Abstract
Background: Deficits in prepulse inhibition may be a common feature in first-episode schizophrenia, bipolar disorder (BD) and major depressive disorder (MDD). We sought to explore the levels and viability of prepulse inhibition to differentiate first-episode schizophrenia, BD and MDD in patient populations.
Methods: We tested patients with first-episode schizophrenia, BD or MDD and healthy controls using prepulse inhibition paradigms, namely perceived spatial co-location (PSC-PPI) and perceived spatial separation (PSS-PPI).
Results: We included 53 patients with first-episode schizophrenia, 30 with BD and 25 with MDD, as well as 82 healthy controls. The PSS-PPI indicated that the levels of prepulse inhibition were smallest to largest, respectively, in the first-episode schizophrenia, BD, MDD and control groups. Relative to the healthy controls, the prepulse inhibition deficits in the first-episode schizophrenia group were significant (p < 0.001), but the prepulse inhibitions were similar between patients with BD and healthy controls, and between patients with MDD and healthy controls. The receiver operating characteristic curve analysis showed that PSS-PPI (area under the curve [AUC] 0.73, p < 0.001) and latency (AUC 0.72, p < 0.001) were significant for differentiating patients with first-episode schizophrenia or BD from healthy controls.
Limitations: The demographics of the 4 groups were not ideally matched. We did not perform cognitive assessments. The possible confounding effect of medications on prepulse inhibition could not be eliminated.
Conclusion: The level of prepulse inhibition among patients with first-episode schizophrenia was the lowest, with levels among patients with BD, patients with MDD and healthy controls increasingly higher. The PSS-PPI paradigm was more effective than PSC-PPI to recognize deficits in prepulse inhibition. These results provide a basis for further research on biological indicators that can assist differential diagnoses in psychosis.
Introduction
Patients with schizophrenia and patients with bipolar disorder (BD) overlap in clinical manifestations, physiology and genetics,1,2 and studies on cognitive impairments, neural circuit disruptions and electrophysiologic deficits indicate notable similarities.3–5 Major depressive disorder (MDD) and BD have also shown close resemblances in phenomenology, neurobiology and pathogenesis. Although schizophrenia, BD and MDD share clinical symptoms, particularly in the mood and psychotic dimensions, they differ in clinical course and progression.6 Unfortunately, no robust biological indicators or pathological methods are currently known to assist the clinical diagnosis of first-episode schizophrenia, BD or MDD, or the evaluation of treatment effect.
The startle reflex can interrupt a variety of ongoing processes, including coherent thought. When a nonstartling prestimulus signal (S1) precedes a stronger, unexpected target stimulus (S2) by an appropriate lead interval, the startle response to S2 is typically reduced. This phenomenon is termed prepulse inhibition. Prepulse inhibition can be used to quantitate sensorimotor gating, a normal brain function, as prepulse inhibition reflects the ability of the sensorimotor gating of the brain to block interfering information.7,8 A deficiency of sensorimotor gating can lead to various psychotic symptoms, such as hallucinations, delusions and disordered speech acts.7,9 In schizophrenia, dysfunctional sensorimotor gating is considered an endophenotype, a stable phenotype with a genetic foundation.10 For testing humans, the stimuli used to measure prepulse inhibition are usually acoustic, and the startle response is the eyeblink reflex, measured by the amplitude and latency of the orbicularis oculi muscle to the startling stimulus.
In 1978, patients with schizophrenia were observed to have lower levels of prepulse inhibition than healthy controls; their startle response to S2 was not as dampened by the prepulse S1.11 Many studies have shown that deficits in prepulse inhibition exist across different stages of schizophrenia, including first episodes, chronic or acute episodes and stable remission.12–14 The degree of prepulse inhibition deficits has been associated with the severity of clinical symptoms, especially illusion and delusion, as measured using the Positive and Negative Syndrome Scale (PANSS).15–17 Deficits in prepulse inhibition have also been noted in the unaffected, first-degree relatives of patients with schizophrenia, suggesting a heredity basis.16,17 Other studies have shown positive associations between cognitive impairments and prepulse inhibition deficits, especially in the attention dimension; patients with poor attention ability had more serious prepulse inhibition deficits.18 Evidence has shown that focusing on the prepulse improves prepulse inhibition.19–24 This verifies that attentional processing has a top–down regulating effect on sensorimotor gating.
