Abstract
Background: Risk-taking behaviours are observed among adults with attention-deficit/hyperactivity disorder (ADHD). We sought to evaluate altered neural processing of stimuli values associated with risk-taking decision behaviour, distinct from learning requirements, among adults with ADHD.
Methods: Overall, 32 adults with ADHD and 32 healthy controls without ADHD underwent a lottery choice task in a functional magnetic resonance imaging (fMRI) experiment. Participants accepted or rejected stakes with explicit information about variable probabilities of winning or losing points at different magnitudes. Outcomes were independent across trials, circumventing reward learning. Data analysis explored group differences in neurobehavioural responses to stimuli values during choice decision-making processing and outcome feedback.
Results: Compared with healthy controls, adults with ADHD had slower response times and tended to accept more stakes with a middle-to-low probability of winning. Adults with ADHD had evidence of lower dorsolateral prefrontal cortex (DLPFC) activity and reduced sensitivity in the ventromedial prefrontal cortex (VMPFC) region of interest in response to linear changes in probability, compared with healthy controls. Lower DLPFC responses were associated with lower VMPFC probability sensitivity and greater risk-taking among healthy controls but not adults with ADHD. Compared with health controls, adults with ADHD showed higher responses to loss outcomes in the putamen and hippocampus.
Limitations: Assessments of real-life decision behaviours are required to further validate the experimental findings.
Conclusions: Our findings explore tonic and phasic neural processing of value-related information that modulates risk-taking behaviours among adults with ADHD. Dysregulated neural computation of the values of behavioural actions and outcomes in the frontostriatal circuits may underlie decision processing distinct from reward learning differences among adults with ADHD.
Clinical trial registration: NCT02642068
Introduction
Attention-deficit/hyperactivity disorder (ADHD) is a common neurodevelopmental disorder, with a worldwide prevalence of 5% in children and adolescents and 2.5% in adults.1–4 Symptoms of ADHD and functional impairments observed in childhood persist to adulthood.5–8 Notably, adults with ADHD have increased access to a greater range of life activities than children, compounding the propensity for risk-taking, such as unsafe driving and sexual behaviours.9–11 In particular, compared with controls, adults with ADHD have been to shown to have poorer financial situations and more impulsive buying behaviours,12,13 and select more disadvantageous options in value-based decision tasks such as the Game of Dice7 and the Iowa Gambling Task.14 Increased risk-taking behaviour in ADHD has been attributed to compromised reinforcement learning that affects behavioural updating based on feedback or inhibition of impulses.15,16 At present, however, it remains unclear whether risk-taking stems from different anticipatory neurocognitive representations of the value of behavioural actions among adults with ADHD compared with healthy controls, apart from learning or inhibitory difficulties.16,17
Previous studies have reported frontal and ventral striatal hypoactivation among adults with ADHD relative to healthy controls during reward processing in the Iowa Gambling Task,14 monetary incentive delay18 and other probabilistic learning tasks that implicate the dopaminergic reward system.19,20 However, in such tasks, behavioural responses are made to irrelevant targets rather than to stimuli reward values,18 or reward information is not stated and must be learned implicitly from past trial outcomes.14
Thus, these approaches have limited ability to distinguish whether lower frontostriatal activity in adults with ADHD reflects differences in anticipation of reward associated with stimuli,18 stimulus–response learning14 or general attention, compared with controls.21 If the latter 2 cases explain risk-taking in ADHD, then making probabilistic information salient may be sufficient to circumvent learning and attention difficulties and resolve the behaviour.14 However, in the case of differences in reward anticipation, more fundamental differences in the neuropsychological representations of value between adults with ADHD and healthy controls are implicated such that modulations of reinforcement learning or attention alone will be insufficient to alleviate risk-taking.18
Studies on impulsivity and risk-taking in ADHD have pre-dominantly focused on children or adolescents.22 Children with ADHD show delays in brain and cognitive development that may contribute to inhibitory control and learning deficits.18 However, in general, inhibitory control and learning abilities should be sufficiently developed for relatively advanced functioning in the adult brain.23,24 Indeed, maturation of the frontal cortices in adult humans is associated with improved control processes, such as the ability for contextual learning and regulation of emotional reactions and impulses.25 This suggests that neurobehavioural differences in value-based decision-making that persist in adults with ADHD may not be fully attributable to learning and attention difficulties, but may also reflect more substantive differences in neural value representations.26–28
