Abstract
Background: Psychological models highlight the bidirectional role of self-referential processing, introspection, worry and rumination in the development and maintenance of insomnia; however, little is known about the underlying neural substrates. Default mode network (DMN) functional connectivity has been previously linked to these cognitive processes.
Methods: We used fMRI to investigate waking DMN functional connectivity in a well-characterized sample of patients with primary insomnia (PI) and good sleeper controls.
Results: We included 20 patients with PI (8 men and 12 women, mean age 42.7 ± 13.4 yr) and 20 controls (8 men and 12 women, mean age 44.1 ± 10.6 yr) in our study. While no between-group differences in waking DMN connectivity were observed, exploratory analyses across all participants suggested that greater waking connectivity between the retrosplenial cortex/hippocampus and various nodes of the DMN was associated with lower sleep efficiency, lower amounts of rapid eye movement sleep and greater sleep-onset latency.
Limitations: Owing to the cross-sectional nature of the study, conclusions about causality cannot be drawn.
Conclusion: As sleep disturbances represent a transdiagnostic symptom that is characteristic of nearly all psychiatric disorders, our results may hold particular relevance to previous findings of increased DMN connectivity levels in patients with psychiatric disorders.
Introduction
Insomnia is one of the most prevalent mental disorders.1 It is associated with a considerably reduced quality of life2 and cognitive impairments.3 Furthermore, insomnia confers an increased risk for psychiatric disorders, especially depression,4 as well as increased cardiovascular morbidity and mortality.5 Thus, insomnia is linked to increased levels of health care consumption, work disability and absenteeism, leading to pronounced direct and indirect economic costs.6
The etiology of insomnia in the absence of any identifiable organic or psychiatric disorder is assumed to be multifactorial, involving cognitive factors (e.g., worry and rumination, selective attention for sleep-related cues, dysfunctional attitudes and beliefs about sleep7,8), maladaptive behaviours (e.g., extending time in bed or daytime napping9) and increased physiologic arousal.10 From a neurobiological point of view, several subcortical and cortical regions, including the ascending reticular activating system, amygdala, hippocampus, caudate nucleus, anterior cingulate cortex (ACC), insula and frontal cortex,11–20 are thought to be involved in the pathophysiology of night- and daytime symptoms in patients with insomnia.
There is some overlap between these regions and the default mode network (DMN). The DMN is a distributed network of brain areas that are more active during rest than during task performance and includes the posterior cingulate cortex (PCC), precuneus (PRC), retrosplenial cortex (RSC), inferior parietal cortex (IPC), ventromedial prefrontal cortex (vmPFC), ACC and hippocampus.21,22 Previous research suggests that the DMN is involved in self-referential processing, introspection, worry and rumination.23,24 As self-focused worry and rumination are common in patients with depression, there is an extensive literature on DMN activity and connectivity in relation to this disorder. Both increased25 and decreased26 functional connectivity within the DMN as well as to other brain areas have been reported in patients with depression, with the majority of studies linking increased connectivity indices to higher levels of rumination.23,27,28
In contrast, little is known about DMN connectivity in patients with insomnia, a disorder that is similarly characterized by high levels of self-referential processing, introspection, worry and rumination and is intimately linked to depression.29–32 A literature review identified 3 resting-state fMRI studies in patients with insomnia that investigated functional connectivity in salience, emotional and memory networks.16,33,34 However, none of these studies focused on between-group differences in the DMN or investigated the association between DMN connectivity and objective sleep parameters.
The present study, therefore, sought to investigate waking DMN functional connectivity in a well-characterized sample of patients with insomnia and good sleeper controls. We hypothesized that the self-referential and ruminative nature of insomnia would be reflected in increased levels of functional connectivity within this network. Given recent findings suggesting that objectively impaired sleep (within insomnia samples) is associated with poorer health and cognitive outcomes35 and that self-reported sleep duration is related to alterations in functional connectivity,36 we also investigated exploratory associations between waking DMN connectivity and sleep continuity/architecture. This study is a primary analysis of data collected during a prospective, observational study at the Department of Psychiatry and Psychotherapy, University of Freiburg Medical Centre, Germany.