Prepulse inhibition has been associated with changes in attention to prepulse, and these changes also correlate with the severity of specific symptoms in schizophrenia.9 Using the perceived spatial separation (PSS-PPI) paradigm — whereby the leading ear given background noise is different from the ear receiving the prepulse stimulus — a study found that the PSS-PPI positively correlated with the speed of processing, attention or vigilance and social cognition in unmedicated, first-episode schizophrenia.25 Among patients with BD, the language domain of the Repeatable Battery for the Assessment of Neuropsychological Status positively correlated with both the PSS-PPI and the perceived spatial co-location (PSC-PPI, in which the leading ear given background noise is the same ear receiving the prepulse stimulus).26 In the same study, double word time and colour interference time negatively correlated with the PSS-PPI. Other studies have reported associations between some cognitive domain functions and prepulse inhibition.27–30
Deficits in prepulse inhibition have been verified repeatedly in studies of schizophrenia,9–11 but also among patients with BD31 and their first-degree relatives,31 as well as patients with obsessive–compulsive disorder,32 attention-deficit/hyperactivity disorder,33 Tourette syndrome34 and Huntington disease.35 However, studies regarding prepulse inhibition deficits in BD and MDD are limited and controversial. Giakoumaki and colleagues31 observed deficits in prepulse inhibition among remitted patients with BD and their unaffected first-degree relatives. This was evidence that prepulse inhibition deficit is a genetically mediated abnormality in BD, yet no associations were found between prepulse inhibition and symptoms, medication or disease severity. Another study reported that patients with euthymic BD had obvious deficits in prepulse inhibition, but with no link to a history of psychotic symptoms. Furthermore, among patients with BD, prepulse inhibition deficits were reflected by performance on tasks related to emotional processing, but with no association with any other clinical or neurocognitive function.36 Among patients with BD without psychotic symptoms, other studies found no deficits in prepulse inhibition, either during acute manic or mixed episodes. Therefore, the authors concluded that prepulse inhibition deficits may be a feature only in BD with psychotic symptoms.37,38
Studies about prepulse inhibition among patients with MDD are especially rare. Some studies reported no significant deficits in prepulse inhibition.39–41 In 2013, a review concluded that no deficiency in prepulse inhibition in MDD has been consistently demonstrated.42 However, a large single study from Japan found that male, but not female, patients with MDD had significant deficits in prepulse inhibition.43
We hypothesized that patients with first-episode schizophrenia, BD and MDD differ regarding the extent of prepulse inhibition deficiency, and if so, this information may be useful toward differentiating these disorders. Thus, we sought to explore the prepulse inhibition deficits of the acoustic startle reflex in patients with first-episode schizophrenia, BD and MDD. Furthermore, we sought to conduct a preliminary analysis to determine the sensitivity and specificity of the effect size of prepulse inhibition deficits to differentiate patients with first-episode schizophrenia, BD and MDD from healthy controls.
Methods
The Ethics Committee of Beijing Anding Hospital, Capital Medical University approved this study (no. 2015127FS-2). All participants or their guardians were informed of the study and provided signed informed consent.
Participants
We recruited outpatients and inpatients aged 18–55 years from Beijing Anding Hospital, Capital Medical University. We required patients in the first-episode schizophrenia group to have a diagnosis of schizophrenia based on screening with the Structured Clinical Interview for DSM-IV Axis I Disorders, Patient Edition (SCID-I/P),44 duration of untreated psychosis of less than 5 years and use of antipsychotic medication for fewer than 14 days.45 Similarly, we required patients in the BD and MDD groups to meet the diagnostic criteria for BD or MDD based on a SCID-I/P screening.44
We required recruits for the healthy control group to have no family history of mental illness, screened through the SCID-I/P to show no mental illness. We matched healthy controls generally with the patient groups in terms of age, gender ratio and education.
