Abstract
Background: Transcranial sonography (TCS) has become a new diagnostic tool in the evaluation of extrapyramidal disorders. Studies of TCS report alterations of the mesencephalic raphe in patients with depression. The aim of this study was to evaluate TCS findings in patients with Huntington disease in correlation with their neurologic and psychiatric status.
Methods: We recruited patients with genetically confirmed Huntington disease. The neurological and psychiatric status of participants was assessed by independent physicians. Echogenicities were investigated according to examination protocol for extrapyramidal disorders using a Siemens Sonoline Elegra system. The sonography examiner was blinded for clinical data.
Results: We included 39 patients in our study; 21 patients (53.8%) showed symptoms of depression at the time of evaluation and, of those, 15 (71.4%) had hypoechogenic raphe structures. Thirty patients (76.9%) had a history of depressive episodes, 19 (63.3%) of them with hypoechogenic raphe structures. All 9 patients without a history of depressive episodes showed normal echogenicity of raphe structures (sensitivity 63.3%, specificity 100%). Twelve (70.6%) of the 17 patients with Huntington disease who showed psychiatric disturbances prior to the occurrence of motor symptoms exhibited pathological raphe echogenicity (sensitivity 70.6%, specificity 68.2%).
Limitations: Most of the patients were taking antichoreatic medication, which particularly influences neurologic status. Thus, a meaningful interpretation of the correlation between TCS findings and neurologic features was limited.
Conclusion: As a novel finding, a relation between mesencephalic raphe echogenicity and depressive state could be identified in patients with Huntington disease. An alteration of the serotonergic brain stem raphe might be involved in the pathogenesis of depression in these patients.
Introduction
Huntington disease is an autosomal dominant disorder caused by an unstable expansion of the trinucleotide repeat, cytosine–adenine–guanine (CAG), on chromosome 4p16.3.1 This CAG expansion leads to a progressive neurodegeneration of the basal ganglia, particularly of the neostriatum.2 However, neuronal loss has been identified in other subcortical and cortical regions.3
Choreatic involuntary movements are the hallmark of Huntington disease, although rigidity, bradykinesia and dystonia may also be present.4 Diagnosis and treatment of Huntington disease typically focuses on movement disorders; however, the clinical phenotype is also associated with psychiatric symptoms like depression, irritability, aggression, anxiety and psychotic and compulsive symptoms.5 The psychiatric disturbances often appear years before the onset of motor symptoms.6 Cognitive decline may even lead to dementia.5
Transcranial sonography (TCS) has become a reliable and sensitive diagnostic tool in the evaluation of extrapyramidal movement disorders, especially in the differentiation of parkinsonian syndromes.7–10 Furthermore, alterations of the brain stem raphe have been observed by TCS in patients with major depression and in depressed patients with Parkinson disease.11–15
Up to now, there has been only 1 TCS study evaluating basal ganglia alterations in patients with Huntington disease.16 The echogenicity of the brain stem raphe was not assessed in this study. This is of particular interest since depression is a frequent psychiatric symptom in patients with Huntington disease.5 Even in neurologically presymptomatic Huntington disease, depressive symptoms appear in 30%–58% of the mutation carriers.6 The suicide rate among patients with Huntington disease varies between 3% and 7.3%.17
The aim of our study was to evaluate echogenicity of the basal ganglia, especially in the brain stem raphe, and to correlate ultrasound findings with psychiatric features in particular, but also with neurologic and cognitive features.
Methods
Participants
We recruited patients with genetically confirmed Huntington disease and a mini mental state examination score greater than 24 points from the Huntington-Center North Rhine-Westphalia in Bochum, Germany. We excluded those with an inadequate temporal bone window. We noted participants’ mean age at onset of motor symptoms and at onset of depression. We calculated toxic load using the following formula: (CAG repeat − 35.5) × age.21 All patients received the best possible medical treatment, including antichoreatic medication. We also recruited age-matched healthy volunteers among hospital staff, the medical student body and their friends and families to form a control group. We determined the existence of a personal or family history of psychiatric or movement disorders by means of a standardized interview, and we excluded people with such history from the control group. After complete description of the study to the participants, we obtained written informed consent. The local university ethics committee approved our study protocol.
