Abstract
Background: Functional underpinnings of cognitive control deficits in unbiased samples (i.e., all comers) of patients with psychotic spectrum disorders (PSD) remain actively debated. While many studies suggest hypofrontality in the lateral prefrontal cortex (PFC) and greater deficits during proactive relative to reactive control, few have examined the full hemodynamic response.
Methods: Patients with PSD (n = 154) and healthy controls (n = 65) performed the AX continuous performance task (AX-CPT) during rapid (460 ms) functional neuroimaging and underwent full clinical characterization.
Results: Behavioural results indicated generalized cognitive deficits (slower and less accurate) across proactive and reactive control conditions in patients with PSD relative to healthy controls. We observed a delayed/prolonged neural response in the left dorsolateral PFC, the sensorimotor cortex and the superior parietal lobe during proactive control for patients with PSD. These proactive hemodynamic abnormalities were better explained by negative rather than by positive symptoms or by traditional diagnoses according to the Diagnostic and Statistical Manual of Mental Disorders Fourth Edition, Text Revision (DSM-IV-TR), with subsequent simulations unequivocally demonstrating how these abnormalities could be erroneously interpreted as hypoactivation. Conversely, true hypoactivity, unassociated with clinical symptoms or DSM-IV-TR diagnoses, was observed within the ventrolateral PFC during reactive control.
Limitations: In spite of guidance for AX-CPT use in neuroimaging studies, one-third of patients with PSD could not perform the task above chance and were more clinically impaired.
Conclusion: Current findings question the utility of the AX-CPT for neuroimaging-based appraisal of cognitive control across the full spectrum of patients with PSD. Previously reported lateral PFC “hypoactivity” during proactive control may be more indicative of a delayed/prolonged neural response, important for rehabilitative purposes. Negative symptoms may better explain certain behavioural and hemodynamic abnormalities in patients with PSD relative to DSM-IV-TR diagnoses.
Introduction
Cognitive dysfunction represents a core deficit in patients with psychotic spectrum disorders (PSDs) and is associated with impairments in everyday functioning1,2 as well as with increased negative symptoms.3–5 A recent trend has aimed to reconceptualize the range of cognitive impairments as reflecting a “common underlying deficit”6,7 in proactive cognitive control (i.e., early selection and sustained maintenance of goal relevant information).5,8 The Dual Mechanisms of Control theory8,9 implicates the involvement of the lateral prefrontal cortex (lPFC) in instantiating and maintaining proactive control during cognitive tasks,10 with reduced lPFC activity typically observed in patients diagnosed with schizophrenia and PSDs.11,12 In contrast, reactive cognitive control (i.e., rapid retrieval of previously encoded contextual information in the presence of competing/conflicting stimuli)8,13 has been reported to be more preserved in schizophrenia.3,12 However, roles of the dorsolateral prefrontal cortex (dlPFC) versus the ventrolateral prefrontal cortex (vlPFC) and their associated temporal activation patterns during cognitive control across PSD remain debated.13,14
Specifically, sustained/tonic activation of the lPFC is posited to occur during proactive control, reflecting active maintenance of task goals via connectivity with sensorimotor regions.8,9,12 Conversely, transient/phasic activation of the lPFC is associated with reactive control, indicating more rapid retrieval of previously encoded contextual information in response to environmental changes.8,13,15 Decreased neural variability has also been described as a marker of poor proactive engagement in schizophrenia11 and healthy controls.16 The AX Continuous Performance Test (AX-CPT) is recommended by the Cognitive Neuroscience Treatment Research to Improve Cognition in Schizophrenia (CNTRICS) initiative to assess behavioural and functional profiles of goal maintenance.17 Two recent studies reported decreased prefrontal and parietal cortex activity for schizophrenia relative to healthy controls during proactive control,12,18 and equivalent activation during reactive control.12 Reduced dlPFC activity during proactive control has also been reported for schizophrenia compared with other PSDs and healthy controls,19–21 is present in those at high risk for developing schizophrenia and other PSDs,21 and represents a stable trait after a first-episode break.22 Conversely, transient activation within the vlPFC and sensorimotor regions is found to be largely preserved in schizophrenia during reactive control,3,11,23 albeit at a reduced level.23
