Abstract
Background: Brain-derived neurotrophic factor (BDNF) influences brain plasticity and feeding behaviour, and it has been linked to anorexia nervosa in numerous studies. Findings in mostly adult patients point to reduced serum BDNF levels in the acute stage of anorexia nervosa and rising levels with weight recovery. However, it is unclear whether this increase leads to normalization or supranormal levels, a difference that is potentially important for the etiology of anorexia nervosa and relapse.
Methods: We measured serum BDNF at admission (n = 149), discharge (n = 130), 1-year follow-up (n = 116) and 2.5-year follow-up (n = 76) in adolescent female patients with anorexia nervosa hospitalized for the first time, and in healthy controls (n = 79). We analyzed associations with body mass index, eating disorder psychopathology and comorbidities.
Results: Serum BDNF was only nominally lower at admission in patients with anorexia nervosa compared to healthy controls, but it increased continuously and reached supranormal levels at 2.5-year follow-up. BDNF was inversely associated with eating disorder psychopathology at discharge and positively associated with previous weight gain at 1-year follow-up.
Limitations: We compensated for attrition and batch effects using statistical measures.
Conclusion: In this largest longitudinal study to date, we found only nonsignificant reductions in BDNF in the acute stage of anorexia nervosa, possibly because of a shorter illness duration in adolescent patients. Supranormal levels of BDNF at 2.5-year follow-up could represent a pre-existing trait or a consequence of the illness. Because of the anorexigenic effect of BDNF, it might play an important predisposing role for relapse and should be explored further in studies that test causality.
Introduction
Patients with anorexia nervosa experience extremely low body weight combined with body image disturbance, drive for thinness, restrictive eating behaviour and often physical hyperactivity.1 Eating disorders are the third most common chronic disease in adolescents, after obesity and asthma.2 Most eating disorders begin in adolescence, and the incidence of anorexia nervosa in adolescent girls is 100 to 200 per 100 000 person-years, higher than that of type 1 diabetes;3 in Europe, the lifetime prevalence anorexia nervosa is 1% to 4%.4 Treatment consists mostly of weight rehabilitation and psychotherapy, but outcomes are not satisfactory and relapse within months after the end of inpatient treatment is a common, unsolved problem. Only one-third to one-half of patients remain weight-restored,5 and of all mental disorders, anorexia nervosa has the highest mortality rate.6 Anorexia nervosa is also a brain-based disease: dramatic brain-volume changes in adolescents, including grey and white matter loss of up to 10%, have been shown in systematic meta-analyses.7 These findings may be driven by astrocyte loss8 and appear to be largely reversible only upon weight rehabilitation. Without achieving weight recovery, brain volumes remain reduced, with potential permanent brain alterations and neuropsychological deficits that manifest in severe and enduring anorexia nervosa.9 Little is known about the underlying pathophysiology and etiology of anorexia nervosa. Specifically, we do not know why these patients lose weight and have such difficulty maintaining a healthy weight.
Brain-derived neurotrophic factor (BDNF) is an important growth factor of the neurotrophin family;10 it has a strong influence on cell proliferation and differentiation, as well as on synaptic plasticity and neuronal survival. In the central nervous system, BDNF is differentially released from neurons in different brain regions (e.g., in the hippocampus and ventral tegmental area) that have been implicated in learning processes and reward11,12 — processes thought to be affected in anorexia nervosa. Furthermore, BDNF neurons in the anterior paraventricular nucleus of the hypothalamus actively inhibit appetite and food intake and enhance physical activity; those in the medial and posterior regions modulate thermogenesis.13 Consequently, there is strong evidence that BDNF interacts with eating behaviour and the regulation of body weight. Cordeira and Rios14 eliminated BDNF postnatally from mouse brains, resulting in hyperactivity and high anxiety levels, symptoms also found in acute anorexia nervosa. However, the authors also reported increased body weight and linear growth (for a recent overview, see Kumar and Singh15). As well, mice with only 1 BDNF allele showed increased food consumption and obesity.16 Central infusion of BDNF in rat brains led to appetite suppression and weight loss in several experiments.14–16 Thus, increased BDNF could be a risk factor for developing anorexia nervosa or inducing anorexia nervosa relapse by reducing appetite and facilitating weight loss.
