Abstract
Objective: It has been hypothesized that abnormal negative feedback of cortisol release in major depressive disorder (MDD) may involve impaired central glucocorticoid receptor (GR) function. Beclomethasone-induced vasoconstriction (BIV) was recently used to test the hypothesis that impaired GR function generalizes to peripheral tissues, and it was reported that BIV was decreased in medicated patients with MDD. The objective was to test the hypothesis that BIV would be reduced in unmedicated women with MDD compared with healthy controls.
Design: Case–control.
Setting: A university womens’ mental health research unit.
Participants: Women aged 18–65 years (n = 19) diagnosed, according to the Diagnostic and Statistical Manual of Mental Disorders, fourth edition, with MDD after a structured interview and clinical assessment. Healthy women pair-matched for age, reproductive and smoking status.
Procedures: BIV was tested using a range of beclomethasone dipropionate concentrations (1–100 μg/mL) applied to the forearm, with vasoconstriction scored visually after 15–18 hours by raters blinded to diagnosis and the randomization of the application sites.
Outcome measure: Visual scores for BIV at each beclomethasone concentration.
Results: No significant differences between patients with MDD and controls were found. Postmenopausal women showed less of a response than premenopausal women or women taking sex-hormone preparations.
Conclusion: The study did not concur with the previous finding that BIV is decreased in MDD. Further research is needed to determine whether the difference in findings is due to medication or to other factors that may have distinguished the samples, including sex, age, reproductive status, illness severity, treatment resistance and setting.
Introduction
Recent reviews suggest that abnormalities of the hypothalamic-pituitary-adrenal (HPA) axis found in major depressive disorder (MDD) are important for understanding the pathogenesis of the disorder.1,2 Findings that include hypercortisolemia and a lack of suppression of cortisol in the dexamethasone-suppression test or the combined corticotropin-releasing hormone dexamethasone test have been postulated to involve an increase in the combined effects of the releasing factors, corticotropin-releasing hormone CRH and vasopressin, and a decrease in negative feedback control. Glucocorticoid receptors (GRs) are sensitive to changes in circulating cortisol and are thought to limit stress-induced increases in cortisol. A defect in GR-mediated feedback has been proposed as an explanation of HPA axis overactivity in depression, and on the basis of a lack of Cushingoid stigmata in hypercortisolemic depressed patients and of abnormalities in GRs in peripheral blood cells, some authors have suggested that GR dysfunction may generalize to tissues outside the HPA axis.1,2 It was recently reported that patients with MDD also showed reduced skin vasoconstriction in response to topical application of the potent glucocorticoid, beclomethasone (beclomethasone-induced vasoconstriction, BIV), further suggesting a peripheral GR abnormality in major depression.3 However, all of the patients studied were receiving antidepressants. The aim of the present study was therefore to test the hypothesis that BIV would be reduced in unmedicated women with MDD compared with healthy controls.
Methods
The study was approved by the university research ethics board. Women aged 18–65 years were recruited via notices and local contacts and gave written informed consent to participate. Subjects were screened for axis I diagnoses using the Mini-International Neuropsychiatric Interview.4 Patients were also fully assessed by an experienced clinician and were included if they met the Diagnostic and Statistical Manual of Mental Disorders, fourth edition, criteria for MDD. All cases scored in the moderate range of severity (17-item Hamilton Depression Rating Scale mean 21, range 17–27) and none met full criteria for atypical features. Controls were free of lifetime axis I disorders. Other exclusion criteria were: psychotic depression; depression or suicide risk of severity that would make it inappropriate to delay treatment until completion of testing in the follicular phase of the menstrual cycle; pregnancy or lactation; a lifetime history of schizophrenia, bipolar disorder or post-traumatic stress disorder (PTSD); history of an alcohol or substance use disorder within 3 months; use of corticosteroids within 3 months. None of the patients had been taking psychotropic medication more recently than 3 months before the study. Reproductive status and hormonal treatments are a source of variance in HPA axis function,5 and controls were therefore pair-wise matched for reproductive status, in addition to age and smoking (1 pair of subjects). Seven pairs were premenopausal, 6 postmenopausal and 6 were on hormonal treatments (birth control or hormone replacement). Subjects did not differ in weekly alcohol intake or body-mass index (Table 1).
