Abstract
Background: Suicide is the most serious consequence of major depressive disorder (MDD). Although the anterior cingulate cortex (ACC; Brodmann area [BA] 24) has been increasingly investigated for its role in the etiology of MDD, there is surprisingly very little information about the possible implication of this brain region in suicide. We hypothesized that changes in BA24 cell densities occur in depressed individuals who commit suicide, possibly reflecting an altered state of cortical plasticity that is thought to occur in depression.
Methods: We investigated cell densities and sizes in BA24 among suicide completers and matched sudden-death controls. We examined a 1-cm3 tissue block from BA24a of the supracallosal ACC in 26 human postmortem brain specimens (13 depressed individuals who committed suicide and 13 controls). We assessed neuronal and glial cell densities as well as neuronal soma sizes in the upper and lower cortical layers using optical fractionator and nucleator 3-dimensional stereological probes.
Results: Glial densities, neuronal densities and soma sizes measured in BA24a did not differ significantly between controls and suicide completers. Secondary analyses showed a significant and robust increase in glial cell densities in BA24a of alcohol-dependent depressed suicide completers compared with depressed suicide completers who were not alcohol-dependent (38%) and, to a lesser extent, controls (30%).
Limitations: Our study, conducted with tissue samples from men only, made use of a nonspecific stain that does not distinguish between neuronal or glial cell subtypes. Furthermore, the quantitative analysis concerned upper and lower cortical contours rather than individual cortical layers.
Conclusion: Our results indicate that in BA24, glial density, neuronal density and soma size are not affected in MDD and suicide. They also suggest that alcohol dependence has an important influence on glial densities in this key limbic structure.
Introduction
Accounting for 10%–12% of annual deaths in North America (World Health Organization; www.who.int), suicide represents a major public health concern and is commonly associated with psychopathology, most notably major depressive disorder (MDD).1,2 It is increasingly clear that individuals who die by suicide may have associated neurobiological processes independent from those related to underlying psychopathology such as MDD.3–7 As part of the recent research efforts to understand the neurobiology of MDD, brain regions implicated in emotional processing of cognitive experiences have been scrutinized using various experimental approaches. One such region is the anterior cingulate cortex (ACC; Brodmann area [BA] 24). Being “the seat of dynamic vigilance by which environmental experiences are endowed with an emotional consciousness,”8 the ACC acts as a bridge between limbic structures and the frontal lobe, integrating cognitive activity with affective experience. Dysfunctions within this network have been described in depressed individuals showing impaired problem solving, maladaptive behavioural regulation and abnormal error monitoring.9 In vivo macroscopic alterations in BA24 associated with depression have also been reported in spectroscopic,10 structural11 and functional studies.12–14 Considering the growing interest in the investigation of ACC alterations in MDD, it is surprising that very little attention has been paid to the implication of this brain region in suicide.
There have been few microscopic studies focused on BA24 cell populations and, to our knowledge, no study has investigated BA24 alterations associated with suicide. Whereas initial reports indicated reductions in BA24 glial cell densities and mean neuronal soma size in patients with MDD compared with controls,15,16 subsequent studies found MDD-related increases in neuronal densities17 and no differences in neuronal densities and soma size.18 Despite discrepancies, these studies suggest that individuals with MDD, some of whom have died by suicide, present alterations in the ACC. However, a more definitive description and a better understanding of such alterations has yet to emerge. Brodmann area 24 cellular alterations in suicide may help explain morphometric changes occurring in the hippocampus,19,20 amygdala21,22 and prefrontal cortical regions,23–30 brain regions that are all directly connected to the ACC and that have also been implicated in MDD and suicide.
