Abstract
Background: Abnormal neural responses to others’ emotions, particularly cues of threat and distress, have been implicated in the development of chronic violence. We examined neural responses to several emotional cues within a prospectively identified group of chronically violent men. We also explored the association between neural responses to social emotions and psychopathic features.
Methods: We compared neural responses to happy, sad, angry, fearful and neutral faces between chronically violent (n = 22) and non-violent (n = 20) men using functional magnetic resonance imaging (fMRI). Participants were prospectively identified from a longitudinal study based on information collected from age 7 to 27 years. We assessed psychopathic features using a self-report measure administered in adulthood.
Results: The chronically violent men exhibited significantly reduced neural responses in the dorsomedial prefrontal cortex to all faces, regardless of the emotional content, compared with nonviolent men. We also observed a hyperactive amygdala response to neutral faces in chronically violent men, but only within the context of viewing happy faces. Moreover, they exhibited a greater dorsomedial prefrontal cortex response to mildly fearful faces than nonviolent men. These abnormalities were not associated with psychopathic features in chronically violent men.
Limitations: It remains unclear whether the observed neural abnormalities preceded or are a consequence of persistent violence, and these results may not generalize to chronically violent women.
Conclusion: Chronically violent men exhibit a reduced neural response to facial cues regardless of emotional content. It appears that chronically violent men may view emotionally ambiguous facial cues as potentially threatening and implicitly reinterpret subtle cues of fear in others so they no longer elicit a negative response.
Introduction
Violence in men is a serious public health problem in the United States, with recent estimates suggesting that more than 2.2 million people are medically treated for violence each year, costing society about $37 billion in medical costs and lost productivity.1 Not included in these estimates are the substantial costs associated with arresting and incarcerating violent offenders and the unmeasurable emotional costs to victims and their families. This makes research aimed at understanding the developmental etiology and biological underpinnings of chronic violence in men a paramount societal concern. Investigations in this area have begun to indicate that dysfunction in the neural systems supporting emotion processing may be an important driver of early emerging violence that persists into adulthood. One theory posits that an attenuated cortico-limbic response (particularly within the amygdala) to cues of emotional distress, such as fear and sadness, are associated with the development of severe and persistent forms of violence.2 This deficit is believed to be most pronounced in individuals with high levels of psychopathic features, which include characteristics such as manipulativeness, conning and a lack of guilt or remorse.3 Another common viewpoint is that chronic violence is partially driven by a hypersensitivity to social cues of direct threat, such as angry faces.4 However, this hypersensitivity to threat is not believed to be uniquely associated with psychopathic features in chronically violent men.2
Indirect evidence linking chronic violence to a reduced neural responsiveness to others’ distress comes from studies indicating that antisocial populations tend to exhibit deficits in recognizing fearful and sad faces in others, although these findings have been less consistent for sad faces.5 There is also evidence that children with elevated conduct problems and psychopathic features show an attenuated right amygdala response to fearful faces,6,7 and psychopathic features have been associated with reduced amygdala reactivity to fearful faces and reduced orbitofrontal cortical reactivity to sad faces in violent adults with schizophrenia.8 However, not all studies have found a link between psychopathy and amygdala reactivity to fearful faces,9 suggesting that further research in this area is needed.