Clinical assessment
Trained and certified investigators (K.S., C.S.) independently evaluated the clinical status of all patients according to the Unified Huntington’s Disease Rating Scale (UHDRS).20 This scale comprises ratings of motor performance, independent functioning and total functional capacity, as well as cognitive assessment. The motor score is the sum of the scores of items 1–15 ranging from 0 (healthy) to 124 points (most severe disease). The sum of the scores of items 70–74 compose the total functional capacity, ranging from 0 (most severe disease) to 13 points (healthy). The independent functioning, and therefore the overall severity of the disease, was graded on the independence scale ranging from 80%–100% (self-care maintained) to 40%–75% (limited self-care) to 0%–35% (dependent). The scale has a 5% step classification, so ratings of 76%–79% and 36%–39% do not exist. The cognitive assessment comprises a battery of neuropsychological tests (verbal fluency, symbol digit, Stroop test). The sum of the scores of these items is the cognitive score (the higher the cognitive performance the higher the score).20
As depression in Huntington disease was a major focus of our study, an independent psychiatrist (D.N.) with substantial experience in caring for patients with neurodegenerative disorders performed a detailed evaluation of depressive symptoms. We also obtained information on the patients’ medical history from a third party, such as their partners or other relatives. Diagnoses were based on the criteria of the DSM-IV.22 The severity of depressive episode at time of evaluation was assessed using the Hamilton Rating Scale for Depression (HAM-D).19 Since there might be an influence of some Huntington disease symptoms on the depression scale, we excluded items quantifying somatic features and psychomotoric functions (items 7–9, 13, 16) from further analysis (HAM-D*). We used the Beck Depression Inventory (BDI)18 as a standardized measure for the self-assessment of depressive syndromes.
Transcranial sonography
Transcranial sonography was performed by a single investigator blinded to the clinical status (C.K.) through the transtemporal bone window using a phased array ultrasound system equipped with a 2.5 MHz transducer (Sonoline Elegra System; Siemens). A penetration depth of 150 mm and a dynamic range of 45 dB were chosen. Image brightness and time gain compensations were adapted as needed. The examination protocol was based on previous published recommendations for TCS.23 Using the transtemporal approach, the midbrain and diencephalic examination planes were visualized in axial sections. In case of hyperechogenicity of the substantia nigra, a planimetric measurement was performed. Sizes of less than 0.20 cm2 were defined as normal, sizes between 0.20 cm2 and 0.25 cm2 were classified as moderate and sizes greater than 0.25 cm2 were graded as markedly hyperechogenic.9 The brain stem raphe was evaluated by bilateral TCS investigation. We used the side with the best visible structure for statistics. Echogenicity of the brain stem raphe was classified semiquantitatively on a 3-point scale using the red nucleus or signal intensity of the basal cisterns as reference points: 0 = raphe structure not visible, 1 = reduced echogenicity (the echogenic line of the brain stem raphe is interrupted or appears abnormally slight and thin) and 2 = normal echogenicity. Echogenicities of the thalami, lentiform nuclei and the heads of caudate nuclei were analyzed from the contralateral side. These structures were graded as hyperechogenic if they were more intense than the surrounding white matter.10 The maximal width of the frontal horns of the side ventricles, the distance between the septum pellucidum and the head of caudate nucleus and the minimal transverse diameter of the third ventricle were measured on a standardized diencephalic examination plane.16 The findings were stored, so that a second experienced TCS investigator (J.E.), who was also blinded to the clinical ratings, performed a second independent “off-line” evaluation. In the case of discrepancies between the ratings, a consensus was reached subsequently.
Statistical analysis
Results are reported as mean values, standard deviations and ranges. Interrater agreement was analyzed by Cohen’s κ, and the agreement was estimated according to a classification proposed by Landis and Koch.24 Statistical comparison of groups and correlation analysis were performed by appropriate nonparametric tests (Mann–Whitney U test, Fisher exact test or Spearman rho test for independent samples) using SPSS 17.0 for Windows.
Results
Clinical and genetic features
We assessed 42 patients with Huntington disease; 3 patients (7.1%) were excluded owing to an inadequate temporal bone window, leaving 39 patients (16 men, 23 women; mean age 47.9, standard deviation [SD] 9.5 yr) for inclusion in the study. Their demographic and clinical characteristics are summarized in Table 1. The mean age at onset of motor symptoms was 42.4 (range 22–61) years, and the mean age at onset of depression was 41.4 (range 18–63) years. Seventeen patients (43.6%) showed symptoms of depression prior to the occurrence of motor symptoms. All patients were taking antichoreatic medication, and 22 (56.4%) were taking antidepressants (Table 2). We enrolled 40 healthy volunteers (19 men, 21 women; mean age 46.3, SD 7.1 yr) in the control group.