We excluded potential participants with neurologic disorders, history of alcohol or drug dependence, or suicidal or violent tendencies, as well as those who received modified electroconvulsive therapy within the previous 6 months or who failed to respond to the startle stimulus (hearing threshold > 40 dB SPL).
Clinical assessments
We collected the demographic information and clinical characteristics of the patients using a questionnaire designed for the study. We assessed patients with first-episode schizophrenia using the PANSS.46 We assessed patients with BD or MDD using the Young Mania Rating Scale (YMRS),47 the Hamilton Depression Rating Scale (HAMD)48 and the Hamilton Anxiety Rating Scale (HAMA).49
Intelligence quotient (IQ) test
We used the Chinese version of the simple Wechsler Adult Intelligence Scale (WAIS-RC) to evaluate the IQ of the participants.50
Prepulse inhibition
We used an Xeye Human Startle Reflex system (Tian Ming Hong Yuan Instruments) with Gaussian white noise, a sampling rate of 48 kHz and a duration of 15 seconds, generated with the “randn ()” packet in MATLAB software (Math-Works). We used low-pass digital filters with a cutoff frequency of 10 kHz to produce broadband noise as background noise. We generated a prepulse stimulus sound with a broadband noise of 150 ms and a startle stimulus sound of 40 ms. To avoid energy spatter, after the introduction of prepulse and startle sounds, we filtered each sound stimulus.
Finally, we set the startle stimulus at 100 dB sound pressure, with a white noise duration of 40 ms. We set the prepulse stimulus to 150 ms, 65 dB sound pressure white noise with 3 ms binaural delay. We set the background noise at 60 dB sound pressure, white noise with 3 ms binaural delay. The interstimulus interval between the prepulse and startle stimulation was 120 ms (then the lead time was 270 ms). We filtered the electromyography signal with a high pass of 10 Hz and a low pass of 500 Hz, and amplified the signal amplitude by a factor of 40 000. At the same time, the sampling frequency was 1000 Hz (collected once in 1 ms) during the recording process, and the acquisition accuracy was 0.008 μV. The data were collected within 250 ms before and 300 ms after the startle stimulus, and were rectified with absolute values.
The experiment required the generation of 6 stimulus sounds. The first 2 stimulus sounds were pulse-alone trials, with binaural delay of the background noise with the right ear leading (RPA) or left ear leading (LPA). The remaining trials used combinations of background noise with right or left ear leading, and prepulse and pulse stimuli with right or left ear leading (RNR, RNL, LNL, LNR). An interstimulus interval of 120 ms was designated as RNR120, LNR120, RNL120 or LNL120, as applicable.
All participants received the same instructions to focus on the prepulse stimulus sounds heard in the right (or left) ear, and count, and then report the number of sounds heard. We told them, “You may hear a few loud sounds, which are irrelevant to the task and can be ignored.” Participants subjectively felt that the 2 sounds (i.e., the background noise and prepulse stimulus) came from different directions, and could use spatial cues to increase the recognition of the target stimulus.
The entire test consisted of 2 blocks (block 1, block 2). The stimulation sequence of each block was a repeating combination of a series of sound stimuli. The background noise of block 1 was always in the left ear leading, persisting throughout the block 1 trials. The LPA, LNL120 and LNR120 trials (with an average interval of 15 s between each trial) were randomly played. For block 2, the background noise was always in the right ear leading, persisting throughout the block 2 trials. The RPA, RNR120 and RNL120 trials (with an average interval of 15 s between each trial) were played randomly.
Stimulation response measurements
We asked participants to sit in a chair in a soundproof room, with their eyes focused on the black spot in the centre of the front screen and to minimize blinking. We did not permit smoking for at least 30 minutes before the test. We used auditory testers to screen the participants with a pure tone audiometry threshold of 40 dB or less and a threshold difference of 15 dB or less between the ears. The bilateral sound stimulus was delivered via a Sennheiser headphone (HD600).
We used the Xeye Human Startle Reflex System (Tian Ming Hong Yuan Instruments) to record the right orbicularis oculi electromyogram of participants and to startle stimulation through an AG/AgCl electrode (inner diameter 4 mm) filled with conductive paste. The recording electrode was located about 1 cm below the pupil of the right eye, and the reference electrode was about 1 cm outside the lateral canthus of the right eye. Each electrode resistance was less than 5 kΩ. The right posterior mastoid electrode was grounded to eliminate the influence of the 50-Hz current.