We conducted the present study to compare BA24 neuronal and glial cell populations in suicide completers and matched sudden-death controls. To decrease variability associated with the underlying psychopathology of suicide, we limited the study to suicide completers with a history of MDD. Our working hypothesis was that changes in BA24 cell densities occur in depressed individuals who commit suicide and that this could reflect an altered state of cortical plasticity that is thought to occur in depression. To test this hypothesis, we used an unbiased 3-dimensional (3-D) stereological counting approach to determine, from a series of Nissl-stained BA24 sections, neuronal and glial cell densities and average neuronal cell body dimensions. In addition, we paid particular attention to alcohol dependence, a trait often comorbid with MDD and suicide that has previously been associated with a number of neurochemical,31,32 neurocellular33–35 and behavioural36 adaptations. Therefore it was of further interest to investigate the multifaceted association among alcohol dependence, depression, suicide and the cellular integrity of BA24.
Methods
Study sample
We obtained postmortem brain tissues from the Quebec Suicide Brain Bank, which is part of the Quebec Brain Bank. The sample comprised specimens obtained exclusively from men aged 18–58 years. In each case, the Quebec Coroner’s Office ascertained the cause of death and performed a toxicological screening with body fluid or tissue samples to detect drug and alcohol use at the time of death. Psychological autopsies were performed for the entire sample as described previously,37 allowing us to have access to detailed information on psychiatric and medical history and other relevant clinical and sociodemographic data. Unfortunately, this did not always allow us to establish precisely the length of alcohol dependence. In brief, a trained interviewer conducted the Structured Clinical Interview for DSM-IV Psychiatric Disorders (SCID-I), nonpatient version,38 with one or more family members of the deceased. A panel of clinicians reviewed SCID-I assessments, case reports, coroner’s notes and medical records to obtain consensus psychiatric diagnoses. The Douglas Hospital Research Ethics Board approved our study, and we obtained written informed consent from next-of-kin.
The suicide group consisted of postmortem tissue samples from men having died by suicide and who had a history of MDD or depressive disorder not otherwise specified. With the exception of alcohol-related disorders, these men had not received diagnoses of any other major psychiatric conditions and they had not taken medication for at least 3 months before death. The control group consisted of postmortem tissue samples from men with no history of major psychiatric diagnoses and who died suddenly (i.e., without agony) from causes that had no direct influence on brain tissue. We matched the groups for age, postmortem interval (PMI) and storage time.
Tissue preparation
We removed tissue samples from the left hemispheres of the brains, which had been flash-frozen upon arrival and kept at − 80°C at the Quebec Brain Bank. We identified an anterior region within the ACC, immediately dorsal to the genu of the corpus callosum (Fig. 1), based on previously defined macroscopic criteria.39,40 We chose the supracallosal BA24a for investigation because it is located ventral to BA24b and BA24c, which have previously been reported to have cytoarchitectonic alterations in individuals with MDD.16,17 Data specifically related to BA24a have been much less consistent,18 therefore it was of particular interest to focus on this region. We removed 1-cm3 tissue blocks containing BA24 and the genu of the corpus callosum and immersed the specimens in formalin overnight, embedded them in paraffin, cut them on a microtome into serial 25 μm–thick sections and mounted them on slides. We systematically and randomly sampled sections, and stained between 3 and 7 sections per specimen with cresyl violet (0.1%) according to standard methods. We coded slides to remain blind to specimens and groups during the analysis. One of us (C.H.) assessed all slides following a consistent and systematic protocol.
Sagittal view of Brodmann area 24a tissue block dissected from above the genu of the corpus callosum.
Identification of cortical laminae
In each section, we identified and selected BA24a of the ACC for analysis according to established microscopic criteria.40 In addition, the genu of the corpus callosum served to orient and specifically identify BA24a, which is located immediately dorsal to this white matter landmark. Under a magnification of × 5, we delimited 2 areas within BA24a for quantification purposes: an upper cortical contour consisting of layers I–III and a lower cortical contour containing layers V (a and b) and VI. The division between these upper and lower cortical contours was based on the upper limit of a prominent layer V, which displays a high density of medium to large neurons and characteristic spindle pyramidal neurons located in layer Vb (Fig. 2).
