow-up was negatively correlated with a change in subjective psychoactivating drug effects (mind racing scores). F: we found no relationship between BPND and behaviour in the occipital cortex. G: BPND (mean ± standard error of the mean [SEM]) in the associative striatum in the placebo group (white bars, n = 8) and in Amph-group participants who did (striped blue bars) or did not (solid blue bars) show behavioural sensitization on the mind racing measure. AUC = area under the curve; BPND = binding potential, non-displaceable; VAS = visual analogue scale.
Post-Amph mGlu5 availability and psychomotor sensitization in humans
We analyzed individual differences in drug response in humans by assessing correlations between BPND values and speech rate, VAS mind racing scores and ARCI Amph scores. We observed no correlation between day 1 BPND and response to dose 1 or change in response from day 1 to day 21 (Appendix 1, Fig. S5). However, the extent of the change in mind racing ratings was negatively correlated with BPND on day 21 in the dorsal striatum (r = −0.73, p = 0.016; Fig. 4C), ventral striatum (r = −0.66, p = 0.037; Fig. 4D), prefrontal cortex (r = −0.70, p = 0.023; Fig. 4E; Appendix 1, Table S3), insula (r = −0.66, p = 0.037) and cingulate (r = −0.67, p = 0.034; Appendix 1, Figure S6). We observed no correlation in the occipital cortex (r = −0.29, p = 0.91; Fig. 4F); this relationship appeared to be specific to the striatum and regions of the cortex implicated in stimulant drug responses and sensitization. We observed nonsignificant trend-level associations with ARCI Amph scores in the prefrontal cortex only (r = −0.60, p = 0.07), and we found no correlation between BPND and change in speech rate (p > 0.16; Appendix 1, Fig. S7). Associations with mind racing were also present in partial correlations with sex included as a covariate (prefrontal cortex r = −0.70, p = 0.036; dorsal striatum r = −0.58, p = 0.010; ventral striatum r = −0.69, p = 0.040) but no longer reached conventional levels of significance in the insula (r = −0.65, p = 0.056) or the cingulate (r = −0.65, p = 0.057).
To explore this effect further, we created subgroups by median split, comparing BPND values in people who showed a high change on mind racing scores from day 1 to day 21 (sensitizers) with those whose scores changed little (non-sensitizers). In the associative striatum, equivalent to the dorsomedial striatum in mice, BPND increased in non-sensitizers (Wilcoxon signed ranks test, Z = −2.0, p = 0.043) but not in sensitizers (Z = 0.0, p = 1.0). On day 21, BPND was higher in non-sensitizers than in sensitizers (Mann–Whitney U test, U = 2.0, p = 0.032; Fig. 4G). This finding suggests that those with a lower propensity for behavioural sensitization experienced drug-induced increases in mGlu5 availability in the associative striatum.
Distribution of mGlu5 in the rodent nucleus accumbens
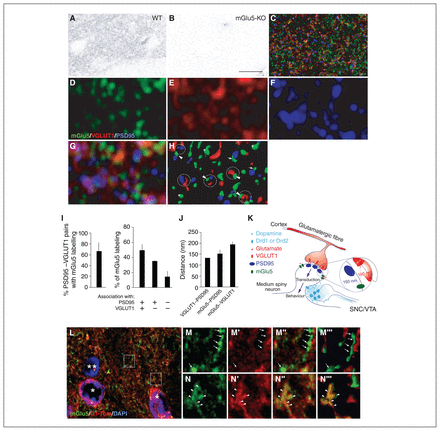
Relationships between mGlu5 availability and sensitization were consistent across species in the striatum. Therefore, we performed immunofluorescence experiments to explore the localization of mGluR5 in mouse striatum relative to synaptic markers. We first validated an mGlu5 antiserum by comparing its labelling in the striatum of wild-type mice and mGlu5-KO mice. In wild-type mice, mGlu5 immunopositive puncta were widely distributed across the neuropil of the nucleus accumbens (Fig. 5A) and the dorsal striatum (not shown). These immunopositive puncta were absent in mGlu5-KO mice (Fig. 5B).