Methods
Participants
We included patients meeting diagnostic criteria for primary insomnia (PI) according to DSM-IV-TR and good sleeper controls in the present study. The groups were matched for age and sex. Patients with PI were referred to our sleep disorders clinic by their primary care providers or medical specialists. Healthy controls were recruited through local advertisements. Six of the patients with PI, all of whom were insomnia non-remitters and received cognitive behavioural therapy, and 19 of the controls have participated in previous neuroimaging studies conducted at our sleep laboratory.18,20,37 For screening purposes, all participants underwent a semistandardized psychiatric and sleep-related interview conducted by an experienced psychiatrist to rule out any lifetime history of psychiatric disorders, shift work or sleep disorders. In addition, all participants underwent a standard physical examination, including electrocardiography, electroencephalography (EEG) and routine blood work (blood cell count, liver, renal and thyroid function); we excluded those with serious medical conditions. All participants were required to be free of any psychoactive medication for at least 2 weeks before and during the study and were required to refrain from alcohol and caffeine consumption and from daytime naps during the recording days. We used polysomnographic recordings and sleep-related interviews to exclude healthy controls if they met the criteria for any sleep disorder. The study was conducted in accordance with the Declaration of Helsinki. The Institutional Review Board of the University Medical Center Freiburg approved our study protocol, and all participants gave informed written consent before inclusion in the study.
Procedure
After the screening procedure and 2 nights of polysomnography (PSG), participants completed a combined EEG/fMRI experiment conducted between 8:00 and 10:00 pm at the Department of Radiology of the University Medical Center Freiburg. Within the available scanner times, the timing of the experiment was a compromise between proximity to bedtime and a reasonable likelihood that most patients could manage to stay awake during the scan. Prior to the fMRI session, participants completed the Stanford Sleepiness Scales (SSS38), and they were instructed to keep their eyes closed and to relax without falling asleep during fMRI acquisition. After the fMRI experiment, all participants were asked to complete the Insomnia Severity Index (ISI),39 the Pittsburgh Sleep Quality Index (PSQI),40 the brief version of the Dysfunctional Beliefs and Attitudes about Sleep Scale (DBAS-16),41 the Glasgow Sleep Effort Scale (GSES),42 the Pre-Sleep Arousal Scale (PSAS),43 the Epworth Sleepiness Scale (ESS),44 the Morningness-Eveningness Questionnaire (MEQ),45 the Beck Depression Inventory (BDI)46 and the State-Trait Anxiety Inventory (STAI).47 The sleep-related questionnaires used in our study are described in detail in Appendix 1, available at jpn.ca. We compared the psychometric data between the groups using Student t tests for independent samples without adjusting for multiple testing.
Polysomnography
All participants underwent 2 consecutive nights of standard PSG sleep monitoring, with the first night serving diagnostic and adaptation purposes. Sleep was recorded for 8 hours from “lights out” (any time between 10:00 and 11:00 pm) until “lights on” (any time between 6:00 and 7:00 am) and was scored visually by experienced raters according to standard criteria.48 Polysomnographic parameters were compared between groups using Student t tests for independent samples without adjusting for multiple testing. We used polysomnographically determined sleep efficiency (ratio of total sleep time:time in bed); sleep-onset latency (time from lights out until sleep onset, defined as the first epoch of stage N2); slow wave sleep (stage N3) percentage; and rapid eye movement (REM) sleep (stage R) percentage of the second sleep laboratory night to investigate the association between objective sleep continuity/sleep architecture alterations and DMN connectivity, as these sleep parameters have been shown to be altered in patients with insomnia.49
EEG recording during fMRI acquisition and EEG analysis
To verify wakefulness during the fMRI acquisition, EEG data were simultaneously recorded using a 64-electrode cap with sintered Ag-AgCl ring electrodes and 2 32-channel MR-compatible EEG amplifiers (BrainProducts GmbH). Recording impedances were kept below 10 kΩ. All signals were recorded against a reference just in front of the Cz electrode. Signal ground was connected to an electrode just behind the Cz electrode. We sampled the EEG signals at 5000 Hz and filtered them between 0.1 and 200 Hz. Imaging artifacts were removed using the method described by Allen and colleagues.50 We performed an independent component analysis (ICA) to remove eye movement and ballistocardiogram artifacts. Owing to technical problems with the MR-compatible EEG amplifiers, EEG data were available for only 13 patients with PI and 15 healthy controls. In these participants, presence of wakefulness during scanning was confirmed by visual scoring according to standard criteria.48
MRI acquisition and preprocessing
We acquired functional echo-planar images (40 axial slices in interleaved order, slice thickness 3 mm with no gap; voxel size 3 × 3 × 3 mm3, echo time [TE] 30 ms, repetition time [TR] 2.49 s, motion and distortion correction by scanner software, 480 scans, total scanning time 19 min 55 s) and anatomic images (magnetization-prepared rapid gradient-echo, TR 2.2 s, TE 2.6 ms, voxel size 1 mm3) on a 3 T scanner (Magnetom TIM-Trio, Siemens), and the images were analyzed using the National Institutes of Health AFNI software (http://afni.nimh.nih.gov/). A neurologist under the supervision of a board-certified neuroradiologist inspected all anatomic scans for the absence of pathological findings.