Nissl-stained section cut from a paraffin-embedded block of Brodmann area (BA) 24a. In this study, upper cortical layers constitute layers I–III, whereas layers Va, Vb and VI constitute the lower cortical layers. Division of upper and lower cortical layers is illustrated by the dashed line. Solid lines above layer I and below layer VI indicate grey matter boundaries of BA24a. CC = corpus callosum; WM = white matter. Scale bar 250 μm.
Morphometric analyses
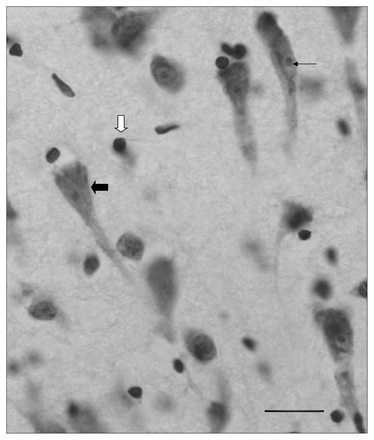
We used an unbiased stereological approach to estimate the number of glial cells and neurons in the upper and lower cortex of BA24a with a light microscope (DM 4000B; Leica) connected to a stereology workstation (Stereo Investigator; MBF Bioscience). We estimated cell densities using the optical fractionator probe allowing for 3-D quantification of both neuronal and glial cells. This unbiased 3-D counting method estimates the total number of objects in a unit of tissue volume with an optical probe providing counts through the z axis that are not distorted by differences in cell size, shape or orientation.41 Among the primary advantages of using the optical fractionator method is that cell counts are independent of section thickness, as they are generated in a defined volume contained entirely within this thickness. The sampling process began by randomly translating a grid formed by 400 μm × 350 μm squares over the section. At each intersection of the grid that fell into the region of interest, we examined a counting frame measuring 225 μm × 150 μm with a 40 × objective (NA 0.75), thus yielding a total of about 120–180 counting frames per specimen on 7 sections. In the few specimens from which fewer evenly spaced sections could be obtained across the whole region, we introduced a correction by altering grid size dimensions. This ensured that the targeted cell count was met and a reliable coefficient of error (CE) was achieved. Using the average of Gunderson (m = 1) and the second estimated CE (Schmitz–Hof) yielded CEs ranging from 0.02 to 0.05 for neurons and glia within each of the defined contours. We identified neurons by the presence of a cresyl violet–stained cytoplasm, a single nucleolus and a large nonspherical outline. We identified glial cells by the absence of stained cytoplasm, the presence of a thicker nuclear membrane and more heterogeneous chromatin within the nucleus (Fig. 3). Consistent with the stereological rules of the dissector probe,42 we counted only cells with a nucleus that fell within the counting frame and that did not contact the exclusion lines when they came into focus within a 9 μm–thick optical dissector centred in the section thickness. To avoid counting cells in nonrepresentative areas of the tissue, we set top and bottom guard zones at 10% of the section thickness. As a result, we counted an average of 1500 glia and 800 neurons per specimen. We expressed cell density as the number of cells per cubic millimetre of tissue, using the total number of cells calculated from the optical fractionator and the volume estimated by Cavalieri’s method.43
Morphological features of Nissl-stained Brodmann area 24a neurons (black block arrow), nucleolus (black arrow) and glial cells (white block arrow); scale bar 25 μm.
To assess neuronal soma size, we applied the stereological isotropic nucleator probe. This probe allows for the mean neuronal area and volume to be estimated from the intersection with the cell boundaries of a set of 4 rays that emerge from the nucleolus.42 For this reason, we applied the nucleator only to neurons that displayed an identifiable nucleolus and a complete and intact cell membrane. We did not assess glial cell volumes, as their cytoplasm is unstained by cresyl violet. Ideally, the nucleator probe should be used to estimate neuronal morphometrics by sampling in tissues cut at random orientations. Owing to technical limitations, we used only sections cut in a coronal plane.
