Abstract
Background: The nucleus accumbens (NAcc) is a crucial brain region for emotionally relevant behaviours. The NAcc is mainly composed of medium spiny neurons (MSNs) expressing either dopamine receptor D1 (D1-MSNs) or D2 (D2-MSNs). The D1-MSNs project to the ventral tegmental area (VTA) and the ventral pallidum (VP), whereas the D2-MSNs project only to the VP. The D1- and D2-MSNs have been associated with depression-like behaviours, but their contribution to anxiety remains to be determined.
Methods: We used optogenetic tools to selectively manipulate D1-MSN projections from the NAcc core to the VP or VTA and D2-MSN projections to the VP during validated anxiety-producing behavioural procedures in naive mice. In addition, we assessed the effects of optical stimulation on neuronal activity using in vivo electrophysiologic recordings in anesthetized animals.
Results: Optogenetic activation of D1-MSN projections to the VTA or VP did not trigger anxiety-like behaviour. However, optical activation of D2-MSN projections to the VP significantly increased anxiety-like behaviour. This phenotype was associated with a decrease in the neuronal activity of putative GABAergic neurons in the VP. Importantly, pretreating D2-MSN–VP animals with the γ-aminobutyric acid modulator diazepam prevented the optically triggered anxiety-like behaviour.
Limitations: The exclusive use of males in the behavioural tests limits broader interpretation of the findings. Although we used optogenetic conditions that trigger quasi-physiologic changes, there are caveats associated with the artificial manipulation of neuronal activity.
Conclusion: The D2-MSN–VP projections contributed to the development of anxiety-like behaviour, through modulation of GABAergic activity in the VP.
Introduction
Increasing evidence points to the existence of alterations in brain regions of the reward circuit in neuropsychiatric disorders such as depression and anxiety.1 Importantly, both of these disorders are characterized by impaired responses to rewarding and aversive events,2,3 which hints at a central role of the reward circuit in the emergence of symptoms of both depression and anxiety.1 One of the key regions of the reward circuit is the nucleus accumbens (NAcc), which is anatomically positioned to integrate and convey limbic and motor information to drive behaviour in response to emotionally relevant events.4
Accumulating evidence shows the involvement of the NAcc in depression-like behaviours;5–9 however, the role of this brain region in anxiety-like behaviour is less well explored. Nonetheless, recent studies suggest an important role of the NAcc in the development and maintenance of aberrant avoidance behaviours in anxiety disorders. For example, in an avoidance task, the degree of activation and deactivation of the NAcc was associated with individual levels of anxiety.10 A larger bilateral NAcc volume has been observed in adults with generalized anxiety disorder and is associated with higher levels of trait anxiety.11 The NAcc volume is also a predictor of patient response to either cognitive behavioural therapy or pharmacologic treatment.12 In addition, an association between NAcc taurine levels and trait anxiety has been observed.13 Accordingly, in animal models of anxiety, pharmacologic and genetic modulation of NAcc activity has resulted in regulation of anxiety-like behaviour.14–16 The above-mentioned findings are not surprising, considering that the NAcc is innervated by the amygdala,17 a key region for emotional processing. In addition, NAcc outputs to the ventral tegmental area (VTA) are key mediators of depression- and anxiety-like phenotypes, 18–20 with the NAcc–VTA circuit underlying emotional, stress-induced anxiety-like behaviour.21
The NAcc is mainly composed of medium spiny neurons (MSNs) expressing dopamine receptor D1 (D1-MSNs) or D2 (D2-MSNs). The D1-MSNs directly project to the VTA (direct pathway), whereas the ventral pallidum (VP) is innervated by both D1- and D2-MSNs (indirect pathway).22 The VP then projects to the VTA through γ-aminobutyric acid (GABA) projecting neurons.22,23 Although there is some consensus about the differential role of these 2 subpopulations in depression-like behaviours, the contribution of D1- and D2-MSNs in anxiety traits is unknown.
Here, we evaluated how D1-MSN projections to the VTA or the VP, and D2-MSN projections to the VP, contribute to the development of anxiety-like behaviours in mice. We activated these projections optogenetically during validated behavioural procedures to observe whether an anxiety-like phenotype could be attenuated or triggered. We also characterized the electrophysiologic response of the downstream target regions (i.e., the VP and VTA) in response to D1- or D2-MSN optical activation.
Methods
Experimental animals
We used male C57BL/6J transgenic heterozygous D1-Cre (founder line EY262, GENSAT Project) and D2-Cre (founder line ER44, GENSAT Project) mice (2–4 mo of age). The animals were housed in groups of 3–5 and were maintained under standard laboratory conditions: 12-hour light/dark cycle (lights on from 8 am to 8 pm), temperature 22 °C ± 1 °C and relative humidity 60%. A standard diet and water ad libitum were provided, except when stated otherwise. All behavioural experiments were performed between 8:30 am and 2:30 pm.
Health monitoring was performed according to guidelines of the Federation of European Laboratory Animal Science Associations. 24 All animal experiments complied with the Animal Research: Reporting of In Vivo Experiments (ARRIVE) guidelines25 and were conducted in accordance with European Union regulations (directive 2010/63/EU).26 Animal facilities and experimenters were certified by the Portuguese regulatory entity, Direção-Geral da Alimentação e Veterinária (DGAV). All protocols were approved by the Ethics Committee of the Life and Health Sciences Research Institute and by DGAV (protocol 19074).
Genotyping
We isolated DNA from tail biopsy samples using DNA isolation kits (Citomed). In a single polymerase chain reaction genotyping tube, the primers Drd1a F1 (5′-GCTATGGAGATGCTCCTGATGGAA-3′) and CreGS R1 (5′-CGGCAAACGGACAGAAGCATT-3′) were used to amplify the D1-Cre transgene (340 bp), and the primers Drd2 F1 (5′-GTGCGTCAGCATTTGGAGCAA-3′) and CreGS R1 were used to amplify the D2-Cre transgene (700 bp). An internal control gene (for lipocalin, 500 bp) was used in the polymerase chain reaction: LCN1 Forward (5′-GTC CTT CTC ACT TTG ACA GAA GTC AGG-3′) and LCN2 Reverse (5′-CAC ATC TCA TGC TGC TCA GAT AGC CAC-3′). Heterozygous mice were differentiated from wild-type mice by the presence of 2 amplified DNA products corresponding to the internal control gene and the transgene. We visualized the gels with Gel Doc EZ imager (Bio-Rad) and analyzed them with Image Laboratory 4.1 software (Bio-Rad).
Virus injection and optical fibre implantation
Mice were anesthetized with 75 mg/kg ketamine (Imalgene, Merial) plus 1 mg/kg medetomidine (Dorbene, CyMedica) and were then subjected to stereotaxic surgery. A 500-nL volume of a Cre-inducible or control virus (AAV5/EF1a-DIO-hChR2( H134R)-eYFP or AAV5/EF1a-DIO-eYFP, respectively; Gene Therapy Centre, University of North Carolina; vector titres 3.7–6 × 1012 viral molecules/mL) was unilaterally injected into the right hemisphere of the NAcc core (coordinates +1.3 mm anteroposterior [AP], +0.9 mm mediolateral [ML] and −4.0 mm dorsoventral [DV]27) using a 30-gauge needle syringe (Hamilton Company), at a rate of 100 nL/min; the needle was left in place for 5 minutes to allow viral diffusion. An optical fibre with 2.5-mm stainless steel ferrule (200 μm core; Thorlabs) was implanted in the right hemisphere of the VP of both D1- and D2-Cre mice (coordinates −0.1 mm AP, +1.6 mm ML, −3.9 mm DV27) or in the right hemisphere of the VTA of D1-Cre mice (coordinates −3.2 mm AP, +0.5 mm ML, −4.5 mm DV27) and was secured to the skull with dental cement (C&B kit, Sun Medical).
For the experiment with optogenetic stimulation plus intracranial delivery of diazepam, D2-Cre mice were subjected to the same surgery for virus injection as described above, with implantation of an optofluid cannula (Doric Lenses) in the right VP, according to the same coordinates as noted above.
For electrophysiologic recordings, animals were subjected to the same virus injection surgery as described above.
After the surgery, mice were given an anesthetic reversal agent (atipamezole 1 mg/kg, Antisedan, Zoetis) and allowed to recover for 4 weeks. All animals were treated 30 minutes before surgery and 6 hours after surgery with an analgesic (buprenorphine 0.1 mg/kg, Bupaq, Richter Pharma), and analgesia was maintained for 72 hours after surgery.
Optogenetic manipulation
Optogenetic stimulation was performed with 5-mW blue light generated by a 473-nm diode-pumped solid-state laser (CNI Laser) and a pulse generator (Master-8; AMPI), delivered through an optic fibre cable (0.22 numerical aperture, 200-μm diameter; Thorlabs) attached to the implanted ferrule. The stimulation was performed as follows: 25-ms light pulses at 20 Hz, 50% duty cycle. The duration of stimulation varied with the behavioural protocol (3 or 5 min).