Few cognitive control imaging studies have consecutively recruited patients with PSDs according to the Research Domain Criteria (RDoC) framework (i.e., an all comers study).24 Other AX-CPT studies18,19,25 have used variable and/or below-chance behavioural thresholds for study inclusion. Inclusion of participants who perform poorly on a task hinders the accurate delineation of specific cognitive deficits26 and makes it challenging to determine the basis for neural activity during task performance (or the lack thereof).27 Finally, few studies have explicitly examined differences in the temporal profile and variability of the hemodynamic response function (HRF) during cognitive control in patients with PSD despite growing evidence of increased sensitivity.28–30
The current study therefore used a multisensory AX-CPT variant to decouple the timing and variability of hemodynamic responses (present/absent; transitory/sustained) to cues and probes. We predicted increased behavioural and hemodynamic abnormalities during contrasts of proactive rather than reactive control for patients with PSD12,18 as well as decreased variability within motor circuitry16 during proactive instantiation.11
Methods
Participants
We consecutively recruited 154 patients with PSD (96 males, 32.00 ± 9.28 years old) in this RDoC study24 from local psychiatric centres and newspaper ads. Patients between the ages of 18 and 65 years were diagnosed with schizophrenia, schizoaffective disorder or bipolar disorder type I with psychotic features by a board-certified psychiatrist using the Structured Clinical Interview for the Diagnostic and Statistical Manual of Mental Disorders Fourth Edition, Text Revision (DSM-IV-TR).31 Exclusion criteria consisted of the following: contraindications for MRI, developmental disorders (e.g., autism spectrum or intellectual disability), a history of neurologic diagnoses, moderate or severe head injury (loss of consciousness > 30 minutes), pregnancy, electroconvulsive shock therapy (scheduled or having occurred within the previous month), or history of substance abuse disorders within the previous 12 months (excluding marijuana use).
We recruited 65 healthy controls (41 males, 33.25 ± 8.15 years old) from local communities through word of mouth and fliers. Additional exclusion criteria for healthy controls included the following: history of an Axis I disorder, recent history of substance abuse, a first-degree relative with a PSD, or elevated depressive symptoms (score > 29 on the Beck Depression Inventory).32
Participants were administered urine-based drug screens and excluded for positive results. Participants provided informed consent according to institutional guidelines at the University of New Mexico School of Medicine.
Clinical and neuropsychological assessments
Primary study measures included the Wechsler Test of Adult Reading, the Measurement and Treatment Research to Improve Cognition in Schizophrenia (MATRICS) Consensus Cognitive Battery (MCCB; overall and Continuous Performance Test–Identical Pairs [CPT-IP] scores), the Executive Abilities: Measures and Instruments for Neurobehavioural Evaluation and Research (EXAMINER) executive composite scale, the University of California San Diego Performance-Based Skills Assessment Brief Version, and the Quality of Life Questionnaire in Schizophrenia 18. Patients with PSD also completed the Positive and Negative Syndrome Scale (PANSS), the Schizo-Bipolar Scale and other measures evaluating psychopathology, medication and motor dysfunction (Appendix 1, available at jpn.ca/190212-a1).
Task description
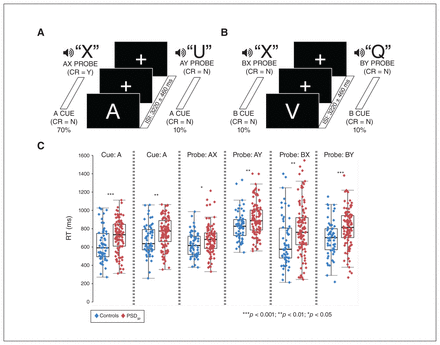
A detailed description of the intermodal AX-CPT (Fig. 1A/1B) can be found in previous studies by our group.16,33 Importantly, these studies evidenced behavioural and functional characteristics similar to the standard intramodal task in healthy controls16 and patients with schizophrenia.33 Additional supporting information in Appendix 1, Table S3, indicates similar performance across groups to previous unisensory implementations of the AX-CPT. Furthermore, previous research has found that multisensory cue–probe tasks do not significantly affect performance relative to unisensory cue–probe tasks,34 in addition to not differentially affecting cognitive control networks across healthy controls and patients with schizophrenia.35,36 Participants observed a series of visual cues (A or non-A letters [collectively B]; duration = 500 ms), followed by a varied interstimulus interval (2760~3680 ms). Next, auditory probes (X or non-X letters [collectively Y]; duration = 500 ms) were presented, followed by a varied intertrial interval (4060~4980 ms). The target response was made during an X probe that was preceded by an A cue, and nontarget responses were made during cue presentation; an X probe preceded by a B cue; or a Y probe preceded by any cue (A or B). The jittering between the interstimulus interval and intertrial interval minimized nonlinear summing of the HRF between cue–probe phases37 and decreased temporal expectations.
