Abstract
Background: Attention-deficit/hyperactivity disorder (ADHD) is an early-onset neurodevelopmental disorder with multiple behavioural problems and executive dysfunctions for which neuroimaging studies have reported a variety of abnormalities, with inconsistencies partly owing to confounding by medication and concurrent psychiatric disease. We aimed to investigate the microstructural abnormalities of white matter in unmedicated children and adolescents with pure ADHD and to explore the association between these abnormalities and behavioural symptoms and executive functions.
Methods: We assessed children and adolescents with ADHD and healthy controls using psychiatric interviews. Behavioural problems were rated using the revised Conners’ Parent Rating Scale, and executive functions were measured using the Stroop Colour-Word Test and the Wisconsin Card Sorting test. We acquired diffusion tensor imaging data using a 3 T MRI system, and we compared diffusion parameters, including fractional anisotropy (FA) and mean, axial and radial diffusivities, between the 2 groups.
Results: Thirty-three children and adolescents with ADHD and 35 healthy controls were included in our study. In patients compared with controls, FA was increased in the left posterior cingulum bundle as a result of both increased axial diffusivity and decreased radial diffusivity. In addition, the averaged FA of the cluster in this region correlated with behavioural measures as well as executive function in patients with ADHD.
Limitations This study was limited by its cross-sectional design and small sample size. The cluster size of the significant result was small.
Conclusion: Our findings suggest that white matter abnormalities within the limbic network could be part of the neural underpinning of behavioural problems and executive dysfunction in patients with ADHD.
Introduction
Attention-deficit/hyperactivity disorder (ADHD) is one of the most common neurodevelopmental disorders in childhood; it has a reported prevalence of 5.3% in school-age children1 and 2.5% in adults2 worldwide. As well as behavioural problems (inappropriate hyperactivity and impulsivity, inattention symptoms), patients show deficits in executive functions (interference control, cognitive flexibility, working memory, planning and organization)3 that can impair academic and vocational performance.4 This disorder is a childhood-onset condition, so the pathophysiological profile in adult patients is more likely to be complicated by interactions with other factors during neuromaturation.5 Studies of children and adolescents with ADHD may therefore give a clearer view of its pathogenesis.
Neuroimaging methods have been useful in exploring ADHD pathophysiology. Much has been learned from macrostructural MRI about abnormalities in grey matter structures, such as the basal ganglia, prefrontal lobe, temporal and parietal cortices,6 and from fMRI about abnormal activation patterns in frontostriatal, frontotemporal and frontoparietal regions.7 This functional work has led to the notion of abnormal large-scale brain circuits in ADHD — notably the frontal–striatal–cerebellar circuit,8 with more recent evidence implicating the default mode (DMN), limbic and visual networks.9 Attention has also turned to white matter microstructural alterations in patients with ADHD8 and to models involving abnormal connections between multiple functional units in neural networks conceived as ensembles of neuronal bodies and axonal pathways.10 The importance of white matter deficits in patients with ADHD is supported by the observation that the effect sizes for volume reductions in total brain and lobar volumes are larger in white matter (0.30–0.64) than in grey matter (0.27–0.35).11
Diffusion tensor imaging (DTI) offers a sensitive and neurobiologically relevant way to characterize microstructural white matter alterations in vivo.12 Of several DTI-derived indices, fractional anisotropy (FA) is the most commonly used, reporting the local and regional directional coherence of water diffusion.13 Additional information is provided by the decomposition of FA into axial diffusivity (AD) and radial diffusivity (RD), which reflect diffusion parallel to and perpendicular to the axons, respectively, and by mean diffusivity (MD), which reflects the magnitude of diffusion in white matter pathways.13 Of the various analytic approaches to DTI, whole-brain analysis provides the best unbiased measure of brain regions that may be missed by region of interest (ROI) analysis based on a priori assumptions and thus offers greater reliability in screening distributed anomalies in patients with ADHD.14
Nine published DTI studies have used whole-brain approaches in children and adolescents with ADHD,5,15–22 reporting altered FA over diverse white matter structures. Such discrepancies may be owing to the differences in sample sizes, demographic characteristics and analysis pipelines (see the Appendix, Table S1, available at jpn.ca). Two of these factors deserve particular attention. Most of these studies included patients who either were currently taking medication or who had a history of taking medication; as medication has been reported to normalize white matter deficits,11 the potential effect of medication is an important confounding factor. The other main confounding factor is comorbid presentation with other psychiatric and/or developmental disorders that may have unique etiological pathways or share pathophysiology with ADHD.23
Another important issue about ADHD is the poorly understood association between the atypical brain circuitry revealed with neuroimaging methods and the executive dysfunctions measured with neuropsychological tests.24 Although fMRI studies have suggested an association with the aberrant frontoparietal circuit,7 debate remains about which core deficits of brain function lead to the impairments of ADHD.25 Diffusion tensor imaging may provide evidence of microstructural abnormalities in the brain circuits underlying specific executive deficits.
