Abstract
Objective: It is thought that an imbalance in serotonergic neurotransmission may underlie many affective disorders. Thus, the serotonin-1A (5-HT1A) receptor is a target for antidepressant and neuroleptic drugs. It has been reported that estrogens modulate serotonergic neurotransmission. Therefore, we investigated the effect of long-term ovariectomy on 5-HT1A receptor–specific binding and G-protein activation in the brain. Correction therapy with estradiol was compared with treatments using the selective estrogen receptor modulators tamoxifen and raloxifene.
Methods: Four months after ovariectomy, Sprague–Dawley rats were treated with vehicle, 17β-estradiol (80 μg/kg), tamoxifen (1 mg/kg) or raloxifene (1 mg/kg) subcutaneously for 2 weeks. Specific binding to 5-HT1A receptors was assessed by autoradiography of brain sections using the 5-HT1A agonist [3H]8-OH-DPAT. 5-HT1A receptor stimulation was measured using R-(+)-8-OH-DPAT-stimulated [35S]GTPγS-binding autoradiography.
Results: Ovariectomy decreased uterine weight, which was corrected by estradiol; tamoxifen and raloxifene partially corrected this decrease. Hormonal withdrawal and replacement left [3H]8-OH-DPAT-specific binding unchanged in the cortex. In contrast, ovariectomy induced a decrease in R-(+)-8-OH-DPAT-stimulated [35S]GTPγS-specific binding in the cortex; this was corrected by estradiol but was not corrected significantly by tamoxifen or raloxifene. In the hippocampus, ovariectomy had no effect on [3H]8-OH-DPAT-specific binding, whereas only 17β-estradiol treatment decreased this binding in a subregion of the CA3. Ovariectomy increased R-(+)-8-OH-DPAT-stimulated [35S]GTPγS-specific binding in the dentate gyrus (but not in the CA1 or CA3); this was corrected by estradiol and raloxifene, but not by tamoxifen. In the dorsal raphe nucleus, ovariectomy increased [3H]8-OH-DPAT-specific binding and R-(+)-8-OH-DPAT-stimulated [35S]GTPγS-specific binding; estradiol corrected this increase, but this was not corrected significantly by tamoxifen or raloxifene.
Conclusions: An overall stimulation by estradiol of 5-HT1A receptor–specific binding and coupling was observed, decreasing raphe somatodendritic receptors and increasing cortical postsynaptic receptors.
Introduction
Estrogens modulate brain activity from development to aging.1 Throughout a lifetime, estrogens influence mood, memory and mental state.2 A large body of data suggests that estrogens could be beneficial in the treatment of mental disorders. 3 In support of this, the risk of depression has been found to be increased in women with low plasma levels of estradiol.4 In addition, estrogen replacement in patients with depression has been seen to mimic the effect of antidepressants.5
Menopause is associated with decreased ovarian hormones. 6 Clinical studies have reported that menopausal women are more vulnerable to mental disorders.4 Experimentally, ovariectomized animals are used as models to investigate the impact of ovarian hormone loss.7 In the central nervous system, ovariectomy was reported to affect the serotonergic, glutamatergic and dopaminergic systems, that is, neurotransmitters implicated in mental disorders.3,7
A decrease in serotonergic function is associated with several mental disorders. The prefrontal cortex and hippocampus are affected in many cognitive and affective disorders.8 Many antidepressants and neuroleptics target the serotonin-1A (5-HT1A) receptor. In the dorsal raphe, the 5-HT1A receptor is an autoreceptor that exerts a negative feedback on the firing activity of 5-HT neurons.9 5-HT1A receptors are present in high density in limbic brain areas, notably, the hippocampus, lateral septum, cortical areas (particularly the prefrontal, cingulate and entorhinal cortex) and the mesencephalic raphe.10 The 5-HT1A receptor activates the Gi/o and Gz proteins family, transmitting inhibitory signals.10 In patients with depression, the 5-HT1A autoreceptor has been reported to be increased, thus, decreasing brain 5-HT function.11