Theoretical models positing an overreactivity to threat cues in chronically violent men have been supported by studies indicating that children exhibiting early forms of violence10 and adult offenders with psychopathic features11 tend to attribute hostile intentions to others in ambiguous conflict situations. However, neuroimaging evidence supporting an increased neural reactivity to threat cues in aggressive and violent adults has been inconsistent. For example, adults with severe aggressive outbursts have been shown to exhibit higher levels of amygdala reactivity and less orbitofrontal (OFC) activation when viewing angry faces in comparison to controls.12 In contrast, a more recent study found that among violent offenders with schizophrenia, higher levels of psychopathic features were associated with lower amygdala, ventrolateral prefrontal cortex and anterior cingulate reactivity to angry faces.8
There is accumulating evidence suggesting that chronic violence may actually be associated with a more general emotion processing impairment than some theoretical models imply. Psychopathic features often found in chronically violent men have been associated with a diminished physiologic arousal to pleasant pictures and sounds,13,14 as well as reduced fusiform gyral activation to both positive pictures15 and happy faces.9 A negative association between psychopathic features and activity in neural regions, including the orbital and dorsolateral prefrontal cortex and amygdala, have also been found in healthy populations even when combining a diverse array of negative and positive emotional faces.16 There is also evidence that adults with psychopathic features exhibit a reduced reactivity in the dorsal and ventral anterior cingulate when processing a diverse array of negative words,17 and violent patients with schizophrenia and elevated psychopathic traits have a reduced amygdala response to facial expressions of disgust.8
Current study
The current study was designed to address several limitations in the existing literature. First, studies showing reduced amygdala reactivity to fearful faces in antisocial populations have been restricted to children6,7 and violent patients with schizophrenia and psychopathic features.8 Second, functional magnetic resonance imaging (fMRI) studies with violent adult populations without severe mental illness have not included a diverse array of positive and negative facial emotions, which is essential to testing the specificity of any neural abnormalities found in this population. Moreover, adult studies have historically relied on relatively small samples of offenders with poorly characterized histories of violence.9,15 In addition, studies in this area have not sufficiently examined whether an attenuated neural reactivity to distress cues is uniquely associated with psychopathic features or more generally associated with persistent forms of violence.
To address these limitations, we recruited a relatively large group of prospectively identified chronically violent and nonviolent men living in the community for participation in a neuroimaging study examining neural responses to different types of facial affect: fearful, sad, angry, happy, neutral. We hypothesized that chronically violent men would exhibit reduced cortico-limbic response to sad and fearful faces as cues of distress in others compared with nonviolent men, with these deficits being associated with psychopathic features. Conversely, chronically violent men were expected to have an increased cortico-limbic reactivity to angry faces as a cue of direct threat in comparison to nonviolent men, with this deficit being unrelated to levels of psychopathic features. We also examined whether there was a more general face processing abnormality in chronically violent men across all emotional expressions combined, and whether this deficit was related to psychopathic features.
Methods
Participants
We recruited 20 chronically violent and 22 nonviolent men from the youngest cohort (n = 503) of the Pittsburgh Youth Study (PYS), a longitudinal study of boys initially recruited from first-graders attending Pittsburgh public schools in 1986–1987. After enrollment in the study, the youngest cohort participated in assessments every 6 months for the first 4 years of the study, with annual assessments occurring over the next 9 years until the boys were about 19 years of age. A recent assessment of this cohort was conducted in young adulthood (mean age 26 yr). Further details regarding sample selection and procedures associated with the larger PYS can be found elsewhere.18,19 The University of Pittsburgh Institutional Review Board approved all procedures for the study, and informed consent was obtained from all participants.
Defining chronically violent and nonviolent men
We selected chronically violent and nonviolent men using criminal record information as well as parent, teacher and self-report assessments collected across the duration of the PYS. Participants were placed into violence groups using assessments collected across 4 developmental blocks: middle childhood (ages 7–9 yr), late childhood (10–12), early adolescence (13–15) and mid/late adolescence (16–19). Parent and teacher data on minor violence (i.e., physical fighting) were available for all phases except the mid/late adolescent block, and were collected using the Child Behavior Checklist20 and Teacher Report Form,21 respectively. Self-report of moderate/severe violence (e.g., attacking with a weapon, robbery) was available for all blocks except middle childhood and was collected using the Self-Report of Delinquency.18 Official criminal conviction data were available from ages 9 to 19 years and were collected using juvenile, state and federal records.22 At the final assessment at age 26, a 15-item lifetime history of violence questionnaire was administered, asking participants to indicate the last time they had committed a series of moderate/serious violent acts (e.g., attacking someone with a weapon, robbery).