We therefore set out to study a relatively large sample of medication-naive children and adolescents with pure ADHD, defining white matter microstructural abnormalities through a whole-brain DTI approach and testing for their correlations with clinical symptoms and executive dysfunctions.
Methods:
Participants
We recruited patients with ADHD from the Department of Psychiatry at West China Hospital, Sichuan University. Diagnosis of ADHD was determined by 2 experienced clinical psychiatrists (L.G. and N.H.; 28 and 5 yr of experience in clinical psychiatry, respectively) using the Chinese modified version of Structured Clinical Interview for DSM-IV-TR Axis I Disorders, Research Version (SCID-I, patient edition).26 We excluded patients with oppositional defiant disorder, conduct disorder, Tourette disorder or any other Axis I psychiatric comorbid disorders. Other exclusion criteria were a full-scale IQ lower than 90 based on an age-appropriate Wechsler Intelligence Scale for Children — Chinese Revision,27 current use of or history of taking psychotropic medication, left-handedness as assessed using the Annett Hand Preference Questionnaire,28 pregnancy or substantial physical illness and standard MRI scanning contraindications.
We recruited healthy controls via advertisement in local schools; controls were also screened using the Chinese modified version of SCID-I (nonpatient edition) to exclude any Axis I psychiatric diagnosis and use of any psychotropic medications. Controls were required to have no family history of psychiatric illness in first-degree relatives. Other exclusion criteria were the same as those for the ADHD group.
Two neuroradiologists (L.C. and X.H., 3.5 and 3 yr of experience in neuroimaging, respectively) inspected conventional MRIs of all participants to exclude gross abnormalities.
Approval for this study was granted by the ethical committee of Sichuan University and the research ethics board at the West China Hospital of Sichuan University. All participants and their parents were fully informed about the purpose and procedures of this study, and written informed consent was obtained from the parents.
Behavioural measures and executive function tests
We used the Chinese version of the revised Conners’ Parent Rating Scale (CPRS)29 to measure the behavioural problems of ADHD patients. The CPRS is a battery of questions to evaluate problematic behaviour across areas such as sleep, temper and peer relationships. The parent rates each behaviour on a 4-point Likert-type scale; the behaviours are aggregated into 6 factors: conduct problems, study problems, psychosomatic, impulsive–hyperactive, anxiety and hyperactivity index. Higher scores indicate more severe problems.
The Stroop Colour-Word (Stroop-CW) Test30 and the Wisconsin Card Sorting Test (WCST)31 are commonly used neuropsychiatric tests of executive abilities. The Stroop-CW Test, in which the participant is shown a list of names of colors printed in mismatching colors and is asked to name the colour instead of the word, is often used to measure executive interference control;32 the dependent variables are number right, number error, number correction and total time. The WCST (64-card version), in which the participant is asked to change matching strategies after correctly matching a card according to a certain stimulus feature (number, colour, form) for 10 consecutive trials, mainly reflects cognitive flexibility,33 involving perceptual learning, set-shifting, working memory and executive control; the dependent variables are perseverative errors, nonperseverative errors, total errors and categories completed.