Statistical analysis
All data are expressed as means and standard errors of the means (SEM). The objective of the statistical analysis was to compare within upper and lower cortical areas 3 morphometric parameters (neuronal density, glial density and neuronal soma size) between the depressed suicide completers and sudden-death controls. We assumed normality after examining each morphometric parameter using Kolmogorov–Smirnov tests. We compared group means using 3 separate (one for each parameter) 2-way repeated-measures analyses of variance (ANOVA), with one within- (upper, lower cortical region) and one between-group (control, suicide) variable. We set type I error at 0.05. We also compared depressed suicide completers who were alcohol-dependent with suicide completers who were not alcohol-dependent and controls on the cellular parameters. We performed 2-way repeated-measures ANOVA on each morphometric parameter of interest with one within- (upper, lower cortical region) and one between-group (control, nonalcohol-dependent depressed suicides, alcohol-dependent depressed suicides) variable followed by post-hoc pairwise comparisons with Bonferroni correction. Two controls had a diagnosis of alcohol dependence and we therefore excluded them from these analyses. We also performed multiple correlation analyses to examine the potential influence of age, PMI and storage time on the dependent variables studied. Since none of these factors was found to be a significant confounder, we did not include them in our analyses.
Results
Study sample
The suicide group consisted of postmortem tissue preparations from 14 men: 13 who had MDD and 1 who had depressive disorder not otherwise specified. Of these, 6 met the criteria for alcohol-dependence. The control group comprised specimens from 14 men without psychiatric disorders who died suddenly. The groups did not differ in age (t24 = − 0.140, p = 0.89), PMI (t24 = − 0.064, p = 0.95) or storage time (t24 = − 1.389, p = 0.17). Likewise, when we further subdivided the depressed suicide group into those who were alcohol-dependent and those who were not, none of the potential confounding variables reached significance between groups: age (F2,21 = 2.013, p = 0.16), PMI (F2,21 = 1.086, p = 0.36), storage time (F2,21 = 0.015, p = 0.99). The tissue samples from 2 men (1 per group) yielded extreme cell density values that were more than 2 standard deviations above the group mean. We removed these specimens from our analyses, thus bringing the number per group to 13. Additionally, we measured section thickness at each sampling site, yielding small, nonsignificant differences in the average mounted thickness between the control and suicide groups (16.74 μm, standard deviation [SD] 3.60 μm v. 14.85 μm, SD 5.09 μm, respectively). Demographics, and histological and clinical information are summarized in Table 1.
Summary of demographic, histological and clinical information for 13 men with major depressive disorder who died by suicide and 13 men with no psychiatric disorders who died suddenly from whom we obtained brain specimens from the Quebec Suicide Brain Bank
Glial densities
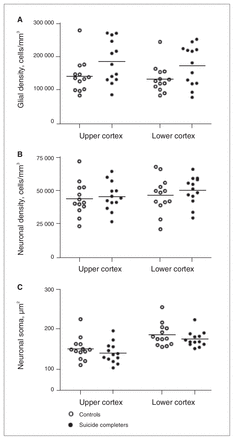
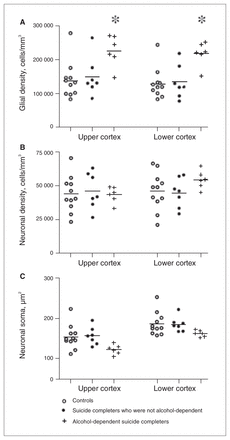
We found no significant differences between the depressed suicide and control groups with respect to BA24a glial cell densities (F1,24 = 3.97, p = 0.06). There was a main effect of cortical region (F1,24 = 5.61, p = 0.026) resulting in upper cortical layers having an overall higher glial density than lower cortical layers (161 702 cells/mm3 v. 151 499 cells/mm3). We observed no interaction (F1,24 = 0.26, p = 0.62; Fig. 4A). Analyzing the influence of alcohol dependence on glial cell densities revealed a main effect of group (F2,21 = 7.30, p = 0.004). Post-hoc pairwise comparisons using the Bonferroni correction showed that specimens from alcohol-dependent depressed suicide completers had higher BA24a glial cell densities than those from suicide completers who were not alcohol-dependent (p = 0.019) and controls (p = 0.004). There was no significant difference in BA24a glial cell densities between controls and depressed suicide completers who were not alcohol-dependent (p = 1.00; Fig. 5A). There was also a main effect of cortical region (F1,21 = 5.62, p = 0.027) with upper cortical layers having a higher glial density than lower cortical layers (170 180 cells/mm3 v. 158 938 cells/mm3). We observed no interaction (F2,21 = 0.21, p = 0.81).