(A and B) These panels graphically represent the task, including the interstimulus interval (ISI) between cues and probes. Correct responses to cues and probes are denoted in panels. (C) Box-and-scatter plots depict reaction time (RT) between healthy controls (n = 58; blue diamonds) and good-performing patients with psychotic spectrum disorder (PSDgp; ≥ 56% accuracy on all conditions; n = 105; red diamonds). ***p < 0.001; **p < 0.01; *p < 0.05. CR = correct response; N = no; Y = yes.
Trials were presented pseudorandomly, each run maintaining an equivalent probability structure (AX = 70%, AY/BX/BY = 10%), resulting in trial counts of AX = 112, AY = 16, BX = 16, BY = 16. The proportion of low probability (10% AY and BX) versus high probability (70% AX) AX-CPT trials necessitates a longer scan session to achieve the minimum trials needed for neural modelling. Prior to entering the scanner, participants were given detailed instructions on the task and performed up to 3 blocks of practice trials (Appendix 1).
We performed behavioural analyses on median reaction time for correct trials (primary), accuracy (% correct; secondary), behavioural shift index (secondary), d’-index and response variability (secondary). For the latter, Pitman–Morgan tests (Appendix 1) assessed whether variance was greater during proactive (BX) relative to reactive (AY) trials.16
Imaging, processing and statistical analyses
We collected MRI data on a Siemens 3 T Tim Trio scanner with a 32-channel head coil. We collected structural scans using a multi-echo magnetization-prepared rapid gradient-echo T1–weighted sequence (5-echo; 1 × 1 × 1 mm voxels). We collected functional data for the AX-CPT using a single-shot, gradient-echo echoplanar pulse sequence with simultaneous multi-slice technology (repetition time 460 ms; multiband acceleration factor = 8; 3.02 × 3.02 × 3.00 mm voxels). We also collected a single band reference image and 2 spin-echo distortion mapping pre-scan sequences (Appendix 1).
We preprocessed time-series data using AFNI and FSL and included despiking, 2- and 3-dimensional motion correction, susceptibility-induced field distortions correction (FSL Topup), spatial normalization and smoothing (6 mm Gaussian kernel). Voxel-wise deconvolution analysis generated a single HRF for each trial type based on the first 14.26 s post-stimulus onset,16 separately modelling correct and incorrect responses along with 6 motion parameters and their first-order derivatives38 relative to the baseline state (visual fixation plus gradient noise). To ensure adequate task performance and proper engagement of relevant neural circuitry for appropriate modelling of AY and BX trials (9/16 trials), all participants were required to achieve a minimum accuracy rate of 56%. Beta coefficients were summed for peak (activation = 3.22–5.06 s post-stimulus) and late-peak (5.06–6.90 s) images and divided by the average constant intercept to capture temporal dynamics associated with cognitive control.16
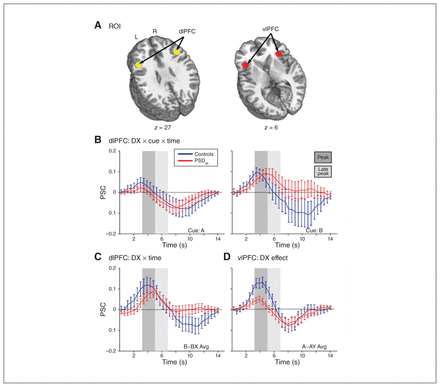
Based on previous studies using the AX-CPT,39,40 we identified a priori, spherical (12 mm radius) regions of interest (ROIs; Fig. 2A) for the bilateral dlPFC (Talairach coordinates, x = ± 41, y = 18, z = 28) and vlPFC (x = ± 36, y = 22, z = 6). A series of 2 × 2 × 2 (group [PSD v. healthy controls] × condition × time [peak v. late peak]) mixed-measures analyses of covariance (ANCOVAs), with mean frame-wise displacement as a covariate, examined functional activation across the following principal contrasts: increased proactive processes during cue presentation (B v. A); reactive versus proactive processes during probe presentation (AY v. BX); proactive (B v. BX) and reactive (A v. AY) control across the entire cue/probe phase. These contrasts were based on previous studies (Appendix 1, Table S1) for cues12,21 and probe3,16,33 models of proactive (B and BX) and reactive (AY) control. We used full cue–probe3,9 contrasts to assess the full time-course of neuronal activation during prototypical proactive (B~BX) and reactive (A~AY) states.