Data acquisition
Whole brain MRI data were acquired in the West China Hospital, Sichuan University, using a Siemens Trio 3 T MRI system with an 8-channel phase-array head coil. Sequences included a spoiled gradient-recalled echo sequence to acquire the T1-weighted images (repetition time [TR] 1900 ms, echo time [TE] 2.26 ms, section thickness 1 mm; matrix 256 × 256; field of view [FOV] 256 mm) and a single-shot echo-planar sequence (TR 6800 ms, TE 93 ms, section thickness 3 mm with no intersection gaps, matrix 128 × 128, FOV 230 mm) to obtain DTIs in 50 axial planes with 20 noncollinear diffusion sensitization gradients (b = 1000 s/mm2) as well as a reference image with no diffusion weighting (b0 image). Earplugs were used, and participants lay supine with their heads fixed by foam pads to minimize head movement. We took several steps to ensure optimal scan quality: good communication with participants and their parents allayed any worries and helped the participants cooperate during scanning. If monitoring detected any movement, the scanning sequence was halted and repeated; the acquired images were checked immediately by 2 neuroradiologists (L.C. and X.H.), and those with suspected artifacts were rescanned. Finally, during preprocessing all DTI maps were checked for artifacts in order to discard all data from the affected participants.
Statistical analysis
Diffusion-weighted images of each participant were analyzed using the Diffusion Toolbox FSL software version 1.1 (FMRIB Software Library). First, the data were corrected for head motion and eddy currents using affine registration to the b0 image volume. Next, brain extraction was performed to delete nonbrain tissue from the whole head image. Finally, we calculated parameter maps for FA, MD, AD and RD using “dtifit,” which fits a diffusion tensor model to each voxel and estimates the principal directions of diffusion. Voxel-based analysis was then performed using SPM8 (Welcome Trust Centre for Neuroimaging; http://fil.ion.ucl.ac.uk) running in MatLab 2012 (MathWorks). Each b0 image was nonlinearly normalized using the echo-planar imaging template supplied with SPM8 to estimate the normalization parameter, which was applied to all parameter maps, each voxel being 2 × 2 × 2 mm3. Finally, the normalized parameter maps were smoothed using an isotropic Gaussian filter (6 mm full-width at half-maximum).
We performed a voxel-wise comparison of FA and MD between patients with ADHD and controls using a 2-sample t test in SPM8 with age and sex as covariates. The threshold was taken as p < 0.05 for false discovery rate (FDR) correction. The resultant clusters were defined as ROIs through the MarsBaR toolbox in SPM8, and we extracted their averaged FA and MD values. We performed a 2-tailed Pearson correlation analysis in the ADHD group between the extracted values and multiple measurements, including CPRS, Stroop-CW Test and WCST results, using SPSS version 20.0 (IBM).
We extracted AD and RD values from clusters identified with significant FA differences through the MarsBaR toolbox and compared them between the ADHD and control groups using a 2 sample t test in SPSS.
As adolescents are undergoing rapid brain development, we performed a subgroup analysis on participants younger than 12 years. To minimize sex influences, we also performed a subgroup analysis on male participants.
Results:
Participants
Our study included 33 patients aged 7–16 years with ADHD (all combined subtype) who were drug-naive and did not have any comorbidities. We also included 35 healthy controls aged 7–14 years. All participants were from the Chinese Han population and were right-handed. As shown in Table 1, demographic variables, including age, sex, IQ scores, birth weight and parental education years were not significantly different between the 2 groups.