Metabotropic glutamate type 5 receptor (mGlu5) expression and localization in the mouse nucleus accumbens. A and B: mGlu5 immunolabelling in sections from wild-type (WT) and mGlu5 knockout (mGlu5-KO) mice. C: mGlu5 (green), VGLUT1 (red) and PSD95 (blue) triple immunofluorescence staining showing widespread mGlu5 expression. D to G: 4× zoom of labelling in C shows (D) mGlu5, (E) VGLUT1, (F) PSD95, (G) merge. H: surface reconstruction of G, showing sites of VGLUT1–PSD95 colocalization, presumed to represent excitatory synapses (circles). mGlu5 is commonly located at these sites (arrowheads), at sites expressing PSD95 but not VGLUT1 (arrows), and at sites not expressing either marker (asterisks). I: left panel, 67% of VGLUT1–PSD95 pairs (putative synapses) had mGlu5 labelling; right panel, of all mGlu5 labelling, 50% occurred at VGLUT1–PSD95 sites, 35% at sites expressing PSD95 but not VGLUT1, and 15% at sites not expressing either label. J: Mean (± standard error of the mean [SEM]) distance between pairs of labels. K: widespread expression of mGlu5 on dendritic spines facilitates glutamatergic inputs to striatal medium spiny neurons. Inset: distance analysis demonstrating predominantly postsynaptic localization of mGlu5. L: mGlu5 labelling (green) and nuclei (blue, Hoechst stain) on sections from Drd1a-tdTomato mice (D1+ neurons in red). M and N: zoomed sections of L, showing mGlu5 labelling separate from (M, arrows) and colocalized with (N, arrowheads) D1-expressing dendrites. M, N, mGlu5; M′, N′, D1-expressing neurons; M″, N″, merge; M‴ and N‴, surface reconstruction of mGlu5 and D1tomato labelling and overlap (yellow). Scale bar in B represents 100 μm in panels A and B; 20 μm in C; 5 μm in D to H; 80 μm in L; and 10 μm in M. SNC = substantia nigra pars compacta; VTA = ventral tegmental area.
Using confocal microscopy and surface reconstruction, we then examined the subcellular localization of mGlu5. Receptor distribution was widespread across the nucleus accumbens and frequently apposed close to both VGLUT1- and PSD95-positive puncta (Fig. 5C to H). Colocalization analyses showed that mGlu5 fluorescent labelling was present at 67 ± 27% of VGLUT1/PSD95-positive sites (Fig. 5H and I). These structures represent putative excitatory synapses. Conversely, 50 ± 14% of mGlu5-positive puncta were located at these VGLUT1/PSD95-positive sites (Fig. 5H), whereas 35 ± 1.3% of mGlu5-positive puncta were located in proximity to PSD95 alone and 15 ± 13% of mGlu5-positive puncta were associated with neither PSD95 nor VGLUT1 labelling. These data suggest that approximately half of the total striatal mGlu5 is associated with VGLUT1-expressing synapses, and half with other excitatory synapses (possibly VGLUT2-positive thalamostriatal synapses), modulatory sites or inhibitory synapses. In the dorsal striatum, 46% of VGLUT1–PSD95 pairs also expressed mGlu5, and 49% of mGlu5 were close to these markers (not shown). Distance analyses showed that on average, mGlu5 was located closer to PSD95 (median edge-to-edge distance 150 ± 12 nm) than to VGLUT1 markers (median distance 190 ± 18 nm; Fig. 5J). This finding was consistent with a predominantly postsynaptic localization of mGlu5 (Fig. 5K, inset). Finally, we investigated the distribution of mGlu5 in D1-positive or -negative (putatively D2-expressing) medium spiny neurons from the nucleus accumbens. We performed immunofluorescent detection of mGlu5 on brain sections from Drd1a-tdTomato mice (Fig. 5L). Immunofluorescent mGlu5 puncta were densely expressed along neurites that did not express D1 (Fig. 5M to M‴, green mGlu5 labelling separate from red D1 labelling) and along D1-positive neurites (Fig. 5N to N‴, mGlu5 labelling colocalized with D1 labelling). These data show that mGlu5 is strategically localized on dendritic spines of medium spiny neurons to modulate excitatory inputs into the striatum (Fig. 5K).
Discussion
In the present work, we measured mGlu5 availability during Amph sensitization in rodents and in healthy humans. An Amph regimen sufficient to induce mild behavioural sensitization was not associated with statistically significant changes in mGlu5 availability. Reductions in receptor binding in the mouse striatum were seen following a slightly more extensive stimulant treatment. Together, these results suggest that although changes in mGlu5 availability develop with continued drug use, these changes are not necessary for the process of sensitization. In both humans and mice, lower mGlu5 availability after drug treatment was associated with greater behavioural sensitization to Amph.
Lower mGlu5 availability has been observed following cocaine self-administration sessions in rodents40 and in people with moderate to severe cocaine use disorders.24,25 Fewer studies have examined the effects of sensitization regimens themselves, in either rodents or humans. In laboratory animals, one Amph sensitization experiment reported reduced mGlu5 mRNA,41 while another found no difference in protein or mRNA levels.42 The present work suggests that regimens of 3 to 5 moderate doses encompass the threshold minimum number of Amph exposures necessary to induce clear changes in mGlu5 availability. This work also presents the first study of mGlu5 binding during sensitization in humans. Because the drug regimen used here is sufficient to induce sensitized striatal dopamine release in humans,3 the present results raise the possibility that dopamine sensitization may occur independently of changes in mGlu5 availability. Therefore, changes in mGlu5 binding availability might not be a component of initial brain changes associated with behavioural sensitization. The reduced mGlu5 binding seen in people who are cocaine-dependent and in rodent models during stimulant withdrawal or abstinence could be a compensatory response to heightened striatal excitability after sensitization.