It has been reported that estrogens modulate 5-HT1A receptors. Autoradiography with specific ligands for the 5-HT1A receptor or in situ hybridization of 5-HT1A receptor mRNA levels in the brains of ovariectomized rats have shown a decrease or no effect of short-term or long-term estradiol treatments in the cortex, the hippocampus and the raphe.12–14 An action of estrogen on lordosis induced by the 5-HT1A agonist 8-OH-DPAT is reported, without changes in 5-HT1A receptor density in the hypothalamus.15 Estradiol was thus hypothesized to act downstream from the receptor. 5-HT1A agonist–stimulated [35S]GTPγS-specific binding was used to investigate estradiol action on 5-HT1A receptor stimulation and as an indication of coupling of this receptor. This assay provides measures of the first step in the signal transduction cascade.16 In nonhuman primates, long-term estradiol treatment decreases 8-OH-DPAT-stimulated 5-HT1A receptors in the dorsal raphe.12 In ovariectomized rats, estradiol (in vivo and in vitro) was reported to rapidly decrease 8-OH-DPAT stimulation of [35S]GTPγS binding in the cortex and hippocampus, whereas no effect was reported with long-term estradiol treatment.17–19
Estrogens modulate cellular functions through 2 estrogen receptors, ERα and ERβ.20,21 These receptors mediate principally genomic actions by activating transcription of target genes.22 Estrogens can also mediate nongenomic effects related to membrane action,1 and accumulating evidence supports the existence of a membrane estrogen receptor.23 In the brain, ERα and ERβ have a specific expression pattern.24,25 The mechanisms involved in estrogen’s beneficial effects related to mental disorders remain to be understood.
Selective estrogen receptor modulators (SERMs) were developed to mimic estradiol’s beneficial action in bone and the cardiovascular system without the increased risk of breast and uterine cancer.26 Tamoxifen, a first-generation SERM, is widely used in the treatment of breast cancer.27,28 Raloxifene, a second-generation SERM, is given to menopausal women to treat osteoporosis.27,28 Few studies are available of the actions of SERMs in the brain.3,29
The aim of the present study was to investigate the impact of long-term ovariectomy (4 months) on brain 5-HT1A receptor–specific binding using the agonist ligand [3H]8-OH-DPAT. An indication of coupling of brain 5-HT1A receptors with Gi proteins was assessed with R-(+)-8-OH-DPAT-stimulated [35S]GTPγS-specific binding. The effect of a correction treatment of 2 weeks with estradiol was compared with tamoxifen and raloxifene. This paradigm was designed to mimic postmenopausal hormonal changes and hormone replacement to maintain a tonic rather than a cyclic fluctuation of estrogens.
Methods
Adult, female Sprague–Dawley rats weighing about 250 g were purchased from Charles River Canada (St. Constant, Que.). The animals were housed 2 per cage in an environment that was light-controlled (12 h light per day, lights on at 7 am) and temperature-controlled (22°C–23°C). The animals had free access to rat food and water. The Laval University Animal Care Committee approved all the animal studies. All efforts were made to minimize the animals’ suffering and to reduce the number of rats used.
The rats were divided into 5 groups of 10–17 animals. The first group comprised 12 intact control rats at random stages of the estrous cycle treated with vehicle (4% ethanol, 4% polyethylene glycol 600, 1% gelatin and 0.9% NaCl). The 4 other groups were rats that had been ovariectomized 4 months before and were treated with either vehicle (11 rats), 80 μg/kg 17β-estradiol (Sigma, St. Louis, Mo.; 10 rats), 1 mg/kg tamoxifen citrate (Sigma, 10 rats) or 1 mg/kg raloxifene (17 rats). Raloxifene was extracted from pills (Evista; Eli Lilly, Indianapolis, Ind.) as previously described.30 The drug doses were the same as in our previous studies.31 The rats were ovariectomized under anesthesia with a 1.5% isoflurane–air mixture. Treatment began 4 months after ovariectomy, once daily for 14 days; 0.5 mL per rat was injected subcutaneously as suspension (in vehicle).