We sought to evaluate risk-taking associated with the differential neural representation of values under minimal learning and attention requirements among adults with ADHD relative to healthy controls. Using a novel lottery choice task (LCT) in a functional magnetic resonance imaging (fMRI) experiment (previously validated in adults without ADHD)29,30 we assessed neurobehavioural responses to stimuli that explicitly and saliently predicted different expected values (EVs) that were independent across trials, among adults with ADHD and controls. Decision behaviours in the LCT to accept or reject stakes with different EVs reflect subjective valuations of immediate stakes with minimal reliance on attention or learning. We previously found differential activity and neural sensitivity to EV variation across medial and lateral frontal, striatal and medial temporal areas that dissociated risk-taking from risk-averse adults without ADHD.29,30 Here, we explored systemic neural response differences to value information in these brain areas that distinguish adults with ADHD from healthy controls, reflecting group differences in the neural representations of value. Overall, we expected greater risk-taking behaviour (specifically, accepting more stakes with low probability of winning) associated with less veridical representations of stimuli values during decision processing in the LCT among adults with ADHD relative to controls. In particular, we expected this behaviour for high-magnitude stakes as low-magnitude stakes may not entail sufficiently sized value to warrant risky behaviour in general. In neural terms, less veridical representations of stimuli values correspond with lower neural sensitivity to stimuli values such that neural responses show less discrimination between different stimuli values in adults with ADHD than controls, particularly in previously implicated regions.30
Methods
Participants
We included adults whose ADHD was clinically diagnosed by an author (S.S.G.) according to diagnostic criteria for ADHD from the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5),31 at the Psychiatry Department, National Taiwan University Hospital (NTUH). One author (S.S.G.) further confirmed diagnoses of ADHD through psychiatric interviews using the Conners’ Adult ADHD Diagnostic Interview.32 We recruited healthy controls adults via advertisements according to the age and sex distributions of the adult ADHD group. They were clinically evaluated by an author (S.S.G.) to confirm absence of any lifetime diagnosis of ADHD. All participants were also interviewed by trained interviewers using the modified adult version of the ADHD supplement33–35 of the Chinese versions of the Kiddie Schedule for Affective Disorder and Schizophrenia, Epidemiological Version (K-SADS-E)36–38 and SADS39,40 for childhood and current diagnoses of ADHD and for other psychiatric disorders, respectively. The exclusion criteria for both ADHD and control groups included any previous systemic medical illness; a history of mood disorders, psychosis, substance use disorder or autism spectrum disorder; current depressive or anxiety symptoms or suicidal ideation; and a full-scale IQ score of less than 80, as assessed by the Wechsler Adult Intelligence Scale, Third Edition (Appendix 1, Method S1, available at www.jpn.ca/lookup/doi/10.1503/jpn.220123/tab-related-content).41
Lottery choice task behavioural paradigm
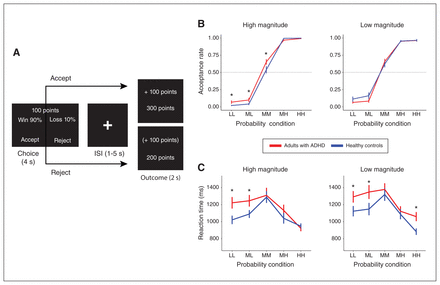
We applied a modified version of the LCT in this fMRI experiment (Figure 1A).29,30,42 In total, 120 trials, each with choice and outcome phases, were distributed equally across 4 scanning runs. During the 4-second choice phase, participants saw text stimuli depicting stakes with varying magnitudes (M) of points (low [ML] 1–12; high [MH] 99–110) and probabilities (P) of winning (low-low [PLL] 4%–15%; middle-low [PML] 24%–35%; middle-middle [PMM] 44%–55%; middle-high [PMH] 64%–75%; high-high [PHH] 84%–95%). The probabilities of winning were simultaneously the reverse probabilities of losing the same stake magnitudes. There were 10 choice conditions, each with 12 trials, that varied EV according to the equation EV = (P × M) + [(1-P) × (−M)]. We also calculated variances (Var = [P × (M-EV)2] + [(1-P) × (−M-EV)2]) of each trial for further analysis. During the 2-second outcome phase, participants saw text stimuli with the outcome points gained (positive), lost (negative) or missed (also positive or negative, but in parentheses) for that trial, as well as the total accumulated points. These outcomes constituted 4 feedback conditions, including acceptance and gain (AG), acceptance and loss (AL), rejection and gain (RG) and rejection and loss (RL) (Appendix 1, Method S2 and Table S3 report trial condition distributions and timings). We computed acceptance rates for 10 choice conditions as the proportion of accepted trials out of all responded trials per condition. We fed acceptance rates and response times of each participant into a repeated-measures analysis of variance (ANOVA) to evaluate the effects of group (ADHD, control), probability (PLL, PML, PMM, PMH, PHH), magnitude (ML, MH) and their interactions (Appendix 1, Method S3).