Quantitative parameters measured in Brodmann area 24a of controls (white circles) and suicide completers (black circles) showing (A) glial densities, (B) neuronal densities and (C) neuronal soma size. Mean bars are shown.
Quantitative parameters measured in Brodmann area 24a of controls (white circles) and depressed suicide completers with† and without alcohol dependence (black circles) showing (A) glial densities, (B) neuronal densities and (C) neuronal soma size. Mean bars are shown. Asterisks denote a main effect of group. Following Bonferroni correction, alcohol-dependent depressed suicide completers had significantly higher glial cell densities than depressed suicide completers who were not alcohol-dependent (p = 0.019) and controls (p = 0.004).
Neuronal densities
We observed no significant differences between controls and depressed suicide completers with regards to neuronal densities in BA24a (F1,24 = 0.39, p = 0.54). There was no effect of cortical region (F1,24 = 3.79, p = 0.06) or interaction (F1,24 = 0.28, p = 0.61; Fig. 4B). When examining the effect of alcohol, results of the 2-way repeated-measures ANOVA revealed a significant interaction of group and cortical region for neuronal density (F2,21 = 4.11, p = 0.031). This was the result of the alcohol-dependent depressed suicide completers having significantly higher neuronal densities in their lower cortical layers compared with their upper cortical layers (F1,21 = 14.03, p = 0.001; 54 931 cells/mm3 v. 43 698 cells/mm3). There was no effect of group (F2,21 = 0.27, p = 0.77; Fig. 5b).
Neuronal soma dimensions
As illustrated in Figure 4C, no significant differences emerged from nucleator measurements between controls and suicide completers (F1,24 = 1.29, p = 0.27). We observed a significant main effect of cortical region (F1,24 = 146.77, p < 0.001), with lower cotical layers having larger somal sizes than upper cortical layers (180 μm2 v. 146 μm2). Neuronal soma dimensions in controls and suicide completers with or without alcohol dependence were significantly different between groups (F2,21 = 3.62, p = 0.045); however, following pairwise comparisons, this effect became nonsignificant when comparing alcohol-dependent suicide completers with suicide completers who were not alcohol-dependent (p = 0.09) and with controls (p = 0.07). We observed no interaction (F2,21 = 0.98, p = 0.39), but there was an effect of cortical region (F1,21 = 132.26, p < 0.001), with lower cortical layers having significantly larger somal sizes than upper cortical layers (178 μm2 v. 145 μm2).
Discussion
In this investigation of BA24a, we found no significant differences in glial and neuronal densities or in neuronal soma size between depressed suicide completers and matched sudden-death controls. However, when taking into account alcohol dependence, depressed suicide completers with this comorbidity had a robust increase in BA24a glial densities and a trend toward a decrease in neuronal soma size compared with suicide completers who were not alcohol-dependent and controls.