The anatomic images were segmented using FreeSurfer version 5.3.0 (http://surfer.nmr.mgh.harvard.edu/), which has demonstrated good accuracy in comparison with manual segmentation.51 Preprocessing of the functional images included slice timing; coregistration with the anatomic image, normalization into Montreal Neurological Institute (MNI) space; smoothing (8 mm full-width at half-maximum); and regressing out the mean white matter signal (left and right cerebral white matter), the mean ventricle signal (third and lateral ventricles) and 6 head motion parameters (3 rigid-body translations and rotations that were estimated during the online motion correction). Within this linear regression, additional movement correction was performed by censoring scans with a framewise displacement sum of greater than 0.5 mm,52 and sine and cosine regressors were used to exclude frequencies greater than 1/10 Hz and below 1/128 Hz. Patients with PI (2.6%) and healthy controls (4.1%) did not differ in the number of censored scans (t38 = 0.70, p = 0.49).
MRI connectivity analyses
For identifying the DMN in all participants, we performed a seed-based analysis using the averaged time series of the PCC (“ctx_*h_G_cingul-Post-dorsal” of the Destrieux parcellation;53 MNI coordinates x, y, z = −0.5, −38.7, 33.5). For each participant, correlation coefficients with the PCC seed region’s time series were calculated voxelwise and converted into Fisher z values.
To study the association between sleep parameters and the degree of DMN connectivity, we used 7 predefined regions of interest (ROIs) obtained using the individual participants’ FreeSurfer segmentations (left and right side combined): 1) ACC (“ctx_*h_G_and_S_cingul-Ant” of the Destrieux parcellation, centre of mass x, y, z = 0.9, 42.0, 10.4); 2) the hippocampus (“Left-Hippocampus” and “Right-Hippocampus” of the FreeSurfer subcortical segmentation, centre of mass of the left part of the ROI x, y, z = −27.0, −21.0, −12.4, centre of mass of the right part of the ROI x, y, z = 26.9, −20.3, −13.4); 3) IPC (“ctx_*h_G_pariet_inf-Angular” of the Destrieux parcellation, centre of mass of the left part of the ROI x, y, z = −46.3, −65.8, 41.1, centre of mass of the right part for the ROI x, y, z = 51.0, −58.8, 38.5); 4) PCC (see previous paragraph); 5) RSC (“ctx_*h_G_cingul-Post-ventral” of the Destrieux parcellation, centre of mass x, y, z = 0.2, −47.4, 9.3); 6) the precuneus (“ctx_*h_G_precuneus” of the Destrieux parcellation, centre of mass x, y, z = −0.5, −59.0, 44.3); and 7) vmPFC (“ctx-*h-medialorbitofrontal” of the Desikan–Killiany parcellation,54 centre of mass x, y, z = −0.7, 39.3, −12.5). In each participant, pairwise correlation coefficients were determined between the fMRI time series of these 7 ROIs and converted into Fisher z values.
We performed a linear regression analysis with the Fisher z values as dependent variables; the centred values of age, sex, BDI and STAI scores as covariates; and no further independent variables to test DMN functional connectivity across all participants (test of the intercept term). The hypothesis-based group analyses were performed using an analysis of variance model with the group (patients with PI v. healthy controls) as the independent variable; the Fisher z values as dependent variables; and the centred values of age, sex, BDI and STAI scores as covariates. Linear regression analyses were used in an exploratory manner to investigate the association between DMN functional connectivity and the following sleep parameters: subjective sleep efficiency and sleep-onset latency derived from the PSQI, and polysomnographically determined sleep efficiency, sleep-onset latency, stage N3 percentage and stage R percentage from the second sleep laboratory night. We considered results to be significant at p < 0.05, 2-tailed, for these analyses (uncorrected for multiple testing).
