Abstract
Background: Neuroticism personality trait is recognized as an important endophenotypic predictor of generalized anxiety disorder (GAD). Furthermore, endophenotype-based pathway approaches have recently been shown to have greater advantages for gene-finding strategies than traditional case–control studies. In the present study, in addition to conventional case–control methods, we used pathway analyses to test whether the tri-allelic serotonin transporter promoter polymorphism (combining 5-HTTLPR and rs25531) is associated with risk of GAD through its effects on trait neuroticism.
Methods: We included 2236 Han Chinese adults in this study, including 736 patients with GAD and 1500 healthy participants. We genotyped the 5-HTTLPR and rs25531 polymorphisms using the polymerase chain reaction restriction fragment length polymorphism method. We used the Neuroticism scale of the Maudsley Personality Inventory (MPI) short version (MPI-Neuroticism) to measure participants’ tendency toward neuroticism.
Results: Using endophenotype-based path analyses, we found significant indirect effects of the tri-allelic genotype on risk of GAD, mediated by MPI-Neuroticism in both men and women. Compared to women carrying the S'S' genotype, women carrying the L' allele had higher levels of MPI-Neuroticism, which in turn were associated with higher risk of GAD. Men, however, showed the opposite pattern. Using traditional case–control comparisons, we observed that the effect of tri-allelic genotype on GAD was significant, but only in women.
Limitations: Participants were restricted to Han Chinese, and we used only 1 questionnaire to assess neuroticism.
Conclusion: These findings are the first to show that the tri-allelic 5-HTTLPR polymorphism is associated with elevated risk of GAD, and that this effect is mediated via increased trait neuroticism, a sex-dependent risk pathway.
Introduction
Generalized anxiety disorder (GAD) is characterized by persistent worry associated with a variety of symptoms, including concentration problems, difficulty sleeping, fatigue, irritability, muscle tension and restlessness. It is a chronic, highly prevalent and debilitating disorder with an estimated lifetime prevalence of 5.1% to 11.9%.1,2 Twin and family research has demonstrated that genetic determinants play an important role in the development of GAD; genetic variance accounts for nearly 32% of risk.3 Therefore, researchers have attempted to identify and verify specific genes that are responsible for the onset of GAD.
Evidence from animals has demonstrated that anxiety-like behaviours are linked to the serotonin transporter (5-HTT),4 which is responsible for serotonergic neurotransmission by recycling serotonin from the synaptic gap back into the presynaptic neuron. In humans, 5-HTT is encoded by the solute carrier family 6 member 4 (SLC6A4) gene, which includes a 5-HTT-linked polymorphic region (5-HTTLPR) polymorphism in the promoter region.5 The 5-HTTLPR variant is a 44 bp insertion/deletion polymorphism with 2 classic allelic forms, the long variant (L) and the short variant (S). The S allele has consistently been shown to reduce 5-HTT mRNA expression and serotonin uptake in vitro.5,6 So far, only a few studies have investigated the association between 5-HTTLPR polymorphism and GAD. The S/S genotype and S allele of the 5-HTTLPR polymorphism have been associated with GAD;7,8 however, no association has been furthermore reported.9 In addition, Verhagen and colleagues10 demonstrated that there is a sex difference in comorbid GAD: male S allele carriers display more GAD than female S allele carriers. The reasons for these conflicting results are unclear.
Recently, research has found that a single-nucleotide polymorphism (rs25531), located upstream of 5-HTTLPR resulting in a substitution of adenosine (A) to guanine (G), influences the transcriptional efficacy of 5-HTTLPR.11 The LA allele is associated with higher levels of 5-HTT than the LG allele; the LG allele is functionally comparable to the S allele and is associated with lower levels of 5-HTT in vitro. The traditional dichotomous splitting of 5-HTTLPR may reduce statistical power due to a failure to distinguish between the LA and LG alleles.12 Therefore, the functional misclassification of 5-HTTLPR alleles in previous studies could have caused inconsistencies. To date, however, no study has attempted to examine the role of the newer tri-allelic (S, LA, LG) 5-HTTLPR polymorphism in GAD.