Brandys and colleagues17 published a meta-analysis on BDNF and anorexia nervosa that included 7 studies and 155 patients. They found lower serum BDNF levels in a population of mostly adult patients with acute anorexia nervosa compared to healthy controls. This finding was similar to observations in other psychiatric disorders, including depression, schizophrenia and bipolar disorder.18–20 Brandys and colleagues17 interpreted this reduction as a marker of disease and possibly of ensuing brain alterations. Interestingly, body mass index (BMI) and measures of eating disorder psychopathology were positively correlated with BDNF in patients with anorexia nervosa.17
However, recent studies have painted a more ambiguous picture. Dmitrzak-Weglarz and colleagues (n = 60),21 Steinhäuser and colleagues (n = 77)22 and Zwipp and colleagues (n = 72)23 found no significant differences between patients with acute anorexia nervosa and healthy controls in a population consisting mostly of adolescents. Eddy and colleagues (n = 72)24 found no significant differences between adults with anorexia nervosa and healthy controls. However, reduced BDNF levels have also been confirmed in 3 studies of adolescents and adults with anorexia nervosa.25–27 Thus, it remains an open question whether BDNF is reduced in acutely ill patients with anorexia nervosa and whether findings in adolescents might differ from those in adults.
Regarding BDNF after weight recovery, 3 of 4 studies that have done cross-sectional comparisons of patients with anorexia nervosa found that patients who had recovered showed significantly higher BDNF levels than those who were acutely ill23,28,29 (but see Steinhäuser and colleagues,22 who found no difference). Five longitudinal studies of short-term weight-recovered patients with anorexia nervosa22,23,27–29 supported these findings, but only with partly trend-level findings and often small sample sizes.
Whether serum BDNF levels normalize in recovered patients with anorexia nervosa (as would be expected) or instead rise to supranormal levels that are significantly higher than that of healthy controls appears less clear. Four studies found nominal increases of 4% to 26% in recovered patients with anorexia nervosa compared to healthy controls, but these increases reached only trend level or were not significant,22,23,28,29 while Tyszkiewicz-Nwafor and colleagues27 found that BDNF levels were still decreased after only partial weight recovery. Thus, there have been indications of an increase in serum BDNF to supranormal levels in recovered patients with anorexia nervosa, but previous studies have lacked the power to provide a definitive answer.
Supranormal BDNF levels in recovered patients with anorexia could point to elevated pre-existing BDNF levels (trait), or they could be a consequence of the illness, the weight gain or the therapeutic process (scar). Because of the anorexigenic effect of BDNF on hunger and appetite, understanding the origins of supranormal BDNF would be of great value for better understanding the pathophysiology of anorexia nervosa and could contribute to relapse prevention.23,29,30
The aim of the present study was to longitudinally and continuously measure serum BDNF in a large, well-characterized sample of adolescent girls with first-onset anorexia nervosa over a 2.5-year follow-up after inpatient treatment and to compare findings with those in healthy controls. Our hypotheses were as follows: BDNF at admission would be reduced in patients with anorexia nervosa compared to healthy controls, it would increase during treatment, and it would rise to supranormal levels compared to healthy controls during follow-up. We also explored associations between BDNF and clinical variables to identify potential interactions.
Methods
Study population
The study sample consisted of 149 female adolescent patients (aged 11 to 18 years) with first-onset anorexia nervosa and 79 age-matched healthy controls. Patients were recruited between 2007 and 2010 within the context of a multicentre study (Anorexia Nervosa Day Patient versus Inpatient Study [ANDI]) at 6 German sites,31 and, of the total 178 ANDI patients, all 149 with available blood serum samples were included. The ANDI patients with anorexia nervosa were hospitalized with a diagnosis of anorexia nervosa according to the Diagnostic and Statistical Manual of Mental Disorders IV32 and had a BMI below the 10th percentile for age.
Patients showed symptoms of anorexia nervosa for 10.13 ± 8.02 months (mean ± standard deviation) before admission to hospital.
Patients with organic brain disease, psychotic or bipolar disorder, substance dependence or abuse, serious self-injurious behaviour, insufficient knowledge of the German language or an IQ of less than 85 were excluded.