Animals were killed by decapitation, and their brains were rapidly removed, flash-frozen in isopentane over dry ice, individually wrapped in aluminum foil and kept at −80°C. The uteri were immediately removed at sacrifice, freed from connective and adipose tissue, and weighed. For cryosection, brains were immersed in Tissue-Tek (Miles, Inc., Elkhart, Ind.) at −20°C, mounted on cryostat chucks and cut into 20-μm coronal slices. Adjacent coronal slices were cut from the anterior region of the prefrontal cortex and cingulate cortex (bregma 4.2 to 3.7), the hippocampus (bregma −3.14 to −3.80), and dorsal raphe nucleus (bregma −7.04 to −7.64) according to the Paxinos and Watson atlas.32 The subregions CA1 stratum oriens, CA1 stratum radiatum, CA3 low subregion, CA3 high subregion (low and high denominations reflect the density of 5HT1A receptor–specific binding in the CA3 region) and the dentate gyrus of the hippocampus were quantified. Slices were thaw-mounted on pre-cleaned slides (Snowcoat X-tra; Surgipath, Richmond, Ill.) and vacuum desiccated at 4°C for 12 hours before storing at −80°C.
Autoradiography of 5-HT1A receptor–binding sites with [3H]8-OH-DPAT and of Gi protein–specific binding with [35S]GTPγS was performed, as previously described.33–35 Sections and calibrated [3H] standards for [3H]8-OH-DPAT binding and [14C] standards for [35S]GTPγS binding (Microscales; Amersham, Arlington Height, Ill.) were exposed for 4 weeks for [3H]8-OH-DPAT and 48 hours for [35S]GTPγS to Kodak BioMax MR film at room temperature. The autoradiograms were analyzed using the software package NIH Image 1.63 on a Power-Macintosh 7100 assisted videodensitometry (Sony camera XC-77).
The percentage of stimulation was determined as follows (Equation 1):
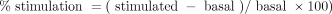
The experimental data were compared using 1-way analysis of variance (ANOVA) using Statviews 4.51 for Macintosh Computer, followed by post hoc pairwise comparisons with a Fisher’s probability of least significant difference test (PLSD). The results were considered statistically significant when p < 0.05. Statistical comparisons of logarithms of uterine weights were used.
Results
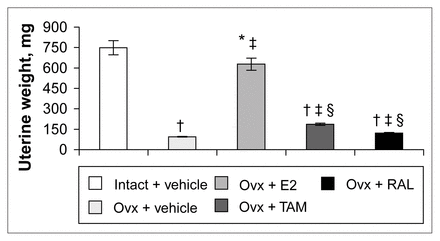
Four months after ovariectomy, uterine weights were decreased by 87% in ovariectomized rats compared with intact rats (Fig. 1). Correction therapy with estradiol for 2 weeks significantly stimulated uterine weight compared with that of ovariectomized rats, but partially (84%) compared with intact rats (Fig. 1). Tamoxifen and raloxifene induced a 49% and a 22% increase, respectively, compared with vehicle-treated ovariectomized rats and significantly less than estradiol treatment (Fig. 1).
Uterine weight of intact rats treated with vehicle and ovariectomized (Ovx) rats treated with vehicle or a correction therapy of estradiol (E2), tamoxifen (TAM) or raloxifene (RAL) for 2 weeks. The results are mean weights (and the standard error of the mean [SEM]). * p < 0.05; † p < 0.001 v. intact rats, vehicle; ‡ p < 0.001 v. ovariectomized rats, vehicle; § p < 0.001 v. ovariectomized rats, estradiol.
