Abstract
Background: Increased automatic processing of threat-related stimuli has been proposed as a key element in panic disorder. Little is known about the neural basis of automatic processing, in particular to task-irrelevant, panic-related, ecologically valid stimuli, or about the association between brain activation and symptomatology in patients with panic disorder.
Methods: The present event-related functional MRI (fMRI) study compared brain responses to task-irrelevant, panic-related and neutral visual stimuli in medication-free patients with panic disorder and healthy controls. Panic-related and neutral scenes were presented while participants performed a spatially non-overlapping bar orientation task. Correlation analyses investigated the association between brain responses and panic-related aspects of symptomatology, measured using the Anxiety Sensitivity Index (ASI).
Results: We included 26 patients with panic disorder and 26 heatlhy controls in our analysis. Compared with controls, patients with panic disorder showed elevated activation in the amygdala, brainstem, thalamus, insula, anterior cingulate cortex and midcingulate cortex in response to panic-related versus neutral task-irrelevant stimuli. Furthermore, fear of cardiovascular symptoms (a subcomponent of the ASI) was associated with insula activation, whereas fear of respiratory symptoms was associated with brainstem hyperactivation in patients with panic disorder.
Limitations: The additional implementation of measures of autonomic activation, such as pupil diameter, heart rate, or electrodermal activity, would have been informative during the fMRI scan as well as during the rating procedure.
Conclusion: Results reveal a neural network involved in the processing of panic-related distractor stimuli in patients with panic disorder and suggest an automatic weighting of panic-related information depending on the magnitude of cardiovascular and respiratory symptoms. Insula and brainstem activations show function-related associations with specific components of panic symptomatology.
Introduction
Recurring sudden panic attacks and anxious apprehension are the 2 core symptoms for diagnosing panic disorder.1 Panic-related anxious apprehension is characterized by worry about future attacks and by fear of bodily and cognitive sensations of anxiety.2 In patients with panic disorder and other anxiety disorders, threat-related stimuli, which are by definition salient, activate neural mechanisms that facilitate fast and preferred processing.3–6 This automatic attention to threat-related stimuli is associated with detection and evaluation mechanisms, reorientation of resources and amplification of processing mechanisms.7
Reviews of functional imaging studies propose a network including the amygdala, brainstem, thalamus, insula, anterior cingulate cortex (ACC), midcingulate cortex (MCC) and medial prefrontal cortex (mPFC) as neural underpinnings of altered threat processing in patients with panic disorder.8–10 However, the neural basis of the processing of panic-related stimuli when these are irrelevant for the task at hand has not commonly been investigated in patients with panic disorder. Using an emotional Stroop task, greater activation in patients with panic disorder than in healthy controls was found in the prefrontal cortex,11 ACC, thalamus, amygdala, hippocampus and inferior parietal cortex.5 With spatially overlapping emotional word–face pairs that required a response to faces only, Chechko and colleagues12 reported decreased ACC and frontal gyrus activation, but increased activation in the amygdala and brainstem in patients with remitted panic disorder for emotionally incongruent compared to congruent pairs. However, studies investigating neural responses to task-irrelevant, ecologically valid panic-related stimuli under experimental conditions in which attention to distracting emotional information is not required at all hardly exist.