In both mice and humans, a pattern emerged in which individual differences in striatal mGlu5 were negatively correlated with Amph-induced behavioural sensitization. In rodents, low post-drug mGlu5 binding was associated with greater initial and sensitized locomotor response to Amph. In humans, this relationship was seen in that subjective psychoactivating drug effects (mind racing) covaried with binding in the cortex and striatum and was not observed with either baseline receptor availability or initial drug response. In the associative striatum, subgroup analyses suggest that this may have been related to a failure in sensitizing individuals to upregulate mGlu5 after drug exposure, although this should be interpreted with caution given the small sample size. Together, these relationships raise the possibility that low endogenous mGlu5 activity could mark or contribute to an individual’s propensity for sensitization, a trait associated with greater drug-seeking behaviours in animal models.43,44 Low mGlu5 early in drug use could therefore be relevant to risk for Amph addiction. The present work suggests that impaired glutamate signalling or a hypoglutamatergic baseline state after the initiation of drug treatment could contribute to elevated responses to Amph, similar to what has been described for cocaine45 but not methamphetamine.46 Notably, in contrast with our finding that low mGlu5 binding is associated with greater sensitization to Amph, blocking mGlu5 signalling in rodents reduces sensitization and reinstatement in cocaine-treated animals. 12–15,47–49 This might reflect different effects of cocaine versus Amph, influences of mGlu5 signalling on reward-related learning rather than sensitization in itself, or an important distinction between the normal variation observed in healthy, drug-naïve participants here and the effects of nonphysiologic pharmacological manipulations of mGlu5 signalling. The roles of mGlu5 in risk for drug-related behaviours in healthy people and in drug-induced plasticity require further study. Pharmacological reduction of mGlu5 activity also leads to impairments in synaptic plasticity and associative learning, including the acquisition and extinction of cocaine seeking and conditioned place preference. 16,19,50,51 This suggests that mGlu5 activity is involved in establishing and updating drug-related associations. We speculate that low mGlu5 availability could contribute to an impaired ability to modulate drug responses and associations, leading to more rigid behaviours in the presence of drug-related stimuli. This model is consistent with the proposition that positive allosteric modulators at mGlu5 could be an effective adjunct to cue-exposure therapy by facilitating the extinction of drug-cue associations and the learning of new goal-directed behaviours.26,52
Immunofluorescence experiments suggest that strategic expression of mGlu5 at excitatory striatal synapses could underlie some of these relationships (Fig. 5K). Previous studies report mixed results regarding localization of mGlu5, including its expression in presynaptic terminals and neuronal cell bodies in the striatum.53–55 In line with our findings, electron microscopy studies of mGlu5 in rats and primates previously suggested that mGlu5 is frequently localized in dendritic spines in the nucleus accumbens, in close proximity to synapses.56,57 The estimated distance between VGLUT1–PSD95 pairs in this study (130 nm) was similar to the 100 nm distance calculated on individual dendritic spines,58 suggesting that our data provide a reasonable estimate of the distribution of mGlu5 at the level of striatal synapses. Our finding that mGlu5 is expressed at approximately two-thirds of excitatory VGLUT1-positive synapses (PSD95- and VGLUT1-positive sites) was in line with that of a previous study, in which mGlu5 labelling was observed on approximately 40% to 50% of cortical inputs and 70% of amygdalar inputs to dendrites in the nucleus accumbens.56 In the present work, VGLUT1-positive sites accounted for half of all mGlu5 labelling. Roughly one-third of mGlu5 labelling was colocalized with PSD95-positive, VGLUT1-negative structures. These sites may represent thalamostriatal inputs, which express VGLUT2 instead of VGLUT1.59 Fig. 5L to N shows mGlu5 labelling along the dendrites of medium spiny neurons in both direct (D1-positive) and indirect (D1-negative) pathways. This expression pattern was consistent with the receptor’s role in mediating associative learning by facilitating glutamatergic activity and plasticity.