To be considered violent in a developmental block, participants had to have been described as “often” engaging in minor violence for 1 or more years, reported engaging in a moderate/serious violent act for 1 or more years or had a criminal conviction for violence. We defined chronically violent men as individuals who exhibited violence in 3 or more developmental blocks and reported engaging in moderate/serious violence in the past 5 years during the last assessment in young adulthood (i.e., at a mean age of 26 yr). In contrast, nonviolent men could not have engaged in any moderate/serious violence or been involved in minor violence in more than 1 developmental block. We excluded men meeting DSM-IV criteria for a past-year diagnosis of mood, anxiety or psychotic disorders according to the Diagnostic Interview Schedule23 from participation. Participants were also excluded if they reported current use of psychotropic medications, a history of neurologic disease, structural brain injury, heart disease, disorders affecting blood flow or the central/peripheral nervous system, or had an estimated full-scale intelligence quotient (IQ) below 70 on the Weschler Abbreviated Scale of Intelligence.24
Measures
We used several scales to assess the comparability of the chronically violent and nonviolent groups on internalizing problems, externalizing problems and recent substance use. To assess state and trait levels of anxiety, participants completed the State-Trait Anxiety Inventory.25 We assessed problems related to DSM-IV diagnoses of depression/dysthymia and attention-deficit/hyperactivity disorder (ADHD) using severity scores from the Adult Self-Report Inventory 4.26 The self-report Substance Use Questionnaire18 was used to assess the typical number of drinks participants consumed when using alcohol over the past year and past-year use of marijuana and hard drugs (e.g., cocaine, heroin, methamphetamine).
The Self-Report of Psychopathy-III (SRP-III; D. Paulhus and C.S. Neumann, Multi-Health Systems: unpublished data, 2010) is a 64-item measure designed as a self-report analogue to the Psychopathy Checklist-Revised (PCL-R). The SRP-III has a 4-factor structure similar to the PCL-R, including sub-scales measuring interpersonal manipulation, callous affect, erratic lifestyle and criminal tendencies.27,28 Each subscale is made up of 16 items where participants indicate how true statements about them are on a 5-point scale (1 = “disagree strongly” and 5 = “agree strongly”). To avoid confounding psychopathy with general antisocial behaviour, items from the criminal tendency subscale were not administered.
To help detect unreported illicit substance use, participants underwent a urine drug screen using the Quest Diagnostics One Step Multi-Drug Screen just before scanning. The screen assessed for metabolites of amphetamines, barbiturates, benzodiazepines, cocaine, marijuana, methadone, methamphetamine, 3,4-methylenedioxymethamphetamine, opiates and phencyclidine. We asked all participants to refrain from substance use for 24 hours before the scan to prevent acute intoxication from influencing the results.
Neuroimaging paradigm
Participants completed 4 event-related experimental runs in which they viewed 10 different facial identities (5 male, 5 female) from a standardized series.29 Within each experimental run, participants viewed faces expressing prototypic emotional intensities and mild intensities achieved by creating a 50% morph of prototpyic and neutral facial expressions, allowing for the examination of the changes in neural response to facial expressions depicting different intensities of a specific emotion. For each experimental run, both mild and prototypic intensities of 1 of the 4 different emotions (i.e., happy, sad, angry, fearful), as well as neutral faces, were presented. The use of emotion-specific runs prevented the neural response to a specific facial emotion from being altered by the emotional context provided by a preceding different facial emotion (e.g., responses to happy faces preceded by fearful faces may be different from responses to happy faces preceded by sad faces). Each identity expressed each level of emotion and neutral faces and was presented twice within a run. There were therefore 60 stimulus presentations in each experimental run. The order of the experimental runs was random across participants. Each face stimuli was presented for 2 seconds followed by an interstimulus interval (ISI) consisting of a fixation cross that varied in duration from 3 to 8 seconds (average ISI 4.9 s). During scanning, participants performed a sex labelling task. This task was used because it has been shown to reliably activate limbic and extra-striate cortical regions in healthy controls30 and elucidate brain function differences between healthy controls and individuals with different forms of psychopathology, including bipolar disorder,31 depression32 and psychopathy.9
Neuroimaging data acquisition
We collected whole-brain fMRI data using a 3.0 T Siemens Allegra MRI scanner (head-only magnet). Blood oxygen level–dependent (BOLD) functional images (T2*-weighted) were acquired with a gradient echo planar imaging sequence covering 36 coronal slices (3.2-mm isotropic voxels, 0-mm gap) encompassing the entire cerebrum and most of the cerebellum: repetition time (TR) 2000 ms, echo time (TE) 25 ms, field of view (FOV) 204 mm, matrix 64 × 64; flip angle 79°. Structural images were obtained using high-resolution T1-weighted 3-dimensional (3-D) gradient echo imaging with a spoiled gradient recalled (SPGR) sequence: axial plane, TR 1630 ms, TE 2.48 ms, flip angle 8°, number of excitations 1, bandwidth 210 Hz/pixel, echo spacing 6.8 ms, matrix 256 × 256, FOV 204 mm, 224 slices, 0.8-mm isotropic thickness, 0-mm gap.