Moreover, and most importantly, it remains to be specified how brain responses under these conditions are associated with symptoms of panic disorder. Patients with this disorder have varied anxiety sensitivity, defined as the fear of anxiety-related bodily sensations derived from beliefs that these symptoms have harmful physical, psychological, or social consequences.13,14 The Anxiety Sensitivity Index (ASI) has been shown to effectively measure fear for physical, psychological, or social aspects associated with the experience of anxiety.15,16 Anxiety severity is associated with the development of spontaneous panic attacks in healthy young adults, and fear of physical concerns (a subcomponent of anxiety severity) plays a particularly important role in predicting the course of panic disorder.17–20 A better understanding of the role of anxiety severity in panic disorder could thus help to design prevention programs, specify therapeutic interventions and might further enrich diagnostic processes. Poletti and colleagues21 reported positive correlations of anxiety severity with neural activation in the PFC, ACC and insula in patients with panic disorder. Thus, preliminary behavioural and neural data underscore the need for further research into the role of anxiety severity sub-constructs in patients with the disorder. It is unknown whether anxiety severity modulates neural effects of automatic, panic-related stimulus processing. Research on anxiety severity has led to revisions in factor structure, currently suggesting 4 subscales (fear of cardiovascular symptoms, fear of respiratory symptoms, fear of loss of control, fear of publicly observable symptoms) in the German ASI-4.15 The ASI-4 factor structure that describes fear of physical symptoms in more detail allows us to disentangle the predictive value of fear of cardiovascular and respiratory symptoms.15
The present event-related functional MRI (fMRI) study aimed to elucidate the neural underpinnings of processing panic-related stimuli in patients with panic disorder when these stimuli are spatially separate from the targets and completely task-irrelevant.7 We used a recently developed standardized set of panic-related and neutral scenes as distractors while participants had to solve a concurrent but spatially nonoverlapping bar discrimination task. We further addressed how neural effects are shaped by panic-related aspects of anxiety sensitivity measured with the ASI-4, using correlation analysis.
Methods
Participants
We recruited patients with panic disorder and healthy controls through public advertisement and in collaboration with an outpatient clinic. Psychiatric medication, neurologic disorders, and fMRI contraindications served as exclusion criteria for both groups. Psychotic or bipolar disorders, suicidal ideations, and drug dependence or abuse within the last 10 years were additional exclusion criteria for the panic disorder group. Controls also had to be free of any psychiatric diagnosis within the last 10 years. Patients had to be free of psychiatric medication for at least 3 months and had to refrain from “as needed” medication for at least 1 week before the fMRI session. An experienced clinical psychologist interviewed all patients and controls using the Structured Clinical Interview for DSM-IV Axis I Disorders (SCID). Participants completed questionnaires on demographic and clinical data. The German ASI-4,15 a 24-item questionnaire, was administered to assess fear of anxiety-related symptoms on fear of cardiovascular symptoms, respiratory symptoms, loss of control and publicly observable symptoms using a 5-point Likert scale (0 = don’t agree at all; 4 = agree completely). All participants gave written informed consent. The study was approved by the ethics committee of the University of Muenster and conforms with the Declaration of Helsinki.
Stimuli
We used the Panic-related Picture Set Münster (PAPS-M), comprising 50 panic-related and 50 neutral scenes. The set was developed in a 3-step pilot study, including an extensive Web search, a clinical expert rating and a patient rating. Panic-related scenes display symptoms related to panic attacks (e.g., shortness of breath, hyperventilation, heart palpitations, chest pain, trembling or shaking, dizziness, fainting), fears (e.g., having a heart attack) and agoraphobia-related situations (e.g., crowded bus, dark tunnel, glass elevator).22
Procedure
During the 6 minute 56 second experimental stimulation, each of the 50 panic-related and 50 neutral distractor scenes was presented once, centred on the screen, in an event-related design. Two circles (target) with a small line inside were presented above and below the concurrently displayed scene (distractor), and participants performed a bar orientation task (Fig. 1; modified from Straube and colleagues23 and Wiens and colleagues24). Lines were either vertical or horizontal and had either the same or different orientation in both circles (target stimuli). Participants indicated by button press with their right index and middle fingers whether orientation was the same or different in both circles. Orientation was the same on half of the trials and different for the other half. Trials were classified as errors when participants gave an incorrect response, missed a trial or pressed both buttons. This task can be categorized as a concurrent but distinct target–distractor task (CDTD) or directed-attention task, which is well suited to investigate distractor-driven attentional processes.7,25 Each scene (distractor) and the 2 circles (target) were presented for 800 ms, separated by a white fixation cross (1280–12200 ms, mean 3360 ms). Neutral and panic-related scenes were presented in random sequence and optimized and counterbalanced with the Optseq algorithm (https://surfer.nmr.mgh.harvard.edu/optseq/), which implements temporal jitter to increase signal discriminability.26 Stimuli were rear-projected onto a screen that the participants viewed through a mirror on the MRI head coil.