Neuroticism, a fairly stable personality trait, is characterized by emotionally unstable over-reactiveness, worry or nervousness.13 Heritability studies have reported that genetic factors have a substantial influence on variability in the neuroticism personality trait.14 In addition, a large-scale twin study has indicated a high degree of shared genetic influence (approximately 80%) on both trait neuroticism and GAD.15 Importantly, this trait has been shown to prospectively predict general anxiety symptoms16 and the development of GAD.17 More importantly, trait neuroticism has recently been recognized as an important endophenotype of GAD — a heritable and state-independent biomarker associated with the illness.15,18 Using a healthy cohort and based on the triallelic approach, we revealed that the 5-HTTLPR polymorphism is associated with the neuroticism personality trait in a sex-dependent manner.19 Men who were homozygous for the S' (including S and LG) allele exhibited higher levels of trait neuroticism than men who carried the L’ (LA) allele. Women, however, showed a nonsignificant association in the opposite direction.
Because endophenotypes link more directly to relevant gene action than complex end point disorders,20 it would seem reasonable to assume that GAD may be conferred by the tri-allelic polymorphism in a sex-specific manner through an effect that is largely mediated by trait neuroticism. Such an endophenotype-based pathway approach may not only have greater advantages for gene-finding strategies than traditional case–control designs,21,22 but it may also provide neuropsychological insights into the complex role of the SLC6A4 promoter variation in GAD. However, to our knowledge, no previous research has explored the possible sex-dependent pathways among tri-allelic 5-HTTLPR variants, trait neuroticism and GAD.
Conducted in a large human sample and using conventional case–control analysis to assess the association between tri-allelic 5-HTTLPR genotypes and GAD, the current study also used a genotype–endophenotype–phenotype pathway model to test whether the functional tri-allelic 5-HTTLPR polymorphism has a sex-specific effect on the neuroticism personality trait, and whether this effect is associated with increased risk of GAD. The theoretical pathway model is presented in Appendix 1, Figure S1, available at jpn.ca/190092-a1.
Methods
Participants
The Institutional Review Board of the Tri-Service General Hospital (TSGH) in Taipei, Taiwan, approved the study protocol. All participants were unrelated ethnic Han Chinese. They provided written informed consent before study procedures began. In total, 2236 adult participants were included in the statistical analysis. We collected data on demographics and lifestyle factors, including body mass index (BMI; kg/m2), weekly exercise level and smoking status. Study participants’ sex was defined based on self-reporting.
Our sample included 736 patients with GAD (315 men and 421 women). They were recruited from inpatient and outpatient settings at TSGH. An attending psychiatrist used the Chinese version of the Mini-International Neuropsychiatric Interview (MINI) to determine each participant’s psychiatric diagnosis, based on the Diagnostic and Statistical Manual of Mental Disorders, fourth edition (DSM-IV-TR).23 All patients had a primary diagnosis of GAD, and none had a comorbid diagnosis of schizophrenia, bipolar disorder, substance use disorder or organic brain disease. However, because GAD has a high rate of comorbidity with depression and other anxiety disorders,24 GAD patients with comorbid diagnoses of depressive disorders (e.g., dysthymia, major depression) or other anxiety disorders (e.g., panic disorder, phobic disorder) were not excluded. We also collected data on participants’ current psychotropic medications (i.e., antidepressants, mood stabilizers, benzodiazepines or antipsychotics taken within 2 weeks before study) and chronic physical illnesses, including cardiovascular disease (e.g., coronary artery disease, myocardial infarction), dyslipidemia, diabetes mellitus and other chronic diseases (e.g., thyroid, liver and kidney disease) based on self-report and medical chart review.
The study sample also consisted of 1500 healthy participants (730 men and 770 women). Recruitment and exclusion processes have been described elsewhere in more detail.25 Briefly, all healthy participants received a medical checkup at TSGH that included biochemical analyses, blood pressure measurement, electrocardiography, physical examination and thoracic radiography. None of them had any organic diseases, including kidney or liver disease, cardiovascular disease, metabolic disorders, neurologic disorders, malignancy or obesity (BMI ≥ 30 kg/m2). They were also screened using the Chinese version of the MINI23 by a well-trained research assistant and were free of mental disorders.