Neuroimaging data analyses
We analyzed all fMRI data using SPM5. All functional images were slice timing–corrected and motion-corrected using a 2-pass procedure. Participants’ estimated motion parameters were examined to ensure they did not move more than 3 mm in any direction. The high resolution structural image was then coregistered to the mean functional image, segmented using an automated procedure and spatially normalized to the Montreal Neurological Institute (MNI) template. We applied the spatial normalization parameters from this procedure to all functional images to warp them into MNI space. All functional images were then smoothed using a 6-mm full-width at half-maximum Gaussian kernel.
We performed statistical analysis of the fMRI data using restricted maximum likelihood estimation of first- and second-level models. At the first level, within-subjects analysis was conducted by specifying separate regressors for each of the 3 conditions (i.e., neutral, mild emotion, prototypic emotion) separately across the 4 emotion-specific runs. These regressors were convolved using a γ-variate hemodynamic response function, with the 6 motion parameters from the realignment procedure entered as covariates. A high pass filter cutoff of 128 seconds was applied to the data to remove slow signal drift. We used an autoregressive (AR1) model to remove serial correlations in the fMRI time series.
For the current study, the focus was on violence group differences between neural responses to faces regardless of emotional content, as well as unique deficits to specific emotional faces (i.e., angry, fearful, sad, happy) and intensities (i.e., mild, prototypic). To examine the former, we averaged the magnitude of the BOLD response indexed using the size of the β weight over all 4 facial affect runs for each individual and then compared the results between the nonviolent and chronically violent groups. Next, we compared the within-individual BOLD response index using the size of the β weight to each level of emotional intensity (i.e., neutral, mild, prototypic) relative to implicit baseline (i.e., fixation) between nonviolent and chronically violent participants separately for each emotion-specific run. Specifically, we conducted a 3 × 2 analysis of variance (ANOVA) with the primary effect of interest being the intensity (i.e., neutral, mild, prototypic) by group (i.e., nonviolent, chronically violent) interaction within each run.
We used a region-of-interest (ROI) approach to examine violence group differences. For the ROI analysis, we examined BOLD responses within the amygdala, OFC (Brodmann area [BA] 11/47), dorsomedial prefrontal cortex (DMPFC; BA 10/32) and medial prefrontal cortex (MPFC; BA 10/32) based on previous research implicating these areas in emotion processing.33 These regions were defined using anatomic masks from the WFU PickAtlas version 2.3.34 To correct for multiple comparisons, we used the Monte Carlo simulation program AlphaSim35 to determine the number of contiguous voxels needed within in each ROI to reach statistical significance (p < 0.05). Voxel-level thresholds were set to p < 0.05 for these a priori ROIs.
For significant clusters, we extracted average BOLD response values and imported them into SPSS for further analyses of relations with clinical variables and potential confounds. Of particular interest were correlations between the BOLD response in these regions and the scores of chronically violent ment on the interpersonal, affective and lifestyle sub-scales of the SRP-III.
Psychopathic features and amygdala response
We conducted a parallel analysis examining the association between the SRP-III subscales and neural responses to each emotion for the entire sample. This analysis focused specifically on the amygdala since reduced activity in this brain region has been implicated in the development of psychopathic traits. This analysis examined voxel-wise correlations between SRP-III subscales and BOLD response values within the amygdala across all participants, using the same contiguity threshold for significance as outlined above.
Results
Behavioural measures
Table 1 presents comparisons between the chronically violent and nonviolent groups on several behavioural measures. We found no significant group differences for IQ or measures of internalizing problems. However, chronically violent men had higher levels of ADHD-related behaviours and were slightly younger than the nonviolent men, although the mean difference between the groups was less than 1 year. A significantly greater proportion of chronically violent men reported hard drug use in the past year and had a positive urine test for marijuana use than nonviolent men. As expected, the groups differed significantly on measures of aggression/violence and psychopathic features. The scores of chronically violent men on the SRP-III subscales were roughly commensurate with those found for incarcerated offenders. Specifically, in a sample of 304 incarcerated men, average scores on the SRP-III subscales were 45 for interpersonal manipulation, 44 for callous affect and 53 for erratic lifestyle (D. Paulhus and C.S. Neumann, Multi-Health Systems: unpublished data, 2010).