Schematic overview of a trial in the concurrent but distinct target–distractor task.
Participants rated all 100 scenes within 7 days after the fMRI experiment in a postscanning rating session. After presentation of a 2 s stimulus, participants rated the scenes with regard to valence, arousal and anxiety. A 9-point Likert scale was used to assess valence (1 = very unpleasant; 5 = neutral; 9 = very pleasant), arousal (1 = not arousing; 9 = very arousing) and anxiety (1 = not anxiety-inducing; 9 = very anxiety-inducing).
Behavioural data analysis
Trials with premature button presses (< 300 ms), delayed responses (+2 standard deviations from the individual log-transformed mean) and erroneous button presses were discarded (315 trials, 6% of all data). To compensate for a skewed reaction time distribution, all reaction time analyses were performed on log-transformed data. Reaction times and rating data were analyzed by means of 2 × 2 repeated-measures analyses of variance (ANOVA) with emotion (panic-related, neutral) as a within-subjects factor and group (panic disorder, healthy control) as a between-subjects factor. We considered results to be significant at p < 0.05, and post hoc comparisons were Bonferroni-corrected for multiple testing.
FMRI acquisition and analysis
We recorded structural brain information and blood oxygen level–dependent (BOLD) responses using a Magnetom PRISMA 3 T MRI scanner with a 20-channel head matrix coil (Siemens Medical Solutions). We recorded a T1-weighted MPRAGE structural volume with 192 slices for anatomic localization. We conducted a run of 225 volumes using a T2*-weighted echo-planar sequence with the following parameters: echo time (TE) 30 ms, flip angle 90°, matrix 92 × 92 voxels, field of view (FOV) 208 mm, repetition time (TR) 2080 ms. Each volume consisted of 36 axial slices (thickness 3 mm, gap 0.3 mm, in-plane resolution 2.26 × 2.26 mm2). The volumes were tilted approximately 20° clockwise from the anterior–posterior commissure line, to minimize susceptibility artifacts in inferior parts of anterior brain areas. A shimming field was applied before functional imaging to minimize external magnetic field inhomogeneities.
We conducted our fMRI data using BrainVoyager QX software version 2.4 (Brain Innovation). The first 10 volumes of each run were discarded from analysis to ensure steady state tissue magnetization. After the last trial, the fixation cross was presented for an additional 15 volumes to allow for data acquisition in randomizations with a very short last interstimulus interval.26 All volumes were realigned to the first volume to minimize artifacts due to head movements and were then resampled to a voxel size of 2 × 2 × 2 mm3. Slice-time correction and spatial (6 mm full-width at half-maximum [FWHM] isotropic Gaussian kernel) and temporal smoothing (high pass filter: 10 cycles in time course [0.023 Hz]; low pass filter: 2.8 s FWHM Gaussian kernel; linear trend removal) were applied. The anatomic and functional images were coregistered and normalized to Talairach space.27 We used the normalization procedure implemented by default in BrainVoyager.
Small volume–corrected and whole-brain analyses
For statistical analyses, we calculated multiple linear regressions modelling the signal time course at each voxel. The expected BOLD signal change for each predictor was modelled with a canonical double γ hemodynamic response function (HRF). Predictors of interest were panic-related and neutral scenes. Only trials in which participants gave a correct answer were included in the analysis. Analogous to reaction-time data analysis, trials including errors were modulated in a separate predictor. This predictor and 6 motion parameters (to account for movement artifacts) were included as regressors of no interest into the model. First, we generated voxel-wise statistical maps and computed percent-standardized predictor estimates (b weights) for each participant. We analyzed predictor estimates across participants by means of t tests in specific regions of interest (ROIs).