(A) Sample trial in the lottery choice task. During the Choice phase, participants are shown the percent probability of winning (and, simultaneously, losing) a given magnitude of points (e.g., 100). After participants decide to accept or reject the stake, feedback is then shown during the outcome phase. In this example, outcome if participants accept the stake, they win the outcome shown in the top number (outcome top box), with the accumulated points updated in the bottom number of this box. If participants reject the stake (outcome bottom box), the missed outcome is given in parentheses, with no change in accumulated points. (B) Acceptance rates across the different levels of probability of winning (or losing) and magnitudes of points for adults with attention-deficit/hyperactivity disorder (ADHD) and healthy controls (Appendix 1, Table S3). (C) Mean reaction times across the different levels of probability of winning (or losing) and magnitudes of points for adults with ADHD and healthy controls. *Between-group pair-wise difference p (uncorrected) < 0.05. HH = high-high probability (84%–95%); ISI = Interstimuli interval; LL = low-low probability (4%–15%); MH = middle-high probability (64%–75%); ML = middle-low probability (24%–35%); MM = middle-middle probability (44%–55%).
Brain imaging acquisition and preprocessing
We acquired MRI data using a 3T MRI system (TIM Trio, Siemens) at NTUH with a 32-channel phased-array head coil. For each participant, we acquired 149 functional scans in each run using an echo-planar imaging sequence (34 axial slices parallel to the anterior–posterior commissural plane, voxel size 3 × 3 × 4 mm, field of view 192 × 192 mm, repetition time [TR] 2 s, echo time [TE] 24 ms, flip-angle 90°). A high-resolution T2 image, coplanar to the functional images, was acquired for co-registration (34 axial slices, voxel size 1 × 1 × 3 mm, field of view 256 × 256 mm, TR 5920 ms, TE 101 ms, flip-angle 90°). A T1-weighted, magnetization-prepared, rapid gradient-echo image was acquired for normalization to standard space (208 coronal slices, voxel size 1 × 1 × 1 mm, in-plane matrix size 256 × 192 mm, TR 2 s, TE 2.98 ms, flip angle 9°).
We conducted preprocessing and analysis of functional brain imaging data using SPM12 (Statistical Parametric Mapping, Wellcome Trust Centre for Neuroimaging, UK). We created a study-specific template using T1 images to avoid biases from group differences in brain structure using the diffeomorphic anatomical registration through exponentiated lie algebra (DARTEL) procedure.43 For each participant, we corrected functional images for motion and slice-time displacements, co-registered to the T2 and then the T1 structural images, and spatially normalized to the study-specific template with resampling to 3 × 3 × 3 mm voxels. We then transformed normalized functional images to the standard Montreal Neurological Institute template space and smoothed them with a 3D 8 mm Gaussian kernel.
Whole-brain data analysis
We first used an exploratory, subject-level general linear model for the purpose of detecting brain areas showing differences in general neural response by choice condition and group (ADHD v. healthy controls). This model included regressors for the onsets of the 10 discrete choice phase conditions (PLLML, PMLML, PMMML, PMHML, PHHML, PLLMH, PMLMH, PM-MMH, PMHMH, PHHMH) and 4 outcome phase conditions (AG, AL, RG, RL), convolved with the canonical hemodynamic response function. All subject-level models included 6 motion covariate parameters and were replicated across the 4 functional runs. Group-level, whole-brain, voxel-wise analysis of the choice phase responses applied a 3-way ANOVA with probability and magnitude as within-subjects independent variables, group as the between-subjects independent variable, and drug usage, age and sex as covariates (Appendix 1, Method S1). Because our previous study found group differences in neural responses driven by stimuli probabilities, we also specifically evaluated brain regions showing linear neural responses to increasing probability (PHH > PMH > PMM > PML > PLL), the linear probability × magnitude effect and group differences in these effects.30 We also identified brain regions showing significant direct group contrasts in neural responses to each of the 4 feedback conditions for the outcome phase. To correct multiple comparisons, the significance threshold for whole-brain contrasts was a cluster-level family-wise error rate of less than 0.05. To implement this, for cortical brain areas, we set a primary voxel threshold of p (uncorrected) less than 0.001, which yielded a cluster size of more than 14 using Monte-Carlo simulation with 10 000 iterations.44 For striatal and medial temporal brain areas with a lower MRI signal-to-noise ratio because of ventricular proximity,45,46 primary voxel threshold was set at p (uncorrected) less than 0.005, which yielded a cluster size of more than 18.