Demographic and clinical characteristics of nonviolent and chronically violent men
The chronically violent group also had an extensive history of arrests and convictions according to official records. As part of the violence history questionnaire, all participants in the chronically violent group acknowledged engaging in at least 1 severe act of violence, including homicide (n = 3, 15.8%), attempted homicide (n = 12, 63.2%), shooting a gun at someone (n = 14, 73.7%), stabbing someone (n = 6, 31%), hitting someone with a weapon (n = 12, 63.2%) and/or sending someone to hospital as a result of violence (n = 18, 94.7%).
Task performance
The average percent accuracy of sex labelling during the fMRI task across the chronically violent and nonviolent groups for the angry, fearful, sad and happy face runs was 95.0% (standard deviation [SD] 0.06), 96.4% (SD 0.05), 94.8% (SD 0.11) and 95.4% (SD 0.06), respectively. The mean reaction times for runs involving angry, fearful, sad and happy faces were 1014 ms (SD 184 ms), 956 ms (SD 158 ms), 951 ms (SD 144 ms) and 965 ms (SD 158 ms), respectively. One participant did not have reaction time or accuracy data for the fearul face run owing to a computer error. For the accuracy data, repeated-measures ANOVA analysis indicated that the main effect of emotion type and group, as well as the emotion by group interaction were nonsignificant (all p > 0.05). For the reaction time data, we found a main effect of emotion type (F3,117 = 6.02, p < 0.001), with post hoc probing indicating that participants responded slower during the angry face run compared with all other runs (all p < 0.05). However, there was no significant violence group or violence group by emotion type interaction for reaction time (all p > 0.05).
Group difference: all faces
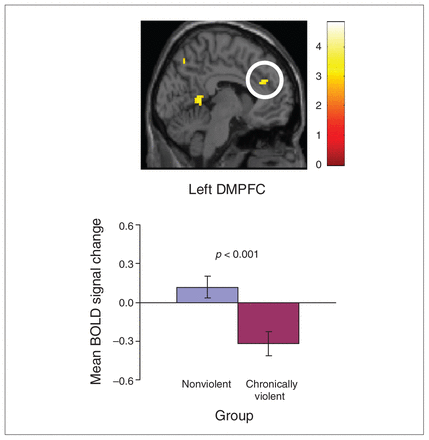
Brain regions exhibiting significant between-group differences for the BOLD response to all faces combined relative to implicit baseline (i.e., fixation cross) are presented in Table 2. For all brain regions, results indicated that nonviolent men exhibited a significantly higher BOLD response than chronically violent men. Significant group differences emerged in ROIs within the MPFC and anterior cingulate gyrus (ACG). Figure 1 presents mean BOLD signal change values for a region located in the DMPFC that significantly differed between chronically violent and nonviolent men. In chronically violent men, all SRP-III subscale scores were negatively correlated with the mean BOLD response to faces for the regions identified, but none of these associations were statistically significant (Table 2).
Group differences between chronically violent and nonviolent men to all faces combined for region within the dorsomedial prefrontal cortex (DMPFC). Cluster in the DMPFC (Montreal Neurological Institute 36, 27, 18) significant at p < 0.05, corrected using AlphaSim threshold for contiguous voxels. Bar graphs depict mean blood oxygen level–dependent (BOLD) response across voxels within the cluster along with standard errors. The p values are based on between-group t tests for extracted mean BOLD change.
Brain regions exhibiting significant violence group differences across all facial expressions combined and associations with psychopathic features*
Group difference: emotion-specific runs
Brain regions exhibiting a significant emotional intensity (i.e., neutral, mild, prototypical) by violence group interaction for each emotion-specific run are presented in Table 3. We found no significant interactions for runs involving angry or sad faces. However, we did find significant interactions for runs involving fearful and happy faces.