Based on the current panic disorder imaging literature, the brainstem, amygdala, insula, thalamus, ACC, MCC, and mPFC served as ROIs.8,9 The ROIs (amygdala, insula, thalamus, mPFC, ACC and MCC) were created based on the Automated Anatomic Labelling (AAL) atlas included in the Wake Forest University (WFU) pick atlas28,29 with a 1 mm dilation factor for the amygdala and insula. For the mPFC, we used the 2 AAL templates “Frontal_Med_Orb” and “Frontal_Sup_Medial.” The ROIs for the brainstem were downloaded from the digitized version of the Talairach atlas (www.talairach.org/nii/gzip/). We converted obtained Montreal Neurological Institute (MNI) coordinates to Talairach space using MATLAB version 8.2 (MathWorks) using the ICBM-152 routine proposed by Lancaster and colleagues.30 Peak voxel labelling was supported by Talairach Dameon software 31 and verified using the Mai atlas.32
For statistical analyses, we used a cluster-based permutation (CBP) approach.33,34 This approach requires no assumptions about the test statistic distribution and has recently been shown to be more valid than classical parametric fMRI analyses and to offer precise control of the false-discovery rate.34 All permutation tests were performed with 1000 permutations.35,36 For each permutation, the individual b maps (panic-related – neutral) were randomly assigned without replacement to 1 of the 2 experimental groups. Voxel threshold was set at pvoxel < 0.005 to balance between type I and type II error. We calculated cluster mass by adding all F values in neighbouring significant voxels. We compared the cluster mass observed in the contrast of interests with the distribution of the maximal cluster mass observed in each of the 1000 permutations. Cluster masses larger or equal to the 95th percentile of the permutation distribution were considered to be statistically significant clusters (i.e., pcluster < 0.05).
We performed correlational analyses for ASI subscale scores (fear of cardiovascular symptoms, fear of respiratory symptoms, fear of publicly observable symptoms, fear of loss of control) to investigate the moderating influence of these scores on neural responses to panic-related versus neutral stimuli in patients with panic disorder. Using the CBP approach, individual b maps (panic-related > neutral) of anatomic regions in which differential between-group effects were detected were randomly assigned to the individual questionnaire subscale scores. The cluster mass was then calculated by adding all correlation coefficients of neighbouring significant voxels. We considered clusters with pcluster < 0.0125 (corrected for number of ASI subscales) to be statistically significant.
Results
Participants
Of the 29 patients with panic disorder originally recruited, 1 aborted the scanning session owing to panic symptoms, and we discarded the data sets of 2 participants for whom 20 trials were classified as errors. This left a final sample of 26 patients with panic disorder (age range 18–46 yr) and 26 controls (age range 19–32 yr) comparable for age, sex and education. The demographic and clinical characteristics of the study groups are shown in Table 1. All participants were native German speakers, had normal or corrected-to-normal vision, and were right-handed, as assessed using the Edinburgh Handedness Inventory. Thirteen of the 26 patients had a primary diagnosis of panic disorder (DSM 300.01) and the other 13 had a primary diagnosis of panic disorder with agoraphobia (DSM 300.21). Six of the 26 patients were undergoing psychotherapy at the time of study participation.
Demographic and clinical characterization of patients with panic disorder and healthy controls
Behavioural data
Patients with panic disorder rated scenes as more unpleasant, more arousing and more anxiety-inducing than healthy controls. Both groups rated panic-related stimuli as more unpleasant, more arousing and more anxiety-inducing than neutral scenes (Fig. 2 and Appendix 1, available at jpn.ca/160226-a1). Arousal and anxiety levels were significantly higher in patients with panic disorder than in controls for panic-related versus neutral scenes.
Mean postscanning scene ratings for valence, arousal and anxiety (disorder-related – neutral) for patients with panic disorder and healthy controls. Ratings were given on 9-point Likert scales as follows: valence, 1 = negative, 5 = neutral, 9 = positive; arousal, 1 = calm, 9 = intense; anxiety, 1 = low, 9 = high.