Before the formal experiment, only the startle stimulus sound was played twice. We excluded participants who did not respond to the startle stimulus. Immediately thereafter, the background noise and prepulse stimulation were played. Participants were required to familiarize themselves with the test sound, and judge the direction of the leading ear of the background noise and the prepulse stimulation. A correct rate of 80% was a qualification to continue to the next experiment. In the formal test, we asked participants to concentrate on hearing the prepulse stimuli in the right (or left) ear and report the number of sounds heard.
Valid trial
We conducted a detailed examination of each trial to remove the myoelectric response caused by automatic blinks, and determined the amplitude mean of the sampling period and maximum peak amplitude of each trial. We considered the trial valid if the maximum peak amplitude was greater than 4 times the amplitude mean of the sampling period or if the amplitude mean of the sampling period was greater than the amplitude mean of the response period.
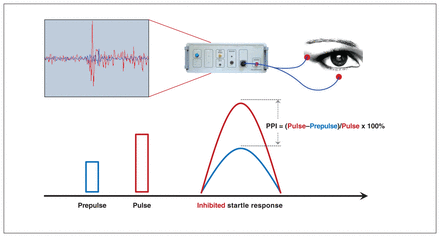
We defined prepulse inhibition as the percentage reduction in startle response that was due to the prepulse; that is, the result of subtracting the startle response after prepulse from the startle response in the absence of a prepulse, divided by the startle response in the absence of a prepulse, multiplied by 100% (Figure 1). We calculated habituation as the result of 1 minus the average amplitude of the acoustic startle response in block 2, divided by the average acoustic startle response in block 1, multiplied by 100. We determined the maximum peak latency according to the specific experimental paradigm, ranging from 50 ms to 550 ms.
Diagram of electrode placement in the prepulse inhibition (PPI) test.
Statistical analysis
We established a database using Epidata 3.1 for Windows and analyzed data with IBM SPSS Statistics 23.0 for Windows software. The valid trial prepulse inhibition data were selected and processed with R language software. We used 1-way analysis of variance (ANOVA) to analyze the general demographic and clinical data, and χ2 tests for enumeration data. When dealing with prepulse inhibition data of the 4 groups (patients and healthy controls), gender, age and education were concomitant variables in the analysis of covariance (ANCOVA). Multiple comparisons used the Bonferroni correction.
Taking the PSS-PPI 120 and PSC-PPI 120 as predictive factors and the healthy controls as the reference, we used receiver operating characteristic (ROC) curve analysis to test the differentiating value of prepulse inhibition. In general, when the area under the ROC curve (AUC) was less than 50%, the diagnostic value was not sufficient. The AUC was used for reference only if values were between 50% and 70%; diagnostic accuracy increases with percentage. We used the Cohen d effect size to distinguish between 2 groups, defined as small (0.2–0.5), medium (0.51–0.8) and large (> 0.8) effects.51 The level of significant difference was p less than 0.05.
Results
Demographics and clinical characteristics
In the patient groups with first-episode schizophrenia, BD and MDD, there were 58, 33 and 27 participants, respectively ( Table 1). We excluded 5, 3 and 2 participants, respectively, because they did not respond to the startle stimulus. The healthy control group comprised 90 people, and 8 of them were excluded for the same reason. The rates of smoking, married and living alone were similar across the 4 groups. There were significant differences among the 4 groups in age (p = 0.006), gender (p = 0.045), education (p = 0.015) and IQ (p < 0.001). Notably, patients in the MDD group were older than those in the other groups. Most patients in the BD group were male (70%), and the reverse was true of the MDD group (68% female). Patients with MDD and healthy controls had more years of education than patients with first-episode schizophrenia or BD. The IQs of patients with BD and first-episode schizophrenia were significantly lower than those of healthy controls. The YMRS scores of the BD group were significantly higher than those of the MDD group, while the HAMD and HAMA scores were lower.