The study had 4 assessment time points. Again, we included only patients for whom serum samples had been obtained: at admission to the hospital (acutely ill patients at admission, n = 149); at discharge from inpatient or 3-week inpatient plus day-patient treatment (both after short-term weight recovery), an average of 4 months after admission (patients at discharge, n = 130); at 1-year follow-up (n = 116); and at 2.5-year follow-up (n = 76).
We recruited female healthy controls via flyers in the Aachen area, age-matched them to the 1-year follow-up group and assessed them at a single time point. Exclusion criteria for the healthy controls were as follows: an IQ less than 85, any lifetime eating disorder or any current other psychological disorders.
Patients, controls and their parents provided written informed consent before participation. The study was implemented in alignment with the Declaration of Helsinki and Good Clinical Practice principles, including independent data management and monitoring. Local ethics committees provided their approval (EK 127/06).
Blood serum collection
Blood was collected in the morning between 0715 h and 0815 h after an overnight fast. In 30 of 79 healthy controls, blood collection was nonfasting and occurred around 1200 h for logistical reasons. After centrifugation and aliquoting, blood serum was stored at −80°C until assessment. Serum BDNF was analyzed via enzyme-linked immunosorbent assay (ELISA) in 2 batches (2012 and 2018) using the Quantikine ELISA (R&D Systems Inc.) at the Institute for Clinical Chemistry and Pathobiochemistry at Otto von Guericke University Magdeburg under the supervision of R.B. The BDNF measurements had intra-assay coefficients of variation of 2.4% to 3.2%, inter-assay coefficients of variation of 4.3% to 7.2% and a lower limit of detection of 1.0 pg/mL. Fasting and nonfasting BDNF values for the healthy controls did not differ (mean ± standard deviation; fasting 13.61 ± 5.93 ng/mL, nonfasting 14.83 ± 7.84 ng/mL; Student t test, p = 0.44), so we used all 79 samples in the analyses.
Because of the long time lapse between the 2 ELISA batch analyses, we compensated for possible batch and storage time effects; we also included age as potential confounder. To do this, we corrected BDNF values by calculating a multiple regression analysis, similar to previous analyses.23,28,33 We entered BDNF values for healthy controls and patients with anorexia nervosa at all 4 time points as dependent variables, and we entered batch, storage time and age as independent variables in a single joint analysis. We labelled the BDNF-standardized residuals from this analysis as “cBDNF” and used this measure for all analyses except the mixed-model analysis, which used the original BDNF results.
BMI-SDS calculation
Because normal BMI differs across development, we calculated BMI standard deviation scores (BMI-SDS) for patients and controls to obtain age-corrected information about weight development in our adolescent sample.34,35 We calculated BMI-SDS for 5 time points: baseline, before the onset of anorexia nervosa (patient recall data at the onset of weight loss), at admission, at discharge, at 1-year follow-up, and at 2.5-year follow-up. Between admission and 2.5-year follow-up, we measured BMI-SDS after an overnight fast; patients were wearing underwear during measurement.
Psychological assessment
To assess mental health and possible comorbidities, patients and healthy controls completed the following questionnaires: the Beck Depression Inventory II,36 the Eating Disorder Inventory 2,37 the Spence Children’s Anxiety Scale38 and the Morgan–Russell Outcome Assessment Schedule.39 The Morgan–Russell Outcome Assessment Schedule contains following subscales: food intake, menstrual pattern, mental state, psychosexual state and socioeconomic state; the maximum possible score is 180, which means no symptoms of illness.
We used the Structured Interview for Anorexia and Bulimia Expert Interview (SIAB-EX) to assess eating disorder symptoms and typical comorbid symptoms.40 The SIAB-EX also includes a number of subscales: body image and slimness ideal, general psychopathology, sexuality and social integration, bulimic symptoms, measures to counteract weight and atypical binges.41 The maximum total score (sexuality and social integration subscales excluded) is 244. We noted any use of antidepressant or antipsychotic medication, because these medications have been shown to increase BDNF.20
Statistical analysis
All analyses were performed using SPSS (version 23; IBM)42 Two-sided p values of less than 0.05 indicated statistical significance.