Demographic and clinical characteristics of study participants
Psychopharmacological medication and daily dose
Twenty-one (53.8%) patients fulfilled the DSM-IV diagnostic criteria for major depressive episode (mean age 50.8, SD 7.4 yr; mean HAM-D score 23.0, SD 7.5; mean HAM-D* score 18.8, SD 7.2; mean BDI score 18.0, SD 9.5). Seventeen (81.0%) of these 21 patients were already taking antidepressive medication. Eighteen (46.2%) patients reported no depressive symptoms (mean age 44.5, SD 10.5 yr; mean HAM-D score 8.8, SD 4.7; mean HAM-D* score 5.5, SD 4.5; mean BDI score 4.3, SD 3.1). Nine (50%) of the currently nondepressed patients had reported experiencing depressive episodes in their medical history, with 7 still taking antidepressants. The CAG expansion and the toxic load correlated with age at onset of symptoms (p < 0.001 and p = 0.015, respectively), irrespective of whether patients showed onset with motor or psychiatric symptoms. Furthermore, the toxic load correlated with the UHDRS motor score (p = 0.025) and UHDRS functional capacity (p = 0.022). There were no correlations between CAG expansion or toxic load and HAM-D* scores. Also, there were no correlations between severity of motor features or disease duration and HAM-D* scores.
Transcranial sonography findings
Owing to partial insufficient bone window in 10 (25.6%) of the 39 patients, an adequate evaluation of the lateral ventricles could not be performed bilaterally, so that only the width of the third ventricle was used for further statistical analysis. In 5 (12.8%) of the patients, the caudate nuclei could not be evaluated sufficiently. In 4 (10.3%) patients, the lentiform nuclei could not be evaluated satisfactorily. Evaluation of the width of the third ventricle, substantia nigra and brain stem raphe could be performed in all 39 patients with Huntington disease.
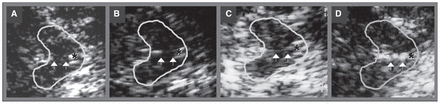
The mean width of the third ventricle was 8.1 mm (SD 2.7, range 3.1–15.8 mm). Nineteen (48.7%) of 39 patients showed pathologic signal of the brain stem raphe. In 14 of these patients, the brain stem raphe echogenicity was reduced (grade 1), whereas in 5 patients, the brain stem raphe was not visible (grade 0). One patient exhibited a continuous but very slight and thin signal of brain stem raphe. Compared with all other patients with a continuous raphe signal (grade 2), this brain stem raphe echogenicity was clearly reduced, so that this unique case was also graded as reduced echogenicity (grade 1; Fig. 1C). Sixteen (41.0%) patients showed hyperechogenicities of the substantia nigra (12 unilateral, 4 bilateral). Of these patients, 6 exhibited marked hyperechogenicity, 2 bilaterally. The median echogenic size of the substantia nigra in all patients with Huntington disease was 0.096 cm2 (SD 0.09, range 0.00–0.38 cm2). Seven (20.6%) of 34 patients exhibited hyperechogenic lesions of the caudate nuclei (4 bilaterally). Six (17.1%) of 35 patients showed hyperechogenicities of the lentiform nuclei (1 bilaterally). In 7 patients, hyperechogenicities were detected in 2 different regions (lentiform nuclei and substantia nigra in 5 patients; caudate nuclei and substantia nigra in 2 patients).
Corresponding mesencephalic axial examination planes in 4 patients with Huntington disease. The butterfly-shaped midbrain is outlined for better visualization. The asterisk indicates the aqueduct. Arrowheads indicate the brain stem raphe. (A) Raphe structure not visible, grade 0, pathologic finding. (B) Echogenic line of the raphe is interrupted, grade 1, pathologic finding. (C) Unique case of continuous but very slight and thin signal of the raphe structure, grade 1, pathologic finding. (D) Normal echogenicity, grade 2, normal finding.