To illustrate the association between polysomnographically determined sleep efficiency and hippocampus connectivity, we performed a further seed-based analysis using the hippocampus as the seed region. Voxelwise regression analysis was conducted with polysomnographically determined sleep efficiency as the independent variable; age, sex, BDI and STAI scores as covariates; and the Fisher z scores of this seed-based analysis as the dependent variables. An overall statistical significance level of p < 0.05 was determined with Monte Carlo simulations on a whole brain mask resulting in a minimum cluster size of 1283 mm3 for a voxel threshold of p < 0.005.
To determine the overlap between the 7 predefined ROIs and a data-driven identification of the DMN in the present sample, we carried out an ICA using the Group ICA of fMRI toolbox (GIFT; http://mialab.mrn.org/software/gift/).55 The number of estimated independent components using the minimum description length criteria was 40. Group components were correlated with a reference DMN mask (ref_default_mode.nii) to identify the component with the highest spatial correlation. Back-reconstruction was used to produce participant-specific spatial maps and time courses for this component. Individual maps corresponding to the group DMN component were entered into a second-level analysis. According to a Monte Carlo simulation, overall statistical significance of p < 0.05 was obtained for this analysis by considering cluster sizes of at least 351 mm3 at a voxel threshold of p < 0.001. We used MRIcron (http://www.mricro.com/mricron/install.html) for 3-dimensional visualization of the results on the ch2better.nii.gz atlas together with the 7 predefined ROIs, as determined by FreeSurfer-based segmentation of the ch2better.nii.gz atlas.
Results
Sample characteristics
Twenty patients meeting DSM-IV-TR diagnostic criteria for PI and 20 good sleeper controls were included in the present study. Demographic and psychometric data of the study sample are presented in Table 1. The groups did not differ significantly in sex distribution, age, body mass index (BMI), ESS, SSS or MEQ scores. The patients with PI had significantly higher scores than controls on the ISI, PSQI, DBAS-16, GSES, PSAS and the trait subscale of the STAI. They also had significantly higher BDI scores, though values were in the healthy range and the group difference was reduced to a statistical trend when excluding the 2 sleep-related questions from the BDI (patients with PI: 4.6 ± 3.6; controls: 2.5 ± 3.2, t38 = −2.00, p = 0.05). Two patients with PI had sleep-onset insomnia, 9 had sleep-maintenance insomnia, and 9 had mixed insomnia. The average duration of PI was 9.4 ± 10.0 years.
Demographic and clinical characteristics of the study population
Polysomnography
Polysomnographic data are presented in Table 2, and the data from the adaptation night are available in Appendix 1, Table S1. Patients with PI had a significantly lower total sleep time and sleep efficiency than good sleeper controls. Additionally, patients with PI had a reduced amount of stage N3 (both in percentage and in minutes).
Polysomnographic data of the study participants
DMN functional connectivity
For all participants, the results of the seed region-based analysis of PCC connectivity are presented in Appendix 1, Figure S1. Of note, these results illustrate a high degree of interindividual stability of waking DMN functional connectivity (see Appendix 1, Fig. S2, for a spatial probability map). The overlap between the 7 predefined ROIs and the ICA-based identification of the DMN in the present sample is presented in Appendix 1, Figure S3.
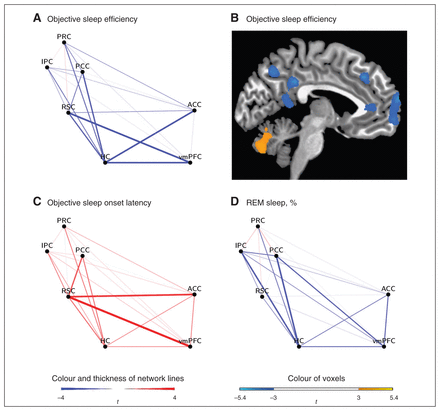
The association between the degree of functional connectivity between the 7 predefined ROIs and insomnia diagnosis, subjective sleep variables and polysomnographically determined sleep variables are presented in Table 3 and Figure 1.
Waking default mode network functional connectivity for the regression analyses with (A, B) polysomnographically determined sleep efficiency, (C) polysomnographically determined sleep-onset latency and (D) stage R percentage. To illustrate the association between polysomnographically determined sleep efficiency and hippocampus (HC) connectivity, (B) the results of a voxelwise regression analysis are presented in which polysomnographically determined sleep efficiency served as independent variable and the Fisher z scores of the seed-based analysis with the hippocampus as seed region served as dependent variables (x = −5, minimum cluster size of 1283 mm3, voxel threshold of p < 0.005). ACC = anterior cingulate cortex; IPC = inferior parietal cortex; PCC = posterior cingulate cortex; PRC = precuneus; REM = rapid eye movement; RSC = retrospinal cortex; vmPFC = ventromedial prefrontal cortex.