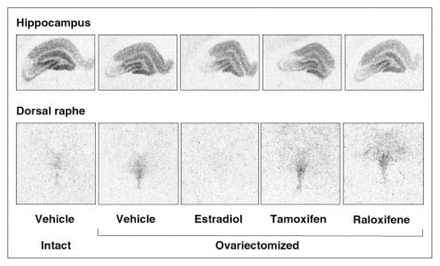
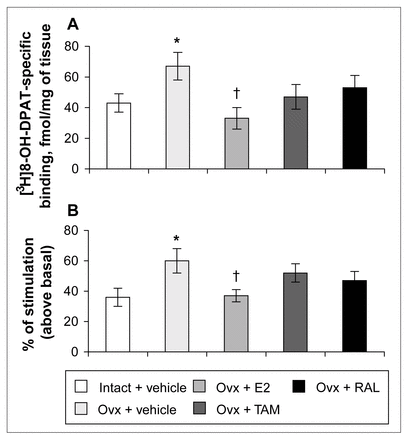
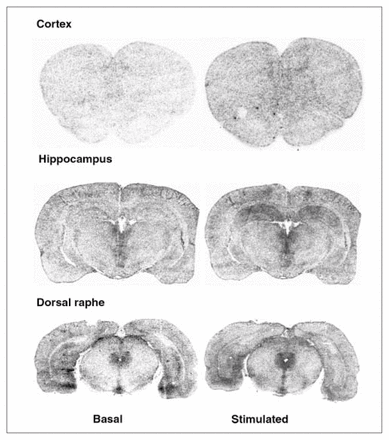
In the dorsal raphe, ovariectomy led to a 36% increase in [3H]8-OH-DPAT-specific binding that was corrected by estradiol (Fig. 2, Fig. 3A). Examples of autoradiograms of basal and stimulated [35S]GTPγS-specific binding in the dorsal raphe are shown in Figure 4. Ovariectomy increased R-(+)-8-OH-DPAT-stimulated [35S]GTPγS-specific binding by 46% compared with that in intact rats (Fig. 3B). Estradiol decreased and restored binding to intact values. In rats treated with tamoxifen and raloxifene, dorsal raphe [3H]8-OH-DPAT-specific binding and R-(+)-8-OH-DPAT-stimulated [35S]GTPγS-specific binding were not significantly different from that in intact or ovariectomized rats (Fig. 3).
Examples of autoradiograms of specific binding of [3H]8-OH-DPAT (2 nmol/L) in the hippocampus and dorsal raphe of intact rats and ovariectomized rats treated with vehicle or with a correction therapy of estradiol, tamoxifen or raloxifene for 2 weeks.
Specific binding of [3H]8-OH-DPAT (2 nmol/L) (A) and specific binding of R-(+)-8-OH-DPAT-stimulated [35S]GTPγS (50 pmol/L) (B) in the dorsal raphe of intact rats treated with vehicle and ovariectomized rats treated with vehicle or a correction therapy of estradiol, tamoxifen or raloxifene for 2 weeks. Results in A are expressed as mean fmol/mg of tissue (and SEM). Results in B are expressed as a percentage of stimulated over basal specific binding (and SEM).* p < 0.05 v. intact rats, vehicle; † p < 0.05 v. ovariectomized rats, vehicle.
Examples of autoradiograms of basal and R-(+)-8-OH-DPAT (1 μmol/L) stimulated [35S]GTPγS-specific binding. Coronal sections at the level of the cortex, hippocampus and dorsal raphe were incubated with [35S]GTPγS (50 pmol/L).