Brain regions exhibiting significant group by facial emotion interactions*
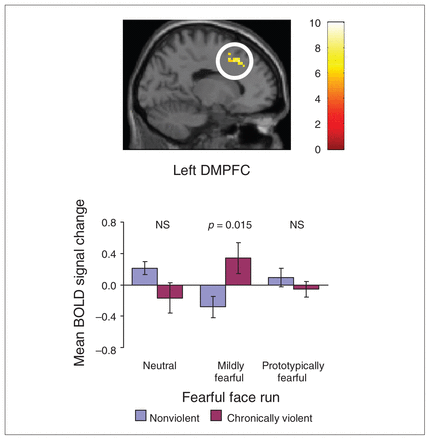
For the fearful face run, significant interactions emerged in the DMPFC/MPFC. In all regions, analysis of mean BOLD signal change values indicated that the violence groups differed primarily in their responses to mildly fearful faces, but not to neutral or intensely fearful faces. In all cases, chronically violent men had higher mean BOLD values than nonviolent men. Figure 2 shows the results for a region in the DMPFC indicating that chronically violent men exhibited a significantly higher BOLD response to mildly fearful faces than nonviolent men, but no difference was found for neutral or prototypically fearful faces. However, correlations between the SRP-III subscale scores in chroncially violent men and the mean BOLD response to mildly fearful faces in the regions identified were nonsignificant (all r from −0.33 to −0.07, all p > 0.14).
Interaction between violence group and emotional intensity within the fearful face run for region in the dorsomedial prefrontal cortex (DMPFC). Region in the DMPFC (Montreal Neurological Institute −15, 24, 42) significant at p < 0.05, corrected using AlphaSim threshold for contiguous voxels. Bar graphs depict mean blood oxygen level–dependent (BOLD) response across voxels within the cluster along with standard errors. The p values are based on between-group t tests for extracted mean BOLD change. NS = nonsignificant.
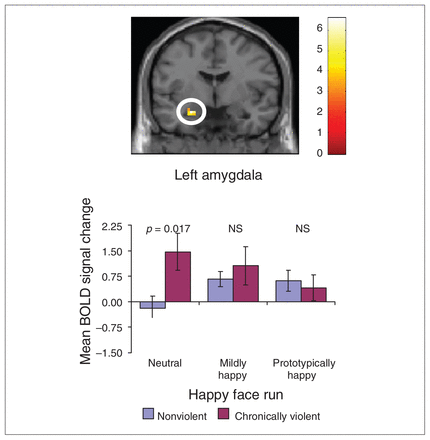
For the happy face run, there was a significant emotional intensity by group interaction in the left amygdala (Table 1). Follow-up analysis indicated that the violence groups significantly differed only in their BOLD responses to neutral faces, not to mildly or prototypically happy faces (Fig. 3). Chronically violent men exhibited a positive BOLD response in the left amygdala to neutral faces, whereas the nonviolent group experienced relatively little BOLD change to neutral faces. However, none of the SRP-III subscales were significantly correlated with amygdala responding to neutral faces in the happy run (all r from −0.33 to −0.07, all p > 0.12).
Interaction between violence group and emotional intensity within the happy face run for region in the amygdala. Region in the amygdala (Montreal Neurological Institute −15, 24, 42) significant at p < 0.05, corrected using AlphaSim threshold for contiguous voxels. Bar graphs depict mean blood oxygen level–dependent (BOLD) response across voxels within the cluster, along with standard errors. The p values are based on between-group t tests for extracted mean BOLD change. NS = nonsignificant.
Parallel analysis: psychopathic features and amygdala response
To supplement the prior analysis, we ran correlations examining the association between psychopathic features sub-scales and the amygdala BOLD response to each emotion versus implicit baseline (i.e., fixation) for all study participants. We found no significant associations between SRP-III subscales and the BOLD response to the different facial emotions presented within the amygdala, including expressions of fear and sadness (all p > 0.05).