The few anatomic studies that have been conducted on BA24 in the context of mood disorders have focused on slightly different subdivisions of this region. In particular, a previous analysis found no evidence for morphological alterations in a subgenual region of BA24, but further examination showed that specimens from individuals with familial mood disorders had substantial reductions (20%) in glial cell numbers.15 The other previous studies conducted on BA24 have mainly examined supracallosal regions. Similar to our findings, Bouras and colleagues18 reported no significant change in BA24a neuronal densities and soma sizes when comparing large samples of specimens from individuals with MDD and controls. These authors, however, did not analyze glial densities and did not report how many of the individuals with MDD from whom they obtained tissue samples had died by suicide. Analyzing layers individually in BA24b, a region dorsal to BA24a, Cotter and colleagues16 reported that, compared with those from controls, postmortem tissues from individuals with MDD (4 of whom died by suicide) displayed substantial decreases in glial cell density (22%) and reductions in neuronal soma size that were limited to layer VI. More recently, using a 2-dimensional approach in their study of BA24c, an area directly dorsal to BA24b, Chana and colleagues17 reported layer V decreases (9%) in neuronal soma sizes and an accompanying increase (24%) in neuronal density. In addition, these authors reported increases in neuronal soma size (30%) and glial nuclear size (10%–13%) in supragranular layers I and II.
Discrepancies between reports could be due to the heterogeneity of the ACC, which displays important cytoarchitectural differences throughout its anteroposterior and dorsoventral extents.39,40 Specifically, one study showed important cellular differences between the subgenual and supracallosal areas of BA24, with glial densities being about 25% lower in the former and neuronal densities higher in the latter.40 Thus, differences between quantitative morphological studies within supracallosal regions of BA24 may be owing, at least in part, to differential sampling of this heterogeneous structure. In the present study, we carefully delineated and sampled the supracallosal BA24a of each specimen according to well-established macroscopic and microscopic criteria.40
There is limited yet growing information in the current literature regarding cellular alterations in the brains of alcohol-dependent individuals. The majority of cell morphological investigations in such a study sample have been conducted on the prefrontal cortex or the hippocampus. To our knowledge, ours is the first study to have examined alcohol dependence as a potential influence on BA24 cells. There is evidence suggesting that specific neuronal subtypes in the brainstem are affected in alcohol-dependent suicide completers. Notably, lower numbers of noradrenergic and serotonergic neurons have been reported in such samples.32,34 In the present investigation, BA24a neuronal cell densities were not significantly altered between groups. These findings are in agreement with previous reports suggesting that neuronal loss is not a generalized phenomenon among alcoholics.44,45 However, and this is the most striking finding of the present study, glial cell densities were significantly increased in alcohol-dependent depressed suicide completers compared with suicide completers who were not alcohol-dependent (38%) and, to a lesser extent, with controls (30%).
Although reductions in glial densities within other cortical regions have previously been observed in samples from alcohol-dependent35,46 and depressed alcohol-dependent individuals,47 there is limited evidence describing increases in glial densities associated with alcohol dependence.33,48 The authors of the latter studies proposed that such increases could reflect glial cell proliferation in response to progressive degeneration and death of cortical neurons. We did not gather any evidence suggesting alterations in BA24a neuron numbers, but our results could indeed reflect increased cortical glial cell proliferation with alcohol dependence. However, caution should be taken when interpreting results based solely on these earlier studies as cells were classified into neurons or glia according to size. Furthermore, in these previous studies, substantial increases (11%) in glial cell densities were observed in a superior frontal region, while no significant changes were found in the ACC of alcoholics.