Drug
Systemic delivery of diazepam (0.5 mg/kg; Labesfal) or vehicle (VEH; 0.9% saline) was accomplished by intraperitoneal (i.p.) administration 30 minutes before the behavioural tests. We used a dosage of diazepam that was previously shown to not have a significant anxiolytic effect under physiologic conditions.28
Intracranial delivery of diazepam (0.075 ng; Labesfal) was performed immediately before behavioural tests through the implanted optofluid cannula using a 5-μL Hamilton syringe (500 nL delivered over 30 s). The drug dose was based on a previous study.29
Experimental cohorts
For experiments with optogenetic stimulation during the behavioural protocols with OFF–ON–OFF epochs, mice were distributed into different groups according to the transgenic line and/or treatment (where ChR2 refers to channelrhodopsin-2 and YFP refers to enhanced yellow fluorescent protein): D1-VP-ChR2 (n = 16), D1-VP-YFP (n = 7), D1-VTA-ChR2 (n = 15), D1-VTA-YFP (n = 7), D2-VP-ChR2 (n = 19), D2-VP-ChR2 vehicle (n = 10), D2-VP-ChR2 diazepam (n = 10), D2-VP-YFP (n = 13), D2-VP-YFP vehicle (n = 9) and D2-VP-YFP diazepam (n = 10).
For experiments with optogenetic stimulation (Stim) during the entire 5-minute behavioural session, mice were distributed into different groups according to the transgenic line and/or treatment: D1-VP-ChR2 Stim (n = 8), D1-VP-YFP Stim (n = 8), D1-VP-YFP no Stim (n = 8), D2-VPYFP Stim (n = 6), D2-VP-YFP no Stim (n = 5), D2-VP-YFP diazepam systemic (n = 6), D2-VP-YFP diazepam intracranial (n = 7), D2-VP-ChR2 Stim (n = 7), D2-VP-ChR2 diazepam systemic (n = 6) and D2-VP-ChR2 diazepam intracranial (n = 7).
For the in vivo electrophysiologic recordings, we used 28 D1-Cre and 40 D2-Cre mice.
Behavioural assessment
Behavioural tests started 4 weeks after surgery. All behavioural tests were performed and analyzed with blinding of the investigators. Mice were transferred to the testing rooms 30 minutes before the beginning of each test to allow acclimation. We analyzed 2 experimental sets: set 1, in which animals performed the behavioural test with periods with and without optical stimulation; and set 2, in which animals underwent optical stimulation during the whole session of the behavioural test.
Set 1: Animals performed the behavioural tests in the following order: day 1, elevated plus maze (EPM) test; day 4, light–dark box (LDB) test; day 6, open-field test (OFT); day 8, novelty-suppressed feeding (NSF) test, session 1; and day 14, NSF test, session 2.
Set 2: Animals performed the behavioural tests in the following order: day 1, EPM test; day 3, LDB test; day 5, OFT; day 10, NSF test.
Elevated plus maze test
The EPM test was adapted from a previously described protocol. 18,30,31 Briefly, each mouse was attached to the optical fibre cable and placed longitudinally in the central platform of the EPM apparatus (2 open and 2 closed arms 50.8 cm × 10.22 cm × 40.6 cm; Med Associates Inc.), always facing the same corner.
In set 1, the EPM test was performed with a duration of 9 minutes, divided into 3 alternating 3-minute epochs: no laser stimulation, laser stimulation, no laser stimulation (OFF–ON–OFF).
In set 2, the EPM test was performed with optical stimulation delivered throughout the full session (5 min). To control for laser off-target effects, we compared the nonstimulated D2-VP-YFP group with the stimulated D2-VP-YFP group.
All sessions were video-recorded and analyzed with Etho-Vision XT software (Noldus). We assessed time spent and number of entries into open and closed arms. Mice showing absence of exploratory behaviour to the open arms in the first OFF epoch (0% time spent) were excluded from the analysis.
Open-field test
The OFT was adapted from a previously described protocol. 18 Briefly, individual mice were attached to the optical fibre cable and placed in the centre of the arena (43.2 cm × 43.2 cm; Med Associates Inc.), always facing the same wall.
In set 1, the session had a duration of 9 minutes, divided into 3 alternating 3-minute epochs: OFF–ON–OFF.
In set 2, optical stimulation was delivered during the full session (5 min).
We measured distance travelled, number of entries and time spent in each zone using an automated video-tracking system (Activity Monitor, Med Associates Inc.). Mice exhibiting absence of exploratory behaviour in the centre of the open field during the first OFF epoch (0% time spent) were excluded from the analysis.
Light–dark box test
The LDB test was adapted from a previously described protocol.30,32,33 Briefly, mice were attached to the optical fibre cable and placed in the centre of the dark zone of an open field arena (43.2 cm × 43.2 cm, half dark and half light; Med Associates Inc.).
In set 1, each session had a duration of 15 minutes, divided into 3 alternating 5-minute epochs: OFF–ON–OFF.
In set 2, optical stimulation was delivered during the full session (5 min).
We measured distance travelled, number of entries and time spent in each zone using the same automated video-tracking system (Activity Monitor, Med Associates Inc.). Mice exhibiting absence of exploratory behaviour to the light zone of the box in the first OFF epoch (0% time spent) were excluded from the analysis.
Novelty-suppressed feeding test
The NSF test was adapted from a previously described protocol. 18,30,34 The apparatus used for the test was similar to the open field arena (Med Associates Inc.); the floor was covered with clean corncob bedding. One pellet of familiar food (Mucedola 4RF21-GLP) was placed in the centre of the arena. Mice that had been deprived of food for 18 hours were attached to the optical fibre cable and placed in a corner of the arena. Latency to reach the food pellet and latency to begin a feeding episode were recorded manually. The session ended when a mouse first fed or after 10 minutes had elapsed without consumption. Immediately after testing, mice were removed from the arena and were housed individually; free food consumption was measured after 30 minutes.
In set 1, stimulation was performed for the entirety of the ON session. Mice showing absence of exploratory behaviour during the 10-minute session (neither exploration to the food pellet located in the centre of the arena nor consumption of the food pellet) were excluded from the analysis.
In set 2, optical stimulation was delivered during the full session (5 min).
In vivo single-cell electrophysiologic recordings
Mice were anesthetized with urethane (1.75 g/kg; Sigma [now Merck KGaA]) and were placed in a stereotaxic frame (David Kopf Instruments) with nontraumatic ear bars (Stoelting). A tungsten recording electrode (tip impedance 3–5 Ω at 1 kHz) coupled with an optical fibre cable (Thorlabs) was placed in the NAcc, VP or VTA (same coordinates as those used during surgery for virus injection and fibre implantation). A ground screw was placed in the skull.
The activity of single neurons was recorded extracellularly, and then amplified and filtered by a NeuroLog amplifier (NL900D, Digitimer Ltd.; low-pass filter at 500 Hz and high-pass filter at 5 kHz). Neuronal activity was recorded in 3 alternating 3-minute epochs (OFF–ON–OFF; same parameters as those used in behavioural testing). Data sampling was performed using a Micro1401 interface and Spike2 software (Cambridge Electronic Design).
Neuronal instantaneous firing was defined as rate of the ith neuron, as given by ri (ak, bk) = h (ui, ak, bk, w), where h is a histogram function over the vector ui that stores the spiking times of the ith neuron in the population, within the time interval (ak, bk), and w is the bin size for h (w = 1 s). Firing rate peri-stimulus histograms were calculated for the first OFF period (180 s before stimulation), the ON period (180 s of stimulation) and the second OFF period, using a bin size of 1 second. To calculate the peristimulus histogram, each recorded spike train from a single neuron was aligned by the onset of optical stimulation. For each neuronal instantaneous firing rate ri, the average activity during baseline was subtracted: ri = ri − avg (ri [t < 180 s]). Neurons were considered responsive to stimulation if their firing rate changed at least 20% from baseline period activity; 35,36 otherwise, they were considered nonresponsive. The percentage change in activity from the first OFF period was calculated using the average activity from the OFF–ON–OFF epochs.
The NAcc neurons were segregated into putative fast-spiking interneurons (basal firing rate > 10 Hz, waveform half-width < 100 μs), tonically active putative cholinergic interneurons (waveform half-width > 300 μs) and putative MSNs (basal firing rate < 5 Hz and not meeting the waveform criteria for putative cholinergic interneurons or putative fast-spiking interneurons).37–39 Given the very small representation of cholinergic interneurons and fast-spiking interneurons in the recordings, only data for MSNs are shown.
Putative GABAergic projection neurons from the VP were identified as having a baseline firing rate of 13.2 ± 7.3 Hz and action potential of 2.4 ± 0.2 ms.40 Other unidentified neurons were excluded from the analysis.