In addition, we used another subject-level general linear model to obtain voxel-wise scalar coefficients per participant that summarized neural response trends to trial-wise continuous variations in P and M in the LCT for use in follow-up analyses of regions of interest (ROIs). This subject-level general linear model included 1 regressor for all choice event onsets convolved with the hemodynamic response function; 6 regressors that modulated the choice event regressor by trial-wise linear and quadratic (to model uncertainty; Appendix 1, Method S4) probabilities, magnitudes, their interactions (P × M, P2 × M) and the previous trial cumulative scores; and 4 regressors for the outcome phase condition onsets convolved with the hemodynamic response function.
Functional ROI analysis
We conducted follow-up whole-brain analyses in functional ROIs. We defined functional ROIs as 8-mm spheres around peak contrast voxels showing significant effects of stimuli values or group differences from the exploratory, group-level, whole-brain general linear model. This approach considers ROIs implicated in neural processing during LCT performance across the whole brain, regardless of hemisphere.47 From these functional ROIs, we focused on those in the frontal, striatal and medial temporal areas that replicated our previous findings using the LCT.30 For choice phase data, we examined group differences in neural responses in each ROI to discrete probability levels and magnitudes in adults with ADHD and healthy controls to qualify the whole-brain results. Specifically, we applied regression analyses of how neural responses and neural sensitivities to LCT variables (based on the summary coefficients from the second subject-level general linear model) related across ROIs to characterize neural processing group differences from a network perspective. We also examined how ROI responses for each discrete choice condition were associated with behavioural acceptance rates in the LCT using correlations and regressions with neural responses, group and their interactions as predictors. Finally, we adopted 2-way ANOVA to evaluate group differences in responses for 4 feedback conditions for feedback-related functional ROIs. In addition, we conducted a post hoc analysis to elucidate the main effects and interactions accordingly. We conducted all ROI statistical analyses using R (version 3.4.0), including the lme4 package (version 1.1–13), the lmerTest package (version 2.0–33) and the lmtest package (version 0.9–38). We set all significance thresholds for all ROI analyses were set at p less than 0.05 with the false discovery rate (FDR) correction for multiple comparisons.
Ethics approval
This work was approved by the Research Ethics Committee of the NTUH, Taipei, Taiwan (no. 201401024RINC; ClinicalTrials.gov number NCT02642068), and all participants provided written informed consent before study implementation.
Results
Participants
Thirty-two adults were clinically diagnosed with ADHD, including 18 males and 14 females, with a mean age of 26.9 (standard deviation [SD] 5.9) years. We also recruited 32 healthy control adults with a mean age of 24.6 (SD 4.6) years, including 19 males and 13 females. We excluded data from 7 participants. Of these, we excluded 2 adults with ADHD and 1 control because of excessive head movement (> 3 mm in any direction; about 1 voxel size), and 1 adult with ADHD and 3 controls because of mean residual noise in time-series data (> 2 SDs) owing to technical system faults.
The final sample included 29 adults with ADHD (mean age 27.6 [SD 7.4] yr; 18 males and 11 females) and 28 healthy controls (mean age 24.5 [SD 4.6] yr; 15 males and 13 females). Demographic and clinical details can be found in Appendix 1, Method S1, Table S1 and Table S2.