Demographic and clinical characteristics of the study population
Prepulse inhibition of the 4 groups
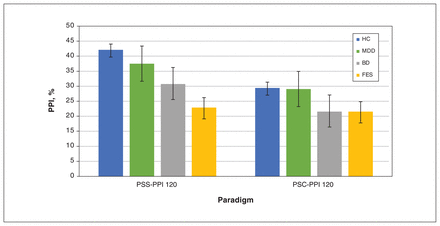
The results of the comparison of prepulse inhibition from the 4 groups are shown in Table 2 and Figure 2. According to the PSC-PPI paradigm, there were no differences in prepulse inhibition across the 4 groups (F = 1.48, p = 0.22), while the differences in prepulse inhibition were significant when the PSS-PPI paradigm was applied (F = 7.61, p < 0.001). Comparisons of the prepulse inhibition of the participants showed no differences between males and females in PSC-PPI (F = 1.756, p = 0.187), PSS-PPI (F = 2.283, p = 0.132) and latency (F = 1.188, p = 0.277). With gender, age and education as covariates, the same conclusion applied, with no significant differences in prepulse inhibition in the PSC-PPI paradigm (F = 1.54, p = 0.21), but significant differences in the PSS-PPI paradigm (F = 6.75, p < 0.001). Under the PSS-PPI paradigm, the levels of prepulse inhibition of the 4 groups in decreasing order were healthy controls, patients with MDD, patients with BD and patients with first-episode schizophrenia. In addition, the latencies of the 4 groups were significantly different (F = 44.71, p < 0.003).
Comparison of prepulse inhibition (PPI) among healthy controls (HC), patients with major depressive disorder (MDD), patients with bipolar disorder (BD) and patients with first-episode schizophrenia (FES). Note: PSC-PPI = perceived spatial co-location paradigm of prepulse inhibition, PSS-PPI = perceived spatial separation paradigm of prepulse inhibition. *p < 0.001.
Comparisons of neuroelectrophysiological measurements
Paired group comparisons of prepulse inhibition
The post hoc Bonferroni test showed that PSS-PPI deficits were more pronounced in the first-episode schizophrenia group than the control group (p < 0.001, d = 0.90), and the latency was significantly different between the BD group and the control group (p = 0.004, d = 0.78; Table 2). However, other paired comparisons of prepulse inhibition among the first-episode schizophrenia, BD, MDD and control groups showed no statistical difference. In the PSS-PPI paradigm, compared with healthy controls, effect sizes for the first-episode schizophrenia, BD and MDD groups were 0.90, 0.46 and 0.19, respectively. In the PSC-PPI paradigm, there were no differences in prepulse inhibition among the paired group comparisons.
ROC curves
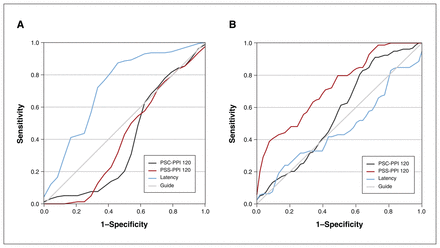
Using PSS-PPI 120, PSC-PPI 120 and latency as predictive factors and healthy controls as the reference, the ROC curve analysis showed that the PSS-PPI 120 had a significant differentiating value between patients with first-episode schizophrenia and healthy controls (AUC = 0.73, p < 0.001; Figure 3), with a sensitivity of 41.3% and a specificity of 92.5%. Latency could differentiate patients with BD from healthy controls (AUC = 0.72, p < 0.001; Figure 3), with a sensitivity of 86.3% and a specificity of 58.3%. However, there were no positive results based on the PSC-PPI 120.
Receiver operating curve of perceived spatial separation paradigm of prepulse inhibition (PSS-PPI), perceived spatial co-location paradigm of prepulse inhibition (PSC-PPI) and latency for discrimination of patients with first-episode schizophrenia (FES) and patients with bipolar disorder (BD).