Assessment of trait neuroticism
We determined propensity to neuroticism using the Chinese version of the Neuroticism scale from the short-form Maudsley Personality Inventory (MPI-Neuroticism).26 This scale consists of 13 self-reported items that were rated on a 3-point Likert scale (no = 0, uncertain = 1, yes = 2). The total scores on the MPI-Neuroticism scale range from 0 to 26; higher scores represent greater levels of tendency toward neuroticism. The MPI-Neuroticism scale has been shown to have a good test–retest reliability and is commonly used in health care and community settings in Taiwan.27,28
Assessment of anxiety symptoms
Patients with GAD were assessed for anxiety over the week preceding the study using the Chinese version of the Beck Anxiety Inventory (BAI), which consists of 21 self-reported items rated on a Likert scale ranging from 0 to 3 (total score range: 0–63).29 The BAI scores are classified as normal anxiety (0–7), mild anxiety (8–15), moderate anxiety (16–25) and severe anxiety (30–63).29 In the present study, GAD patients with BAI scores ≤ 7 were identified as having remitted GAD; the others (BAI > 7) were identified as having current GAD.
Genotyping
We isolated genomic DNA from venous blood samples using the QIAamp DNA Mini Kit (Qiagen). We genotyped the 5-HTTLPR (S/L) and rs25531 (A/G) polymorphisms via polymerase chain reaction (PCR)–restriction fragment length polymorphism assay as described previously.19 In short, the PCR product resulting from the 5-HTTLPR S allele was 486 bp to 487 bp long, and the product from the L allele was 509 bp to 529 bp long.30 The PCR products were then digested using restriction endonuclease MspI31 to determine the A or G allele (rs25531) on the L allele. An LG allele indicated that the digested products contained a fragment of 166 bp; an LA allele indicated the lack of such a fragment.30,31
Because there is no functional difference between the SA and SG alleles, they were both recorded as the S allele.31 We then reclassified the 5-HTTLPR variant as lower-expressing S’ (S, LG) and higher-expressing L’ (LA) alleles for data analyses. In this study, because only 48 participants had the L’L’ genotype, those carrying at least 1 L’ allele were grouped together (L’ allele carriers, coded as “0”) and compared with the S’ allele homozygotes (coded as “1”). This method has been used previously to deal with skewed genotypic distributions.32
Covariates
We entered age as a covariate in all analyses. We also dummy-coded psychiatric diagnoses (i.e., depressive disorders and other anxiety disorders) and entered them as covariates in all analyses. We created a covariate for chronic medical conditions (e.g., cardiovascular disease, diabetes or thyroid disease; 0 = no, 1 = yes) in all of our statistical models. We also treated current use of psychotropic drugs as a covariate (0 = no, 1 = yes) in all analyses. We used lifestyle factors, including BMI, weekly exercise level (0 = nil, 1 = 1–2 times/week, 2 = ≥ 3 times/week) and smoking status (0 = no, 1 = yes), as covariates in all analyses.
Statistical analysis
We tested the Hardy–Weinberg equilibrium for the 5-HTTLPR and rs25531 polymorphisms using the χ2 test. We used the Student t test (stratified by sex) to compare continuous data, and the χ2 test for categorical data among the tri-allelic 5-HTTLPR genotypes. We substituted the Fisher exact test for the χ2 test when sample sizes were smaller than expected (≤ 5 participants). In the case–control analyses, we applied logistic regression models to determine the association between the tri-allelic genotypes and GAD. According to modern approaches to (moderated) mediation in path analyses, demonstrating that the initial variable (i.e., tri-allelic 5-HTTLPR genotypes) is correlated with the outcome (i.e., GAD) is not required to establish (moderated) mediation.33,34 Instead, it is recommended that (moderated) mediation analyses be conducted with the significance tests of the indirect effect, which more directly address (moderated) mediation.35 Thus, using Hayes’ PROCESS,36 a regression-based modelling approach (model 1), we initially tested whether sex moderated the associations between tri-allelic genotypes and trait neuroticism based on the bootstrap (n = 10 000) bias correction method. Then, we used logistic regression models to assess the effect of MPI-Neuroticism on the risk of GAD. Lastly, using Hayes’ PROCESS36 model 8, we examined whether sex moderated indirect (mediation) effects of tri-allelic genotypes on GAD via trait neuroticism based on a bias-corrected bootstrapping procedure (10 000 bootstrap samples). The path coefficients between studied variables were reported using unstandardized β regression coefficients (B). We performed post hoc analyses, separated by sex, to check the 95% bootstrapped confidence intervals (95% CIs). If the 95% CI did not contain zero, we concluded that the results were statistically significant (i.e., p < 0.05).36 We conducted all statistical analyses using SPSS statistics software (version 24; IBM).
