To date, the pathogenesis of schizophrenia remains poorly understood. However, neurodevelopmental theories have been used to explain this complex disorder, as converging lines of evidence suggest abnormal trajectory of brain development as a key pathological process.1 To understand the pathogenesis through a neurodevelopmental lens, sex differences observed between men and women, including age at onset, prevalence and clinical presentations, are important. This editorial uses the neurodevelopmental hypothesis as the main framework to reflect on and examine the variation of incidence between the sexes across the life span, with some emphasis on late-onset schizophrenia (LOS). We think that the neurodevelopmental theory may prove valuable in accounting for sex differences in early-onset schizophrenia (EOS), as sex differences in early brain development may point to differing ways the brains of women and men handle in-utero insults. However, this theory is limited in accounting for the neurobiological mechanisms underlying sex differences across the entire life span, as it may fail to explain the predominance of women with LOS. We think that the widely discussed role of estrogen in delaying the age of onset of schizophrenia in women may not be restricted to the “estrogen protection” hypothesis; rather, estrogen’s modulation of key brain structures and processes in the adult brain, perhaps through epigenetic changes, may be more intimately connected to LOS in women. Therefore, the classical neurodevelopmental theories may not be sufficient to allow a full understanding of the sex differences in patients with schizophrenia.
What are the differences between men and women?
The incidence of schizophrenia in men is approximately 40% higher than in women,2 and men have an earlier peak age at onset.3 Sex differences are especially relevant in LOS. Manfred Bleuler first described LOS in 1943, and it remains distinct in its clinical profile compared with EOS.3 Late-onset schizophrenia has a 1-year prevalence rate of 0.6% when accounting for patients in whom the disorder is diagnosed after age 45, and it is reported that 23% of patients with schizophrenia had an onset of symptoms after age 40.4 While men appear to be at a greater risk than women for EOS, women have consistently accounted for a greater proportion of patients with LOS than men.4 A study examining LOS reported that women make up 66%–87% of patients with LOS, with onset after the age of 40–50 years.5 Further, women with LOS may have less severe negative symptoms and present with more positive symptoms, particularly sensory hallucinations and persecutory delusions.6 Because of this clinical profile and a better preservation of the cognitive functions, LOS is generally considered to have a better prognosis than EOS.4 Neuroimaging studies in patients with LOS show brain abnormalities similar to those in patients with EOS, such as higher ventricular:brain ratio, but these findings are not conclusive.4 These key symptomatic differences as well as the observation that women account for a greater proportion of patients with LOS than men suggest that sex plays an important role in elucidating the etiology of both EOS and LOS.
The second peak of incidence in women: Is it so obvious?
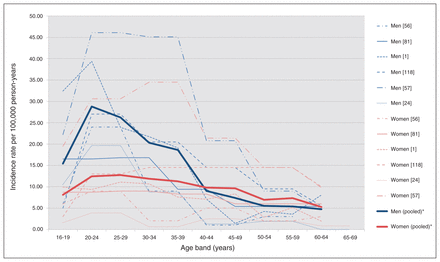
A meta-analysis conducted by Kirkbride and colleagues7 compiling schizophrenia incidence rates over a 6-year period in England noted a “secondary peak” of incidence in women near the age of 45 years. This concept has also been discussed in other reports.8 However, consideration of Figure S4 in the meta-analysis by Kirkbride and colleagues7 does not show a peak of incidence in middle-aged women (Fig. 1). Men have a notable peak of incidence in late adolescence and a subsequent sharp decline into middle age. However, incidence rates in women remain largely flat across all ages, with the exception of a minor peak in adolescence that corresponds in time with the peak in men. Thus, it appears that this “second peak” in women is not actually a peak. Rather, the higher proportion of women affected by LOS may be explained by the drastic drop in incidence rates in men at this stage of life, giving the appearance of a second peak in women. Further, what was particularly striking in the meta-analysis is the different trajectory of incidence rates in men and women, especially before middle age (Fig. 1). Therefore, the role of sex is not limited to LOS, and perhaps it is more important to explain differences in incidence rates across all ages.
Incidence of schizophrenia by age and sex in England, 1950–2009, pooled and per relevant citation. Reproduced with permission from Kirkbride and colleagues.7
How can we explain the sex differences?