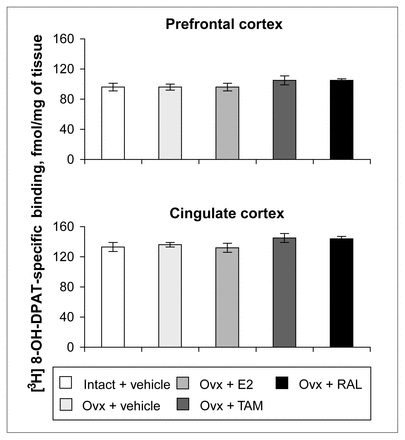
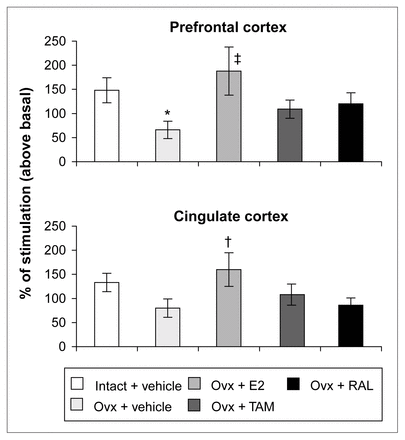
Ovariectomy and treatments left [3H]8-OH-DPAT-specific binding unchanged in the prefrontal and cingulate cortices (Fig. 5). Examples of autoradiograms of basal and stimulated [35S]GTPγS-specific binding in the cortex are shown in Figure 4. In cortical subregions, ovariectomy decreased R-(+)-8-OH-DPAT-stimulated [35S]GTPγS-specific binding by 55% and 40%, respectively, compared with intact rats (Fig. 6); this finding reached statistical significance in the prefrontal cortex. Estradiol treatment restored R-(+)-8-OH-DPAT-stimulated [35S]GTPγS-specific binding to intact values both in the prefrontal and cingulate cortices (Fig. 6). In rats treated with tamoxifen and raloxifene, cortical [3H]8-OH-DPAT-specific binding and R-(+)-8-OH-DPAT-stimulated [35S]GTPγS-specific binding were not significantly different from that in intact or vehicle-treated ovariectomized rats (Fig. 6).
Specific binding of [3H]8-OH-DPAT (2 nmol/L) in the cortex of intact rats treated with vehicle and ovariectomized rats treated with vehicle or a correction therapy of estradiol, tamoxifen or raloxifene for 2 weeks. Results are expressed as mean fmol/mg of tissue (and SEM).
R-(+)-8-OH-DPAT-stimulated [35S]GTPγS-specific binding (50 pmol/L) in the cortex of intact rats treated with vehicle and ovariectomized rats treated with vehicle or a correction therapy of estradiol, tamoxifen or raloxifene for 2 weeks. Results are expressed as percentage of stimulated over basal specific binding (and SEM). * p < 0.05 v. intact rats, vehicle; † p < 0.05, ‡ p < 0.01 v. ovariectomized rats, vehicle.
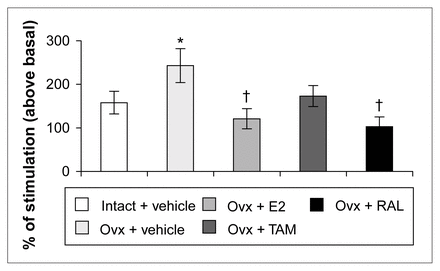
Ovariectomy and treatments left [3H]8-OH-DPAT-specific binding unchanged in the CA1, CA3 high subregion and the dentate gyrus of the hippocampus (Table 1). In the CA3 low subregion, estradiol treatment decreased this binding by 12% compared with that in intact and ovariectomized rats (Fig. 2, Table 1). Autoradiograms of basal and stimulated [35S]GTPγS-specific binding in the hippocampus are shown (Fig. 4). Ovariectomy and treatments did not alter R-(+)-8-OH-DPAT-stimulated [35S]GTPγS-specific binding in the CA1 and CA3 subregions (Table 2). In the dentate gyrus, ovariectomy increased R-(+)-8-OH-DPAT-stimulated [35S]GTPγS-specific binding by 56% compared with that in intact rats; this was corrected with estradiol and raloxifene treatments, but not with tamoxifen (Fig. 7).