Behavioural acceptance rates and response times
Acceptance rates across magnitude and probability conditions for ADHD and control groups are shown in Figure 1B, with means and SDs reported in Appendix 1, Table S4. Because we expected possible group differences in risk-taking for low-probability, high-magnitude stakes, we conducted planned pairwise comparisons between adults with ADHD and controls focused on the PMM, PML and PLL conditions at high magnitude. We found that adults with ADHD had higher acceptance rates than healthy controls during the PMMMH (t60 = 1.76, p [uncorrected] = 0.042), PMLMH (t60 = 1.69, p [uncorrected] = 0.049) and PLLMH (t60 = 1.88, p [uncorrected] = 0.034) conditions, spanning middle-to-low probabilities of winning high magnitudes; these were no longer significant after correction for multiple comparisons. Repeated-measures ANOVA showed a significant main effect of probability (F4,620 = 262, p < 0.001) with an interaction between probability and group that only approached significance (F4,620 = 1.98, p = 0.090); no other pair-wise comparisons or ANOVA effects were significant.
Mean responses times and SDs are shown in Figure 1C and are reported in Appendix 1, Table S5. Repeated-measures ANOVAs showed that the mean response times had significant main effects with regard to probability (F4,620 = 19.1, p < 0.001) and group (F1,620 = 14.8, p = 0.001), with no other significant effects (Figure 1C). Simple effects analyses qualified these ANOVA results as owing to slower response times during the PMM condition (mean 1324 [SD 429] ms) than during the PHH (mean 953 [SD 234] ms; PMM v. PHH t197 = 8.59, p FDR < 0.001) and PLL (mean 1165 [SD 361] ms; PMM v. PLL t247 = 3.22, p FDR < 0.001) conditions, and slower response times among adults with ADHD (mean 1203 [SD 400] ms) relative to healthy controls (mean 1095 [SD 349] ms; ADHD v. control t626= 3.65, p FDR < 0.001) (Appendix 1, Result S1, Figure S1).
Neural responses during choice phase
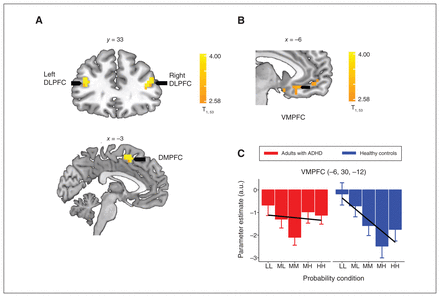
Mean neural responses during the LCT choice phase were significantly higher among healthy controls than adults with ADHD in the bilateral dorsolateral prefrontal cortex (DLPFC), left dorsomedial prefrontal cortex (DMPFC), pre-central gyrus, rolandic operculum, middle frontal gyrus and right inferior parietal lobule (Appendix 1, Table S6). We also observed significant negative-going neural responses across both groups to a linearly increasing win probability in the ventromedial prefrontal cortex (VMPFC), bilateral frontal and supplementary motor areas, and the left putamen, as well as the temporal, postcentral and occipital responses (Appendix 1, Table S7). We found interaction effects between a linear probability increase and magnitude on neural responses across the insula, temporal and parietal areas (Appendix 1, Table S8). Neither brain areas showed higher mean neural responses during the choice phase among adults with ADHD compared with healthy controls, and we did not observe group differences in the linear effect of probability or in the interaction effect between linear probability and magnitude in this exploratory whole-brain analysis.
We then conducted ROI analyses focusing on the bilateral DLPFC, DMPFC and VMPFC areas, identified as functional ROIs (Figure 2A and 2B; Appendix 1, Tables S5 and S6), which replicated and are implicated in value-based processing in our previous findings using the LCT.30 In these ROI analyses, the VMPFC showed a significant group difference in the linear probability response, which was significantly more negative among healthy controls compared with adults with ADHD (control: t247 = −4.84, p FDR < 0.001; ADHD: t256 = −0.56, p FDR = 0.571; control v. ADHD: t503 = −3.03, p FDR = 0.003) (Figure 2B, 2C). Neural responses in the VMPFC increased as the probability of winning decreased (and as the probability of losing increased) in among controls but were not modulated by linear changes in probability among adults with ADHD. No other ROIs had evidence of group differences concerning neural responses associated with probability.