The 2 sonographers agreed independently in 34 (87.2%) of the 39 brain stem raphe gradings, resulting in a substantial agreement (Cohen’s unweighted κ = 0.782). There was unambiguous consent in the classifications of the substantia nigra, lentiform nuclei and caudate nuclei findings.
Comparison of TCS findings between patients with Huntington disease and healthy controls
The width of the third ventricle in patients with Huntington disease was significantly larger than in the control group (p < 0.001, Table 3). Seven (17.5%) controls had hyperechogenic lesions of the substantia nigra, 2 (5.0%) had hyperechogenic lesions of the caudate nuclei and 3 (7.5%) had hyperechogenic lesions of the lentiform nuclei. Patients with Huntington disease significantly more often had hyperechogenicities of the substantia nigra than healthy controls (p = 0.027). Additionally, we observed a trend toward greater frequency of caudate nuclei hyperechogenicities in patients with Huntington disease (p = 0.07). Four (10.0%) of the 40 controls exhibited reduced brain stem raphe echogenicity, and in 2 people (5.0%) the brain stem raphe was not visible. Thus, a pathologic brain stem raphe echogenicity was detected significantly more often in patients with Huntington disease than in controls (p = 0.002). The detected echogenicities of both groups are summarized in Table 3.
Evaluation of brain parenchyma echogenicity in patients with Huntington disease in comparison to healthy controls
Comparison of TCS findings between subgroups of patients with Huntington disease
A significant correlation between the width of the third ventricle and the cognitive score, as well as the functional capacity, could be observed in patients with Huntington disease (p < 0.001, rho = −0.57 and p = 0.025, rho = −0.37, respectively; Fig. 2).
Widths of the third ventricle in relation to cognitive and functional scores. Widths (in millimetres) of the third ventricle in patients with Huntington disease, measured with transcranial sonography in relation to (A) the cognitive scores and (B) functional capacity. A correlation between cognitive score and widths of the third ventricle (Spearman correlation, rho = −0.57, p < 0.001) as well as between functional capacity and widths of the third ventricle (Spearman correlation, rho = −0.37, p = 0.025) was observed.
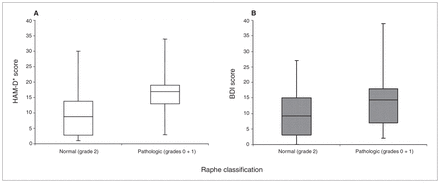
We found no correlation between clinical symptoms and hyperechogenicities of the substantia nigra, caudate nuclei or lentiform nuclei. Specifically, we observed no correlations between hyperechogenic caudate nuclei or lentiform nuclei and disease severity or special clinical features like dystonia in this patient population. Pathologic findings of the brain stem raphe correlated significantly with the presence of depressive symptoms depicted by HAM-D* and BDI scores (p = 0.014 and 0.031, respectively; Fig. 3). Fifteen of the 21 currently depressed patients with Huntington disease showed hypoechogenicity in the brain stem raphe, whereas among the 18 patients without depression at time of evaluation, a pathologic signal was depicted in 4 patients (p = 0.004, sensitivity = 71.4%, specificity = 77.8%; Table 4). All 9 patients without a history of depressive episodes had a regular echogenic signal of the brain stem raphe, whereas 19 of the 30 patients with at least 1 previous depressive episode exhibited hypoechogenicities in the brain stem raphe (p = 0.001; Table 4). Twelve (70.6%) of the 17 patients with Huntington disease who showed psychiatric disturbances prior to the occurrence of motor symptoms exhibited pathologic hypoechogenicity of the brain stem raphe, whereas 7 (31.8%) of the 22 patients showing motor symptoms as disease onset exhibited a pathologic signal (p = 0.025, sensitivity = 70.6%, specificity = 68.2%; Table 4). No correlations were seen between high UHDRS motor score and hypoechogenic brain stem raphe.
Echogenicity of the brain stem raphe in relation to depression scale scores. Normal and pathologic sonographic findings of mesencephalic raphe structures in patients with Huntington disease in relation to (A) the Hamilton Rating Scale for Depression* (HAM-D*)19 scores and (B) Beck Depression Inventory18 scores. A significant difference between normal or pathologic raphe-findings and HAM-D* scores (Mann–Whitney U, p = 0.014) as well as BDI scores (Mann–Whitney U, p = 0.031) was observed. HAM-D* excludes items 7–9, 13 and 16.