Subject characteristics and waking and afternoon salivary cortisol concentrations for women with major depressive disorder (MDD) and healthy matched controls
Procedures
Self-ratings were made for the last week on the Mood and Anxiety Symptoms Questionnaire short form (MASQ), a 62-item 5-point scale that discriminates depressive anhedonia, general distress and anxious arousal,6 and for the last month on the Perceived Stress Scale, a 14-item 5-point scale for rating non-specific appraisals of stress.7 Subjects refrained from alcohol for 48 hours before the tests, and BIV was tested in the follicular phase (day 6–10 after onset of menses), where appropriate. Tests used beclomethasone dipropionate (Sigma-Aldrich, Canada), dissolved in 95% ethanol to final concentrations of 0 μg/mL (vehicle control), 1 μg/mL, 3 μg/mL, 10 μg/mL, 30 μg/mL and 100 μg/mL. Solutions were prepared and stored under conditions known to give stable test responses.8 In the late afternoon, at least 2 hours after the last meal,9 subjects collected a salivary cortisol sample using a dental roll for 2 minutes (Salivette; Sarstedt Inc., Newton, NC). Six 2-cm grease rings were applied to the volar aspect of each forearm, and 10-μL aliquots of test solution were applied at random locations and then occluded with plastic wrap and tube gauze. After 15–18 hours, BIV was assessed visually on a 5-point scale under standard fluorescent lighting by the same 2 trained observers who underwent regular inter-rater assessment and feedback and who were blinded to the study design, diagnosis and randomization. The BIV scale was: 0 = no blanching, 1 = faint blanching, 2 = obvious blanching, 3 = intense blanching, 4 = intense blanching that extends outside test ring.8 The interrater rank correlation was 0.88, and BIV scores for each concentration were averaged from the 2 observers. To correspond with the analysis of Cotter et al,3 an additional “median BIV score” was calculated as the median score for each subject from the 3–100 μg/mL concentrations. Subjects collected a saliva sample 30 minutes after waking the next morning, before any vigorous activity, smoking, eating, tooth brushing or going outdoors, and after rinsing their mouth with plain water.10 Samples timed in relation to waking are correlated with diurnal cortisol and are more reliable than those collected at set time points.11 Saliva samples were centrifuged, stored at −80°C, and salivary cortisol was assayed in duplicate using a highly specific enzyme-linked immunosorbent assay kit (Alpco Milenia Cortisol; Dako Diagnostics Canada, Mississauga, Ont.), with inter-assay and intra-assay coefficients of variation ≤ 2% at 1.0 μg/dL and ≤ 8% at 5.0 μg/dL.
Statistics
Parametric data (including salivary cortisol and body mass index after log-normalization) were compared using paired t tests. BIV ratings at each beclomethasone concentration, median BIV scores, alcohol intake, MASQ general distress and anxious arousal were compared using Wilcoxon signed-rank tests. Spearman rank correlations were used to test for associations between salivary cortisol concentrations and BIV scores. Data are given as means (and standard deviations [SD] back-transformed for the normalized data), medians and quartiles; 95% confidence limits for the differences between means are given for the non-transformed parametric data.
Results
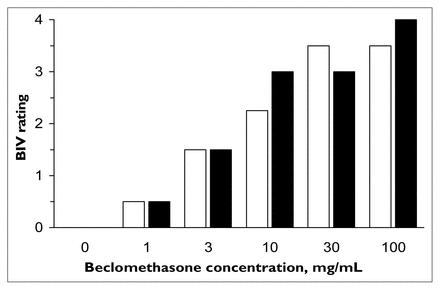
BIV scores did not differ significantly between the MDD patients and controls at any beclomethasone concentration (Fig. 1). In addition, “median BIV scores” from the 3–100 μg/mL concentrations did not differ between depressed patients (mean 3.0, 95% confidence limits [CL] 2.5–3.5) and healthy controls (mean 2.75, 95% CL 2.5–3.5) (z = −1.02, p = 0.31). The number of subjects with at least faint blanching at each concentration did not differ significantly, and 100% of subjects showed obvious or greater blanching at 10 μg/mL beclomethasone or above. In the combined sample, there was a significant effect of reproductive status on the median BIV score (Kruskal-Wallis χ2 = 6.025, p = 0.049). Postmenopausal women (mean 2.5, 95% CL 2.25–2.9) showed less vasoconstriction than women who were premenopausal (mean 3.125, 95% CL 2.56–3.53) or taking hormones (mean 3.0, 95% CL 2.56–3.5).
Median ratings of beclomethasone-induced vasoconstriction (BIV) in women with major depressive disorder (black bars) and healthy controls (white bars). Wilcoxon signed-ranks tests: 1 μg/mL: z = −0.44, p = 0.66; 3 μg/mL: z = −0.17, p = 0.87; 10 μg/mL: z = −1.16, p = 0.25; 30 μg/mL: z = −0.83, p = 0.41; 100 μg/mL: z = −0.56, p = 0.58.
There were no significant differences in salivary cortisol concentrations (Table 1). Median BIV scores were positively correlated with afternoon cortisol in MDD patients (r = 0.49, p = 0.035) but were not correlated with waking cortisol in patients (r = 0.29, p = 0.23) or with waking (r = −0.25, p = 0.31) or afternoon cortisol (r = 0.03, p = 0.92) in controls.