Neurons of the VTA were segregated into putative dopaminergic (basal firing rate < 10 Hz, waveform duration > 1.5 ms) and putative GABAergic (basal firing rate > 10 Hz, waveform duration < 1.5 ms).41,42 Other unidentified neurons were excluded from the analysis.
Immunofluorescence
Mice were deeply anesthetized and then transcardially perfused with 0.9% saline followed by 4% paraformaldehyde. Brains were removed, postfixed in 4% paraformaldehyde for 24 hours, transferred to 30% sucrose and sectioned coronally at a thickness of 40 μm in a vibrating microtome (VT1000 S, Leica). Sections containing the NAcc, VP or VTA were washed with phosphate-buffered saline, treated with citrate buffer, permeabilized with phosphate-buffered saline containing 0.3% Triton X-100 (PBS-T), blocked with PBS-T plus 10% fetal bovine serum and incubated with the primary antibody goat anti-green fluorescent protein (dilution 1:500; ab6673, Abcam). After washes with PBS-T, sections were incubated with the secondary fluorescent antibody Alexa Fluor 488 donkey anti-goat (dilution 1:500; A-11055, Invitrogen). All sections were stained with 4′,6-diamidino-2-phenylindole (1 mg/mL; Invitrogen) and mounted using PermaFluor (Invitrogen). Images were collected and analyzed by confocal microscopy (FLUOVIEW FV3000, Olympus).
Quantification of viral infection in each area was performed with ImageJ version 1.42 software.43 For the NAcc, VP and VTA, the percentage of area labelled with green fluorescent protein was calculated with the Analyze Particles tool, through percentage of labelled area within a given selection, obtained by defining a pixel intensity threshold for the background.
Statistical analysis
Before doing any statistical comparisons between groups, we tested for the presence of outliers (using a box-and-whisker graph, followed by the Tukey test). We removed any outliers before advancing in the data analysis. We then performed tests of normality (Shapiro–Wilk) and conducted statistical analyses accordingly.
For histology data, we compared the 2 groups using the Student t test; when normality assumptions were not met, we used the Mann–Whitney U test instead.
For behavioural data, we used 2-way analysis of variance (ANOVA; mixed model) for within- and between-group comparisons (OFF v. ON v. OFF; ChR2 v. YFP; interactions). For the analysis of percentage change from the OFF session in the NSF test, we used 1-way ANOVA. When normality assumptions were not met for the 2-way and 1-way ANOVAs, we performed a mixed-effects model with the Geisser–Greenhouse correction or a Kruskal–Wallis test, respectively.
For behavioural data with diazepam treatment, we used 3-way ANOVA for within- and between-group comparisons (OFF v. ON v. OFF; ChR2 v. YFP; diazepam v. vehicle; interactions).
For comparing neuronal populations that responded by increasing, decreasing or not changing activity, we performed 1-way ANOVA with repeated measures. When normality assumptions were not met, we performed the Friedman test, followed by the Dunn multiple-comparison test. For the analysis of electrophysiologic temporal variation, for each time bin, the activity during stimulation was considered significant if, on that time bin, the activity was out 95% of the distribution of the baseline activity. We performed the Kolmogorov–Smirnov test for 2 samples to determine differences in distribution between the stimulus period and baseline. To assess differences in the percentage change in activity during the optical excitation period between the D1-VP-ChR2 and D2-VP-ChR2 groups, we performed a χ2 test.
In all of the ANOVA analyses, a Bonferroni test of post hoc multiple comparisons was applied to determine between-group differences.
Results are presented as mean ± standard error of the mean and were considered statistically significant for p ≤ 0.05. All of the statistical details of experiments and final numbers of animals (excluding outliers and nonperformers) are presented in the Results section and in Appendix 1, Table S1 and Table S2 (available at www.jpn.ca/lookup/doi/10.1503/jpn.220086/tab-related-content); these tables include the statistical tests used and exact p values. Appendix 1, Table S3 shows information about animals that were excluded from the tests.
All statistical analyses were performed using GraphPad (Prism 8.0.2).
Results
To explore whether D1-MSN projections to the VTA or VP, or D2-MSN projections to the VP, could contribute to the development of anxiety-like behaviours, we optogenetically activated (using 20-Hz, 25-ms light pulses) these projections during a battery of validated behavioural procedures, namely the EPM test, the OFT, the LDB test and the NSF test.
These behavioural tests take advantage of the conflict between rodents’ exploratory nature in novel environments and their innate aversion to brightly illuminated or elevated areas. Rodents prefer dark spaces, such as the closed arms of the EPM, the dark side of the LDB or the corners of the OFT arena, but they also like to explore novel contexts. More anxious animals will spend less time in anxiogenic zones such as the open arms or illuminated spaces. To ensure that we could compare performance within the same animal between epochs with (ON) and without (OFF) optical stimulation, we used versions of the behavioural tasks that included alternating OFF–ON–OFF epochs. When this option was not possible because of task design, the same behavioural test was performed twice for each animal (ON and OFF sessions, with the order counterbalanced).
To further confirm the impact of optical stimulation on anxiety-like behaviour, we tested another set of animals in the same behavioural tests, with stimulation throughout the whole session.
Optical activation of D1-MSN projections to the VTA does not alter anxiety-like behaviour
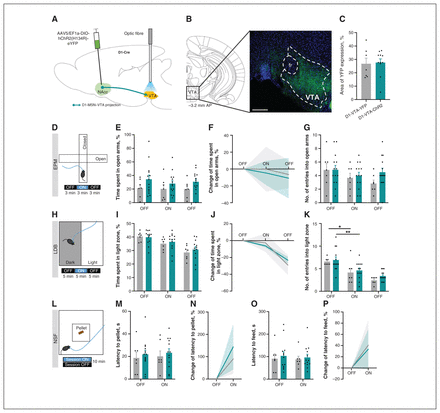
To investigate whether D1-MSN–VTA projections are involved in the induction of anxiety-like behaviour, we unilaterally injected a Cre-dependent construct containing ChR2 fused with enhanced YFP (AAV5-EF1A-DIO-hChR2(H134R)-eYFP) or a control YFP virus into the NAcc of D1-Cre mice (Figure 1A). We placed an optical fibre in the VTA, to allow optical activation of D1-MSN terminals in the VTA during behavioural testing. About 27% of the VTA area presented YFP innervation (D1-MSN terminals) (Figure 1B and 1C; NAcc transfection region in Appendix 1, Figure S9).
Optogenetic stimulation of medium spiny neurons expressing dopamine receptor D1 (D1-MSNs) projecting to the ventral tegmental area (VTA) did not induce anxiety-like behaviour. (A) The D1-MSNs in the nucleus accumbens (NAcc) of D1-Cre mice were injected with Cre-dependent virus expressing either channelrhodopsin-2 (ChR2) or yellow fluorescent protein (YFP). Blue light for optical stimulation was delivered through an optical fibre implanted in the VTA to allow terminal stimulation (473 nm, 20 Hz, 25-ms pulses, 50% duty cycle). AAV5/EF1a-DIO-hChR2(H134R)-eYFP = virus injected into NAcc. (B) Coronal brain slice showing expression of YFP by immunostaining. Numbers represent distance to bregma (mm). AP = anteroposterior; fr = fasciculus retroflexus; scale bar = 200 μm. (C) About 27% of the VTA region presented YFP expression, indicative of D1-MSN terminals. (D) The elevated plus maze (EPM) was divided into 3-minute epochs: OFF–ON–OFF. (E) Optical stimulation of D1-MSN projections to the VTA (D1-MSN–VTA) did not change time spent in the open arms. (F) Change of time spent in open arms of the EPM, expressed as the absolute percentage and normalized to the first OFF epoch (baseline), showed no differences in behaviour. (G) Number of entries into the open arms was similar between groups. (H) The light–dark box (LDB) test was divided into 5-minute epochs: OFF–ON–OFF. No differences were observed in (I) time spent in the light zone of the arena, (J) absolute percent change in time spent in the light zone or (K) number of entries into the light zone. (L) Two sessions of the novelty-suppressed feeding (NSF) test were performed: ON and OFF (counterbalanced); optical stimulation was performed throughout the ON session. The D1-VTA-ChR2 mice showed (M and N) similar latency to reach the food pellet located in the centre of the arena and (O and P) similar latency to feed in the ON and OFF sessions. Grey bars = D1-VTA-YFP; teal bars = D1-VTA-ChR2. Data are presented as mean ± standard error of the mean. *p ≤ 0.05; **p ≤ 0.01.
In the EPM test, the percentage of time spent in, or the number of entries into, the open arms during the ON period was similar between ChR2 and YFP (control) mice (Figure 1D, 1E and 1G and Appendix 1, Figure S1A–D). We also calculated the percent change in time spent in the open arms from the first epoch (baseline). As depicted in Figure 1F, no significant differences were observed between the ChR2 and YFP groups.