In the final sample, on average 94 of 100 (range 87–98) trials per person were included. The number of trials excluded owing to errors or outliers did not differ between groups (average number of excluded trials: 5 in controls v. 7 in patients with panic disorder; t50 = 1.47, p = 0.15). Analysis of log-transformed reaction times showed neither significant main effects (emotion: F1,50 = 0.083; group: F1,50 = 0.669) nor a significant group × emotion interaction (F1,50 = 0.767, all p > 0.1; reaction times in the panic disorder group: disorder-related = 900.22 ms ± 96.67 ms, neutral = 894.32 ms ± 83.45 ms; reaction times in healthy controls: disorder-related = 876.02 ms ± 97.71 ms, neutral = 878.29 ms ± 94.83 ms).
fMRI data
Small volume–corrected analysis
Patients with panic disorder showed stronger emotion effects (always referring to panic-related > neutral scenes) than healthy controls in the amygdala, brainstem, insula, thalamus (lateral nucleus), ACC and MCC (Table 2 and Fig. 3). In the mPFC, activation in response to panic-related versus neutral scenes did not differ between patients with panic disorder and controls. No hyperactivations in controls versus patients for panic-related versus neutral scenes were observed.
Differential brain activation for disorder-related compared with neutral distractor scenes in patients with panic disorder versus healthy controls in a priori–defined regions of interest (panic disorder > control, panic-related > neutral, all p < 0.005, uncorrected; p < 0.05, corrected; only selected effects are shown). ACC = anterior cingulate cortex; L = left; MCC = midcingulate cortex; R = right.
Significant hyperactivations for disorder-related versus neutral scenes across all patients relative to healthy controls revealed by small volume–corrected analysis (p ≤ 0.05, corrected)
Whole-brain analysis
Whole-brain analysis yielded 2 clusters of greater activation in patients with panic disorder versus controls for panic-related versus neutral scenes. The first cluster encompassed brainstem regions, as shown in small volume–corrected analysis, and extended to cerebellar and posterior cingulate cortex (PCC) regions. The second cluster encompassed the insula effect and extended to inferior frontal and temporal regions (Table 3). Healthy controls showed no clusters of activation higher than patients with panic disorder for the contrast panic-related versus neutral stimuli.
Significant hyperactivations for disorder-related versus neutral scenes across all patients relative to healthy controls revealed by whole-brain analysis in 2 clusters (p ≤ 0.05, corrected)*
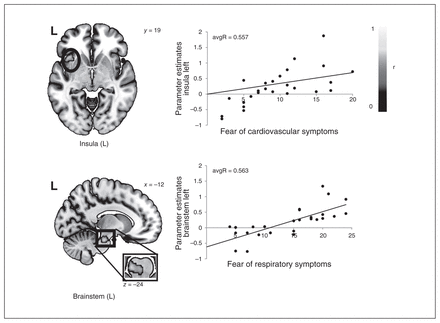
The investigation of the influence of ASI-4 subscales on differential brain activation in patients with panic disorder resulted in a significant positive correlation between fear of cardiovascular symptoms and anterior insula activation (peak voxel: x, y, z = −41, 19, −5; maxR = 0.718, avgR = 0.557, k = 246 voxels, p = 0.003) and between fear of respiratory symptoms and brainstem activation (peak voxel: x, y, z = −12, −24, −24; maxR = 0.692, avgR = 0.563, k = 136 voxels, p = 0.004; Fig. 4).
Correlation of panic disorder parameter estimates in the left insula (panic-related > neutral) and fear of cardiovascular symptoms as well as correlation of panic disorder parameter estimates in the brainstem (panic-related > neutral) and fear of respiratory symptoms. L = left.
Discussion
The present study investigated in patients with panic disorder the neural correlates of automatic processing of panic-related visual scenes that were task-irrelevant and spatially distinct from task targets. Comparing panic-related versus neutral scenes, patients with panic disorder showed greater activation than controls in the amygdala, brainstem, thalamus, insula, ACC and MCC, whereas behavioural responses did not differ between the groups. Fear of cardiovascular symptoms was the strongest predictor of emotion effects (always referring to panic-related > neutral) in the insula, whereas fear of respiratory symptoms predicted emotion effects in the brainstem in patients with panic disorder.