Sonographic evaluation of mesencephalic raphe nuclei in patients with Huntington disease and correlation to clinical data of depressive symptoms
Discussion
The main sonographic finding of our study is that patients with Huntington disease exhibiting a pathologic echogenicity of the brain stem raphe significantly more often exhibit depressive symptoms in the course of their disease than patients with normal raphe echogenicity. In our study, all patients with Huntington disease without any history of depressive episodes showed normal echogenicity of the raphe structures. Furthermore, in patients who showed psychiatric disturbances prior to the occurrence of motor symptoms, a pathologic raphe echogenicity was detected significantly more often.
The analysis of the relation between brain stem raphe echogenicity and mood status was the main focus of our study. It could be shown that patients with Huntington disease with depressive symptoms significantly more often exhibited reduced echogenicity of the brain stem raphe than patients without depressive symptoms. Such observations have been reported in patients with unipolar depression and in depressed patients with Parkinson disease.11–15 The precise pathophysiological and morphological interpretation of brain stem raphe hypoechogenicity is still speculative. A correlation of this finding to signal alteration on magnetic resonance imaging (MRI) studies has been reported previously, suggesting that hypoechogenicity reflects a structural disruption of the mesencephalic raphe structure.12
Positron emission tomography studies analyzing neuro-biological bases of emotional disturbances in patients with Huntington disease demonstrate a widespread reduction of activity within the frontal and parietal lobes.25 Mesencephalic raphe nuclei form complex serotonergic interconnections with forebrain structures, making an association between disrupted serotonergic projections at the midbrain level and frontal hypoactivity conceivable.12 However, 1 neuropathological study did not reveal neuronal pathology within the dorsal raphe nucleus in patients with Huntington disease.26 On the other hand, raphe hypoechogenicities have also been reported in patients with adjustment disorders and depressed moods.14 Studies have shown that major depression can be a consequence of being genetically tested for Huntington disease.27 High rates of affective disturbances have also been reported in patients’ family members who do not carry the genetic mutation, leading to the hypothesis that depressive symptoms arise in response to emotional stressors, such as being at risk for Huntington disease or the burden of growing up in a family with affected members.28 From this point of view, brain stem raphe hypoechogenicity might be a finding reflecting a predisposition for depressive reaction, independent from the neurodegenerative process in Huntington disease. In the patients included in our study, the occurrence of depressive symptoms before motor symptoms was related to the sonographic finding of raphe echogenicity. This might support the idea of an increased liability to depressive response to emotional stressors. Previous findings in patients with adjustment disorders and depressed moods support this interpretation.14 Interestingly, in our study, all 9 patients with Huntington disease without previous depression exhibited a regular raphe echogenicity. To our knowledge, there have not been studies that use TCS for early detection of depression or to identify prospectively those at risk for depression in any population.
Substantia nigra hyperechogenicities were first described in patients with Parkinson disease.7 A review of 31 subsequent studies revealed that substantia nigra hyperechogenicities were present in 87% of the 1334 published cases.29 These hyperechogenicities are marked in 73%–79% and moderate in 20%–25% of the cases.9,23 It is assumed that substantia nigra hyperechogenicity is based on increased amounts of iron bound to proteins other than ferritin.23,30
Although these findings are characteristic for idiopathic Parkinson disease, less frequently, hyperechogenicities of the substantia nigra were reported in other conditions, such as spinocerebellar ataxia, depression and Huntington disease.15,16,31
In a study of 45 patients with Huntington disease, Postert and colleagues16 observed hyperechogenicites of the substantia nigra in 12 (27%) patients, of the caudate nuclei in 6 (13%) patients and of the lentiform nuclei in 4 (9%) patients. These authors reported that the number of CAG repeats and the severity of the disease correlated with the identification of hyperechogenicities of the substantia nigra. We could not confirm a significant correlation between CAG repeats and substantia nigra hyperechogenicity (p = 0.57). Since we excluded patients with higher grades of dementia from our study, the cohort was different and a replication of reported findings was not applicable.