Although cresyl violet staining did not allow the identification of the glial cell type(s) responsible for the density increases observed in alcohol-dependent depressed suicide completers, it is tempting to speculate that this phenomenon results from an immune response leading to microgliosis, as recently observed in multiple brain regions of suicide completers, including the ACC, regardless of their psychiatric conditions.49 As these authors point out, increases in microglia could be the result of stress-related changes that are independent from suicidal behaviour. A similar mechanism could be occurring in alcohol-dependent suicide completers whereby a combination of immunomodulatory effects and stress-related factors would contribute to the observed increases in glial cell densitites. There is evidence suggesting that alcoholics experience chronically increased levels of cortisol, the main hormone mediating the stress response.31 Similarly, a 2-fold increase in the microglial markers, ionized calcium binding adaptor protein–1 and glucose transporter type 5, were recently observed within the ACC in postmortem tissue of alcohol-dependent individuals compared with controls.50 These authors also reported substantially higher levels of monocyte chemoattractant protein-1, a key cytokine mediating central nervous system inflammation and alcohol-drinking behaviour, in the brains of alcoholics across several limbic regions, including the hippocampus and amygdala. Furthermore, recent studies have shown that low concentrations of ethanol promote inflammatory processes involving glial cells in the brain by upregulating certain responses to inflammatory injury, such as the release of cytokines and the activation of associated signalling pathways.51,52 Future comparisons of BA24a glial cells between sudden-death controls with or without alcohol dependence but not other psychopathologies should help determine whether the present observations result from alcohol dependence alone.
Limitations
A few technical limitations need to be kept in mind when interpreting our findings. First, cresyl violet does not distinguish between subtypes of neuronal or glial cells. This raises the possibility that changes in specific cell subpopulations might have gone unnoticed in our analyses. Likewise, the significant increase in glial cell density measured in alcohol-dependent depressed suicide completers could not be attributed to a specific glial subtype. Second, to gain confidence in our analyses, we chose to delineate upper and lower cortical contours instead of individual layers. Some previous studies have suggested that statistically significant BA24 cell alterations are, for the most part, layer-specific. Therefore, the possibility remains that alterations occurring within particular layers between suicide completers and controls were missed because of our delineation. Lastly, the sample comprised specimens exclusively from men, thus we were unable to assess potential sex differences. Despite these limitations, our data strongly suggest that MDD and suicide do not affect glial and neuronal densities in BA24a. Furthermore, the results showing that glial cell densities across BA24a are robustly and specifically increased in alcohol-dependent depressed suicide completers suggest that alcohol dependence alone, or the comorbidity of alcohol dependence with MDD, has a profound influence on glial cells in this key limbic structure. Interestingly, previous studies using a similar approach in other brain regions have illustrated that in depression, glial cell populations display the most robust changes.22,23,27 Although we observed an increase in glial cell density in alcohol-dependent depressed suicide completers, it is important to mention that this sample size was fairly small in our study (n = 6). Future work should include larger samples to determine whether this robust effect is still present. Apart from these confines, it is increasingly clear that glial cells are major players in the altered brain plasticity thought to occur in depression and associated comorbidities. Future investigations are warranted to determine the glial cell subtype(s) affected by these conditions in the ACC and other relevant brain regions.
Acknowledgements
We gratefully acknowledge the collaboration of the Quebec Coroner’s Office, as well as the cooperation and support of the next-of-kin of the deceased. We also thank Simon Spanswick for his technical advice and Dr. Joseph Rochford for his statistical expertise. The Quebec Brain Bank is supported in part by the Fonds de la recherche en santé du Québec (FRSQ). This research was supported by a Canadian Institutes of Health Research grant to Dr. Turecki and by a Douglas Institute start-up grant to Dr. Mechawar. Dr. Mechawar is also an FRSQ scholar.
Footnotes
Competing interests: None declared.
Contributors: Ms. Hercher, Dr. Parent, Ms. Canetti, Dr. Turecki and Dr. Mechawar designed the study. Ms. Hercher carried out the experiments, acquired the data and performed the statistical analyses. Ms. Hercher and Dr. Mechawar wrote the manuscript. Drs. Parent, Flores and Turecki also analyzed and interpreted the data. All authors reviewed the article and approved its publication.
- Received September 9, 2008.
- Revision received December 28, 2008.
- Revision received January 7, 2009.
- Accepted January 8, 2009.