Limitations
The human PET study used BPND as an outcome measure, although previous studies have found evidence of specific binding of [11C]ABP688 in the cerebellum.60 We previously found that BPND correlates well with volume of distribution (VT) values derived using an arterial input function;61 nevertheless, changes in cerebellar mGlu5 may have masked (or amplified) observed effects in this study. In the human study, variability in BPND was high, although lower than in previous studies.62 This finding, combined with the modest sample size, limited the study’s power to detect more subtle changes in mGlu5 binding. Further, novelty seeking scores were higher in the placebo group than the Amph group (p = 0.04; Appendix 1, Table S1), which may have limited our ability to induce sensitization with this moderate regimen.3 An Amph dosing regimen that produced relatively mild acute and sensitized psychomotor effects likely compounded this issue (i.e., only modest changes in mGlu5 binding might be expected from a modest drug regimen). This is also relevant to behavioural measures in humans: although we observed a robust increase in speech rate on day 21 in the Amph group, sensitization effects on subjective self-report scales were mild or at trend level in most cases and did not survive correction for multiple comparisons. Nevertheless, because our mouse experiments with lower overall variability yielded results that were highly consistent with our findings in humans, they gave greater confidence in both the negative and positive results.
In comparing across species, several protocol differences should be noted. We measured receptor binding before the challenge dose in humans and immediately after the final dose in mice, allowing for the possibility that acute effects of Amph on glutamate or on mGlu5 affected binding measures. We observed no difference in mGlu5 binding after acute Amph in a group of saline-pretreated animals (Appendix 1, Fig. S3), concordant with previous studies, 41,42 but this distinction between the 2 protocols should be kept in mind. Conversely, making primary comparisons in the mouse study between saline/saline-treated and Amph/Amph-treated animals to best compare with the human study was atypical for sensitization studies and may limit comparison with other literature. Second, measurements taken in vivo in humans and postmortem in animals are subject to different influences; binding in the human study could conceivably have been influenced by stress or other physiologic factors. Similarly, binding comparisons in animals were performed between subjects, not within subjects. A parsimonious explanation is that the relationship between sensitization and mGlu5 binding is consistent across species, although we do not rule out the possibility that the convergent observations arose from different influences and mechanisms in the 2 species. Finally, the use of only male mice was an important limitation, given baseline sex differences in mGlu5 binding in humans.38 The human study was not sufficiently powered to detect sex differences in sensitization effects on mGlu5, but these might exist. Female rodents show a greater propensity to develop behavioural sensitization to psychostimulants, 63 and estradiol interacts with mGlu5 to potentiate cocaine sensitization.64
Conclusion
These studies suggest that large changes in mGlu5 availability are not a component of the earliest neural adaptations contributing to Amph sensitization. Nevertheless, the experiments in mice indicate that they may begin to develop with higher levels of drug exposure. Further, low mGlu5 following initiation of drug use was associated with a tendency to develop stronger drug responses, which may be associated with greater drug-seeking behaviours. Expression of mGlu5 at the majority of excitatory corticostriatal inputs may contribute to these effects.
Acknowledgments
The authors thank Kevin Larcher for assistance with PET imaging and analysis, Arturo Aliaga for assistance with the rodent experiments and Dr. Nicolas Heck for technical assistance on the immunofluorescence experiments and distance analyses. This study used the services of the Molecular and Cellular Microscopy Platform in the Douglas Institute and the Cell Biology and Image Acquisition Core at the University of Ottawa.
Footnotes
* These authors contributed equally to this work.
Dr. Chawki Benkelfat passed away on Jan. 7, 2020, during preparation of this manuscript for publication.
Funding: This research was supported by funds from the Canadian Institutes for Health Research (C. Benkelfat and M. Leyton, MOP-119509), Brain Canada Multi-Investigator Research Initiative (S. El Mestikawy), Fond de Recherche Santé du Québec (S. El Mestikawy, FRQS 30582), Natural Sciences and Engineering Research Council (NSERC, RGPIN/386431-2012) Discovery (DG) Grants (S. El Mestikawy), ERANET-Neuron Joint Transnational Call for “European Research Projects on Mental Disorders” and research projects on synaptic dysfunction in disorders of the central nervous system JTC 2013 and 2017 (S. El Mestikawy). The sponsors had no role in the design or conduct of the study, in data collection, analysis or interpretation, in manuscript preparation, approval or review, or in the decision to submit the manuscript for publication.
Competing interests: M. Leyton is an associate editor of JPN. He was not involved in reviewing or the decision to accept this paper for publication.
Contributors: K. Smart, M. Milella, M. Favier, E. Vigneault, S. El Mestikawy, M. Leyton and C. Benkelfat designed the study. K. Smart, D. Yae Sakae, M. Favier, L. Louie, A. Hamilton, S. Ferguson and M. Leyton acquired the data, which K. Smart, A. Nagano-Saito, M. Favier, E. Vigneault, L. Louie, P. Rosa-Neto, S. Narayanan, S. El Mestikawy, M. Leyton and C. Benkelfat analyzed. K. Smart, S. El Mestikawy, M. Leyton and C. Benkelfat wrote the article, which all authors reviewed. All authors approved the final version to be published and can certify that no other individuals not listed as authors have made substantial contributions to the paper.
- Received September 23, 2019.
- Revision received December 21, 2019.
- Accepted February 3, 2020.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is non-commercial (i.e. research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/