R-(+)-8-OH-DPAT-stimulated [35S]GTPγS-specific binding (50 pmol/L) in the dentate gyrus of intact rats treated with vehicle and ovariectomized rats treated with vehicle or a correction therapy of estradiol, tamoxifen or raloxifene for 2 weeks. Results are expressed as a percentage of stimulated over basal specific binding (and SEM). * p < 0.05 v. intact rats, vehicle; † p < 0.01 v. ovariectomized rats, vehicle.
[3H]8-OH-DPAT*-specific binding to serotonin-1A (5-HT1A) receptors in subregions of the hippocampal formation of intact rats and ovariectomized rats† treated once daily, subcutaneously, with either vehicle or a replacement therapy of 17β-estradiol (80 μg/kg), tamoxifen (1 mg/kg) or raloxifene (1 mg/kg) for 2 weeks
R-(+)-8-OH-DPAT-stimulated [35S]GTPγS-specific binding in subregions of the hippocampal formation of intact rats and ovariectomized rats* treated once daily, subcutaneously, with either vehicle or a replacement therapy of 17β-estradiol (80 μg/kg), tamoxifen (1 mg/kg) or raloxifene (1 mg/kg) for 2 weeks
Discussion
Our results showed that ovariectomy and estradiol differentially affect [3H]8-OH-DPAT-specific binding and R-(+)-8-OH-DPAT-stimulated [35S]GTPγS-specific binding in the prefrontal and cingulate cortices and the hippocampus compared with the dorsal raphe nucleus. [35S]GTPγS-binding autoradiography used in the present experiments has been called “functional autoradiography” and has the advantage of providing functional information in an anatomical context.16 The essence of the method is to visualize agonist-activated receptors by labelling receptor-coupled G-proteins with a radiolabelled analogue of guanosine triphosphate (GTP). This assay measures the first step in the signal transduction cascade and is an indication of coupling to the receptor.
In the dorsal raphe, 4 months after ovariectomy, 5-HT1A receptor density and agonist stimulation were increased compared with those in intact rats. This suggests that ovariectomy, modelling hormonal withdrawal in menopause, leads to a state of 5-HT1A inhibitory autoreceptor hyperactivity in the dorsal raphe. This receptor decreases neuronal firing and serotonin release. The risk of depression has been found to be increased in women with low plasma levels of estradiol.4 In addition, patients with depression have an increase of [3H]8-OH-DPAT-specific binding to 5-HT1A receptors in the dorsal raphe.11
This 5-HT1A autoreceptor hyperactivity was corrected (decreased to intact rat values) by an estradiol treatment of 2 weeks but was not corrected significantly with tamoxifen or raloxifene. This estrogenic effect has similarities to the effects of long-term treatment with the antidepressants fluoxetine, clorgyline and ipsapirone, as well as the 5-HT1A receptor partial agonist buspirone, which also decrease dorsal raphe 5-HT1A-receptor-stimulated [35S]GTPγS binding.36 Hence, these data suggest that estrogens could be a beneficial adjunct to antidepressant treatment in postmenopausal women with mood disorders, by decreasing the 5-HT1A auto-receptor–specific second messenger pathway.
In the dorsal raphe, the effects of hormonal status on receptor density and coupling were similar, suggesting that a change in coupling reflects a change in receptor availability. In the other brain regions investigated, this was not the case.