Group comparisons of cBDNF
First, to analyze whether cBDNF levels were different between patients with anorexia nervosa and healthy controls at the different assessment points, we compared cBDNF values between groups using Student t tests. To exclude any potential effects of selective serotonin reuptake inhibitor (SSRI) use, antipsychotic medication use or anorexia nervosa subtype, we repeated this analysis excluding all patients taking an SSRI, all patients taking olanzapine and all patients of the binge-purge subtype, respectively.
Longitudinal analyses of cBDNF
To analyze longitudinal changes in the anorexia nervosa group only, we calculated a repeated-measures analysis of variance (ANOVA) of cBDNF at admission, discharge, 1-year follow-up and 2.5-year follow-up for patients with complete data at all 4 time points.
To validate the above analyses and use the full patient sample, we also assessed longitudinal changes in the anorexia nervosa group using a linear mixed model calculation with uncorrected BDNF values and independent correction for age, batch effect and storage time (this could not include healthy controls because we had data from only 1 time point for this group). We computed models with different repeated-measures covariance structures and compared them with likelihood ratio tests to find the best model in terms of fit and parsimony. For this procedure, we estimated models once with maximum likelihood and once with restricted maximum likelihood. Both strategies resulted in compound symmetry with a heterogeneous variance–covariance structure. We estimated the final model with restricted maximum likelihood. Our sample size allowed us to detect even small effects of d > 0.14 with 80% power in the longitudinal ANOVA and d > 0.10 in the mixed-model analyses. We repeated both analyses with restrictive-subtype patients only.
Parameters influencing cBDNF
To identify any potential influences of clinical parameters on the longitudinal course of BDNF, we repeated the linear mixed model described above. We included the following at each time point as independent variables, separately for each influencing factor: BMI-SDS, SSRI use, olanzapine use, anorexia nervosa subtype, menstruation status, hormonal contraceptive use and total scores on the Beck Depression Inventory II, Spence Children’s Anxiety Scale, Morgan–Russell Outcome Assessment Schedule, Eating Disorder Inventory 2 and SIAB-EX.
To further analyze which variables could influence cBDNF at each time point, we used backward linear regression analyses, including the following potential influencing factors from the same or previous time points: BMI-SDS, weight loss before admission, illness duration, and total scores on the Beck Depression Inventory II, Morgan–Russell Outcome Assessment Schedule, Spence Children’s Anxiety Scale and SIAB-EX. For a full list of included parameters, please see Appendix 1, available at jpn.ca.
Influence of cBDNF on weight development and eating disorder psychopathology
We also determined whether prior cBDNF measurement would help predict BMI-SDS or SIAB-EX total score at a later time point.
First, we performed 3 backward linear regressions using BMI-SDS as the dependent variable and previous cBDNF measurements, illness duration, current BMI-SDS, previous weight change and questionnaire data from previous assessment points as independent variables.
Then, we calculated whether previous cBDNF measurements would predict SIAB-EX total score at a later time point using the same strategy, using SIAB-EX as the dependent variable and cBDNF, previous BMI-SDS variables and illness duration as independent variables. Because of high intercorrelation, we did not include data from the other questionnaires.
Finally, we tested whether cBDNF could help predict relapse, conceptualized as underweight (BMI less than the 10th percentile or BMI-SDS less than −1.28) at 1-year and 2.5-year follow-up using logistical regression analysis with all previous cBDNF measurements as independent variables. We further compared previous cBDNF values of relapsed patients with those of non-relapsed patients at 1-year and 2.5-year follow-up using Student t tests.
Results
For participant clinical characteristics and BDNF and cBDNF values, see Table 1. The results are reported as mean ± standard deviation.