In the LDB test, the D1-VTA-ChR2 mice spent a similar amount of time and entered into the light zone at levels similar to those of the D1-VTA-YFP (control) animals (time: F1,12 = 0.2, p = 0.69; number of entries: F1,12 = 1.4, p = 0.25; Figure 1H–K and Appendix 1, Figure S1E–G). Although no differences were observed between the experimental groups, there was a significant decrease in overall exploration of the apparatus over the course of the experiment (time spent in light zone: F1,24 = 46.6, p < 0.001; number of entries into light zone: F2,24 = 38.7, p < 0.001; Figure 1H–K and Appendix 1, Figure S1E–G).
In the NSF test, no differences in latency to reach the pellet or latency to feed were observed due to D1-MSN–VTA stimulation (Figure 1L–P and Appendix 1, Figure S1H).
The D1-VTA-ChR2 mice showed no changes in locomotion as measured by the OFT (Appendix 1, Figure S1I–N).
These data show that acute optical activation of the D1-MSN–VTA projections did not affect anxiety-like behaviours.
Optical activation of D1-MSN projections to the VP does not alter anxiety-like behaviour
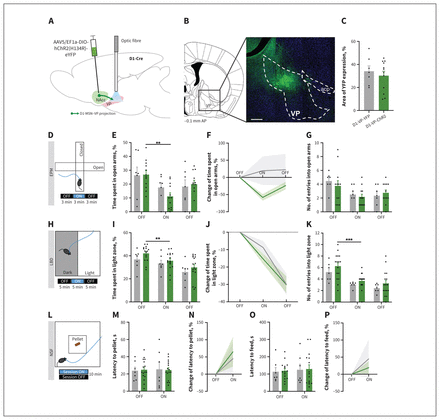
To investigate whether D1-MSN–VP projections are involved in the induction of anxiety-like behaviour, we used a similar approach to that described above for D1-MSN–VTA projections, but we placed the optical fibre in the VP, to allow optical activation of D1-MSN terminals in this region during behavioural testing (Figure 2A). About 30%–34% of the VP area presented YFP innervation (D1-MSN terminals) (Figure 2B and 2C; NAcc transfection region in Appendix 1, Figure S9).
Optogenetic stimulation of medium spiny neurons expressing dopamine receptor D1 (D1-MSNs) projecting to the ventral pallidum (VP) did not have a major effect on anxiety-like behaviour. (A) The D1-MSNs in the nucleus accumbens (NAcc) of D1-Cre mice were injected with Cre-dependent virus expressing either channelrhodopsin-2 (ChR2) or yellow fluorescent protein (YFP). Blue light for optical stimulation was delivered through an optical fibre implanted in the VP to allow terminal stimulation (473 nm, 20 Hz, 25-ms pulses, 50% duty cycle). AAV5/EF1a-DIO-hChR2(H134R)-eYFP = virus injected into NAcc. (B) Coronal brain slice showing expression of YFP by immunostaining. Numbers represent distance to bregma (mm). acp = anterior commissure; AP = anteroposterior; scale bar = 500 μm. (C) About 30%–34% of the VP region presented YFP expression, indicative of D1-MSN terminals. (D) The elevated plus maze (EPM) was divided into 3-minute epochs: OFF–ON–OFF. (E) Optical stimulation of D1-MSN projections to the VP (D1-MSN–VP) induced a slight decrease in the time spent in the open arms. (F) Change of time spent in the open arms, expressed as the absolute percentage, showed no significant differences between groups. (G) Number of entries into the open arms was similar between groups. (H) The light–dark box (LDB) test was divided into 5-minute epochs: OFF–ON–OFF. No differences were observed in (I) time spent in the light zone of the arena, (J) absolute percent change in time spent in the light zone or (K) number of entries into the light zone. (L) Two sessions of the novelty-suppressed feeding (NSF) test were performed: ON and OFF (counterbalanced); optical stimulation was performed throughout the ON session. The D1-VP-ChR2 mice showed (M and N) similar latency to reach the food pellet located in the centre of the arena and (O and P) similar latency to feed in the ON and OFF sessions. Grey bars = D1-VP-YFP; green bars = D1-VP-ChR2. Data are presented as mean ± standard error of the mean. **p ≤ 0.01; ***p ≤ 0.001.
In the EPM test, the percentage of time spent in the open arms in the ON epoch was similar between ChR2 and YFP (control) mice (Figure 2D and 2E and Appendix 1, Figure S2A–C). However, the D1-VP-ChR2 mice spent less time in the open arms of the EPM during the ON than the first OFF epoch (Figure 2D and 2E; F2,22 = 8.8, p = 0.002; OFF v. ON p = 0.002). However, given that we observed an overall trend for a decrease in the percentage of time spent in the open arms in both groups, this may indicate that the decrease observed in ChR2 animals was not due to optical stimulation per se.
We also calculated the percentage change in time spent in the open arms relative to the first OFF epoch. As depicted in Figure 2F, no significant differences were observed between the YFP and ChR2 groups. Similarly, no significant differences were observed in the number of entries into the open arms of the maze (Figure 2G and Appendix 1, Figure S2D).
In the LDB test, the D1-VP-ChR2 mice spent a similar amount of time (F1,13 = 3.1, p = 0.15) and entered into the light zone at similar levels to the D1-VP-YFP (control) animals (F1,15 = 1.4, p = 0.26; Figure 2H–K and Appendix 1, Figure S2E–G). As in the EPM test, the D1-VP-ChR2 mice showed a decrease in exploratory activity in the light zone during the ON period (Figure 2H and 2I; OFF–ON–OFF, F2,30 = 37.8, p < 0.001). However, this decrease was also observed in the YFP (control) animals (Figure 2J).
In the NSF test, no significant differences in the latency to reach the pellet or latency to feed were observed (Figure 2L–P and Appendix 1, Figure S2H).
Importantly, relative to the D1-VP-YFP mice, the D1-VPChR2 mice did not show differences in time spent or distance travelled in the OFT arena (Appendix 1, Figure S2I–N), which suggests that the optical stimulation protocol adopted in this study caused no gross locomotor alterations.
These data show that acute optical activation of the D1-MSN–VP projections did not trigger an evident anxiety-like behaviour.
Optical activation of D2-MSN projections to the VP drives anxiety-like behaviour
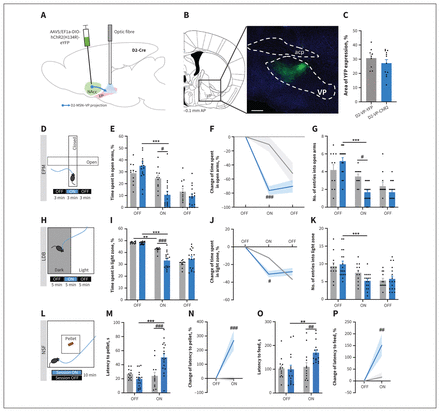
Next, to determine the influence of D2-MSNs projecting to the VP in the induction of anxiety-like behaviour, we unilaterally injected the NAcc of D2-Cre mice with a Cre-dependent construct containing ChR2 fused with YFP or the control (YFP) virus (Figure 3A); an optical fibre was placed in the VP to allow optical activation of the D2-MSN terminals. About 27%–30% of the VP area presented YFP innervation (D2-MSN terminals) (Figure 3B and 3C; NAcc transfection region in Appendix 1, Figure S9).
Optogenetic stimulation of medium spiny neurons expressing dopamine receptor D2 (D2-MSNs) projecting to the ventral pallidum (VP) triggered anxiety-like behaviour. (A) The D2-MSNs in the nucleus accumbens (NAcc) of D2-Cre mice were injected with Cre-dependent virus expressing either channelrhodopsin-2 (ChR2) or yellow fluorescent protein (YFP). Blue light for optical stimulation was delivered through an optical fibre implanted in the VP (473 nm, 20 Hz, 25-ms pulses, 50% duty cycle). AAV5/EF1a-DIO-hChR2(H134R)-eYFP = virus injected into NAcc. (B) Coronal brain slice showing expression of YFP by immunostaining. Numbers represent distance to bregma (mm). acp = anterior commissure; AP = anteroposterior; scale bar = 500 μm. (C) About 30% of the VP region presented YFP expression, indicative of D2-MSN terminals. (D) The elevated plus maze (EPM) was divided into 3-minute epochs: OFF–ON–OFF. (E) Optical stimulation of D2-MSN projections to the VP (D2-MSN–VP) significantly decreased the time spent in the open arms. (F) Change of time spent in the open arms, expressed as the absolute percentage, was reduced in the D2-VP-ChR2 animals. (G) The number of entries into the open arms of the EPM was also lower in the D2-VP-ChR2 animals. (H) The light–dark box (LDB) test was divided into 5-minute epochs: OFF–ON–OFF. (I) Time spent in the light zone of the arena was significantly reduced in the D2-VP-ChR2 animals during the ON period. (J) Change in the percentage of time spent in the light zone, relative to the first OFF epoch, was reduced. (K) The number of entries into the light zone was also reduced. (L) Two sessions of the novelty-suppressed feeding (NSF) test were performed: ON and OFF (counterbalanced); optical stimulation was performed throughout the ON session. The D2-VP-ChR2 mice showed significantly higher latencies to (M and N) reach the food pellet and (O and P) consume the pellet relative to YFP mice and relative to the OFF session. Grey bars = D2-VP-YFP; blue bars = D2-VP-ChR2. Data are presented as mean ± standard error of the mean. Asterisks represent significant results for comparison of ChR2 results between the first OFF session and the ON session; number signs represent significant results for comparison between YFP and ChR2 results during the ON session. #p ≤ 0.05; ** or ##p ≤ 0.01; *** or ###p ≤ 0.001.