In nonhuman primates, long-term estradiol treatment was also reported to decrease 5-HT1A receptor density and stimulation. 12 Hence, these results in ovariectomized monkeys and rats could explain why estradiol is seen to improve the mental state of postmenopausal women by increasing 5-HT firing, as antidepressants do.5 The decrease in 5-HT1A stimulation could be achieved by a decrease in receptor density and by the modulation of Gi protein expression. Indeed, in nonhuman primates, estradiol was reported to decrease the Gαi3 subtype in the midbrain.12 Fluoxetine was also reported to decrease Gi2 and Go subunit protein expression in the rat midbrain. 37 In the dorsal raphe nucleus, ERβ is expressed in 5-HT neurons, whereas ERα is expressed in non-5-HT neurons.38 It is likely that ERβ mediates the estradiol decrease in 5-HT1A receptor density and coupling. Rats treated with tamoxifen and raloxifene had R-(+)-8-OH-DPAT-stimulated GTPγS binding in the dorsal raphe, which was neither different from that in intact nor ovariectomized rats, suggesting that these drugs have no estrogenic agonist activity or weak estrogenic agonist activity on the coupling to this receptor. In the raphe of ovariectomized macaques, treatment for 30 days with estradiol, raloxifene or arzoxifene left 5-HT1A mRNA unchanged, whereas tryptophan hydroxylase mRNA was increased and serotonin transporter mRNA decreased.39 Hence, SERMs are likely candidates to modulate serotonin neurons at different steps of neurotransmission.
No effect of hormonal withdrawal and replacement was measured on the 5-HT1A receptor labelled with [3H]8-OH-DPAT in the prefrontal and cingulate cortices. We observed similar results in short-term ovariectomized rats.40
In the prefrontal and cingulate cortices, long-term ovariectomy decreased R-(+)-8-OH-DPAT stimulated [35S]GTPγS-specific binding. This result suggests that hormonal withdrawal can reduce 5-HT1A signalling. Many affective disorders are linked to a hypofunction of the serotonergic system in the cortex.41 This observation may be implicated in menopausal women’s vulnerability to mental disorders.4
This is the first report to show that estradiol treatment for 2 weeks increases R-(+)-8-OH-DPAT-stimulated [35S]GTPγS-specific binding in the prefrontal and cingulate cortices and counteracts the effect of ovariectomy. Mize and Alper17 reported that a 14-day estradiol treatment of ovariectomized rats left 8-OH-DPAT-stimulated GTPγS binding unchanged. However, they used a lower dose of estradiol and membrane from the anterior cortex, which could have diluted an effect concentrated in the anterior prefrontal and cingulate cortices. 17 In addition, in the present experiments, estradiol treatment was started 4 months after ovariectomy, whereas it was started 2 weeks after ovariectomy in the study by Mize and Alper.17 Short-term and in-vitro estradiol treatment was shown to decrease the percentage of 5-HT1A receptor stimulation in the anterior cortex.17,19 Long-term estradiol treatment could modulate 5-HT1A receptor functionality. Indeed, it has been suggested that in-vitro estradiol, by activating protein kinase A and C, may lead to 5-HT1A phosphorylation and uncoupling. 18 These effects observed with short-term estradiol treatments were mimicked by the membrane-impermeable bovine serum albumin–estradiol (E2-BSA), reinforcing the idea of the involvement of a membrane estrogen receptor.19
As in the raphe, rats treated with tamoxifen and raloxifene had cortical R-(+)-8-OH-DPAT-stimulated GTPγS binding that was neither different from that in intact nor ovariectomized rats, suggesting that these drugs have no estrogenic agonist activity or weak estrogenic agonist activity on the coupling to this receptor. Mize et al19 previously reported that tamoxifen, in a dose-dependent manner, decreased the percentage of stimulation of 5-HT1A receptors. This decrease was observed with a high dose of tamoxifen (10−6 mol/L),19 which is unlikely to have been reached in our in-vivo experiment using 1 mg/kg subcutaneously of tamoxifen. In the cortex, ERα and ERβ are expressed, with a higher proportion of ERβ;25,42,43 therefore, the estrogenic effects observed could involve either of these receptors.