Discussion
We found that patients with first-episode schizophrenia exhibited significant deficits in prepulse inhibition, relative to healthy controls. Patients with first-episode schizophrenia had the lowest level of prepulse inhibition, followed by patients with BD, patients with MDD and healthy controls. In addition, compared with the PSC-PPI 120, PSS-PPI 120 was a more effective paradigm for differentiating patients with schizophrenia. The PSS-PPI 120 appears to be a promising, objective approach for identifying schizophrenia. However, deficits in prepulse inhibition are a feature of psychiatric diseases generally, with variations of degree in each, and there is insufficient evidence that prepulse inhibition can be used for diagnosis. In future studies, more specific modifications of prepulse inhibition, or combining other biomarkers with prepulse inhibition, may prove more helpful toward diagnoses in psychosis.
Deficits in prepulse inhibition may be considered a biomarker among patients with psychosis. In the present study, the 4 groups (patients with first-episode schizophrenia, patients with BD, patients with MDD and healthy controls) displayed different levels of prepulse inhibition. Patients with first-episode schizophrenia had prominent deficits in prepulse inhibition relative to the control group. This is consistent with a previous study.11 After Bonferroni correction, we observed that levels of prepulse inhibition of patients with BD and patients with MDD were statistically comparable to those in healthy controls. This concurs with some other studies.38–41,52,53 However, although studies regarding prepulse inhibition deficits in schizophrenia have been relatively consistent, there is controversy concerning BD and MDD. Patients with MDD have shown normal levels of prepulse inhibition, which may be why prepulse inhibition studies in MDD are scarce. Inconsistencies could be related to different paradigms or disease states, including stages of disease such as mixed episodes or stable remission.37,52,54
One study found genetic associations in 5 major mental diseases (schizophrenia, BD, MDD, autism spectrum disorders and attention-deficit/hyperactivity disorder), with specificity ranging from 17% to 28%. The lack of prepulse inhibition specificity may then be attributed to overlapping genetic variants that are common to psychiatric disorders.55 Many patients who display symptoms on a continuum between schizophrenia and BD have traditionally been considered schizoaffective. A dimensional approach to the psychosis spectrum has been used between BD and schizophrenia.5 In the present study, we found different degrees of prepulse inhibition in MDD, BD and first-episode schizophrenia. This provides support for studying these diseases as a continuous spectrum.
Deficits in prepulse inhibition may be related to a psychopathological mechanism that is common to various mental diseases.32,36,56,57 Therefore, prepulse inhibition deficits are a relatively specific biomarker for the presence of psychotic disorders, but lack differentiating specificity. We did find that the prepulse inhibition levels of the 4 groups decreased from healthy controls to patients with MDD, patients with BD and, finally, patients with first-episode schizophrenia. Other research using cognitive and structural magnetic resonance imaging (MRI) has found similar patterns. A structural MRI study found that the grey matter volumes of the temporal pole were different across the 4 groups, from largest to smallest among healthy controls, patients with MDD, patients with BD and patients with schizophrenia.58 The 4 groups also showed neurocognitive differences (i.e., speed of processing, attention and vigilance, working memory, Hopkins Verbal Learning and composite T scores), with scores from highest to lowest among healthy controls, patients with MDD, patients with BD and patients with first-episode schizophrenia.59 We found similar phenomena in neuroelectrophysiology, neuroimaging and neuropsychology; the 3 disease groups showed the same trend in these different deficit fields. Additional similar studies are needed to provide a basis for clinical differential diagnoses and objective biological indicators in the field of psychiatry.
Previous studies reported that prepulse inhibition could be enhanced when the participants focused on the prepulse;24,60 attention is important for identifying relevant spatial locations and task-related objects.61 The PSS-PPI may enhanced among healthy controls because of intact selective attention to the focused stimulus, which is severely disrupted among patients.62 The PSS-PPI paradigm has been studied among animals and patients with schizophrenia.27,63 In the current study, we used both the PSC-PPI and PSS-PPI to measure prepulse inhibition. The PSS-PPI paradigm integrated selective attention and auditory preference, improving the effectiveness for measuring deficits in sensorimotor gating. Only the PSS-PPI paradigm showed significant differences among the 4 groups; there was no positive result in the PSC-PPI. This is consistent with other studies showing that patients with schizophrenia had significant deficits in prepulse inhibition, especially when applying the PSS-PPI paradigm.11,42,64–66 The same conclusion was found in studies involving patients with BD, in which patients showed significant deficits in prepulse inhibition compared with healthy controls using the PSS-PPI paradigm, but with no statistical differences when using the PSC-PPI paradigm.26
In animal studies, it has been observed that perceptual spatial separation can increase prepulse inhibition by enhancing selective attention.67,68 In humans, higher education levels, attention scores and total composite scores of neurocognition tasks have been shown to contribute to PSS-PPI, and patients with recurrent episodes of schizophrenia showed poorer PSSPPI.27 Correlational and stepwise regression analyses revealed that the underlying mechanisms of PSS-PPI included modulating attention.27 Therefore, the PSS-PPI is more effective and advantageous than the PSC-PPI for recognizing deficits in prepulse inhibition.