Interestingly, optical stimulation of the D2-MSN–VP projections significantly decreased the time spent in the open arms of the EPM, as evidenced by the time spent in the open arms during the ON epoch for D2-VP-ChR2 and D2-VP-YFP mice (F2,36 = 39.1, p < 0.001; ChR2 v. YFP, p = 0.041; Figure 3D and 3E and Appendix 1, Figure S3A–C). The ChR2 animals spent less time in the open arms during the ON than the OFF epoch (F2,28 = 29.3, p < 0.001; ChR2 OFF v. ON, p < 0.001), an effect that did not occur in YFP animals.
The percent change in time spent in the open arms was significantly different, with ChR2 animals spending less time in the open arms relative to the YFP mice (F1,14 = 17.2, p = 0.001; ChR2 v. YFP, p < 0.001; Figure 3F).
In addition, the number of entries into the open arms was lower in the ChR2 group (F1,17 = 1.8, p = 0.20; ChR2 v. YFP, p = 0.05; Figure 3G and Appendix 1, Figure S3D).
These data are supported by the LDB results, given that ChR2 mice also showed decreased percentage of time spent in the light zone during the ON period (F2,32 = 120.3, p < 0.001; ChR2 v. YFP in the ON epoch, p < 0.001; Figure 3H–J and Appendix 1, Figure S3D and S3E). The ChR2 mice spent less time in the light zone of the arena than the YFP mice (F1,16 = 1.3, p = 0.26; ChR2 v. YFP, p = 0.003; Figure 3J and Appendix 1, Figure S3E). The number of entries into the light zone was similar between groups (Figure 3K).
In further support of an anxiogenic role of D2-MSN–VP projections, there was a significant increase in latency to reach the pellet (F1,36 = 10.2, p = 0.003; OFF v. ON, p < 0.001; Figure 3L–M) and to initiate feeding (F1,36 = 5.6, p = 0.024; OFF v. ON, p = 0.002; Figure 3O) during the ON epoch of the NSF session relative to the OFF epoch. In addition, the D2-VP-ChR2 mice had signicantly greater latency than the D2-VP-YFP control group to both reach the pellet and initiate a feeding episode in the centre of the arena (D2-VP-ChR2 v. D2-VP-YFP; latency to pellet: F1,20 = 5.0, p = 0.037; ChR2 v. YFP in the ON epoch, p < 0.001; latency to feed: F1,20 = 4.4, p = 0.05; ChR2 v. YFP in the ON epoch, p = 0.017).
We also normalized the behavioural performance of the ON session to the OFF session and observed that ChR2 mice had an increased percentage of time to reach the pellet in the centre of the arena (U362,103 = 25, p < 0.001; Figure 3N) and to start a feeding episode of the pellet (U274,131 = 25, p = 0.008; Figure 3P), relative to the YFP group. No differences in food consumption after the NSF test were observed (Appendix 1, Figure S3H).
Importantly, optical activation of D2-MSN–VP projections did not induce locomotor alterations in the OFF epoch (Appendix 1, Figure S3I–N).
The behavioural tests described above were longer than the usual tests, to ensure OFF and ON epochs during the same session. However, we observed a decrease in exploratory behaviour throughout the EPM and LDB sessions, which could be a confounding factor for interpretation of the data. Thus, in another set of animals, we performed optical stimulation throughout the whole session of the behavioural test in YFP and ChR2 groups.
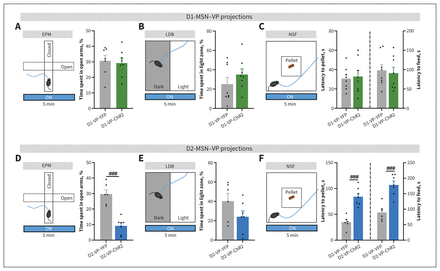
We observed no significant effect of stimulation of D1-MSN–VP projections in the EPM, LDB and NSF behavioural tests (Figure 4A–C and Appendix 1, Figure S4A–D, I–K).
Optogenetic stimulation of medium spiny neurons expressing dopamine receptor D2 (D2-MSNs) projecting to the ventral pallidum (VP), but not medium spiny neurons expressing dopamine receptor D1 (D1-MSNs), during the whole session triggered anxiety-like behaviour. (A–F) A Cre-dependent virus expressing channelrhodopsin-2 (ChR2) or yellow fluorescent protein (YFP) was delivered to the nucleus accumbens of D1- or D2-Cre mice. Blue light for optical stimulation was delivered through an optical fibre implanted in the VP (473 nm, 20 Hz, 25-ms pulses, 50% duty cycle). Each behavioural test consisted of a 5-minute session with the laser ON. Animals stimulated with YFP were used as the control group. Optical excitation of D1-MSN–VP terminals did not trigger an anxiety-like effect in (A) the elevated plus maze (EPM), (B) the light–dark box (LDB) test or (C) the novelty-suppressed food (NSF) test. Conversely, D2-MSN–VP optical stimulation induced (D) a significant decrease in the percentage of time spent in the open arms of the EPM and (F) an increase in the latency to reach the food pellet and initiate a feeding episode in the NSF. (E) In the LDB test, there was no difference between mice subjected to D2-VP-ChR2 and D2-VP-YFP conditions. Data are presented as mean ± standard error of the mean. ###p ≤ 0.001 for comparison between YFP and ChR2 results during the ON session.
Conversely, optical excitation of D2-MSN–VP projections significantly decreased the time that ChR2 mice spent in the open arms of the EPM (t11 = 6.2, p < 0.001; Figure 4D). No significant between-group effect was observed in the LDB (Figure 4E). In the NSF, and in accordance with EPM data, optically stimulated D2-VP-ChR2 mice presented a significant increase in latency to reach the pellet and to start feeding in comparison to optically stimulated D2-VP-YFP mice (latency to pellet: t11 = 5.8, p < 0.001; latency to feed: t11 = 5.2, p < 0.001; Figure 4F and Appendix 1, Figure S4E–H, L–N).
Altogether, these data show that acute optical activation of D2-MSN–VP projections (but not D1-MSN–VP projections) elicits anxiety-like behaviour.
Electrophysiologic correlates of D1-MSN stimulation
To understand the effects of MSN stimulation, we performed extracellular single-cell electrophysiologic recordings in anesthetized D1-Cre or D2-Cre mice previously injected with a Cre-dependent ChR2 or YFP virus in the NAcc (Figure 5A and 5B and Appendix 1, Figure S5A and S6). A recording electrode glued to an optical fibre was descended to the target region: the NAcc for cell somas or the VP or VTA for terminal-evoked activity. We applied the same OFF–ON–OFF epoch stimulation protocol that was used during behavioural testing (3 min OFF; 3 min ON [20 Hz, 25-ms light pulses, 50% duty cycle, 5 mW at tip of fibre]; 3 min OFF).
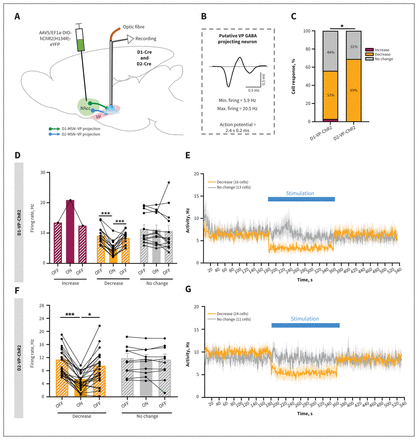
Electrophysiologic responses of the ventral pallidum (VP) to optical stimulation of projections from medium spiny neurons expressing dopamine receptor D1 or D2 (D1-MSN–VP or D2-MSN–VP). (A) Schematic representation of VP electrophysiologic recordings. NAcc = nucleus accumbens; AAV5/EF1a-DIO-hChR2(H134R)-eYFP = virus injected into NAcc. (B) Representative waveform of a VP putative GABAergic neuron. GABA = γ-aminobutyric acid. (C) Bar chart showing the percentage of each type of neuronal response to stimulus. There was a higher percentage of VP putative GABAergic neurons with decreasing activity during stimulation of D2-MSN–VP projections relative to D1-MSN–VP projections. ChR2 = channelrhodopsin-2. (D) Bar graph representing net firing rate before, during and after optogenetic stimulation of D1-MSN–VP projections. (E) Temporal variation of VP activity in response to D1-MSN–VP stimulation. There was a decrease in activity during optical stimulation (blue bar). (F) Bar graph representing net firing rate before, during and after optogenetic stimulation of D2-MSN–VP projections. (G) Temporal variation of VP activity in response to D2-MSN–VP stimulation. There was a decrease in activity during optical stimulation (blue bar). Data are presented as mean ± standard error of the mean. *p ≤ 0.05; ***p ≤ 0.001.