In the hippocampus, long-term ovariectomy had no effect on [3H]8-OH-DPAT-specific binding; estradiol treatment decreased this binding in the CA3 low subregion of the hippocampus without affecting the other subregions. These results are consistent with a previous report concerning long-term treatment of rats with estradiol,14 whereas no effect of ovariectomy and estradiol treatment was observed on 5-HT1A receptors in the hippocampus in recently ovariectomized rats.40 Long-term estradiol treatment induced a decrease in 5-HT1A receptor–specific binding and mRNA in the CA3, and single administration of estradiol had no effect.13,14 This could involve a downregulation by estradiol of 5-HT1A receptor gene transcription. The 5-HT1A receptor gene contains an Sp1 sequence where estradiol could interact.44,45 Tamoxifen and raloxifene did not mimic the estradiol effect in the CA3, indicating that they have no estrogenic properties on 5-HT1A receptor–specific binding. ERα and ERβ are both expressed in the subregions of the hippocampus;46 however, on the Sp1 promoter, ERβ was reported to be ineffective.47,48 It seems likely that estradiol decreased 5-HT1A receptor expression in the hippocampus through an ERα pathway.
Ovariectomy increased R-(+)-8-OH-DPAT-stimulated GTPγS binding only in the dentate gyrus of the hippocampus. This increase was corrected by estradiol. A previous study reported no effect of long-term estradiol treatment in the whole hippocampus.17 Similarly, if our results for R-(+)-8-OH-DPAT-stimulated [35S]GTPγS binding are quantified for the whole hippocampus, no effect of ovariectomy and estradiol treatment is observed, masking the subregional effect reported here. Short-term administration of estradiol has been reported to decrease 5-HT1A receptor coupling.17 It was proposed that this effect involved an increase of protein kinase A and protein kinase C activities.18 In the hippocampus, estradiol was previously shown to enhance the 5-HT1A signal in hippocampal CA1 pyramidal cells by c-fos activation and GAD65 inhibition.49–52
Long-term treatment with fluoxetine has been reported to leave 5-HT1A receptor–specific binding unchanged in all the brain regions investigated, including the raphe, the cortex and the hippocampus.53,54 Long-term treatment with antidepressants like fluoxetine and imipramine, however, induces an increase in agonist-stimulated [35S]GTPγS binding in the hippocampus of ovariectomized rats, whereas clorgyline, ipsapirone and buspirone did not change this binding.36,55 More specifically, fluoxetine increases or leaves unchanged 5-HT1A receptor stimulation in the CA1 and dentate gyrus.53,54 Hence, in the hippocampus, 5-HT1A receptor stimulation is either not modulated or is differently modulated in some subregions by estradiol and antidepressants. Raloxifene, but not tamoxifen, decreased the percentage of stimulation. This is unlikely to be because of their absence or a concentration that is too low in the brain, because both tamoxifen and raloxifene were previously reported to have estrogenic activity and mimic the estradiol effect on N-methyl-D-aspartate (NMDA) and 2-amino-3-hydroxy-5-methyl-4-isoxazole propionate (AMPA) receptors.56–58
In conclusion, the present data show regional specific changes in 5-HT1A receptor stimulation by long-term ovariectomy that are corrected with estradiol treatment. The receptor coupling changes observed with gonadal hormone withdrawal and replacement are not always associated with changes in 5-HT1A receptor–specific binding. The estradiol effect on 5-HT1A receptors in the brain regions investigated, decreasing raphe somatodendritic and increasing cortical postsynaptic receptors, is generally either only modestly reproduced or not reproduced with tamoxifen and raloxifene treatments. Interestingly, in the dorsal raphe, estradiol decreases 5-HT1A receptor coupling, as do antidepressants.36,55
Acknowledgements
A grant from the Canadian Institutes of Health Research (CIHR) to Dr. Di Paolo supported this research. Ms. Le Saux holds a studentship from the Fondation de l’Universtité Laval.
Footnotes
Medical subject headings: estrogens; G proteins; models, animal; raloxifene; rats; receptors, serotonin; tamoxifen.
Competing interests: None declared.
- Received December 29, 2003.
- Revision received April 1, 2004.
- Accepted April 6, 2004.