(A) Brain regions in which healthy controls had significantly higher neural activity than adults with attention-deficit/hyperactivity disorder (ADHD). (B) Sagittal view of ventromedial prefrontal cortex (VMPFC) cluster, in which there were negative-going neural responses to a linearly increasing win probability (PHH > PMH > PMM > PML > PLL) across both adults with ADHD and healthy controls during the choice phase. C) Neural response estimates across probability levels, extracted from the VMPFC region of interest (identified from the whole-brain contrasts). Contrasts testing linear (Plinear; solid black lines) effects of increasing probability on neural responses depict significantly more negativegoing neural responses to linearly increasing win probability in controls relative to adults with ADHD. The whole-brain, cluster-wise significance threshold was set at p < 0.05 (adjusted for multiple comparisons using a family-wise error rate). A.u. = arbitrary units; dorsolateral prefrontal cortex = DLPFC; dorsomedial prefrontal cortex = DMPFC; HH = high-high probability (84%–95%); LL = low-low probability (4%–15%); MH = middle-high probability (64%–75%); ML = middle-low probability (24%–35%); MM = middle-middle probability (44%–55%).
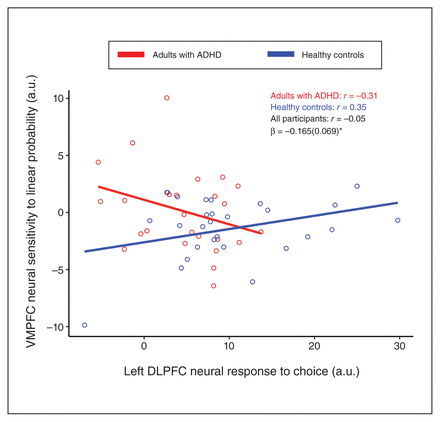
Given the observed ROI findings, we conducted regression analyses that further examined how mean choice phase responses in the individual left and right DLPFC, and in the DMPFC, were associated with each other and with VMPFC linear neural sensitivity to probability in a way that differed between ADHD and control groups. Specifically, for each of the 4 ROI responses, we regressed the effects of the other ROI responses, group, and the group by ROI response interaction, in separate models. This showed a significant interaction effect for group by left DLPFC choice response on neural sensitivity to linear probability in the VMPFC (β [standard error of the mean] = −0.165 [0.069]; t49 = −2.40, p FDR = 0.020) (Figure 3). This interaction was such that healthy controls with lower engagement of the mean left DLPFC during the choice phase showed more negative VMPFC sensitivity to linear probability (r = 0.352, p FDR = 0.036, 1-tailed), but this association was not present among adults with ADHD (r = −0.312, p FDR = 0.939, 1-tailed).
Scatterplots depicting correlations between the mean neural response of the left dorsolateral prefrontal cortex (DLPFC) to choice and the neural sensitivity of the ventromedial prefrontal cortex (VMPFC) to linear probability in adults with attention-deficit/ hyperactivity disorder (ADHD) and healthy controls. Correlation coefficients (r) are shown for adults with ADHD, healthy controls and for both groups together, along with the interaction coefficients of group by left DLPFC response (β, with standard error of the mean in parentheses). A.u. = arbitrary units. *denotes p < 0.05 (adjusted for multiple comparisons using false discovery rate).
Associations between LCT neural responses and behavioural data
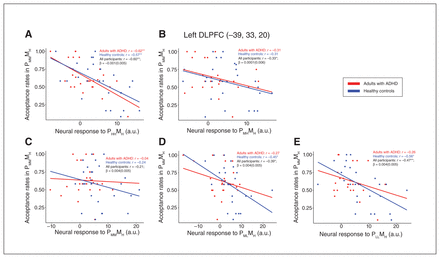
We evaluated the relationships between the ROI neural responses and LCT behaviour. We found that lower acceptance rates for the PMMMH condition (i.e., more conservative decision behaviours) correlated with higher neural responses in the left DLPFC among healthy controls for the PHHMH (r = −0.576, p [FDR] = 0.002), PMLMH (r = −0.449, p FDR = 0.019) and PLLMH (r = −0.557, p FDR = 0.003) conditions (Figure 4). By contrast, among adults with ADHD, this association was only present for the PHHMH condition (r = −0.623, p FDR < 0.001). No other correlations between acceptance rates and ROI neural responses were significant. Regression analyses of the interactive effects between ROI neural responses by group on acceptance rates also did not yield significant results.