Demographic characteristics, behavioural measures and executive functions of patients with ADHD and healthy controls
Behavioural measures and executive function tests
Table 1 shows the results of behavioural measures and executive function tests. As expected, patients with ADHD had more severe behavioural problems and worse executive function than controls. In the CPRS, patients scored higher than controls in all indices (p < 0.001) except anxiety (p = 0.30). In the Stroop-CW test, patients had fewer right answers (p < 0.001), more errors (p < 0.001) and corrections (p < 0.001) and longer total time (p < 0.001) than controls. In the WCST, patients achieved fewer total correct reponses (p = 0.002) and categories completed (p = 0.001) and made more total errors (p = 0.001), perseverative errors (p = 0.001) and nonperseverative errors (p = 0.006) than controls.
Group differences in diffusion characteristics and correlation analysis
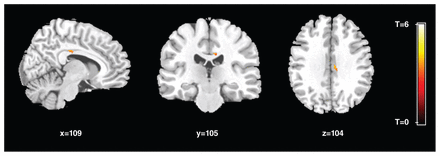
As shown in Figure 1, relative to controls, patients with ADHD had 1 cluster showing increased FA in the left posterior cingulum bundle (MNI coordinates: x, y, z = −6, −20, 32, t = 5.23, cluster size 8 voxels). In no instance did patients with ADHD show significantly lower FA than controls, and there were no significant differences in MD between the ADHD and control groups. For the cluster with significant FA differences, the ADHD group had significantly higher AD and lower RD than controls (AD: ADHD 12.00 ± 0.60 v. control 11.42 ± 0.57, p < 0.001; RD: ADHD 7.17 ± 0.53 v. control 7.42 ± 0.40, p = 0.036; all units 10−4 mm2/s).
Regions with significantly increased fractional anisotropy (FA) in patients with attention-deficit/hyperactivity disorder (ADHD). One cluster with increased FA in the left posterior cingulum bundle in patients compared with controls, corrected for multiple comparison (p < 0.05 after false-discovery rate correction).
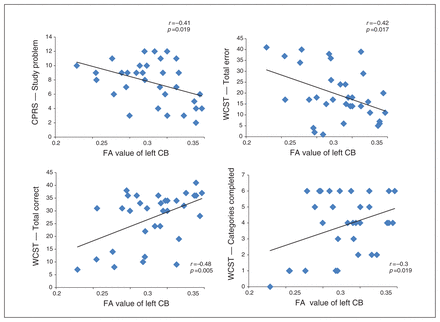
As shown in Figure 2, the average FA of the cluster in the left posterior cingulum bundle showed significant negative correlations with the scores for the CPRS study problems factor (r = −0.41, p = 0.019) and the WCST total errors factor (r = −0.41, p = 0.017) and positive correlations with the WCST total correct factor (r = 0.48, p = 0.005) and the categories completed factor (r = 0.36, p = 0.046). No other significant correlation was detected.
Correlations of fractional anisotropy (FA) in left posterior cingulum bundle (CB). The averaged FA of the cluster in the left posterior CB showed significant negative correlations with the study problem scores on the Conners’ Parents Rating Scale (CPRS) and the total errors scores on the Wisconsin Card Sorting Test (WCST; upper panels) and positive correlations with the total correct and categories completed scores on the WCST (lower panels).
Subgroup analysis
Our subgroup analysis of participants younger than 12 years included 26 patients (mean age 8.7 ± 1.3 yr, all boys) and 23 controls (age 9.7 ± 1.2 yr, all boys); there was a significant age difference between the 2 groups (p = 0.013). Voxel-wise comparison of FA between the 2 groups revealed no cluster with significant FA differences after multiple comparison correction (p < 0.05 for FDR correction). At a relaxed threshold of p < 0.001 for voxel level, 1 cluster was revealed to have higher FA in patients with ADHD than controls and was located close to the cluster identified by the whole group analysis in the left posterior cingulum bundle (MNI coordinates: x, y, z = −6, −22, 32, t = 4.17, cluster size 20 voxels). In the whole-group analysis the larger sample size probably enhanced the effect size of this cluster so that it survived multiple comparison correction.