Stimulation of D1-MSNs in the NAcc led to increased neuronal activity in this region, in the range of physiologic levels (Appendix 1, Figure S5A; change in firing rate from about about 2 Hz to about 4.6 Hz during the ON period).
Next, we evaluated the neuronal activity of putative dopaminergic and GABAergic neurons of the downstream target VTA41,42 (Appendix 1, Figure S6B) in response to optical stimulation of D1-MSN–VTA terminals. Stimulation of the D1-MSN–VTA terminals decreased the firing rates for 59% of VTA putative dopaminergic neurons (23/39 neurons; F2,22 = 38.6, p < 0.001; OFF v. ON, p < 0.001) and 33% of putative GABAergic neurons (4/12 neurons; Appendix 1, Figure 6C–G), indicating an overall decrease in activity of the VTA.
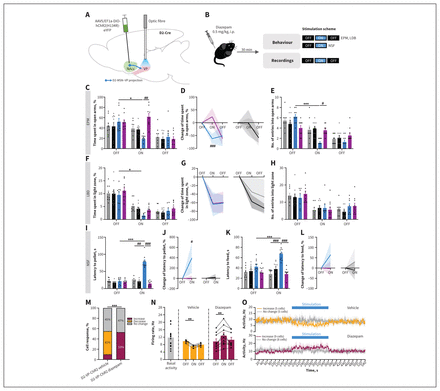
Systemic pretreatment with diazepam prevented the anxiety-like phenotype induced by optogenetic stimulation of projection terminals from medium spiny neurons expressing dopamine receptor D2 into the ventral pallidum (D2-MSN–VP). (A) The D2-Cre mice were transfected with a Cre-dependent virus expressing channelrhodopsin-2 (ChR2) (or yellow fluorescent protein [YFP] in the control group) in the nucleus accumbens (NAcc), and an optical fibre was implanted in the VP to allow D2-MSN terminal stimulation. AAV5/EF1a-DIO-hChR2(H134R)-eYFP = virus injected into NAcc. (B) Four weeks later, mice were injected with diazepam (0.5 mg/kg intraperitoneal [i.p.]) or vehicle 30 minutes before the elevated plus maze (EPM), the light–dark box (LDB) test and the novelty-suppressed feeding (NSF) test. Each behavioural session included laser ON and laser OFF sessions. Behavioural tasks were performed 72 hours apart. (C) Diazepam treatment significantly decreased the time spent in the open arms of the EPM. The D2-VP-ChR2 conditions led to a reduction in (D) the absolute percent change from baseline in time spent in the open arms and (E) number of entries into the open arms of the EPM. (F and G) Similarly, in the LDB test, diazepam treatment reversed the decrease in time spent in the light zone of the arena. (H) The number of entries into the light zone was unaltered. Diazepam treatment also reversed the anxiety-like phenotype of the NSF test, with significant decreases in latency to (I and J) reach the centre of the arena and (K and L) consume the food pellet. In all of the behavioural tests, diazepam treatment did not change basal anxiety states in control mice (subjected to YFP optical stimulation). (M–O) Electrophysiologic recordings following pretreatment with diazepam (or vehicle) were performed in D2-VP-ChR2 animals. Neuronal activity was evaluated during the OFF and ON sessions, similar to the behavioural experiments. (M) Percentage of each type of neuronal response to stimulus in both diazepam- and vehicle-treated mice. The diazepam-treated D2-MSN–VP mice presented a higher percentage of VP putative GABAergic neurons increasing activity during stimulation relative to the vehicle-treated group. (N) Bar graph representing the net firing rate before, during and after optogenetic stimulation of D2-MSN–VP diazepam and vehicle. (O) Temporal variation in VP neuronal activity. Grey bars = D2-VP-YFP vehicle; black bars = D2-VP-YFP diazepam; blue bars = D2-VP-ChR2 vehicle; purple bars = D2-VP-ChR2 diazepam. Data are presented as mean ± standard error of the mean. Asterisks represent significant results for comparisons between the first OFF session and the ON session, and number signs represent significant results for comparison between groups during the ON session: * or #p ≤ 0.05; ** or ##p ≤ 0.01; *** or ###p ≤ 0.001.
Next, we evaluated the neuronal activity of putative GABAergic projecting neurons of the VP40 in response to D1-MSN terminal stimulation (Figure 5A and 5B). Optical stimulation of the D1-MSN–VP projections significantly decreased the average firing rate for 53% of VP neurons (decrease: F2,15 = 35.2, p < 0.001; OFF v. ON, p < 0.001; Figure 5C–E and Appendix 1, Figure S5B and S5C); a small proportion (3%) showed increased activity, and 44% of recorded neurons had no change in activity. The average neuronal activity of VP neurons returned to baseline after the D1-MSN–VTA stimulation period (Figure 5E).
Optical stimulation in YFP mice did not trigger any evident change in activity in the VP or VTA (Appendix 1, Figure S5D, S5H and S5I).
These data show that D1-MSN optical manipulation significantly altered neuronal activity in the NAcc, VP and VTA, although it did not trigger major changes in anxiety-like behaviour.
Electrophysiologic correlates of D2-MSN stimulation
In the behavioural tests, optogenetic stimulation of D2-MSN–VP projections triggered an anxiety-like phenotype. We therefore performed extracellular single-cell electrophysiologic recordings in D2-MSN–VP animals (Figure 5A, 5B, 5F and 5G).
Stimulation of D2-MSNs in the NAcc led to increased neuronal activity in this region, in the range of physiologic levels (Appendix 1, Figure S5D; change in firing rate from about 2 Hz to about 4.6 Hz during the ON epoch).
Optical stimulation of D2-MSN–VP projections significantly decreased the average firing rate for 69% of VP putative GABAergic projecting neurons (24/35 neurons; F2,23 = 7.9, p < 0.001; OFF v. ON, p < 0.001; Figure 5C, 5F and 5G and Appendix 1, Figure S5E and S5F), while 31% of recorded neurons had no change in activity. The average neuronal activity of VP neurons returned to baseline after the D2-MSN–VTA stimulation period (Figure 5F and 5G).
Optical stimulation in YFP mice did not trigger a significant change in VP activity (Appendix 1, Figure S5C and S5F), which confirms that laser stimulation alone did not change overall neuronal activity.
Because both D1- and D2-MSNs project to the VP, we compared the stimulation-evoked responses in this brain region. More than two-thirds (69%, 24/35) of VP cells had reduced activity in response to D2-MSN activation, whereas 53% (16/30) had decreased activity in response to D1-MSN activation. Importantly, the percent change in activity was higher with stimulation in the D2-MSN–VP than in the D1-MSN–VP group (χ2 = 7.4, p = 0.025; Figure 5C). These data suggest that D2-MSN manipulation triggered more evident changes in VP GABAergic activity than did D1-MSN manipulation, which may partly explain the anxiogenic effect observed with D2-MSN manipulation.
Pretreatment with diazepam prevents anxiety-like phenotype induced by D2-MSN–VP stimulation
The preceding results showed an anxiety-like behaviour triggered by D2-MSN–VP stimulation and suggested a GABA-mediated effect of D2-MSN terminal stimulation in the VP, leading to a reduction in net VP activity. Therefore, we treated animals systemically with a low dose of diazepam, a conventional anxiolytic drug that is a positive allosteric modulator of the GABA type A receptors.44 As noted above, the dosage used was previously shown to not have a significant anxiolytic effect under physiologic conditions.28 For this experiment, we injected the NAcc of a new set of D2-Cre mice with ChR2 (or YFP in control group), implanted an optical fibre in the VP to allow optical stimulation, and injected diazepam (0.5 mg/kg) or vehicle i.p. 30 minutes before behavioural testing (Figure 6A and 6B and Appendix 1, Figure S10).
In the EPM test, diazepam had no effect in YFP (control) mice (Figure 6C–E). Conversely, D2-VP-ChR2 mice treated with diazepam spent significantly less time during the ON epoch in the open arms of the EPM than did vehicle-treated mice (F2,68 = 8.5, p < 0.001; ChR2 vehicle v. ChR2 diazepam, p = 0.004; Figure 6C and Appendix 1, Figure S7A–C).