Scatterplots depicting correlations between acceptance rates in the PMMMH (middle-middle probability, high magnitude) condition and individual neural responses to (A) PHHMH (high-high probability, high magnitude), (B) PMHMH (middle-high probability, high magnitude), (C) PMMMH (middle-middle probability, high magnitude) (D) PMLMH (middle-low probability, high magnitude) to (E) PLLMH (low-low probability, high magnitude) condition in the left dorsolateral prefrontal cortex (DLPFC) among adults with attention-deficit/hyperactivity disorder (ADHD) and healthy controls. Correlation coefficients (r) are shown for adults with ADHD, healthy controls and for both groups together. A.u. = units. *p < 0.05 (uncorrected), **p < 0.05 (adjusted for multiple comparisons using false discovery rate).
Neural responses during outcome phase
Neural responses to the AL outcome condition were higher among adults with ADHD than healthy controls across several brain areas (Appendix 1, Table S9), particularly in the right putamen (t51 = 2.75, p FDR = 0.008) and left hippocampus (t51 = 3.16, p FDR = 0.003), areas that were implicated in our previous study (Figure 5).29 No group differences were observed for the AG, RG and RL conditions.
(A) The subcortical brain regions in which neural responses to feedback of acceptance and loss (AL) differed between adults with attention-deficit/hyperactivity disorder (ADHD) and healthy controls. The whole-brain, cluster-wise significance threshold was set at p < 0.05 (adjusted for multiple comparisons using family-wise error rate). (B) Neural response estimates across outcome conditions, extracted from the right putamen and left hippocampus regions of interest (identified from the whole-brain statistical volume). *p < 0.05 (adjusted for multiple comparisons using false discovery rate). AG = acceptance and gain; a.u. = arbitrary units; RG = rejection and gain; RL = rejection and loss.
Discussion
Our study characterizes neural correlates underlying risk-taking during value-based decision-making in the LCT among adults with ADHD relative to age-matched healthy controls. We observed differences even though the stakes were salient to participants, and the task did not require inhibitory control or stimulus–response learning. During lottery decisions, adults with ADHD showed lower frontoparietal activity than controls. Moreover, individual differences in DLPFC activity modulated VMPFC sensitivity to probability among controls but not among adults with ADHD, who showed minimal VMPFC sensitivity to differential value information. Individual differences in DLPFC activity also modulated risk-taking behaviour among controls but not adults with ADHD, who showed greater risk-taking trends.
Unlike previous implications of learning or inhibitory difficulties in ADHD,15,16 our findings from this experimental task suggest a differential way by which adults with ADHD and controls anticipate action values.16,17 Specifically, LCT stimuli values were explicit in simple text and participants freely decided based on subjective preferences, with no need to consider past or future outcomes in each trial. Although ADHD core symptoms assessed by the adult ADHD supplement of K-SADS-E were significantly higher among adults with ADHD than controls, minimal differences in IQ or general cognition suggest that LCT processing did not stem from cognitive differences. Moreover, adults with ADHD took more time in making decisions than controls, ruling out impulsive, spontaneous responses. Taking these findings together, we did not find evidence to support that decision processing in our sample of adults with ADHD can be explained by differential learning ability or inhibition from controls.
Further, we found differences in neural responses to prediction error, specifically to unanticipated losses in the putamen and hippocampus among adults with ADHD relative to controls. Performance differences in the LCT stemming from compromised feedback learning in ADHD should be associated with a general hyposensitivity to feedback.16,18,48,49 Instead, neural responses to prediction errors were only different between adults with ADHD and controls for the AL condition, suggesting group differences in predicting value during the choice phase. Lower AL responses among controls may reflect lower neural responses for unanticipated losses.50 By contrast, higher AL responses among adults with ADHD may suggest reduced pre-emptive anticipation underlying the chosen behavioural actions so that there is greater attentional salience instead of surprising negative events.51 Overall, these findings reflect less distinctive neural engagement for predicting differential outcomes associated with actions during value-based decision processing among adults with ADHD.