It is advantageous to evaluate sex differences under the neurodevelopmental hypothesis, as this remains a dominant view in explaining the etiology of schizophrenia. Under the classical variant of this hypothesis, schizophrenia is the result of genetic and/or environmental insults in utero or during the perinatal period that lead to alterations in neural development and connectivity.9 The theory has gained momentum, as neuroimaging studies provide support for brain abnormalities in first-episode patients and in patients with chronic schizophrenia that appear (in a large number of studies, but not all) to remain static and nonprogressive. Excluding the rare childhood onset cases, schizophrenia commonly manifests in late adolescence/early adulthood, and it is not completely clear why the major signs and symptoms of the disorder remain silent from birth until about adolescence. One explanation is that brain maturational processes continue from embryonic periods to adolescence and that early insults may not impart their effects until the brain has completed its development.10 Therefore, when trying to understand sex differences in schizophrenia, the developing brain must be examined both in prenatal life and in early childhood. Analyzing the data presented in the meta-analysis by Kirkbride and colleagues7 that plotted incidence rates against age, we see the data from men follow a much clearer neurodevelopmental trajectory than those from women. The sharp peak in a discrete time period in late adolescence followed by a steep decline corresponds well to a disorder linked with abnormal brain development. However, it is more difficult to establish this trend in women owing to the flatter incidence rates. Women may follow a neurodevelopmental pattern, but they appear to be at lower risk owing to the smaller peak (Fig. 1). If men are at a greater risk for EOS, how can this be explained from a neurodevelopmental perspective?
Neuroimaging studies have found significant sex differences in brain development trajectories during childhood and adolescence in healthy participants.11 Grey matter volumes peak at an earlier age in women than in men, and the pattern is consistent across many brain structures. One interpretation of these differences could be that boys have longer childhood periods of dynamic brain changes. Studies in animal models examining neurodevelopmental trajectories after an in utero immune challenge found that structural abnormalities in brain development showed sex specificity in rats.12 Therefore, is it possible that there is a higher probability of early insults manifesting as schizophrenia in men due to longer periods of dynamic brain changes during the critical years of brain maturation? Sex differences in neurotransmitters and growth factors that guide neuronal pathways may also have implications for how the developing brain handles environmental insults. For example, dopaminergic neurons migrate and differentiate in early fetal life, and sexual dimorphisms in dopaminergic neurons during rat brain development have been reported.13 In addition, developmental differences in amino acid neurotransmitter levels in the rat brain have shown sex specificity.14 These differences are important for neurodevelopment, as glutamate promotes dendritic growth in hippocampal pyramidal neurons and γ-aminobutyric acid increases neuronal migration. Sex differences in the central nervous system colonization and function of microglia may also be a contributing factor to vulnerability windows in neural development.15 It has been proposed that boys and girls may respond differently to a challenge to the prenatal immune system, as studies found microglia with a more activated phenotype in female rats.15 Taken together, sex differences in the developing brain may provide plausible explanations for a greater proportion of men affected by EOS due to a greater susceptibility to insults in utero. This may connect sex differences observed in patients with EOS with the neurodevelopmental hypothesis.
While the discussed sexual dimorphisms in brain development provide insight to account for the trajectory in men, what about the predominance of women with LOS? The widespread hypothesis remains that of “estrogen protection,” suggesting that estrogen delays the age of onset of schizophrenia possibly by acting as an anti-dopamine agent16 and that 17-β estradiol may be an antipsychotic-like agent.17 Therefore, estrogen decline during menopause may lead to psychosis and the onset of schizophrenia. The estrogen protection theory can be used to explain LOS as a neurodevelopmental disorder by proposing that estrogen masks developmental insults until menopause. However, it is difficult to believe that estrogen could mask the disorder for such a substantial period of an individual’s life. Furthermore, if estrogen were merely delaying the onset of a neurodevelopmental form of schizophrenia, one would assume that the cognitive and negative symptoms would show greater similarity to those of EOS. However, it has been consistently reported that patients with LOS have less severe negative symptoms and a greater tendency toward hallucinations and delusions. Thus, the neurodevelopmental framework and the estrogen protection theory seem limited in accounting for sex differences in patients with LOS.
Estrogen may still have a role in explaining LOS owing to its interaction with stress and adult neurogenesis. Studies suggest that women may have a greater vulnerability to stress due to sexual dimorphism in the hypothalamus–pituitary–adrenal (HPA) axis. Estradiol is a key modulator of the HPA axis, and postmenopausal women are reported to show an enhanced cortisol response to the corticotropin releasing hormone test compared with young controls.18 Thus, declining estrogen in postmenopausal women may create a more maladaptive response to stressful stimuli. The role of gonadal and stress hormones on adult hippocampal neurogenesis may also prove valuable in understanding LOS in women.19 Although adult neurogenesis may be considered to fall within the extended neurodevelopmental window encompassing both fetal and adult lives, it is not generally invoked in the context of classical neurodevelopmental theories of schizophrenia. It is then logical to consider that women may be at greater risk for LOS owing to a genetic predisposition interacting with declining estrogen and stress hormones whose effects may include an impact on ongoing adult neurogenesis. In a sense, this suggests an epigenetic cause of LOS in women, which can be supported by sex differences seen in the epigenetic response to stress.20 Taken together, we believe that a late, detrimental combination of a gene × environmental interaction may be more plausible than a hidden neurodevelopmental disorder masked by estrogen when describing LOS in women. Perhaps it may be more valuable to study the mechanistic underpinnings of the origins of LOS as a separate category.21
Acknowledgements
Writing of this editorial was supported by a Canadian Institutes of Health Research grant to L. Srivastava.
Footnotes
Competing interests: None declared.