Results
Participants: demographics and clinical characteristics
The distributions of 5-HTTLPR and rs25531 genotypes did not significantly deviate from Hardy–Weinberg equilibrium expectations in our cohort (data not shown). The demographics and clinical characteristics of our sample, stratified by sex, are presented in Table 1 based on tri-allelic 5-HTTLPR genotype. In the total sample, we found no significant differences in age, BMI, weekly exercise levels or smoking status across the triallelic 5-HTTLPR genotypes in either men or women. In patients with GAD, we found that male S’ allele homozygotes had a significantly higher rate of current GAD than male L’ allele carriers (χ2 = 6.17, p = 0.013), and this effect became more robust after adjustment for covariates (B = 1.07, p = 0.006). Female L’ allele carriers had a higher rate of current GAD than female S’ allele homozygotes; however, the effect did not reach statistical significance. Other parameters — including BAI score, depressive disorders, other anxiety disorders, chronic medical conditions or psychotropic medication use — did not differ significantly between the tri-allelic 5-HTTLPR genotypes in male or female patients with GAD.
Participant characteristics according to tri-allelic 5-HTTLPR genotype (5-HTTLPR/rs25531) and sex
In the total sample, with regard to sex differences, women were older than men (t = 9.49, p < 0.001). In addition, women had lower BMI (t = 17.0, p < 0.001), current smoking rate (χ2 = 202.9, p < 0.001) and weekly exercise levels (χ2 = 43.4, p < 0.001) than men. Among patients with GAD, women exhibited higher rates of comorbid depressive disorders than men (χ2 = 6.93, p = 0.008). We found no significant differences in BAI score, other anxiety disorders, chronic medical conditions or psychotropic medication use between male and female patients with GAD.
Finally, patients with remitted GAD had significantly lower MPI-Neuroticism scores than patients with current GAD (t = 10.4, p < 0.001). In addition, patients with GAD who were taking any psychotropic medication had significantly higher MPI-Neuroticism ratings than those who were not taking psychotropic medication (all p < 0.05).
Case–control analyses of association between tri-allelic 5-HTTLPR genotypes and GAD
Based on logistic regression analysis and analyzed in the total sample, the risk of GAD was not significantly influenced by the tri-allelic 5-HTTLPR genotypes (B = −0.20, p = 0.13). However, further analyses by sex showed that women who were L' allele carriers had a significantly higher risk of GAD than women with the S'S' genotype (B = −0.36, p = 0.043). Conversely, men carrying the S'S' genotype had a higher incidence of GAD than men carrying the L' allele, although the difference did not reach significance (B = 0.05, p = 0.81; Table 2).
Case–control analyses of associations between tri-allelic 5-HTTLPR genotypes and GAD*†
Pathway analyses of tri-allelic 5-HTTLPR genotypes, trait neuroticism and GAD
Moderating effect of sex on association of tri-allelic genotypes with trait neuroticism
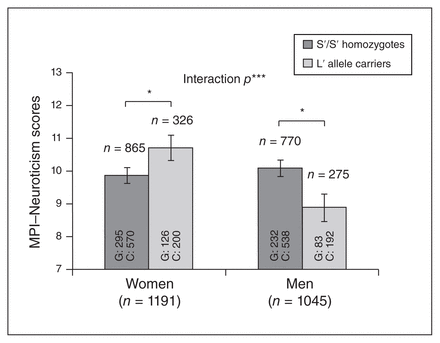
In the total sample, MPI-Neuroticism scores were significantly affected by sex (B = −1.62, p = 0.006) and tri-allelic 5-HTTLPR genotype (B = −0.99, p = 0.029), but this finding was qualified by a significant genotype × sex interaction (B = 2.17, p = 0.001). Furthermore, even without adjusting for covariates, the interaction effect of 5-HTTLPR genotype and sex on MPI-Neuroticism was significant (B = 1.82, p = 0.012). The adjusted effect (B = 2.17, p = 0.001) was larger than the unadjusted effect (B = 1.82, p = 0.012), indicating that covariate adjustment was needed to improve the accuracy of the model in this study. The moderating effect of sex on the associations between the tri-allelic polymorphism and MPI-Neuroticism is plotted in Figure 1. Follow-up analyses showed that men with the S'S' genotype had significantly higher MPI-Neuroticism ratings than men carrying the L' allele (B = 1.18, p = 0.016). Women, however, exhibited a significant inverse pattern, demonstrating that carriers of the L' allele had higher MPI-Neuroticism scores than S' allele homozygotes (B = −0.99, p = 0.029).