Critically, differences in LCT decision processing involved differential engagement of the DLPFC and VMPFC during choice processing between the 2 groups. Previous studies report reduced orbitofrontal activity among adults with ADHD relative to controls during outcomes in the Iowa Gambling Task.14 We further show a reduction of dynamic associations between DLPFC tonic activity, VMPFC phasic linear dissociation of stimuli values and decision behaviours among adults with ADHD relative to controls, which reflects suboptimal neural tuning for anticipatory value computations in these neural circuits. Specifically, DLPFC activity during LCT choice phases is tonically lower among adults with ADHD than controls across choice conditions. Among controls but not adults with ADHD, VMPFC activity had evidence of more phasic responses that linearly distinguished trials with an increasing probability of losing. Differential left DLPFC activity levels covaried with VMPFC neural sensitivity to probability and risk-taking behaviours among controls but less so among adults with ADHD. Interestingly, further supplementary analysis indicated that VMPFC sensitivity to the EV (jointly integrating probability and magnitude) during choice processing distinguished decision behaviours of controls from adults with ADHD beyond stimuli EVs, and was associated with overall severity of ADHD symptoms (Appendix 1, Method S5, Result S2, Table S10). We speculate that such nondynamic neural responses to different stimuli values among adults with ADHD indicate difficulty in prioritizing relative utilities across different levels or domains of actions and decisions.52,53 Such neural dysregulations, implicating frontal processing, among adults with ADHD may stem from hypoactive dopaminergic anticipatory signaling of reinforcing cues, as in dopamine transfer deficit theory.15,16,54 In addition, the inefficacy of communication about stimuli salience between the thalamus and frontal areas may also be implicated.55 Future studies are required to validate these considerations by manipulating dopaminergic activity in the DLPFC and VMPFC, or by modulating stimuli salience processing for patients with ADHD and assessing the perceived utility of different actions.
Limitations
We note that our findings of group differences in behavioural acceptance rates were at a trend level. Furthermore, the group interactive effects on neural responses to stimuli values were based on ROI analyses. Differences between adults with ADHD and controls for whole-brain, voxel-wise contrasts were limited to main effects for choice in general and to specific comparisons of feedback conditions. Nevertheless, our planned pairwise comparisons of acceptance rates were in expected directions. Moreover, ROI analyses found a consistently lower neural sensitivity to stimuli values associated with greater risk-taking behaviour in the LCT among patients with ADHD relative to controls. Some of our findings might be limited owing to this clinical study’s relatively small sample size. At the very least, given that adults with ADHD lead relatively functional lives in society, the difference between ADHD and healthy control groups during neural processing of value-based decisions appears minimal but is present in specific regional brain responses. Future studies with larger samples that focus on decisions about low-value stakes may be more efficient at evaluating decision processing among adults with ADHD.
Conclusion
Our study provides insight into the processing of mixed-lottery decisions among adults with ADHD, in which adults must simultaneously weigh the prospect of gain against the possibility of loss, a common situation in life. Among adults with ADHD, we found a trend of accepting more losing stakes that is not easily accounted for by impulsivity or learning difficulty, given our experimental task. Critically, neural findings among adults with ADHD indicated an underlying lack of predictive anticipatory distinction between the values of different behavioural actions. Clinical interventions for this population that target preemptively accentuating the consequences of actions (rather than feedback) during choice tasks are encouraged. These might take the form of behavioural training to elaborate or enhance the salience of cues in decision contexts.
Footnotes
Competing interests: Joshua Oon Soo Goh reports support for presentations from National Cheng Kung University, National Tsinghua University, National Yang Ming University, National Cheng-Chi University, National Central University, National Chung Cheng University and Brandeis University. He is an editor with Neurobiology of Aging and Frontiers in Human Neuroscience. No other competing interests were declared.
Contributors: All of the authors conceptualized the study. Susan Shur-Fen Gau conducted clinical assessments of all the participants and provided clinical and imaging data. Chun-Yi Lee collected the functional magnetic resonance imaging data of the lottery choice task. Chun-Yi Lee and Joshua Oon Soo Goh analyzed the data. Chun-Yi Lee and Joshua Oon Soo Goh drafted the manuscript. All of the authors revised it critically for important intellectual content, gave final approval of the version to be published and agreed to be accountable for all aspects of the work.
Funding: This study was partially supported by the Ministry of Science and Technology, Taiwan (MOST103-2314-B-002-021-MY3, 103-2410-H-002-082-MY2, 105-2420-H-002-MY2, 105-2410-H-002-055-MY3, 107-2410-H-002-124-MY3), the Chen-Yung Foundation, National Health Research Institute, Taiwan (NHRI-EX101-10008PI, NHRI-EX102-10008PI, NHRI-EX103-10008PI) and the National Institutes of Health, United States (R01 AG061886-01).
- Received July 4, 2022.
- Revision received November 2, 2022.
- Revision received November 29, 2022.
- Revision received December 23, 2022.
- Accepted December 24, 2022.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/