We could demonstrate that pathologic signal of the caudate nuclei was more frequent in patients with Huntington disease than healthy controls (p = 0.07). An association between caudate nuclei hyperechogenicity and increased signal intensity in T2-weighted MRIs in patients with Huntington disease has been reported previously.16 It is known that gliotic lesions account for high signal intensity in MRI.32 Hence, this may support the hypothesis that sonographic detection of hyperechogenicity reflects neurodegeneration. On the other hand, signal alteration in MRI is also caused by accumulation of metals.33 Neuroradiological examinations revealed increased ferritin iron levels in the basal ganglia of patients with Huntington disease.34 This condition may lead to changes of iron–protein binding, which might cause echogenic alterations similar to substantia nigra hyperechogenicities in Parkinson disease.23 Moreover, molecular findings indicate that huntingtin plays an essential role in cellular iron homeostasis.35 Additionally, postmortem studies showed that, in addition to iron, copper levels are increased in patients with Huntington disease.34 Copper accumulation is supposed to cause hyperechogenicities primarily of the lentiform nuclei in patients with Wilson disease.10 Lentiform nuclei hyperechogenicitiy is also a frequent finding in patients with cervical or upper-limb dystonia.36 Neuropathological examinations in patients with idiopathic dystonia revealed significantly increased copper levels in the globus pallidum and putamen compared with control brain samples.37 Further neuropathological and multimodal imaging studies are needed to elucidate the pathogenesis of the hyperechogenicities in patients with Huntington disease.
Regarding brain atrophy, the sonographic evaluation of the ventricular system, including ventricular enlargement, has been described previously.38 The width of third ventricle serves as a marker of cerebral atrophy in the sonographic discrimination between corticobasal degeneration and progressive supranuclear palsy.39 As expected, the patients with Huntington disease in our study showed significantly larger widths of the third ventricle than healthy controls. Furthermore, we documented a correlation between the extent of the ventricular enlargement and cognitive score as well as functional capacity. Recently, voxel-based morphometric studies have described a correlation between cognitive dysfunction and cerebral atrophy.40
Limitations
Most of the patients in our study were taking antichoreatic medication, which particularly influences neurologic status. Thus, a meaningful interpretation of the correlation between TCS findings and neurologic features was limited. Moreover, there are some general limitations of transcranial sonography that need to be taken into account. Owing to insufficient transtemporal bone window, the midbrain structures in 5%–10% of white people can be only partially assessed.8 In addition, the area that can be evaluated sonographically is limited to deep grey matter structures, therefore other relevant structures, such as cortical regions, are excluded from the examination. Furthermore, the reliability of the findings is dependent on the quality of the ultrasonography system and on the qualification of the investigator.
Conclusion
Our study reveals a particular pattern of hyperechogenicities in patients with Huntington disease. We observed a correlation between the sonographic evaluation of ventricular enlargement and cognitive and functional features. Moreover, a relation between brain stem raphe echogenicity and depressive symptoms could be identified. An alteration of the serotonergic brain stem raphe might be involved in the pathogenesis of depression in patients with Huntington disease. Transcranial sonography is a commonly available, noninvasive and inexpensive diagnostic tool that provides reliable information about the morphology of the brain in Huntington disease, even in agitated patients who do not tolerate other imaging techniques.
Future research on this topic should focus on examinations of presymptomatic patients with Huntington disease, so that the value of TCS in detecting patients at risk for depressive syndromes can be tested in a prospective and longitudinal design.
Footnotes
* Drs. Ninphius and Saft contributed equally to this work.
Competing interests: None declared for Drs. Krogias, Strassburger, Norra and Ninphius. Dr Eyding declares having received speaker fees from Bracco. Dr. Gold declares board membership with Elan, Biogen Idec, TEVA, MerckSerono and Bayer; he has consulted for them and has received grant funding and speaker fees from them; he has a patent filed with Biogen. Dr. Juckel declares having received grants and speaker fees from Janssen-Cilag, Lilly, AstraZeneca, Pfizer, Lundbeck, BMS, Wyeth and Bayer Vital. Dr. Saft declares having received grants from TEVA, speaker fees from Temmler Pharma, travel assitance from the European Huntington’s Disease Network and compensation from NeuroSearch for the ACR-16 study and from Novartis for the AFQ-056 study.
Contributors: Drs. Krogias, Saft and Ninphius designed the study. Drs. Krogias, Strassburger, Eyding, Saft and Ninphius acquired the data, which all authors analyzed. Dr. Krogias wrote the article, and the other authors critically reviewed it. All authors approved publication of the article.
- Received April 11, 2010.
- Revision received July 15, 2010.
- Accepted July 16, 2010.