(A) A priori regions of interest (ROIs; 12 mm sphere) within the bilateral dorsolateral prefrontal cortex (dlPFC; x = ± 41, y = 18, z = 28) and ventrolateral prefrontal cortex (vlPFC; x = ± 36, y = 22, z = 26) based on the Talairach atlas (z = axial slice location). Line graphs with standard error bars represent the percent signal change (PSC) from baseline for the average hemodynamic response (B) within the dlPFC across cue conditions, and averaged across (C) proactive (B~BX) trials within the dlPFC and (D) reactive (A~AY) trials within the vlPFC contrasting healthy control (n = 58; blue line) and good-performing patients with psychotic spectrum disorder (PSDgp; n = 105; red line) groups. Panel background shading designates peak (dark grey) or late peak (light grey) phases of the hemodynamic response. L = left; R = right.
Group effects and interactions are presented here; condition effects and interactions are briefly discussed in Appendix 1. We corrected voxel-wise results for false positives (p < 0.05) based on 10 000 Monte-Carlo simulations (p < 0.001) and spherical autocorrelation estimates41 according to the latest recommendations.42 Volume thresholding was set at 513 μL. Finally, all significant (PSD v. healthy controls) behavioural and/or functional results underwent additional ANCOVAs within PSD, including both DSM-IV-TR diagnosis and PANSS positive and negative subscales to determine whether diagnostic category or positive/negative psychotic symptoms accounted for unique variance.43 We predicted spectrum-dependent (schizophrenia > schizoaffective disorder > bipolar disorder type I) behavioural and functional deficits during proactive control conditions,19 primarily mediated by negative clinical symptoms.43,44
Results
Demographics and clinical data
Surprisingly, 46 PSD patients and 6 healthy controls were excluded from behavioural analysis for poor performance (< 56% accuracy on at least 1 trial type) despite practice before scanning. Three additional PSD patients and 1 healthy control participant were excluded for excessive (3 times interquartile range) head motion relative to their group. Therefore, the functional MRI analyses consisted of 105 good-performing patients with PSD (PSDgp; schizophrenia = 65, schizoaffective disorder = 9, bipolar disorder type I = 31; 69 males; 31.58 ± 9.32 years old) and 58 healthy controls (34 males; 32.90 ± 8.25 years old). Clinical and behavioural comparisons including poor-performing patients with PSD (PSDpp; schizophrenia = 34, schizoaffective disorder = 3, bipolar disorder type I = 9; 26 males; 32.57 ± 9.30 years old) are also presented for full disclosure (Appendix 1).
We found no significant differences in sex, age or premorbid intelligence (all p > 0.05) between PSDgp and healthy controls (Table 1). The PSDgp group had lower educational attainment, executive composite score, MCCB overall cognitive battery score, MCCB CPT-IP score, quality of life and performance total scores, and elevated nicotine dependence relative to healthy controls (all p ≤ 0.01; Table 1). However, PSDgp presented higher educational attainment, premorbid intelligence, executive composite score, MCCB overall cognitive battery score, MCCB CPT-IP score and performance total scores relative to the PSDpp. The PSDgp group also demonstrated lower levels of clinical symptoms (PANSS subscales) and were less medicated (olanzapine equivalence) than the PSDpp group (all p ≤ 0.01; Table 1).
Demographic characteristics and cognitive and clinical measures
Behavioural results
We observed significant group effects across all 4 principal reaction time analyses (PSDgp > healthy controls; all p < 0.01; Fig. 1C) in conjunction with nonsignificant group × condition interactions (all p > 0.10), indicative of overall slowing for PSD. Expected main effects of condition were exhibited for cue (F1,165 = 47.92, p < 0.001; B > A) and probe (F1,165 = 69.90, p < 0.001; AY > BX) contrasts.3,16 Cue–probe effects were present for A versus AY (F1,165 = 324.87, p < 0.001; A < AY) and absent for B versus BX (p = 0.88). We repeated analyses using all PSD to align with previous investigations,3,12,21 and results indicated a proactive deficit driven primarily by PSDpp (Appendix 1, Figure S1).
Secondary analyses indicated group differences in accuracy (healthy controls > PSDgp; all p < 0.05) for A/AX/BX/BY conditions, as well as the d’-index. We found no group differences in response time variance during the AY versus BX contrast (p > 0.10). Finally, supplemental assessments replicated previous observations16 that the behavioural shift index was explained primarily by BX (PSDgp R2 = 73%; healthy controls R2 = 80%) rather than AY (PSDgp R2 = 2%; healthy controls R2 = 8%) variance in both groups.