We also calculated the absolute percent change in behaviour, by determining the change in the percentage of time spent in the open arms, relative to the first OFF period. As depicted in Figure 6D, treatment with diazepam had no effect in YFP animals. However, diazepam treatment normalized the behaviour of ChR2 animals (F1,34 = 3.9, p = 0.05; ChR2 vehicle v. ChR2 diazepam, p < 0.001). In agreement with an anxiogenic profile, vehicle-treated ChR2 mice had fewer entries into the open arms of the EPM (F2,68 = 45.1, p < 0.001; OFF v. ON, p < 0.001; Figure 6E and Appendix 1, Figure S7D), an effect that was not observed in YFP animals (OFF v. ON, p = 0.10).
In the LDB test, D2-MSN–VP optical activation caused a decrease in the percentage of time spent in the light zone of the compartment (F2,47 = 122.9, p < 0.001; ChR2 vehicle OFF v. ChR2 vehicle ON, p = 0.017), an effect that was no longer observed after treatment with diazepam (Figure 6F–H and Appendix 1, Figure S7E–G). However, no differences were observed in the percentage of time spent in the light zone of the LDB, when results were normalized to the first OFF epoch (Figure 6G).
In the NSF test, treatment with diazepam prevented the anxiety-like phenotype caused by optical stimulation of D2-MSN–VP projections. Importantly, in the ON session, diazepam-treated D2-VP-ChR2 mice displayed a significant decrease in the latency to reach the pellet in the centre of the arena (F1,33 = 49.1, p < 0.001; ChR2 diazepam v. ChR2 vehicle, p < 0.001; Figure 6I). Importantly, when latency to reach the pellet was normalized to the OFF epoch, vehicle-treated D2-VP-ChR2 mice displayed a significant increase in the percentage change in latency (F3,33 = 14.5, p < 0.001; ChR2 vehicle v. YFP vehicle, p < 0.001; ChR2 vehicle v. ChR2 diazepam, p < 0.001; Figure 6J). Similarly, diazepam-treated ChR2 mice also presented a decreased latency to initiate feeding (F1,33 = 49.7, p < 0.001; ChR2 diazepam v. ChR2 vehicle, p < 0.001; Figure 6K) relative to vehicle-treated D2-VP-ChR2 mice, and comparable to vehicle-treated D2-VP-YPF (control) mice. Nonetheless, no significant differences were found in the normalized percentage of time to initiate a feeding episode (Figure 6L), and no effect of diazepam was observed for any parameters in YFP (control) animals. Food consumption after the NSF test was similar among all groups (Appendix 1, Figure S7H).
Diazepam had no sedative effect in the OFT (Appendix 1, Figure S7I–N).
We further extended these data by analyzing effects of diazepam in animals subjected to optical stimulation during the whole session (Figure 7A–G and Appendix 1, Figure S8). In addition, we assessed the effect of intracranial administration of diazepam (0.075 ng) in D2-VP-ChR2 and D2-VP-YFP animals.
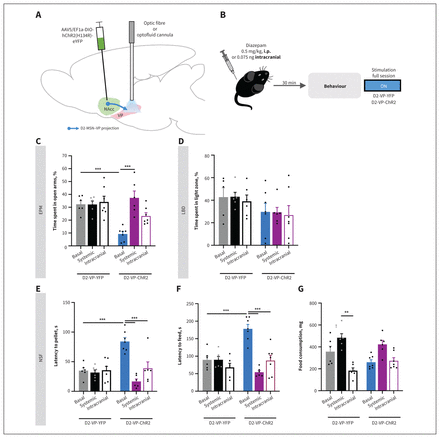
Systemic and intracranial administration of diazepam in the ventral pallidum (VP) blocked the anxiety-like phenotype induced by optogenetic stimulation of projection terminals from medium spiny neurons expressing dopamine receptor D2 into the VP (D2-MSN-VP) during the full session. (A) The D2-Cre mice were transfected with a Cre-dependent virus expressing channelrhodopsin-2 (ChR2) (or yellow fluorescent protein [YFP] in the control group) in the nucleus accumbens (NAcc), and an optical fibre or optofluid cannula was implanted in the VP to allow D2-MSN terminal stimulation or optical stimulation plus drug delivery, respectively. AAV5/EF1a-DIO-hChR2(H134R)-eYFP = virus injected into NAcc. (B) Four weeks after implantation surgery, the mice underwent systemic injection with diazepam (0.5 mg/kg intraperitonal [i.p.]) 30 minutes before or intracranial injection (0.075 ng) immediately before behavioural evaluation with stimulation during the full session. The anxiogenic phenotype caused by D2-MSN-VP optical excitation observed in D2-VP-ChR2 (basal) mice relative to D2-VP-YFP (basal) mice was reversed by both systemic and intra-VP (intracranial) administration of diazepam, measured by (C) percentage of time spent in the open arms of the elevated plus maze (EPM), (D) percentage of time spent in the light arena of the light–dark box (LDB) test, and latency to (E) reach the food pellet and (F) start a feeding episode in the novelty-suppressed feeding (NSF) test. (G) Neither treatment changed consumption of food after the NSF, with the exception of YFP mice treated intracranially with diazepam. Data are presented as mean ± standard error of the mean. **p ≤ 0.01; ***p ≤ 0.001.
In support of our previous data, D2-VP-ChR2 mice spent a significantly lower percentage of time in the open arms of the EPM than did YFP mice (F2,33 = 8.1, p = 0.001; ChR2 basal v. YFP basal, p < 0.001; Figure 7C). This anxiogenic phenotype was blocked by both systemic (ChR2 basal v. ChR2 systemic, p < 0.001) and intracranial (ChR2 basal v. ChR2 intracranial, p < 0.001) administration of diazepam.
In the NSF test (Figure 7E–G), ChR2 basal mice showed significantly higher latency both to reach the pellet (F2,32 = 12.3, p < 0.001; YFP basal v. ChR2 basal, p < 0.001; Figure 7E) and to initiate the first feeding episode (F2,32 = 14.2, p < 0.001; YFP basal v. ChR2 basal, p < 0.001; Figure 7F) relative to non-treated YFP mice. Systemic or intracranial administration of diazepam also reversed the phenotype observed in the NSF test (latency to pellet: ChR2 basal v. ChR2 systemic, p < 0.001; ChR2 basal v. ChR2 intracranial, p < 0.001; latency to feed: ChR2 basal v. ChR2 systemic, p < 0.001; ChR2 basal v. ChR2 intracranial, p < 0.001).
No significant differences were observed in the LDB test (Figure 7D).
These results confirm that systemic or intracranial administration of diazepam significantly reversed the anxiogenic-like phenotype induced by D2-MSN–VP activation.
Pretreatment with diazepam changes the VP response to D2-MSN terminal stimulation
To understand how diazepam could prevent the anxiolytic effect of D2-MSN–VP activation, we performed electrophysiologic recordings in ChR2 and YFP animals treated with vehicle or diazepam (Figure 6M–O). The diazepam was administered at 0.5 mg/kg i.p., and 30 minutes later, VP recordings were obtained with OFF–ON–OFF epochs similar to those used in behavioural testing. During the ON epoch, 45% (5/11) of GABAergic neurons in the VP of vehicle-treated mice showed excitation (Figure 6M–O), similar to what was previously reported in nontreated animals (Figure 5F), and 10% (1/11) showed a decrease in activity. Interestingly, diazepam-treated mice showed a completely different response, with excitation of 53% (9/17) of GABAergic neurons in the VP (χ2 = 74.4, p < 0.001; Figure 6M). Average VP activity returned to baseline after stimulation (Figure 6O).
In control experiments, we found no differences in basal VP activity in terms of the firing rate before and after treatment (Figure 6N). Similarly, diazepam treatment did not trigger any significant changes in VP activity in YFP mice (Appendix 1, Figure S7O–R).
Overall, these results indicate that diazepam treatment prevented the stimulation-triggered changes in VP neuronal activity but had no significant effect in control animals.
Discussion
In this work, we showed that acute optogenetic activation of D2-MSN–VP projections elicited a transient anxiety-like phenotype. Optical activation of D2-MSNs triggered a decrease in VP GABAergic activity. We further showed that modulating GABAergic activity through systemic administration of the anxiolytic drug diazepam prevented the anxiety-like state triggered by stimulation of D2-MSN–VP projections. Manipulation of NAcc D1-MSN–VP or D1-MSN–VTA projections had no observable effects in terms of anxiety-like behaviour.
There is sparse human evidence linking the NAcc to anxiety. Neuroimaging studies have shown that patients experiencing social anxiety disorder have increased activity in the NAcc.45 Also, in an avoidance task, accumbal activation was associated with anxiety behaviour.10 Interestingly, deep brain stimulation of the NAcc has been shown to be effective in reducing anxiety in patients with depression.5 It is well known that animal models present inherent limitations for the study of psychiatric conditions; however, given the existence of mensurable behavioural readouts indicative of anxiety-like phenotypes, these models are valuable for studying the mechanisms underlying anxiety.