Associations between tri-allelic 5-HTTLPR genotypes and trait neuroticism, stratified by sex. Data were adjusted for age, lifestyle factors, depressive disorders, other anxiety disorders, chronic medical conditions and use of psychotropic drugs. C = controls; G = generalized anxiety disorder; MPI = Maudsley Personality Inventory (short form). *p < 0.05; ***p = 0.001.
In a separate analysis of the healthy participants (n = 1500; adjusting for age, BMI, smoking status and weekly exercise levels), the interaction effect of sex and tri-allelic 5-HTTLPR polymorphism on MPI-Neuroticism remained significant with a similar pattern (B = 1.41, p = 0.037). Likewise, when we analyzed only patients with GAD (n = 736; adjusting for all relevant covariates), we still observed a significant moderating effect of sex on the associations between the tri-allelic 5-HTTLPR polymorphism and MPI-Neuroticism (B = 2.20, p = 0.044).
Effect of trait neuroticism on GAD
Based on a logistic regression model, higher MPI-Neuroticism scores were significantly associated with a higher risk of GAD (B = 1.25, p < 0.001). Both men and women had similar significant results (B = 1.26 and 1.24, respectively; both p < 0.001).
Indirect effect of tri-allelic genotypes on GAD via trait neuroticism
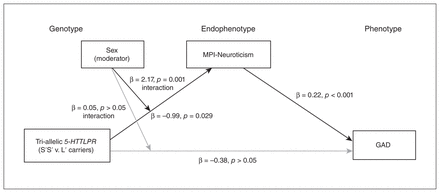
We further tested whether the tri-allelic 5-HTTLPR polymorphism-driven variability in MPI-Neuroticism was indirectly related to incidence of GAD in a sex-dependent manner. The major findings of our model were that the tri-allelic 5-HTTLPR variation was significantly associated with higher MPI-Neuroticism scores in a sex-specific way (B = 2.40, p = 0.001). Levels of MPI-Neuroticism, then, were positively and significantly associated with elevated risk for GAD (B = 0.89, p < 0.001; Fig. 2). Post hoc analyses stratified by sex revealed that the indirect effects of tri-allelic 5-HTTLPR genotypes on GAD risk via trait neuroticism were significant in both men and women, but with an opposite pattern (B = 0.264 [95% CI = 0.048 to 0.478] and −0.221 [95% CI = −0.417 to −0.023], respectively; Table 3).
Sex moderates the pathways between tri-allelic 5-HTTLPR genotypes, MPI-Neuroticism and GAD. Tri-allelic 5-HTTLPR genotypes coded as 0 = L' allele carriers, 1 = S' allele homozygotes. Data were adjusted for age, lifestyle factors, depressive disorders, other anxiety disorders, chronic medical conditions and use of psychotropic drugs. Black arrows indicate significant pathways; grey arrows represent nonsignificant pathways. GAD = generalized anxiety disorder; MPI = Maudsley Personality Inventory (short form).