Functional task results
The PSDgp group displayed greater mean frame-wise displacement relative to healthy controls (p < 0.001; Z = −4.48; r = 0.35). We performed all subsequent functional analyses with mean frame-wise displacement as a covariate of non-interest. Mixed-measures ANCOVAs of dlPFC ROI activity exhibited a significant group × cue × time interaction in the A versus B contrast (F1,159 = 4.16, p = 0.043; ηp2 = 0.03; Fig. 2B). Follow-up tests indicated no group differences across the HRF in either the A or B cue, and within-group tests indicated a similar temporal pattern of activation (peak > late peak) across groups following A cues. In contrast, whereas healthy controls (p = 0.003) exhibited a similar pattern following B cues (peak > late peak), patients with PSD (p = 0.87) exhibited statistically equivalent activation across both HRF periods (peak ≈ late peak). We observed a significant group × time interaction for the B versus BX contrast (F1,160 = 5.86, p = 0.017; ηp2 = 0.04; Fig. 2C), with no group differences across both HRF periods (all p > 0.05). We observed time-dependent differences in activity within groups for healthy controls (peak > late peak; p < 0.05) but not PSDgp (peak ≈ late peak; p > 0.10). The probe (AY v. BX) and A versus AY contrasts did not demonstrate group effects or interactions for dlPFC activity.
The vlPFC ROI exhibited a significant group effect (F1,160 = 4.03, p = 0.046; ηp2 = 0.03; Fig. 2D) for the A versus AY contrast; healthy controls showed increased overall activity relative to PSDgp. All other vlPFC contrasts were nonsignificant for group effects or interactions (all p > 0.05).
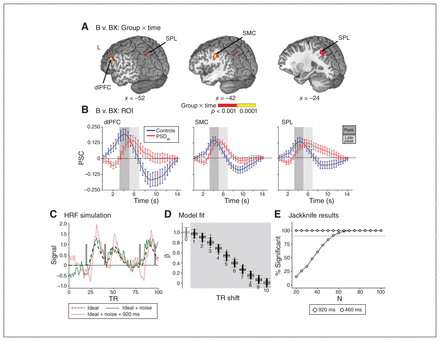
Whole-brain analyses for the B versus BX contrast resulted in significant group × time interactions (Fig. 3A) in the left dlPFC (599 μL; Brodmann area [BA] 9), posterior sensorimotor cortex (SMC), extending into the left inferior parietal lobe (804 μL; BA 2/40) and the posterior portion of the left superior parietal lobe (SPL), extending in the precuneus (602 μL; BA 7/19). The SMC and SPL interactions were characterized by statistically equivalent activation across groups during the peak period, coupled with decreased activation in healthy controls relative to PSDgp in the late peak period (Fig. 3B). In contrast, we found no group differences for the dlPFC. Instead, we observed an inverse pattern of activity in the dlPFC for healthy controls (peak > late peak) relative to patients with PSD (peak < late peak), indicative of a delayed HRF.
(A) Significant group × time interaction for the B versus BX contrast within the left dorsolateral prefrontal cortex (dlPFC), sensorimotor cortex (SMC) and superior parietal lobe (SPL) based on the Talairach atlas (x = sagittal slice location). The significance level of the omnibus test is denoted by colour (red: p < 0.001; yellow: p < 0.0001). (B) Line graphs with standard error bars represent the percent signal change (PSC) from baseline for the average hemodynamic response averaged across B and BX conditions within the dlPFC (left), SMC (centre) and SPL (right) between healthy controls (n = 58; blue line) and good-performing patients with psychotic spectrum disorder (PSDgp; n = 105; red line). Panel background shading designates peak (dark grey) or late peak (light grey) phases of the hemodynamic response. (C) The design matrix for AX probes (black square wave; up: stimulus on; down: stimulus off) was convolved with a double γ variate function to form a simulated ideal HRF (dashed black line). Realistic noise was added to the ideal hemodynamic response function (HRF; green line) based on empirical data. The coral line represents a simulated HRF time shifted by 920 ms (2 TRs) with experimental noise. Importantly, noise was randomly applied for all simulations, so the data presented in panel A are only a representative example. (D) Box-and-scatter plot of decreasing β coefficients corresponding to the ideal HRF regressed upon the 10 time-shifted data averaged over 100 iterations, with each shift corresponding to 460 ms. (E) Percentage of significant (p < 0.001) results from 1000 permutations when comparing shifted versus unshifted data across sample sizes ranging from 20–100. The horizontal dotted line indicates cases where 90% of permutations were significant. Presented results are limited to 460 ms (circle) or 920 ms (diamond) shifts, given that all tests at all sample sizes achieved statistical significance thereafter. L = left; TR = repetition time.