Participant characteristics
Group comparisons of cBDNF
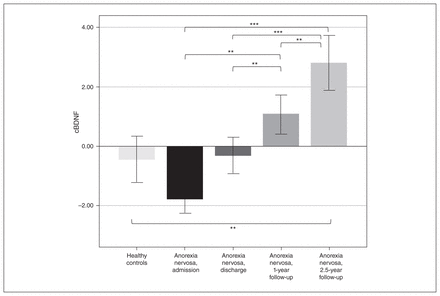
We found that the cBDNF levels of patients with anorexia nervosa at admission were only nominally lower than those of healthy controls (p = 0.15). cBDNF levels did not differ at discharge (p = 0.90) or at 1-year follow-up (p = 0.14), but they were increased significantly at 2.5-year follow-up (p = 0.008; see Figure 1 and Appendix 1, Table S1).
cBDNF levels in healthy controls (n = 79) and in patients with anorexia nervosa at admission (n = 149), at discharge (n = 130), at 1-year follow-up (n = 116), and at 2.5-year follow-up (n = 76). Lower bracket: Student t test of healthy controls versus patients with anorexia at different time points; only healthy controls versus patients with anorexia nervosa at 2.5-year follow-up reached significance. Upper brackets: post hoc tests, repeated-measures analysis of variance in 68 patients with anorexia nervosa using available data at all time points. **p < 0.01; ***p < 0.001. cBDNF = brain-derived neurotrophic factor corrected for age, storage time and batch effect.
Longitudinal changes in cBDNF
cBDNF levels in patients with anorexia nervosa increased continuously from hospital admission until 2.5-year follow-up, as evidenced by repeated-measures ANOVA (all post hoc tests p < 0.05 except for patients at admission versus patients at discharge; see Figure 1 and Appendix 1, Table S2).
Our mixed-model analysis in only patients with anorexia nervosa showed that when we calculated the model using uncorrected BDNF and correcting for age, batch and storage time separately, we found significant time effects of rising BDNF between all 4 time points, except between discharge and 1-year follow-up (see Appendix 1, Figure S1, and Table S3). The exclusion of patients taking an SSRI or olanzapine, or of patients of the binge-purge subtype, did not significantly alter group comparisons or longitudinal results (for those with restrictive anorexia nervosa only, see Appendix 1, Tables S4 to S6).
Parameters influencing cBDNF
We found no significant longitudinal influences on cBDNF when we separately added the following as independent variables to the mixed-model analysis above: BMI-SDS, SSRI use, olanzapine use, menstruation status, hormonal contraceptives or total scores on the Beck Depression Inventory II, Spence Children’s Anxiety Scale, Morgan–Russell Outcome Assessment Schedule, Eating Disorder Inventory 2 or SIAB-EX.
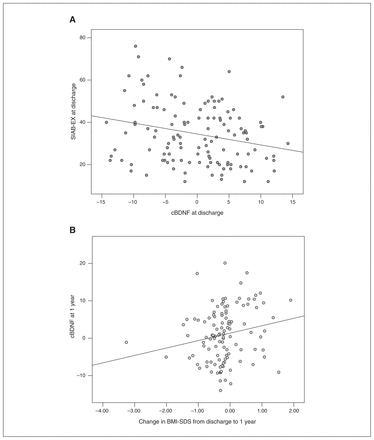
We also conducted linear regression analyses for factors associated with cBDNF at each time point separately. We found that cBDNF at discharge was significantly inversely associated with eating psychopathology (SIAB-EX at discharge; F = 7.201, R2 = 0.071, β = −0.267, p = 0.009), and cBDNF at 1-year follow-up was associated with previous weight gain between discharge and 1-year follow-up (change in BMI-SDS [discharge − 1-year follow-up]; F = 4.058, R2 = 0.038, β = 0.195, p = 0.047; Figure 2).
Bivariate scatterplots of factors that significantly influenced cBDNF in the regression analysis. (A) Negative association between cBDNF and SIAB-EX at discharge. (B) Increased BMI-SDS from discharge to 1-year follow-up predicted cBDNF at 1-year follow-up. Spearman ρ correlation, 2-tailed. *p < 0.05. BMI-SDS = body mass index standard deviation score (age-adapted BMI); cBDNF = brain-derived neurotrophic factor corrected for age, storage time and batch effect; SIAB-EX = Structured Interview for Anorexia and Bulimia Expert Interview.
Our tests of whether cBDNF could help predict BMI-SDS or eating psychopathology (SIAB-EX total score) based on backward linear regressions revealed no significant effects of cBDNF, nor did cBDNF significantly predict relapse in the logistical regression analysis. Interestingly, previous cBDNF values were nominally higher in relapsed patients, but the difference compared to nonrelapsed patients did not reach significance (cBDNF at discharge: −0.33 ± 7.09 [relapse at 1 year, n = 40] versus −1.35 ± 6.87 [no relapse at 1 year, n = 74], p > 0.05; cBDNF at 1 year: 2.07 ± 6.13 [relapse at 2.5 years, n = 21] versus 0.58 ± 7.69 [no relapse at 2.5 years, n = 53], p > 0.05).