Sex-specific indirect effects of tri-allelic 5-HTTLPR genotypes on GAD via trait neuroticism*†
Discussion
To our knowledge, this was the first study to investigate possible sex-specific pathways between tri-allelic 5-HTTLPR polymorphism, trait neuroticism and GAD. After adjusting for all the confounding variables, our main findings showed that women carrying the L' allele exhibited higher levels of MPI-Neuroticism than women carrying the S'S' genotype. However, we found the opposite effect in men: S'S' genotype carriers had higher MPI-Neuroticism scores than those with at least 1 copy of L' allele. Furthermore, in the endophenotype-based pathway model, we observed that in women, 5-HTTLPR L' allele carriers were high in MPI-Neuroticism, which in turn was associated with increased risk of GAD. However, men carrying the S'S' genotype had higher degrees of MPI-Neuroticism, which were in turn associated with a higher incidence of GAD. Using a traditional case–control approach, we could find a significant result for the association between tri-allelic genotype and GAD in women, but not in men. We also found that the tri-allelic 5-HTTLPR genotype was associated with current GAD, but only in men with GAD. These findings were in line with growing evidence for sex-specific effects of 5-HTTLPR on different brain functions and related behaviours.32,37–40
Accumulated evidence has shown that the function of the serotonergic system is differentially regulated in men and women, such as via different levels of serotonin metabolism41 and rates of serotonin synthesis,42 which are largely determined by the effect of 5-HTT. In addition, estrogen treatment, has been reported to upregulate the expression of the SLC6A4 gene, but not androgen treatment.43 Notably, recent research has shown that LL 5-HTTLPR female rhesus monkeys showed greater prolactin response to acute citalopram administration than S-variant female monkeys when estrogen and progesterone levels were high.44 However, Josephs and colleagues45 demonstrated that SS homozygotic participants revealed higher cortisol reactivity to a variety of stressors than participants homozygous for the L allele, but only in participants with high testosterone. Our sex-opposite results for the tri-allelic 5-HTTLPR genotype on trait neuroticism and GAD were in accordance with previous findings from distinct sex steroids.44,45
In a mentally and physically healthy cohort (n = 1139: 550 males and 589 females), we showed that men carrying the 5-HTTLPR S'S' genotype had higher neuroticism ratings than male L' allele carriers.19 Women, however, showed an opposite but statistically nonsignificant pattern. In the present study, using a sample almost twice as large (n = 2236: 1045 males and 1191 females) that included healthy participants and patients with GAD, we replicated the significant finding with respect to the tri-allelic 5-HTTLPR effect on trait neuroticism in men. In addition, we further identified a significant inverse effect in women, demonstrating that women who were L' allele carriers had higher levels of MPI-Neuroticism than women with the S'S' genotype. Thus, the previous nonsignificant result in women19 may indeed have been due to the limited sample size. As well, in the present study, even analyzed separately for the GAD and healthy participant groups, the effects of 5-HTTLPR on MPI-Neuroticism were also consistently sex-specific. The findings of the present study are in line with those of previous studies that have reported sex-specific associations between conventional bi-allelic 5-HTTLPR polymorphism and trait anxiety/neuroticism.32,46–48
As mentioned in the introduction, the SS genotype and S allele of the bi-allelic 5-HTTLPR polymorphism have been shown to be a risk factor for GAD. However, inconsistent results have been reported. In the present study, using a traditional case–control approach, the effect of the tri-allelic 5-HTTLPR genotype on GAD was significant only in women. Men showed a nonsignificant opposite pattern. However, when analyzing male patients with GAD, we found that the tri-allelic 5-HTTLPR genotype was related to current status of GAD; female patients with GAD showed an inverse but nonsignificant pattern. Importantly, because the tri-allelic approach provides better estimates than the traditional bi-allelic method,12 and because endophenotypes reflect more proximal effects of genes involved in complex psychiatric phenotypes,20 integratively analyzing trait neuroticism and taking the moderator role of sex into consideration may facilitate discovery of the role of 5-HTTLPR variation in GAD. Indeed, our genotype–endophenotype–phenotype pathway model, which met the criteria for moderated mediation,33,34 revealed a sex-dependent effect of tri-allelic 5-HTTLPR polymorphism on trait neuroticism, which, in turn, was associated with increased risk for GAD. When stratified by sex, the indirect effects of the tri-allelic 5-HTTLPR variant on GAD via trait neuroticism were both significant, with inverse patterns in men and women. These results were partly in line with the findings of Verhagen and colleagues:10 they reported that male S allele carriers were associated with increased comorbid GAD than female S allele carriers. Taken together, our findings may complement previous case–control studies and provide a potential neuropsychological insight into the association of 5-HTTLPR polymorphism with GAD. Finally, the finding that tri-allelic genotype-driven effects run in opposite directions in men and women may explain the inconsistent results with respect to the associations between 5-HTTLPR and GAD in previous studies that had different ratios of male and female participants.