We observed a group × time interaction for the probe (AY v. BX) contrast in the right fusiform gyrus extending into the cerebellum (545 μL; BA 19; Appendix 1, Fig. S2), indicating group differences during the late-peak phase (PSD > healthy controls) but not the peak phase of the HRF. We found no group effects or interactions for cue (A v. B) or A versus AY contrasts during whole-brain analyses.
Activity within all significant ROI and whole-brain clusters were not associated with medication load (olanzapine equivalence), nicotine dependence or extrapyramidal symptoms among patients with PSDgp (all p > 0.05).
Motor variability analyses
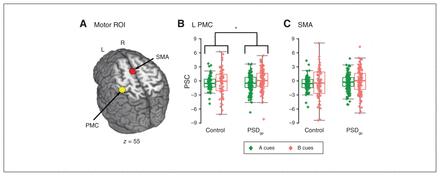
Following our previous study,16 variability analyses focused on activity in the left primary motor cortex and bilateral supplementary motor area during the after-peak phase (6.90–8.30 s) for cue comparisons and pre-peak phase for probe contrasts (1.38–3.22 s post-probe; Fig. 4A). Findings indicated that healthy controls (r = −0.55) exhibited significantly greater post-peak variability for B relative to A cues in the left primary motor cortex (Z = −2.07, p = 0.038; Fig. 4B) compared to patients with PSDgp (r = −0.26), but nonsignificant findings in the supplementary motor area (p = 0.18; Fig. 4C). We also independently replicated previous findings16 of greater post-peak variability following B relative to A cues in the supplementary motor area (r = −0.47, p < 0.001) across both groups and greater pre-peak variability observed for BX relative to AY probes in the left primary motor cortex (r = −0.49, p < 0.001) and supplementary motor area (r = −0.54, p < 0.001).
(A) A priori regions of interest (12 mm sphere) within the supplementary motor area (SMA; x = −2, y= −7, z = 55) and the left primary motor cortex (PMC; x = −39, y= −38, z = 55) based on the Talairach atlas (z = axial slice location). Box-and-scatter plots present percent signal change (PSC) from baseline for the average hemodynamic response within (B) the left PMC and (C) SMA across A (green diamonds) and B (coral diamonds) cue conditions separately for healthy controls (n = 58) and good-performing patients with psychotic spectrum disorder ( PSDgp; n = 105). *p ≤ 0.05. L = left; R = right; ROI = region of interest.
Relationships with DSM diagnosis and symptomatology
Finally, ANCOVAs for patients with PSD only indicated that DSM diagnosis and positive and negative symptoms were not associated with behavioural slowing (A, B, AY, BX reaction times; all p > 0.05) or with abnormalities in hemodynamic activity within dlPFC and vlPFC ROIs (p > 0.05). However, negative symptoms demonstrated a positive relationship with magnitude difference across peak and late-peak periods for both the left SPL (F1,99 = 4.45, p = 0.037; ηp2 = 0.04) and dlPFC (F1,99 = 5.41, p = 0.022; ηp2 = 0.05) clusters; increased late-peak relative to peak activity was associated with elevated negative symptoms, but DSM diagnosis was not associated for either cluster (all p > 0.05).
Simulation analyses
Based on current and previous results,11,12 simulations were conducted to examine the effects of a delayed (i.e., time-shifted) hemodynamic response on resultant β coefficients (Appendix 1, Fig. S3C to E). Briefly, canonical HRFs were shifted from 1 to 10 repetition times (TRs; 460 ms), followed by the addition of realistic noise. Simulated data were permutated 100 times and compared using jackknife resampling across sample sizes ranging from 20 to 100 for 1000 iterations. Results indicated that delays of a single TR resulted in significantly reduced β coefficients relative to non-delayed HRF with sample sizes of 30 in both groups at thresholds of p ≤ 0.001 on 34.2% of the iterations. However, a delay of 2 TRs (920 ms) was significant for 100% of the jackknife samples (n = 20–100). Thus, previous studies that did not explicitly model temporal HRF characteristics during cognitive-control tasks may have interpreted a delayed/abnormal HRF response as being indicative of hypoactivation.