Exploratory analysis
We conducted an exploratory Pearson correlation analysis of the remaining time points to check for further associations between cBDNF and either BMI-SDS or SIAB-EX total score, which were significant in the regression analyses above. This analysis revealed additional associations between previous SIAB-EX and cBDNF levels: change in SIAB-EX (discharge – admission) was negatively correlated with cBDNF at discharge (r = −0.281, p = 0.002) and SIAB-EX at 1-year follow-up was negatively correlated with cBDNF at 2.5-year follow-up (r = −0.354, p = 0.002, see Appendix 1, Figure S2).
Discussion
The aim of this study was to observe serum BDNF development in the largest longitudinal sample to date (consisting of 149 female adolescent patients with first-time onset anorexia nervosa) over a period of 2.5 years, and to compare these findings to those for 79 age-matched healthy controls. At admission, cBDNF levels in patients showed only nominally lower values compared to those of healthy controls, and then cBDNF values increased continuously to supranormal levels at 2.5-year follow-up. cBDNF values were inversely associated with eating disorder psychopathology (SIAB-EX total score) at discharge and positively associated with previous weight gain at 1-year follow-up.
Increased cBDNF levels at 2.5 years after first hospitalization is a new and important finding. Because BDNF has a known anorexigenic effect on appetite and satiety — in addition to potentially positive effects on neuronal growth and synapse formation — it might make patients more susceptible to weight loss and relapse.
The nominally lower cBDNF values we found at admission point in the same direction as a previous, similarly powered meta-analysis by Brandys and colleagues,17 which found significantly lower serum BDNF values in 155 mostly adult patients with acute anorexia nervosa compared to healthy controls. However, 4 recent studies did not find significant differences in a total of 209 adolescents and 72 adults21–24 (but see divergent results25–27). This raises the possibility that BDNF reduction might not be as pronounced in adolescent patients with anorexia nervosa, who tend to be less chronically ill. We could neither support nor dismiss this explanation, because we found no correlation between cBDNF and duration of illness in our study. However, this finding might have been because of a lack of variability of illness duration in this cohort of first-onset patients and definitely warrants further research. As mentioned above, lower BDNF levels have also been reported in other chronic mental disorders, including schizophrenia, bipolar disorder and depression.18–20 BDNF plays an important role as a growth factor in terms of long-term potentiation and synaptic plasticity, which have been shown to be altered in these diseases.43,44 It could certainly also be true for anorexia nervosa, in which striking brain volume reduction, astrocyte reduction, neuron-associated light chain protein deviations and (moderate) neuropsychological deficits have been well documented.8,9,45,46
Our results showed that BDNF levels increased continuously to supranormal values over 2.5 years of observation in patients with anorexia nervosa. The increase in BDNF over the course of weight rehabilitation and recovery fits well with the pre-existing literature.22,23,27–29 This increase has been interpreted by previous authors as a reconstruction process, indicating repair mechanisms and reversal of damage induced by starvation.17,23 However, this explanation does not appear to capture the whole process, because it does not explain our new finding of a supranormal endpoint at 2.5-year follow-up. Indeed, BMI-SDS and eating disorder psychopathology did not change much, on average, between discharge and 2.5-year follow-up in our study, disputing a continuous process of repair and improvement or a rebound effect. On the other hand, the possibility of truly increased levels of BDNF after recovery has been hinted at by previous studies,22,23,28,29 which found that BDNF values were increased by 4% to 25% above those of controls. However, these results did not reach significance, partially because of limited power (n = 18–62). In the present study, we found significantly increased cBDNF, even in a mixed sample of patients who had recovered and were still battling with disease, making our result relatively stronger.