Anxiety disorders are classically maintained by maladaptive approach–avoidance conflict behaviours.46 Avoidance can be considered a motivated behaviour in response to stimuli that threaten the individual’s well-being (e.g., fear or pain), whereas approach can be considered a drive motivated by stimuli or events that ensure well-being (e.g., through reward). Importantly, these features can be assessed in rodents through behavioural tests, namely the EPM, LDB and NSF tests,18,32 which rely on the premise that animals with an anxiety-like phenotype will avoid threatening/unsafe open and bright spaces. To date, studies evaluating the contribution of NAcc MSNs to anxiety have been performed in animal models already presenting an anxiety-like phenotype, mostly triggered by exposure to stress. However, in the current work, we modulated NAcc activity in naive animals to observe whether an anxiety-like phenotype could be elicited.
We found that acute D1-MSN–VTA activation did not lead to major changes in anxiety-like behaviour in the EPM, LDB and NSF tests. In addition, D1-MSN–VP input activation did not significantly alter anxiety-like behaviour. In terms of neuronal correlates, a decrease in activity of dopaminergic and GABAergic VTA neurons was triggered by D1-MSN–VTA activation. In a recent study, chemogenetic activation of VTA dopaminergic neurons directly triggered anxiety-like behaviour, whereas chemogenetic inhibition promoted resilience to stress-induced anxiety-like behaviour.21
Regarding projections to the VP, activation of D1-MSN terminals did not robustly alter anxiety-like behaviour. Electrophysiologic correlates showed a decrease in net VP activity in response to D1-MSN–VP stimulation, although it was less evident than in response to stimulation of D2-MSN terminals in this region. These data are in agreement with the fact that VP neurons have higher innervation from D2-MSNs than from D1-MSNs.22
Interestingly, acute D2-MSN–VP input activation triggered an anxiety-like phenotype in the EPM, LDB and NSF tests in animals subjected to the OFF–ON–OFF experiments. These data were recapitulated in animals with optical stimulation during the entire session, with the exception of the LDB test. In addition, D2-MSN–VP activation was associated with a concomitant decrease in VP GABAergic activity, consistent with the inhibitory GABAergic nature of D2-MSNs. Our data align with those of other studies showing that inhibition of GABA neurons in the VP induces place aversion and a sense of threat, as well as increasing defence and avoidance behaviours.23,47–49 Importantly, the decrease in VP GABAergic activity observed here may lead to increased VTA GABAergic activity,50,51 which would suppress VTA dopaminergic neurons and can promote a generalized anxiety-like phenotype.20
Although we stimulated MSN terminals during behavioural testing, we wanted to confirm that the optical stimulation parameters used (which were based on previous work18,32) altered accumbal neuronal activity within the range of what has been observed in vivo under physiologic conditions in animals engaged in learning tasks,52,53 which was indeed the case. In addition, both D1- and D2-MSN terminal activation resulted in significant changes in downstream targets, the VP and VTA, which were previously shown to contribute to rewarding and aversive conditioning.50,54 Importantly, although recordings were obtained in different animals, D2-MSN manipulation triggered more evident changes in neuronal activity in the VP than occurred with D1-MSN activation. This finding is in line with anatomic data showing that the VP has greater innervation with D2-MSNs,22 and can partially explain the anxious phenotype observed in D2-MSN–VP (but not D1-MSN–VP) stimulated animals.
Given that the data pointed to activity changes in GABAergic neurons in the VP, we administered a low dose of diazepam 30 minutes before behavioural assessment. This systemic pretreatment prevented the anxiety-like behaviour caused by optogenetic activation of D2-MSN–VP inputs. Also, because the diazepam was administered systemically, its effects were not restricted to the NAcc–VP circuitry and might have affected other brain regions as well. Yet this circuit appears to be relevant for the observed phenotype, given that intra-VP diazepam administration also prevented the anxiety-like behaviour triggered by D2-MSN–VP stimulation. Importantly, in both experiments, we selected a low dose of diazepam that did not exert an observable effect in control animals,28,29 because we aimed to abolish optically induced GABA-mediated changes in stimulated animals, rather than exerting a robust and nonspecific/general anxiolytic effect.
Interestingly, systemic diazepam administration robustly changed the type of VP-evoked responses. In animals that received vehicle, D2-MSN stimulation led to inhibition of most VP cells (45% inhibited, 10% excited), whereas in diazepam-treated animals, most cells displayed increased activity (53% excited). This shift in VP neuronal activity may explain the absence of anxiety-like behaviour in D2-stimulated diazepam-treated animals. However, the electrophysiologic changes are only correlative with the observed behaviour, given that recordings were obtained from anesthetized animals. In this context, it would be interesting to record the activity of D2-MSN terminals and VP neurons in animals performing the anxiety tests, by means of calcium imaging, for example.
Although the data appear to support a prominent role of GABA in the development of the anxiety-like phenotype, we cannot exclude the contribution of other neurotransmitters. For example, in previous work, we showed that prolonged stimulation of MSNs can lead to endogenous opioid release in downstream regions, namely the VTA and VP.50 It would thus be important to evaluate whether endogenous opioids contribute to the electrophysiologic effects observed in the VP and/or the anxiety-like phenotype observed with D2-MSN stimulation.
Limitations
In this work, we initially adopted a strategy that allowed the assessment of anxiety-like effects of stimulation within the same animal, by applying OFF and ON epochs in the same session. Several others have used the same approach to show that the amygdala, the prefrontal cortex and the VTA can modulate anxiety-like behaviours.18,21,32 However, because most of the tests performed here rely on the drive to explore or avoid unfamiliar arenas, some of the conclusions could be partially compromised by the use of longer test sessions. In fact, we observed an overall decrease in exploratory behaviour throughout most of the tests. Therefore, we performed additional experiments in which we stimulated animals throughout the whole session, thus shortening the duration of each session, as in the original protocols. This confirmed that D2-MSN–VP activation (but not D1-MSN–VP activation) indeed triggered anxiety-like behaviour.
Another caveat is our use of unilateral manipulation, which may lead to partial effects on the behavioural response. It would be interesting to assess the effect of bilateral optogenetic manipulation in anxiety-like behaviour.
Although we used optogenetic stimulation conditions that triggered changes in neuronal activity similar to those observed under physiologic conditions, the findings of this study should be interpreted with caution. In this context, electrophysiologic or calcium imaging recordings in freely behaving animals would further contribute to determining the role of NAcc neurons in anxiety-like behaviours.
The use of male animals for all of the tests also limits a broader interpretation of the findings.
Conclusion
In this study, optical activation of NAcc D2-MSN–VP inputs triggered a transient anxiety-like phenotype that could be prevented by pretreatment with diazepam. Diazepam pre-treatment also shifted the neuronal activity changes in the VP due to D2-MSN terminal activation. These findings highlight the need for additional studies to investigate how distinct subpopulations of NAcc neurons contribute to the development of anxiety-like behaviours.
Footnotes
Competing interests: None declared.
Contributors: A.J. Rodrigues and C. Soares-Cunha designed and supervised the work. R. Correia, B. Coimbra, A.V. Domingues, M. Wezik, N. Vieitas-Gaspar, R. Gaspar and C. Soares-Cunha acquired and analyzed the data. R. Correia, B. Coimbra, A.V. Domingues, N. Sousa, L. Pinto and C. Soares-Cunha interpreted the data. R. Correia, B. Comibra and C. Soares-Cunha wrote the first draft of the manuscript. All authors critically revised the manuscript, provided final approval of the version to be published and agreed to be accountable for the work. A.J. Rodrigues, N. Sousa and C. Soares-Cunha secured the study funding.
Funding: This work was funded by BIAL Foundation grants 30/2016 and 175/2020 and by Portugal’s Foundation for Science and Technology (FCT) under the scope of projects PTDC/MED-NEU/29071/2017, PTDC/SAU-TOX/6802/2020 and PTDC/MED-NEU/4804/2020. Part of the work received funding from “la Caixa” Foundation (ID 100010434), under agreement LCF/PR/HR20/52400020. The project also received funding from the European Research Council under the European Union’s Horizon 2020 research and innovation funding program (grant agreement no. 101003187). Part of the work was also funded by the ICVS Scientific Microscopy Platform, member of the national infrastructure Portuguese Platform of BioImaging (PPBI-POCI-01-0145-FEDER-022122) and by national funds, through the FCT (projects UIDB/50026/2020 and UIDP/50026/2020). Raquel Correira holds an FCT PhD fellowship (2021.06818.BD). Carina Soares-Cunha and Bárbara Coimbra have Scientific Employment Stimulus Contracts from the FCT (CEECIND/03887/2017; CEECIND/03898/2020), and Ana Verónica Dominques has a PhD grant from the FCT (SFRH/BD/147066/2019).
- Received June 27, 2022.
- Revision received December 13, 2022.
- Revision received January 10, 2023.
- Accepted January 11, 2023.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/