Discussion
This study failed to replicate the finding of Cotter et al3 that MDD was associated with reduced BIV. Differences between the study populations or rating methods could be relevant to this discrepancy. The sample in the Cotter et al study was 41% male, whereas all of our subjects were female. Second, in the current study, subjects were matched for reproductive factors, including hormone treatments and cycle phase. Sex and reproductive factors may be relevant in view of the present finding that postmenopausal women showed reduced BIV and the literature on sex differences in HPA axis function.5 Third, patients were older in the Cotter et al study, and age may have contributed to the reduced response in postmenopausal women in this study. Fourth, all of the patients in this study were psychotropic-free, whereas Cotter et al’s patients were all taking antidepressants. Although antidepressants have been reported to increase, not decrease, GR function,2 it is possible that BIV may be influenced by effects of antidepressants on vascular tone downstream from GRs. For example, glucocorticoids potentiate noradrenaline-induced vasoconstriction,12 and noradrenergic function may be increased by antidepressants.13 Fifth, the subjects in this study were moderately depressed outpatients recruited via public notices, whereas Cotter et al studied inpatients with major depression, half of whom were taking lithium. This suggests that factors such as severity, treatment-seeking, treatment-resistance, admission to hospital or suicide risk may be relevant, particularly because we excluded patients needing urgent treatment. Psychotically depressed patients, who may have more frequent or marked HPA axis abnormalities,14 were excluded from both studies. In the current study, patients with PTSD were excluded because of evidence that BIV may be increased in this population.15 In most of the patients in the Cotter et al study, but none in this study, blanching was undetectable at 3–10 μg/mL. A lower threshold of the current raters for detecting faint blanching could produce this result, but this would not explain the lack of group differences across the whole range of beclomethasone concentrations and on the “median BIV scores.”
Visual-ratings are the most sensitive method to detect faint steroid-induced vasoconstriction and are reliable within laboratories if consistent conditions and trained raters are used.16,17 However, comparisons between laboratories cannot be readily made without standardization. There is good evidence that BIV is mediated by GRs. First, steroid-induced vasoconstriction has shown clear dose–response relations,18 and vasoconstrictive potency is associated with clinical efficacy in the treatment of psoriasis, allergy and asthma.19–21 Second, vasoconstriction was inhibited by the nonselective GR antagonists, progesterone, deoxycorticosterone and mifepristone (RU486).8,22,23 Third, pharmacological blockade of the intracellular metabolism of cortisol by 11-β-hydroxysteroid dehydrogenase (11-β-HSD), using glycyrrhetinic acid, increased cortisol-induced vasoconstriction.24 11-β-HSD does not metabolize beclomethasone, and so differences in 11-β-HSD would not contribute directly to differences in BIV.25 Fourth, budesonide-induced vasoconstriction is influenced by genetic variants of GRs in healthy subjects26 that are also associated with risks for glucocorticoid-related disorders, including familial hypertension, hyperinsulinemia and abdominal obesity. 27–29 Fifth, direct studies of BIV have also shown an association with familial hypertension and hyperinsulinemia30 and with clinical resistance to steroids in the treatment of asthma.8 These data also suggest that although GRs are differentially regulated in different tissues, alterations in GRs or in post-receptor mechanisms might exert effects across multiple tissues.
There was a mild increase in waking cortisol in patients compared with controls. Although this was not significant with 2-tailed tests selected a priori, it might be argued that 1-tailed tests would have been appropriate on the basis of prior evidence for cortisol hypersecretion in depression, although this may not be as common in outpatient or community populations as in patients in hospital.1,2,30 Waking cortisol was not correlated with BIV. Afternoon salivary cortisol and BIV were correlated, but in the opposite direction to that predicted for receptor down-regulation. This analysis was not the primary objective of the study and was uncorrected for multiple comparisons. The sample was small, and the timing of the saliva collection and control of the subjects’ preceding activities were not as rigorous for the afternoon sample as for the waking sample.
The possibility that decreased BIV might be associated with more marked cortisol hypersecretion requires further study. Steroid-resistant asthma patients have shown lower BIV than steroid-sensitive patients taking long-term prednisone,8 and BIV was not correlated with plasma cortisol in hypertension,31 suggesting that reduced BIV may occur as a result of mechanisms other than GR down-regulation. In patients with MDD, impaired GR function may be a more consistent abnormality than reduced GR binding in peripheral blood cells.32
In conclusion, the present study did not find the predicted reduction in BIV in unmedicated patients with MDD, despite mild cortisol hypersecretion compared with the controls. Recent findings in PTSD15 and marked differences between the samples of patients with MDD studied to date suggest that further research is warranted. Although such samples may be difficult to recruit, MDD patients who are unmedicated, but also have severe, chronic, resistant depression may need to be studied.
Acknowledgements
Our thanks to raters Gail Rauw and Jordyce van Muyden, and to Jesse Chan, Heidi Choi, Evelyn Mitchell, Carla Morden, Kelly Shinkaruk, Sona Sihra for assistance with the study. Thanks to Brian Walker at the Western General Hospital, Edinburgh, Scotland for advice on the BIV method. Thanks to David Watson and Lee Anna Clark for permission to use the MASQ. Funding is gratefully acknowledged from the Alberta Heritage Foundation for Medical Research (NC, KH), the NeuroScience Canada Foundation (NC) and the Canadian Institutes of Health Research (KH).
Footnotes
Medical subject headings: beclomethasone; depressive disorder; glucocorticoid; hydrocortisone; receptors, vasoconstriction; women.
Competing interests: None declared.
- Received July 24, 2002.
- Revision received December 4, 2002.
- Accepted March 11, 2003.