We were unable to show significant associations between cBDNF levels and BMI-SDS in general or with underweight status at 1-year and 2.5-year follow-up, making a simple relationship between cBDNF and starvation or body weight recovery unlikely. However, predating cBDNF levels at discharge and 1 year were both nominally higher in later relapsed patients compared to nonrelapsed patients, compatible with the role of cBDNF as a risk factor for relapse. Two overlapping effects could have been at play: on one hand, cBDNF could be a (positive) marker of brain repair processes during acute weight restoration; on the other hand, increased cBDNF levels after weight restoration could be a (negative) marker for a higher risk of relapse.
Several confounders could influence serum BDNF over time. For example, the increase could be an effect of time, because BDNF is known to increase with age.47,48 However, we corrected cBDNF for age and included age in the mixed-model analysis, where it was not significant, so an effect of time was not likely. Exercise also has a positive effect on BDNF excretion in patients, healthy controls and animals.49–51 Theoretically, an increase in exercise over time in our previous patients could have influenced BDNF values. Because we did not include objective exercise measures, we could not rule out this effect.
We are aware that BDNF does not pass the blood–brain barrier because of its size,52,53 so peripheral BDNF levels do not directly influence synaptic plasticity during recovery. However, multiple studies have suggested a strong association between BDNF levels in the blood and the brain (for example, see Klein and colleagues54 and Cao and colleagues55); our hypotheses about BDNF in the brain and abnormal eating behaviour are based on these findings.
A possible underlying mechanism for rising BDNF levels could be abnormalities in estrogen-signalling pathways in anorexia. Sohrabji and colleagues56 reported that estrogen replacement in rats led to increasing BDNF expression in several brain regions. Wessels and colleagues57 showed that treatment with estradiol in ovariectomized mice significantly increased BDNF in the mouse uterus, and Cui and colleagues58 showed behavioural disturbances in mice deficient in estrogen-related receptor α. Increasing blood levels of estrogen, provoking an aberrant brain response during puberty, could be involved in anorexia nervosa and explain why mainly young women are affected by anorexia nervosa.59
Another possible underlying mechanism for elevated BDNF levels is linked to leptin, which is strongly reduced in patients with anorexia nervosa.60 Li and colleagues61 suggested that BDNF-expressing neurons were targets of leptin, and that leptin deficiency decreased BDNF gene expression. Milos and colleagues62 treated 3 female patients off-label with metreleptin and observed decreasing hyperactivity and decreasing weight phobia in 2 patients and improved mood in all 3 patients. Finally, antidepressant medication has been shown to increase reduced BDNF values in depressive patients, sometimes up to normalization.20 Because of the high comorbidity of depression and anorexia nervosa, antidepressant use could have played an important role in our cohort, but it was not a significant covariate in the mixed model. We excluded all patients taking antidepressant or antipsychotic medications, repeated the analyses and had similar results, making it unlikely that antidepressants were confounders.23
Interestingly, patients with autism spectrum disorder also show increased levels of BDNF, as shown in 2 large meta-analyses with more than 1200 and 1700 participants, respectively.63,64 Because patients with anorexia nervosa are known to portray more autistic traits,65 a common developmental pathophysiologic mechanism in the brain could be at work. Many patients with autism also tend to be “picky eaters,”66 and high-functioning (but not low-functioning) patients with autism are also known to weigh less and have a higher chance of being underweight,67–69 pointing to a possible eating-related effect of increased BDNF in autism and warranting future investigation.
Because BDNF has pleiotropic functions that differ in varying tissues and states in patients with anorexia nervosa, any overlapping effects of the above-described (positive) initial repair mechanisms, altered effects on brain development and potentially negative endpoint effects on hunger suppression do not exclude each other. Truly increased levels of BDNF could predispose patients to future weight loss because of its hunger-suppressing properties, or even represent a predisposing factor (trait) for anorexia nervosa if increased BDNF is present before the illness.23,29
In our association analysis, we found that lower eating-disorder psychopathology and higher weight gain were associated with higher cBDNF. This fit well with the literature, in which several studies have found BDNF correlations with BMI28,70–72 and illness markers such as Eating Attitude Test subscales,72 Eating Disorder Inventory subscales and the Global Severity Index.28 Nevertheless, we were surprised not to find more associations, given the power of this large longitudinal study. The pleiotropic, partly antagonistic functions of BDNF and the multiple factors influencing them47 could add too much unexplained variation to detect further determining factors.