Our subgroup analysis of boys only included 32 patients (mean age 9.6 ± 2.3 yr) and 33 controls (mean age 10.6 ± 1.7 yr), with no significant age difference between the 2 groups (p = 0.07). After multiple comparison correction (p < 0.05 for FDR correction), 1 cluster was revealed to have elevated FA in the ADHD group and was located close to the cluster identified by the whole group analysis in the left posterior cingulum bundle (MNI coordinates: x, y, z = −6, −22, 32, t = 5.28, cluster size 14 voxels).
No cluster with decreased FA was found in the ADHD group in either subgroup analysis.
Discussion
Using whole brain DTI analysis we have characterized white matter microstructural abnormalities in a relatively large sample of medication-naive children and adolescents with pure ADHD. Compared with healthy controls, patients showed increased FA in the left posterior cingulum bundle. Furthermore, the elevated FA in this cluster also showed significant correlations with behavioural measures on the CPRS and executive function scores on the WCST, supporting a role for white matter microstructure abnormalities in the pathophysiology of ADHD. This anomalous microstructure was also found in subanalyses of boys only and of children younger than 12 years, suggesting the independence of sex and maturation effects.
The cingulum bundle is the longitudinally oriented aggregate of myelinated axons underlying the cingulate cortex, whose projections connect different parts of the cingulate cortex with other brain areas, especially the medial frontal lobe, the temporal lobe and the parahippocampal gyrus.34 As an important component of the limbic system, the cingulum bundle is involved in emotions and cognitive functions, including attention, memory and motivation,35,36 and shows abnormalities in psychiatric disorders, including schizophrenia, major depression and obsessive–compulsive disorder.37–39
Previous DTI studies of ADHD have reported FA abnormalities in the cingulum bundle, but the results have been inconsistent. Four studies have reported decreased FA.10,40–42 Makris and colleagues10 found decreased FA in the right cingulum bundle in adults with childhood ADHD compared with healthy controls, suggesting the persistence of structural abnormalities into adulthood. Konrad and colleagues40 replicated this finding with a larger sample of medication-naive adult patients.40 Two studies using diffusion spectrum imaging tractography41 and DTI tractography,42 respectively, also found lower FA in the bilateral cingulum bundle in youths with ADHD. In contrast, our study and 2 others16,32 reported increased FA of the cingulum bundle in children and adolescents with ADHD; 1 study,16 based on a detailed analysis of eigenvalues (λ1, λ2 and λ3), further proposed reduced neural branching as an explanation for this FA increase. Finally, several studies have reported no abnormalities in the cingulum bundle in patients with ADHD.43–45 These discrepancies might be explained by variations in methodology and demographic characteristics (e.g., medication status, different comorbidities and subtypes) of participants. To minimize such confounding, the present study included only drug-naive patients with the combined ADHD type, and participants were carefully screened for comorbidities.
Our finding of increased FA in the cingulum bundle at the posterior cingulate gyrus region is therefore consistent with those of 2 previous studies.16,21 Little consideration has been given in the pathophysiology of ADHD to dysfunction of the posterior cingulum bundle, although it is a plausible hypothesis given that this carries the major efferent and afferent fibres to the posterior cingulate cortex, which participates in the regulation of attention46 and internally directed cognition47 and is a key component of the DMN. The DMN is a brain network associated with self-reflection, self-referential cognitions and mind-wandering. It is active in a state of rest and deactivated during tasks that make cognitive demands, with stronger deactivation corresponding to increasing attentional demands.48,49 Increasingly, ADHD is considered as a DMN disorder50 in which attentional lapses derive from an inappropriate persistence of DMN activation49; for example, a study using a stop signal task found that failure to suppress the DMN was associated with greater errors.51 Recent pathophysiological models of ADHD recognize it as a disorder encompassing large-scale neural networks, such as the cognitive control network and limbic networks, not only abnormal connectivity within the DMN itself. For instance, studies have found a negative correlation between the cognitive control network and the DMN in controls, whereas this inverse correlation was attenuated in patients with ADHD.52 In this conception, the interplay of the DMN with other neural networks is likely crucial to suboptimal neural functioning.9 As the posterior cingulum bundle is a junction containing reciprocal connections between the prefrontal cortex, anterior cingulate cortex and posterior cingulate cortex,53 the abnormality we have identified is in agreement with this hypothesis.