Discussion
An unexpected finding from the current study was that approximately 30% of our consecutively recruited PSD sample could not perform the AX-CPT above chance (≥ 56% accuracy), even with adequate training before scanning. The poor-performing subgroup also presented with increased cognitive and everyday functioning deficits, global clinical symptomatology, positive and negative symptoms and greater deficits in proactive control, and they were more medicated than the good-performing PSD group. While the relationship of poor performance on a complex cognitive task with other indices of impairment is expected, these findings collectively question the utility of the AX-CPT to appraise goal maintenance/cognitive control with functional neuroimaging as recommended by CNTRICS.17 We observed overall response slowing and decreased accuracy for PSDgp during proactive and reactive cognitive control relative to controls, with no clear statistical differences based on DSM diagnosis. The results from good performing patients with PSD were therefore more consistent with generalized cognitive control deficits,23,45 rather than with a specific deficit in proactive control.12,18 In contrast, supplemental analyses indicated that a more specific deficit in proactive control was observed when all patients with PSD were included in behavioural analyses.3,12,21
Previous AX-CPT imaging studies implemented lower performance thresholds (e.g., > 10% accuracy on AX/AY/BX or > 50% accuracy on BY;18 > 44% accuracy on AX and > 50% accuracy on BY19) or did not report performance criteria to differentiate good versus poor performers.11,12 While lower-than-chance cutoffs can be used when focusing on behavioural impairments, neural activity cannot be appropriately modelled using such low thresholds for several reasons. First, error trials have different neural signatures and associated variance relative to correct trials.38 Second, the power of discriminating specific from overall cognitive deficits in clinical groups is reduced when using below-chance performance on a task,26 leading to inherently biased samples that could hinder reverse inference of the investigated region (i.e., is the brain region really involved in the task),27 and to unbiased differences in activity between groups. Therefore, although previous AX-CPT studies have used below-chance accuracy rates in schizophrenia18 and PSDs,19 determining the neural basis of these performance deficits for any clinical sample may not be feasible.
Current methodologies (rapid temporal sampling in conjunction with deconvolution) permitted a fine-grained temporal analysis of hemodynamic activity separately for correct cues and probes, with results indicating different types of functional abnormalities during proactive (B cue and BX-probe) relative to reactive (A and AY) control. During proactive control, the HRF was delayed and/or prolonged for patients with PSD in the left dlPFC, SMC and SPL. The dlPFC is commonly implicated in goal maintenance and top–down bias for processing upcoming stimuli,8,11 whereas both the SMC and SPL are involved in interference conditions for both proactive46,47 and reactive13,16 control.
Previous studies in schizophrenia3,11,12,18,20,23,40 and PSDs19 typically report hypoactivity during proactive control conditions. Other studies report abnormal delays in the temporal profile of the HRF in lieu of magnitude differences for patients with PSD relative to healthy controls.28–30 This includes the absence of the post-stimulus undershoot,29 which was also qualitatively evident in the current findings (Fig. 2 and Fig. 3). Subsequent simulations clearly demonstrated how a delayed HRF could manifest as “hypoactivation” in more common functional MRI (i.e., fixed shape) modelling techniques. The delayed and/or prolonged activity in higher-order cognitive control regions could therefore be attributed to increased cognitive demand in PSD,48 or it could be the result of overall behavioural slowing across proactive trials.49 This delineation is important because hypoactivity suggests a cortical processing deficit that hinders performance of the task, whereas a delayed hemodynamic response suggests more typical but slowed cortical reactivity. Similar to a recent study on connectivity profiles,50 these 2 responses would likely involve different therapeutic strategies.