Next to the general hyperactivation in several brain regions in patients with panic disorder during processing of task-irrelevant, panic-related stimuli, activation in 2 key regions — the insula and brainstem — was modulated by specific aspects of panic symptomatology. The insula, associated with interoceptive processing and the processing of one’s own bodily symptoms, plays an important role in panic disorder.9,37,38 Hyperactivation in the anterior insula in our patients is best explained by fear of cardiovascular symptoms. Earlier studies found anxiety severity to correlate positively with insula hyperactivation in response to masked fearful faces in healthy controls and with insula activation in patients with panic disorder during emotional processing of facial affect expressions.21,39 Killgore and colleagues39 emphasized the role of specific subscales of anxiety severity and reported fear of physical symptoms to significantly correlate with anterior insular activation. Our findings of insular activation, as driven by fear of cardiovascular symptoms, match this result and extend it to a clinical sample that is characterized by its enhanced sensibility to bodily symptoms relative to other anxiety disorders.40 The insula hyperactivity of patients with panic disorder when processing disorder-related scenes may thus be specifically associated with their sensibility to and monitoring of bodily symptoms of anxiety.41–43 Findings suggest that those who fear physical symptoms more also pay them more attention, and vice versa. Notably, we observed greater panic-related insula activation despite panic-related scenes requiring no explicit processing, which indicates a low threshold for interoceptive processing in patients with panic disorder — a marker that might present an etiological or maintaining factor in panic disorder.
Furthermore, our results showed that task-irrelevant panic-related scenes trigger greater brainstem activation in patients with panic disorder than in healthy controls (panic-related > neutral), and fear of respiratory symptoms predicted differential brainstem activation in patients with panic disorder. Brainstem alterations in patients with panic disorder have been reported in anatomic44–46 and functional imaging studies.12,47–49 Aberrant activation in the brainstem, a site of homeostatic integration, may be closely linked to the changes in chemoreception and cardiorespiratory control perceived by patients with panic disorder.10,50–52 Using a carbon dioxide challenge, Goossens and colleagues53 reported increased brainstem activation in response to hypercapnia in patients with panic disorder compared with healthy controls. Although limited resolution does not allow for specific localization of subregions, coordinates suggest an involvement of pontine nuclei and locus coeruleus, a carbon dioxide/H+-sensitive brain site involved in communication of respiratory- and stress-induced activation changes.54,55 The present study adds to what is known about the association between the brainstem and respiratory symptoms by linking increased brainstem activation to the subjective fear of respiratory symptoms. In his “false suffocation alarms” hypothesis, Klein56 postulated that patients with panic disorder have a pathologically altered suffocation alarm monitor that results in a carbon dioxide hypersensitivity, forming the basis for sudden panic attacks. Integrating findings by Goossens and colleagues53 as well as the present findings, brainstem hyperactivation and its association with the subjective fear of respiratory symptoms might present a neurobiological substrate of such a monitoring system. Our results thus support the idea of increased brainstem activation in patients with panic disorder as a marker of an oversensitive alarm system that might predispose patients to the development of panic attacks. Replication of results, a more detailed postscanning interview on feelings of suffocation, a measurement of end-tidal carbon dioxide as well as more fine-grained analysis of brainstem subregional findings are necessary to further explore the neural basis of the false suffocation alarm hypothesis.