Neural responses and potentially confounding demographic and clinical variables
Because chronically violent and nonviolent participants differed in age, past year hard drug use, positive urine screen for marijuana and ADHD symptoms, we used correlation analyses to examine whether these factors were significantly related to the mean BOLD signal change in the brain regions that differentiated the 2 violence groups. For regions presented in Table 2, we found nosignificant associations between mean BOLD change and age, past year hard drug use, positive urine screen for marijuana and ADHD symptoms. For the regions that differed between the violence groups for mildly fearful faces (Table 3), we found no significant associations between mean BOLD change and age, past year hard drug use, and positive urine screen for marijuana. However, ADHD symptoms were significantly related to an increased BOLD response in the right DMPFC (MNI 9, 57, −9) and MPFC (MNI 27, 51, −3). When violence group membership and ADHD symptoms were simultaneously entered in a regression model predicting BOLD change to mildly fearful faces, both violence group and ADHD symptoms failed to independently predict the neural response in these regions (all p > 0.07). We found no significant correlations between amygdala reactivity to neutral faces within the happy run and age, past year hard drug use, positive urine screen for marijuana and ADHD symptoms.
Discussion
This study examined whether a prospectively identified group of chronically violent men living within the community exhibited abnormal neural responses when viewing several types of facial affect (i.e., fearful, sad, angry, happy, neutral). Findings indicated that chronically violent men exhibit a reduced neural response across all facial expressions in the DMPFC and ACG. In comparison to nonviolent men, chronically violent participants exhibited higher levels of neural responding to mildly fearful faces in the DMPFC, as well as greater amygdala reactivity to neutral faces within the context of viewing happy faces. However, none of these abnormalities was related to psychopathic features within chronically violent men. In contrast to prior studies,6–8 there was no evidence that psychopathic features were associated with an abnormal amygdala response to emotional expressions of distress.
To our knowledge, this is the first study to show that chronically violent men exhibit abnormal neural responses to faces regardless of emotional content within regions of the DMPFC/ACG. Because these differences were not associated with psychopathic features, they appear to be characteristic of chronically violent men as a whole rather than specific to those men with psychopathic traits. It is also important to point out that the differences observed may not be specific to chronically violent men’s processing of faces since the contrast condition was an implicit baseline (i.e., fixation cross). It is possible that similar deficits may occur when examining differences between chronically violent and nonviolent men’s responses to other stimuli, making face-specific interpretations of this finding speculative. With this limitation in mind, evidence does indicate that regions of the DMPFC are intimately involved in attending to and processing information related to social relationships.36 Given that chronically violent men may have relatively little interest in connecting with others, they may attend less vigilantly to social cues involving facial features and exhibit less neural reactivity to these cues as a result.
Contrary to expectations, chronically violent men exhibited a significantly greater neural response to faces depicting mild fear in comparison with nonviolent men, particularly within regions of the MPFC and DMPFC. Research has consistently implicated the DMPFC in subprocesses related to both voluntary and involuntary emotion regulation, including attention redirection and reappraisal.33 It is possible that men exhibiting chronic violence implicitly reinterpret more ambiguous cues of fear in others so they no longer elicit a negative response. Although this may partially contribute to chronically violent men’s lack of emotional responsivity to distress cues in others, there was no evidence that psychopathic features were related to increased DMPFC responding to mildly fearful faces. This suggests that this neural abnormality may be characteristic of chronically violent men, regardless of their level of psychopathic features. The examination of potential confounds indicated that ADHD symptom severity was also associated with increased responding to mildly fearful faces in some, but not all, of the DMPFC regions identified. However, regression models suggested that neither chronic violence nor ADHD symptoms significantly predicted DMPFC responding after statistically controlling for their overlap. As a result, it remains unclear which of these 2 factors may best account for the neural abnormalities found in these brain regions.
An interesting finding from the current study was that chronically violent men exhibited a significantly greater amygdala response to neutral faces, but this was confined to the emotional run that included happy faces. This suggests that when neutral faces are presented alongside positive emotional stimuli they may be seen as potentially threatening or ambiguous to chronically violent men. This interpretation is consistent with studies indicating that severely antisocial children10 and adults11 tend to attribute hostile intentions to others when presented with ambiguous conflict situations. Moreover, greater than normal amygdala reactivity to neutral faces has been reported in studies with other psychiatric populations, including patients with schizophrenia,37 social anxiety disorder38 and pediatric bipolar disorder.39 The latter study further showed that children with bipolar disorder perceived neutral faces as more hostile than healthy children. Furthermore, persecutory delusions in patients with schizophrenia have been associated with a tendency to classify neutral stimuli as unpleasant.40 The current evidence indicates that an elevated amygdala response to neutral faces in the context of positive emotional stimuli may be characteristic of chronically violent men, regardless of their level of psychopathic features.