In the present study, the PSS-PPI 120 showed great specificity, a large effect size and medium sensitivity for differentiating patients with first-episode schizophrenia from healthy controls. Latency could significantly distinguish patients with BD from healthy controls with moderate sensitivity and specificity. Prepulse inhibition was not sufficiently objective as a biological indicator for differential diagnosis, because the necessary correlation between prepulse inhibition and severity of clinical symptoms was lacking and because prepulse inhibition deficits are a feature of many mental disorders.
A previous study suggested that emotional learning (e.g., auditory fear) can enhance prepulse inhibition, with top–down modulation.67 Considering the influence of emotional factors, such as fear conditioning in regulation of prepulse inhibition, was helpful to the specificity of prepulse inhibition testing for BD and MDD, which are mainly characterized by emotional or mood changes.69 Previous studies have been too limited to confidently advocate prepulse inhibition as an objective index to predict and differentiate mental diseases. More studies are needed to explore objective biological indicators. Combining different paradigms of prepulse inhibition to increase relevance in specific situations may be fruitful.
Limitations
The 4 study groups were not ideally matched by demographic characteristics, such as age, education and IQ. In analyses, these variables were corrected. We did not perform any cognitive assessments. In addition, patients were receiving psychotropic drugs (e.g., mood stabilizers, antipsychotics, antidepressants) singly or in combinations, and equivalent dosages were too complex to calculate. We cannot rule out the possible effect of medications on levels of prepulse inhibition. The analysis did not account for onset latency. Comprehensive assessments and strict control of all influencing factors are required to examine the clinical and scientific value of prepulse inhibition.
Conclusion
This study determined that patients with first-episode schizophrenia displayed distinct deficits in prepulse inhibition compared with controls, whereas patients with BD and patients with MDD showed no significant prepulse inhibition deficits. Although using deficits in prepulse inhibition to distinguish among specific psychiatric disorders is challenging, the present study groups did differ, with the first-episode schizophrenia group having the lowest degree of prepulse inhibition, followed by the BD group and then the MDD group. In addition, the PSS-PPI paradigm was more effective than the PSC-PPI for recognizing prepulse inhibition deficits. By applying the PSS-PPI paradigm, the larger effect size enabled differentiating patients with first-episode schizophrenia from healthy controls. Latency had value for differentiating patients with BD from healthy controls, with moderate effect size. This study contributes to research on biological indicators for differential diagnosis. Future studies should include larger samples and assess the predictive validity of combining other biomarkers with prepulse inhibition to help in the diagnosis of psychosis.
Acknowledgements
The authors thank all study participants.
Footnotes
↵* These authors contributed equally to this work.
Competing interests: None declared.
Contributors: Yue Sun, Qijing Bo, Liang Li and Chuanyue Wang contributed to study conception and design. Zhen Mao and Qing Tian acquired the data. Fang Dong analyzed and interpreted the data. Yue Sun, Qijing Bo and Liang Li drafted the article. All of the authors revised it critically for important intellectual content, gave final approval of the version to be published and agreed to be accountable for all aspects of the work.
Funding: This work was supported by Beijing Hospitals Authority Clinical Medicine Development of Special Funding (no. ZLRK202335) and National Natural Science Foundation (no. 81971250).
- Received May 25, 2023.
- Revision received August 10, 2023.
- Revision received September 18, 2023.
- Accepted September 27, 2023.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/