Our correlation analysis provides further support for a pathophysiological role of the posterior cingulum bundle in patients with ADHD and is consistent with the hypothesis that components of executive functions may be associated with the limbic system in addition to the prefrontal regions.54,55 However, it is interesting to note that in our study patients with higher FA displayed better executive functions, albeit still worse than those of healthy controls. This phenomenon was first noted in one of our previous studies,19 in which we found a positive correlation between increased FA of right frontal white matter and the Stroop-CW Test scores in another group of children with ADHD.
As for the reason why FA is increased in patients with ADHD, further analysis of AD and RD showed that this resulted from both increased water mobility along the principal fibre direction (AD) and decreased mobility perpendicular to the fibre direction (RD), with the former component being predominant. Numerous factors, such as fibre myelination, fibre crossings, fibre density and caliber, influence AD and RD, so the exact pathological mechanism of the increased FA is uncertain. Some previous studies identifying increased FA in patients have suggested abnormally decreased dendritic branching or anomalous white matter development.16,21 However, as both the present study and our earlier study published in 201019 found better executive function in patients with higher FA, we are more inclined to postulate a compensatory mechanism in children and adolescents with ADHD; for (hypothetical) example, excessive myelin hyperplasia in initially defective fibres, perhaps partially compensating for the cognitive deficits.
Limitations
Our study has some limitations. First, the sample size was small, although it was larger than those of previous whole brain DTI studies in children and adolescents with ADHD. Second, the adoption of FDR correction enhanced the impact of our result, but the small cluster size (8 voxels) limited its interpretation. FInally, a cross-sectional study cannot shed light on the development of the condition; longitudinal studies are therefore required to confirm and extend our conclusions.
Conclusion:
We found that medication-naive children and adolescents with ADHD (combined type) have increased FA in the left posterior cingulum bundle and that the FA value of this cluster was associated with behavioural problems as well as executive dysfunctions. Our results suggested that, beyond the frontal–striatal–cerebellar circuit, children and adolescents with ADHD also have pathophysiological abnormalities in the limbic network. Moreover, such microstructural alterations might be a potential substrate for abnormal behaviours and executive deficits in patients with ADHD.
Acknowledgments
This study was supported by the National Natural Science Foundation (Grant Nos. 81030027, 81227002,81171488 and 81220108013), National Key Technologies R&D Program (Program No. 2012BAI01B03) and Program for Changjiang Scholars and Innovative Research Team in University (Grant No. IRT1272) of China. Dr Gong would also like to acknowledge his Visiting Adjunct Professor appointment in the Department of Psychiatry at the Yale School of Medicine, Yale University, USA.
Footnotes
↵* These authors contributed equally to this work.
Competing interests: None declared.
Contributors: L. Chen, X. Huang, L. Guo and Q. Gong designed the study. L. Chen, X. Huang, D. Lei, N. He, X. Hu, Y. CHen, Y. Li and J. Zhou acquired the data, which L. Chen, X. Huang, D. Lei, N. He, L. Guo, G. Kemp and Q. Gong analyzed. L. Chen and X. Huang, wrote the article, which all authors reviewed and approved for publication.
- Received July 18, 2014.
- Revision received November 15, 2014.
- Accepted December 2, 2014.