Results of the statistical tests evaluating functional connectivity between ROIs as well as the association between sleep parameters and waking DMN functional connectivity*
No significant between-group differences were found for waking DMN connectivity. However, there was a significant negative association between waking DMN connectivity and polysomnographically determined sleep efficiency. Specifically, this association was found for hippocampus–ACC (r2 = 0.24), hippocampus–PCC (r2 = 0.28), hippocampus–vmPFC (r2 = 0.26) and RSC–vmPFC (r2 = 0.24) connectivity. There was also a significant positive association between waking DMN connectivity and polysomnographically determined sleep-onset latency (RSC–ACC: r2 = 0.26; RSC–PCC: r2 = 0.17; RSC–vmPFC, r2 = 0.31). Furthermore, waking DMN connectivity (hippocampus–IPC: r2 = 0.22; hippocampus–PCC: r2 = 0.31) was significantly negatively related to stage R percentage. In contrast to these findings, subjective sleep efficiency, subjective sleep-onset latency and polysomnographically determined stage N3 percentage were not associated with waking DMN connectivity.
Discussion
In the present study, we investigated waking DMN functional connectivity in individuals with PI, a disorder characterized by high levels of self-referential processing, introspection, worry and rumination. Two main findings emerged. First, we did not find any differences in waking DMN connectivity between patients with PI and good sleeper controls. Second, in exploratory analyses across both groups, polysomnographically determined sleep continuity and sleep architecture disturbances, namely lower sleep efficiency, lower stage R percentage and higher sleep-onset latency, were significantly associated with greater waking connectivity between the RSC/hippocampus and various nodes of the DMN.
Although we found no significant difference in waking DMN connectivity between the groups, the secondary findings of our study suggest an association between polysomnographically determined sleep continuity and sleep architecture disturbances and greater waking connectivity. Previous within-subjects studies in healthy, good sleepers have reported reduced DMN connectivity during non-REM (NREM) sleep in contrast to wakefulness56–58 after light sedation with midazolam59 or after a single night of sleep deprivation.60 In contrast to these findings, the current study used a between-subjects design and suggests that objectively determined sleep continuity and architecture disturbances are associated with greater waking connectivity between the RSC/hippocampus and several nodes of the DMN.
As sleep disturbances are evident in most psychiatric disorders and, thus, transdiagnostic in nature,61 our results may, at least partially, explain previous findings of increased DMN connectivity levels in patients with psychiatric disorders.25,62 Therefore, it appears to be important to measure and control for sleep when investigating waking DMN connectivity in the psychiatric field. This would also be in line with the purpose of the Research Domain Criteria (RDoC) project, which suggested focusing on dimensions of observable behaviour and neurobiological measures for classifying psychopathology instead of focusing on psychiatric diagnoses.
The significant correlations between sleep parameters and waking functional connectivity were found only in specific combinations of brain areas with both greater local and long-range connectivity being associated with sleep continuity and sleep architecture disturbances. In particular, all significant findings involved either RSC or hippocampal connectivity to several other areas of the DMN, namely the vmPFC, ACC, PCC and IPC. This finding is in line with those of previous studies showing that both RSC and hippocampal connectivity to other DMN areas decrease specifically with increasing sleep depth,57,63 supporting the postulated roles of RSC connectivity in the regulation of consciousness64 and hippocampal connectivity in memory retrieval during wakefulness.65 The present study suggests that RSC and hippocampal connectivity may covary with disturbances of sleep continuity and sleep architecture across participants, potentially identifying an “at-risk signature” that deserves attention in future studies. Of note, potential associations between sleep-related variables and RSC and hippocampal connectivity to other brain areas cannot be ruled out since the analysis was restricted to the DMN. Of particular importance, the amount of rapid eye movement sleep was specifically associated with hippocampal connectivity, which further supports the proposed role of this sleep stage for sleep disturbances.66
Several reasons might account for the lack of significant difference between patients with insomnia and controls. It is conceivable that patients with insomnia do not have any functional deficits in the DMN. While this would be surprising in light of the literature on DMN connectivity and self-referential processing, introspection, worry and rumination in individuals with depression, several studies have also reported that the ruminative nature of insomnia is markedly different from that of depressed individuals.67,68 Consequently, the neurobiological mechanisms of the above-mentioned cognitive processes may differ between individuals with insomnia and those with depression.