For the reactive control condition, the current study observed minimal vlPFC activity (i.e., true hypoactivation) for patients with PSD relative to healthy controls across the cue-probe (A and AY) period during ROI analyses. Although some studies have observed reduced anterior insula/vlPFC activity in PSD and schizophrenia,3,51 others report preserved activity,11,20,52 with findings seemingly dependent on the type of contrast employed. In the context of the current study, the reduced vlPFC activity most likely reflects differences in the ability of patients with PSD to inhibit a prepotent motor response relative to healthy controls.53 Finally, crus I of the cerebellum demonstrated increased activity in patients with PSD relative to healthy controls across both proactive and reactive probe conditions. This region of the cerebellum has previously been observed to be involved in the “executive task” network54 and has connections with the dlPFC.55 The increased activity across probes could represent impairment in making connections of contextual demands, as well as error response modulation in patients with PSD.54
We have previously demonstrated16 and now replicated increased behavioural variability for BX relative to AY probes, with BX probes also accounting for the majority of variance in the behavioural shift index. Similarly, we observed increased variance for healthy controls relative to patients with PSD in motor circuitry during proactive (B cue and BX-probe) relative to reactive (A and AY) control. Increased neural variability within frontostriatal–motor systems likely reflect individual differences in the use of diverse strategies during proactive control relative to a more homogeneous approach during reactive trials.56 The observed group difference in variability therefore represents increased attentional and varied proactive responding in healthy controls relative to less variable and more reactive responding in patients with PSD during proactive conditions.8
This study implemented RDoC-style recruitment and analyses to investigate the contribution of symptom burden (here positive and negative symptoms) versus DSM diagnoses in cognitive control.24 A previous study by Smucny and colleagues19 demonstrated a linear trend of performance among participants with PSD: participants with schizophrenia and bipolar disorder type I performed worse than healthy controls, but participants with bipolar disorder type I performed better than participants with schizophrenia on behavioural and functional outcomes. However, other work in a similar sample indicates that positive and negative symptom burden explains more variance for both everyday functioning deficits (replicated across cohorts) and cognitive dysfunction (observed for a single cohort) relative to traditional DSM diagnoses.43 Similar results have been observed for disorganized symptoms in schizophrenia3,12 and bipolar disorder type I.19 In the current study, negative symptom load was associated with abnormal activity within the dlPFC and SPL, irrespective of DSM diagnosis and positive symptom burden, indicating the importance of negative symptomatology on performance and compensatory effort in PSDs.50 We observed null findings for functional abnormalities in terms of reactive control for both symptom burden and DSM diagnoses. Therefore, both current and previous results43,44,50 suggest that symptom burden may have greater effects on cognitive and neural performance above and beyond traditional diagnostic categories.
Limitations
The current experiment was limited by several factors. First, functional MRI may be inherently restrictive in truly delineating sustained versus transient activity during proactive/ reactive cognitive control due to the temporally sluggish nature of brain hemodynamics, even with the more rapid sampling scheme used in the current experiment. Second, a primary aim of the study was to implement RDoC-style recruitment and analysis. However, a large number of patients with PSD were subsequently excluded for poor performance (schizophrenia = 34; schizoaffective disorder = 3; bipolar disorder type I = 9), contributing to limited sample size and power, further highlighting issues using the AX-CPT for inclusive hemodynamic modelling of cognitive performance. Finally, future studies will aim to include additional cognitive tests to better ascertain domain ability.
Conclusion
Current results indicate delayed and/or prolonged dlPFC, SMC and SPL activity for a subset of patients with PSD (i.e., who performed the task above chance levels) relative to healthy controls, potentially representing aberrant activation of the dlPFC in terms of subsequent signalling to the SMC and SPL during proactive control. Hemodynamic abnormalities were better explained by negative symptom burden relative to DSM diagnosis. In contrast, decreased vlPFC activation may indicate impairment in the inhibition of prepotent motor responses during reactive control. Current and previous findings57 suggest that selective impairment in proactive control may be limited to lower-functioning PSD, with unique contributions of specific regions of the lPFC to cognitive control strategies.13,14 Previous findings of a “hypoactive” profile may be the result of more restrictive approaches to hemodynamic modelling that fail to assess the temporally delayed aspect of the HRF in schizophrenia and PSDs.28–30
Acknowledgments
This work was supported by the National Institutes of Health (grant number 1RO1MH101512-03 to ARM). The authors thank Diana South and Catherine Smith for their assistance with data collection.
Footnotes
Competing interests: None declared.
Contributors: D. Stephenson, R. Yeo and A. Mayer designed the study. N. Shaff, J. Bustillo, A. Dodd, C. Wertz, F. Hanlon, S. Stromberg, D. Lin, S. Abrams and A. Mayer acquired the data, which D. Stephenson, A. El Shaikh, N. Shaff, A. Dodd, S. Ryman, F. Hanlon, J. Hogeveen, J. Ling, S. Abrams and A. Mayer analyzed. D. Stephenson, A. El Shaikh, N. Shaff, A. Dodd, C. Wertz, S. Stromberg and A. Mayer wrote the article, which all authors reviewed. All authors approved the final version to be published and can certify that no other individuals not listed as authors have made substantial contributions to the paper.
- Received February 27, 2020.
- Revision received April 13, 2020.
- Accepted May 23, 2020.