Previous research has demonstrated that several variables, such as age, lifestyle factors (e.g., cigarette smoking),49 physical illness (e.g., cardiovascular diseases),50 psychological disorders (e.g., affective disorders)51 and/or use of psychotropic drugs (e.g., antidepressants)52 may influence the ratings of trait neuroticism. Failure to avoid these confounding effects may bias the data; however, our study had a carefully controlled protocol for adjusting the possible effects of these non-genetic confounders. In addition, although remitted patients with GAD had lower MPI-Neuroticism ratings than current GAD patients, this may not have affected our findings of 5-HTTLPR and sex on MPI-Neuroticism. Indeed, when patients with remitted GAD (n = 79) were excluded from analysis (i.e., using 657 current GAD and 1500 controls for analysis), we observed that the results still showed a very similar and significant pattern (for interaction: B = 2.10; p = 0.002). Furthermore, there are large racial differences in the distribution of 5-HTTLPR variants. The frequency of the low-expressing (S') allele was much higher in our Asian sample (~0.76) than in white cohorts (~0.22); other populations had intermediate values.11 Therefore, admixture of different racial groups may lead to problems in determining the genetic effects of the SLC6A4 gene. However, all of the participants in our study were Han Chinese adults recruited from a genetically homogeneous population pool in Taiwan.53 Moreover, testing in samples with greater genetic homogeneity has been demonstrated to increase the power of gene-finding in studying complex traits or diseases.54 Taken together, the findings of the present study may precisely reveal the sex-specific effects of tri-allelic 5-HTTLPR polymorphism on trait neuroticism and GAD without confounding or ethnic stratification bias.
Limitations
The present study had several limitations. Although the neuroticism personality trait is a well-established risk factor of GAD, the explanation of the temporal order of the relationship between trait neuroticism and GAD should be a concern. However, our previous research,19 conducted in healthy participants only, has already shown that the tri-allelic 5-HTTLPR polymorphism is associated with trait neuroticism in a sex-specific manner. This finding was further confirmed in the present study using a larger healthy cohort. Thus, trait neuroticism may be an endophenotype of people with this particular sex-dependent genetic vulnerability to GAD (tri-allelic 5-HTTLPR genotype), rather than the result of the onset of illness, providing support for the temporal relationships between trait neuroticism and GAD in our path model. Despite this, future studies with a prospective design are needed to validate our cross-sectional findings. Furthermore, to avoid population stratification biases, the participants recruited for the present study were all Han Chinese. Therefore, our findings may be of limited generalizability to other populations. Further research should be conducted in ethnically diverse populations to verify this issue. Finally, we used only the MPI to determine the presence of the anxiety-related trait, neuroticism. Future studies are needed that use additional standardized questionnaires to measure anxiety-related traits such as harm avoidance55 to validate the findings of the present study.
Conclusion
Our findings demonstrate that the tri-allelic 5-HTTLPR polymorphism is associated with increased risk of GAD, and that this effect is mediated through increased levels of the neuroticism personality trait, a sex-specific risk pathway.
Acknowledgments
The authors thank Ms. Hsiao-Hsin Hung for her assistance in preparing this manuscript. This study was supported in part by grants from the Medical Affairs Bureau of the Ministry of National Defense, Taipei, Taiwan (MAB-104-074), the Tri-Service General Hospital, National Defense Medical Center, Taipei, Taiwan (TSGH-C105-122) and the Ministry of Science and Technology of Taiwanese Government (MOST-107-2314-B-016-050).
Footnotes
Competing interests: None declared.
Contributors: H.-A. Chang, W.-H. Fang and C.-C. Chang designed the study. H.-A. Chang, W.-H. Fang, T.-C. Chang and C.-C. Chang acquired the data, which H.-A. Chang, W.-H. Fang, Y.-P. Liu, N.-S. Tzeng, J.-F. Shyu, F.-J. Wan, S.-Y. Huang and C.-C. Chang analyzed. H.-A. Chang, W.-H. Fang and C.-C. Chang wrote the article, which all authors reviewed. All authors approved the final version to be published and can certify that no other individuals not listed as authors have made substantial contributions to the paper.
- Received May 7, 2019.
- Revision received October 3, 2019.
- Accepted January 12, 2020.