Several further ROIs, such as the amygdala, thalamus, ACC and MCC, showed increased activation in response to threat in patients with panic disorder, but without an association with symptom scores. Amygdala hyperactivation in patients with panic disorder has been interpreted as hyperresponsivity to environmental cues, eliciting full-scale threat-related responses.57 Remarkably, findings of greater amygdala activation in patients with panic disorder than in healthy controls are inconsistent,9,58 and panic attacks have even been observed in patients with amygdala lesions.59–61 Thus, the amygdala seems to be associated with relevance detection and salience processing,62,63 initiating a cascade of panic-related activations without being necessarily involved in (all) full-blown panic attacks.60 The thalamus, as a sensory relay station, is important for the fast processing of incoming visual input and weighing its relevance. Its activation is modulated by selective attention, which gains special significance in the present study owing to the task-irrelevance of panic-related stimuli.64,65
For the resolution of conflict caused by task-irrelevant panic-related stimuli, which closely links emotion and cognition, the cingulate cortex has been proposed as a major neural structure.66,67 The dorsal cingulate cortex plays an important role in overcoming interference due to emotional distraction,68 which is relevant to understanding the co-occurrence of elevated emotion effects in the fear network of patients with panic disorder in the absence of significant differences on the behavioural level. Heightened cingulate cortex activation has also been linked to enhanced sensory sensitivity, including exaggerated scanning for threats (i.e., hypervigilance).66,69 The dorsal anterior cingulate cortex has been linked to the active monitoring of emotions (e.g., the appraisal of an emotion-inducing stimulus), whereas the ventral cingulate cortex has mainly been associated with emotion regulation.70 With regard to the present data, this suggests that patients with panic disorder might consciously attribute threat to the disorder-related stimuli, but lack the capacity to activate emotion regulation processes associated with more ventral cingulate cortex activations. According to Seeley and colleagues,6 the dorsal cingulate cortex represents the key node in the salience network that reflects paralimbic emotional salience processing and is modulated by subjective anxiety ratings.6
The whole-brain analysis showed larger effects in response to panic-related stimuli in patients with panic disorder than in healthy controls in a brainstem/cerebellum/PCC cluster and an anterior insula/inferior-frontotemporal cluster. The hyperactivation for panic-related scenes in visual processing regions in patients with panic disorder underlines the anxiety-related relevance of these scenes for patients, given that the affective significance of stimuli enhances their visual processing.71–73 Emotional salience and simultaneous attentional influences have been observed to modulate sensory processing.74
Limitations
Some limitations of our study need to be considered. Although the sample of 26 patients can be considered large in the field of clinical affective neuroscience, greater sample sizes are needed to increase power in statistical analyses. Six of the 26 patients with panic disorder were undergoing psychotherapy at the time of study participation; however, the Panic and Agoraphobia Scale scores of these 6 patients indicated mild to moderately severe impairment. We implemented a recently developed set of complex visual disorder-related stimuli. However, panic disorder is characterized by strong sensations, including multiple sensory modalities. Thus, anticipation of disorder-related stimuli on other sensory modalities should be compared with or combined with visual emotional processing in future studies. In addition, future studies should also include general threatening stimuli in order to disentangle the effects of generally anxiety-inducing and panic-related stimuli. The additional implementation of measures of autonomic activation, such as pupil diameter, heart rate, or electrodermal activity, would have been informative during the fMRI scan as well as during the rating procedure. More precisely, a concurrent measurement of autonomic measures might help us gain further insight into the involvement of brainstem substructures, such as the locus coeruleus. Future studies should implement such measures to further investigate how subjective and objective measures from anxiety-related body systems translate to the brain.
Conclusion
Taken together, the present study provides new insights about neural correlates of automatic processing and their modulation by key aspects of symptomatology in patients with panic disorder. The disorder can be described as the subjective sense of danger to the core survival systems, such as respiration and heartbeat. Findings in the insula and brainstem suggest that fear of cardiovascular and fear of respiratory symptoms represent core features of panic disorder and are associated with neural activation in regions that are important for monitoring body states and for signalling a potential state of alarm. The present study greatly adds to the knowledge on the neurobiological basis of these fears by revealing an association of subjectively reported panic disorder–specific fears and altered activation in the insula and brainstem in an automatic context.
Footnotes
Funding: This work was supported by grants awarded to T. Straube by the German Research Society (Deutsche Forschungsgemeinschaft, (DFG; SFB/TRR 58: C06, C07).
Competing interests: None declared.
Contributors: K. Feldker, C. Heitmann, P. Neumeister, P. Zwisterlood and T. Straube designed the study. K. Feldker, C. Heitmann and P. Neumeister acquired the data, which all authors analyzed. K. Feldker and L. Brinkmann wrote the article, which all authors reviewed and approved. All authors approved the final version to be published and can certify that no other individuals not listed as authors have made substantial contributions to the paper.
- Received November 24, 2017.
- Revision received March 30, 2017.
- Revision received April 28, 2017.
- Accepted May 1, 2017.