However, it should also be noted that the present sample size of 20 participants per group provides sufficient power only for the detection of large effect sizes in a between-subjects design. Small or medium-sized effects, which have been often reported in the literature on physiologic alterations in patients with PI,10 cannot be ruled out with the present investigation. It should also be noted that the psychometric and polysomnographic data suggest that the PI sample consisted of individuals with mild to moderate psychophysiological insomnia with comparably high levels of daytime sleepiness. Therefore, both greater insomnia severity and lower levels of daytime sleepiness might have led to different results.
Of particular relevance for power considerations, PI is a heterogeneous disorder that may involve reduced quality, duration or efficiency of sleep with multiple possible psychological and neurobiological etiological pathways.69 Furthermore, levels of self-referential processing, introspection, worry and rumination may also substantially differ among patients with insomnia.29–32 Thus, the investigation of dimensional behavioural or neurobiological variables as predictors of waking DMN connectivity may result in a greater proportion of explained variance than the one observed for the insomnia diagnosis. In addition, future studies should investigate waking DMN connectivity in larger samples of well-defined subgroups of patients with insomnia (e.g., those with objective short sleep duration, treatment-naive patients or those with high levels of self-referential processing, introspection, worry and/or rumination).
Limitations
Several limitations of the present investigation have to be acknowledged. First, owing to technical problems, EEG data were not available for 7 patients with PI and 5 controls. Thus, it cannot be ruled out that some of these participants fell asleep during the fMRI investigation (see also the recent article by Tagliazucchi and Laufs70), which may have affected our results. However, none of our participants subjectively fell asleep, and all participants were explicitly instructed to stay awake during the scanning session. While the effects of this instruction may be questioned, the SSS data suggest that none of our participants was excessively sleepy during the investigation. Second, there was a low spatial overlap between the individual hippocampus segmentations and the group DMN map, as obtained using ICA. Therefore, strictly speaking, the hippocampal connectivity results may be regarded as concerning functional connectivity between the DMN and the hippocampus instead of functional connectivity within the DMN. Third, we did not systematically control for light intensity in our routine sleep laboratory procedure, and there was a certain deviation between habitual and sleep laboratory bedtimes in some participants. This may have increased the error variance of polysomnographically determined sleep parameters. In addition, sleep diary data would have been helpful for determining subjective sleep parameters, and we did not directly investigate self-referential processing, introspection, worry and rumination in our sample to correlate these measures with DMN connectivity. Furthermore, owing to the cross-sectional nature of the study, conclusions about causality cannot be drawn, and the direction of the association between sleep continuity and sleep architecture disturbances and greater waking connectivity remains unclear. Finally, it should be noted that the exploratory analyses were not corrected for multiple testing. Thus, the results of these analyses have to be replicated in future confirmatory studies.
Conclusion
We did not find any between-group differences in waking DMN connectivity or any significant association between subjective sleep parameters and waking DMN connectivity. However, polysomnographically determined sleep continuity and sleep architecture disturbances were significantly associated with greater waking connectivity between the RSC/hippocampus and several nodes of the DMN across the entire sample. Thus, our results give relevance to objective sleep alterations when studying the neurobiology of sleep and psychiatric disorders. Further studies may investigate the impact of a successful treatment of objective sleep disturbances in a within-subject design to gain more knowledge about possible causal links between sleep and DMN connectivity with increased statistical power.
Footnotes
Funding: This work was supported by the Else Kröner-Fresenius-Stiftung grant (grant number 2011_A208) awarded to D. Riemann and K. Spiegelhalder.
Competing interests: C. Nissen has received speaker honoraria from Servier. D. Riemann has received personal fees from Abbvie. No other competing interests declared.
Contributors: W. Regen, C. Nissen, D. Riemann and K. Spiegelhalder designed the study. W. Regen, C. Baglioni and K. Spiegelhalder acquired the data, which all authors analyzed. W. Regen, S. Kyle, J. Hennig and K. Spiegelhalder wrote the article, which all authors reviewed and approved for publication.
- Received October 6, 2014.
- Revision received June 1, 2015.
- Accepted September 1, 2015.