For this reason, the solid finding of supranormal cBDNF at 2.5-year follow-up must be valued even more highly. Future research is needed to analyze the causal underpinnings (for example, by longer-term animal experiments with follow-up after starvation) and discern the repair and scarring hypotheses. As well, serum BDNF levels in high-risk groups (such as healthy siblings of patients with anorexia nervosa or preadolescent offspring of former patients with anorexia nervosa) could be compared with serum BDNF levels in healthy controls to identify whether increased BDNF might be a genetic trait. If corroborated, increased BDNF in groups at high risk for anorexia nervosa might have the potential to become a clinical marker of predisposition. Interestingly, Mercader and colleagues73 analyzed plasma BDNF and found significantly increased levels in 21 patients with anorexia nervosa compared to their siblings. Unfortunately, they did not analyze BDNF in unrelated healthy controls.
Limitations
A limiting factor of the present study was substantial attrition: the number of participants who provided blood serum samples decreased over time. However, similar losses to follow-up are common in longitudinal research.74 To ensure that we could still use data from all patients longitudinally, we conducted an additional mixed-model analysis to include all available information at each point of measurement. We also compared patients who were lost to follow-up with those who had complete data. Patients who were lost to follow-up were slightly older and had a longer illness duration, a lower BMI-SDS and Morgan–Russell Outcome Assessment Schedule score, but slightly higher BDNF and cBDNF values at admission (Appendix 1, Table S7). However, losing patients with higher initial BDNF values would be expected to lower the group mean, strengthening our finding of a continuous increase and elevated cBDNF levels at 2.5-year follow-up in the remaining participants. For logistical reasons, we had to analyze blood serum samples in 2 different ELISA runs at different time points, and this introduced a risk of batch and storage time effects. Indeed, we found a significant batch effect, but no effect of additional storage time; however, we corrected all calculations for this confounding factor. Finally, we evaluated healthy controls at only 1 time point, so they could not be included in the mixed model. However, we were able to compare each time point separately after correcting for age.
Conclusion
To our knowledge, this is the largest and longest longitudinal study of BDNF development in adolescent first-time patients with anorexia nervosa to date. It answers the important question of whether BDNF normalizes or increases above values in healthy controls over time. We showed that cBDNF levels rose continuously in patients with anorexia nervosa over treatment and long-term follow-up, leading to significantly elevated cBDNF compared to healthy controls after 2.5 years. Furthermore, we found inverse associations between cBDNF and eating disorder psychopathology and positive correlations between cBDNF and previous weight gain. Supranormal BDNF 2.5 years after first-onset anorexia nervosa may have come from a reinstated pre-existing elevation (state) or the long-term consequences of the disease or rehabilitation period (scar). The anorectic effect of BDNF on hunger and food consumption could contribute to pathophysiological models of the initial development of eating disorders and to the prevention of relapse. Further research should explore the underlying mechanisms and causality of our observations.
Acknowledgments
We especially want to thank all patients who participated in the ANDI study, as well as their parents. We also want to acknowledge all staff who took part in caring for these patients and supporting the ANDI study. Funding for the ANDI study and these serum analyses were provided by the German Ministry for Education and Research (grants 01GV0602 and 01GV0623).
Footnotes
Competing interests: K. Egberts reports membership in the ANDI-Konsortium. N. Timmesfeld reports grants from Bundes-ministerium für Bildung und Forschung during the conduct of the study. No other competing interests declared.
Contributors: B. Borsdorf, K. Konrad, N. Timmesfeld, B. Herpertz-Dahlmann and J. Seitz designed the study. B. Dahmen, K. Buehren, K. Egberts, S. Ehrlich, C. Fleischhaker, R. Schwarte and C. Wewetzer acquired the data, which B. Borsdorf, A. Dempfle, S. Ehrlich, R. Biemann, W. Scharke, B. Herpertz-Dahlmann and J. Seitz analyzed. B. Borsdorf wrote the article, which all authors reviewed. All authors approved the final version to be published and can certify that no other individuals not listed as authors have made substantial contributions to the paper.
- Received March 26, 2021.
- Revision received June 14, 2021.
- Accepted July 1, 2021.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/