There was no evidence that psychopathic features were associated with reduced amygdala responding to distress cues in others, including fearful and sad faces. Whereas findings from the current study are inconsistent with previous studies showing lower amygdala reactivity to fearful faces among antisocial populations,6–8 there are several possible explanations for this discrepancy. First, these prior studies focused on children or patients with schizophrenia exhibiting elevated levels of antisocial and psychopathic features assessed at a single time point, rather than examining emotion processing in chronically violent men. Second, although the current investigation was strengthened by its examination of several different types of facial emotions, the relatively small number of faces presented for each emotion type may have limited our ability to detect significant effects. It is also possible that the use of a self-report measure of psychopathic features rather than clinical interviewer ratings may have contributed to the nonsignificant findings. However, self-report measures of psychopathic features have been successfully used with normal adult populations in prior studies.16,41
Limitations
There are some limitations to the present study. First, it is difficult to determine whether differences in neural response between chronically violent and nonviolent men represent developmental precursors to, or consequences of, persistent violence. To our knowledge, there are no longitudinal studies beginning in early neurodevelopment that have examined whether facial processing abnormalities prospectively predict persistent violence or the emergence of psychopathic features. It is also possible that the observed group differences between chronically violent and nonviolent men can be accounted for by some third unmeasured variable. The sample used in the present study was also exclusively male. It is therefore unclear if the current findings are generalizable to girls and women exhibiting violent behaviour. Moreover, some of the group differences that emerged were unanticipated, such as differences in amygdala response to neutral faces in the happy face experimental run, making these findings in need of further replication. It is also possible that differences in task design across studies may lead to divergent results. Whereas the current study used a fast event-related design with emotion-specific experimental runs, previous fMRI studies with antisocial youth have used block design paradigms focusing on fearful faces7 or intermixed angry, fearful and neutral faces within the same experimental run.6 Similarly, the decision to use emotion-specific experimental runs prevented us from comparing neural responses to different emotional expressions presented within the same experiment, which is an area in need of future study. However, the task employed has been validated in several previous investigations with both healthy30 and clinical populations,31 including one study on psychopathy.9
Conclusion
To our knowledge, this is the first study to examine the neural response to several different facial emotions among chronically violent men living in the community. The findings suggest that these men may exhibit a reduced response within brain regions involved in processing a wide variety of facial expressions, as well as a greater amygdala response to ambiguous social cues (i.e., neutral faces) and an increased DMPFC response to mildly fearful faces. The hypothesis that persistent forms of violence, as well as psychopathic features in particular, are associated with reduced amygdala reactivity to fearful or sad faces was not supported in the present study. This finding suggests that the neural basis of psychopathy is more complex than previously postulated. It also emphasizes the importance of replicating findings across multiple studies with diverse populations before drawing firm conclusions about the neural basis of both chronic violence and psychopathic features.
Acknowledgements
Neuroimaging data collection was supported by grants awarded to Dr. Pardini from the National Institute on Mental Health (1K01MH078039–01A1), Centers for Disease Control and Prevention (R49/CCR323155) through the National Center for Injury Prevention, and National Science Foundation (SES-0215551) through the National Consortium on Violence Research. The larger PYS study was supported by grants awarded to Dr. Rolf Loeber from the National Institute on Drug Abuse (DA411018), National Institute on Mental Health (MH 48890, MH 50778) and the Office of Juvenile Justice and Delinquency Prevention (96-MU-FX-0012). Any opinions, findings, conclusions or recommendations expressed in this paper are those of the authors and do not necessarily reflect the views of the National Science Foundation. Special thanks to Rolf Loeber, Magda Stouthamer-Loeber and to the PYS participants for their assistance on this project.
Footnotes
Competing interests: See above for Dr. Pardini. None declared for Dr. Phillips.
Contributors: Drs. Pardini and Phillips both designed the study, analyzed the data, reviewed the written article and approved its publication. Dr. Pardini acquired the data and wrote the article.
- Received February 25, 2010.
- Revision received May 11, 2010.
- Accepted May 11, 2010.