Abstract
Background: Depression is a prevalent nonmotor symptom in Parkinson disease and can greatly reduce the quality of life for patients; the dopamine receptors found in glutamatergic pyramidal cells in the medial prefrontal cortex (mPFC) play a role in regulating local field activity, which in turn affects behavioural and mood disorders. Given research showing that glial cell–derived neurotrophic factor (GDNF) may have an antidepressant effect, we sought to evaluate the impact of exogenous GDNF on depression-like behaviour in mouse models of Parkinson disease.
Methods: We used an established subacute model of Parkinson disease in mice involving intraperitoneal injection of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP), followed by brain stereotaxic injection of GDNF into the mPFC region. Subsequently, we assessed depression-like behaviour using the sucrose preference test, forced swimming test and tail suspension test, while also evaluating protein expression in the mPFC.
Results: We included 60 mice, divided into 3 groups, including a control group (saline injection), an MPTP plus saline injection group and an MPTP plus GDNF injection group. We found that exogenous GDNF injection into the mPFC led to an increase in dopamine receptor D1 (DRD1) protein levels. We also observed that activating the protein kinase A pathway through DRD1 produced a prolonged antidepressant response. Under GDNF stimulation, the expression of dopamine receptor D2 (DRD2) remained constant, suggesting that the DRD2 signal was ineffective in alleviating depression-like symptoms. Moreover, our investigation involved Golgi staining and Western blot techniques, which found enhanced synaptic plasticity, including increased dendritic branches, dendritic spines and retrograde protection after GDNF treatment in Parkinson disease models.
Limitations: A subtle motor phenotype became evident only toward the conclusion of the behavioural testing period. The study exclusively involved male mice, and no separate control group receiving only GDNF treatment was included in the experimental design.
Conclusion: Our findings support a positive effect of exogenous GDNF on synaptic plasticity, mediated by DRD1 signalling in the mPFC, which could facilitate depression remission in Parkinson disease.
Introduction
Parkinson disease is the world’s second most common degenerative neurologic disorder, and nonmotor symptoms cannot be ignored in most patients with Parkinson disease, including cognitive decline, sleep disturbance, fatigue, anxiety, and depression. Depression, as one of the most common comorbidities of Parkinson disease, occurs at any stage of Parkinson disease and usually precedes the clinical motor symptom phase.1,2 A subacute mouse model of Parkinson disease induced by 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) has shown depression-like behaviour in about 60% of the mice.3
Depression with Parkinson disease is thought to arise from damage to the mesocorticolimbic system, affecting dopamine secretion.4 The medial prefrontal cortex (mPFC), a crucial brain region that regulates emotions and sensations, has shown increased activity in social cognition, depression and self-related tasks.5 Imaging studies have shown that increased mPFC activity can inhibit negative emotions.6 Abnormal medial prefrontal lobe function closely correlates with dysfunction in visceral regulation, emotional cognition, self-focused thinking and other functional disorders among patients with depression.7 Furthermore, a decrease in prefrontal volume among patients with depression and bipolar disorder has been linked to a reduced number of prefrontal glial cells, resulting in weakened nutritional support provided by glial cells and functional and morphological changes in medial prefrontal pyramidal cells.8 Studies have shown that the dopamine receptor D1 (DRD1) and dopamine receptor D2 (DRD2) of mPFC pyramidal neurons in 6-hydroxydopamine-injured rats have abnormal functions.9 Experiments have shown that patients with depression have lower levels of dopamine and 5-hydroxytryptamine,10 so the dopamine signal pathway in mPFC may play an antidepressant role.
Glial cell–derived neurotrophic factor (GDNF) plays a vital role in promoting the survival of distinct neuronal subpopulations during different stages of nervous system development.11 Glial cell–derived neurotrophic factor and its receptors are expressed in the prefrontal cortex and hippocampus.12 Animal experiments have shown that GDNF exhibits neuroprotective and reparative effects on dopamine neurons and can also enhance various functions.13 Studies have found that GDNF family receptor α-1 can be mediated by signalling from neural cell adhesion molecules to induce dendritic growth and dendritic spine formation in the hippocampus.14 In investigating the impact of exercise on depressive symptoms, researchers have found that changes in the number of excitatory synapses may be indicative of alterations in depressive symptoms.15 Stress-induced dendritic spine loss has been linked to decreased synaptic currents, reduced levels of synaptic proteins and increased depression-like behaviours.16 Notably, during stress periods, dopaminergic transmission in the mPFC can lead to the retraction of apical dendrites, highlighting the crucial role of dopaminergic transmission in maintaining the regular morphology of dendrites.17 Our recent study reaffirmed that, in the initial phases of Parkinson disease, nonmotor symptoms can manifest even in the presence of relatively subtle changes to dopaminergic terminals in the prefrontal cortex, which are attributed to a deficiency in GDNF. Furthermore, our findings underscore the role of GDNF in enhancing dopaminergic synaptic transmission within the mPFC, shedding light on its potential significance in addressing cognitive impairments associated with Parkinson disease.12 Here, we sought to address the effect of GDNF on dopamine postsynaptic receptor-mediated signalling and whether it is related to Parkinson disease with depression.
We aimed to examine the impact of exogenous GDNF on depression-like behaviour in MPTP-induced Parkinson disease models. We had 2 objectives, namely to affirm the antidepressant efficacy of prefrontal GDNF supplementation in Parkinson disease mice models, and to elucidate the underlying mechanism through which GDNF ameliorates depression. These investigations are built on previous observations, which noted a more pronounced reduction of GDNF levels within the prefrontal cortex of MPTP-induced Parkinson disease mice exhibiting heightened anhedonic and selective depressive-like behaviours.18 Furthermore, our findings could have important implications for the prevention and management of depression among people with Parkinson disease.
Methods
Animals and drug administration
We used 8-week-old male C57BL/6J mice (20–25 g), purchased from the Experimental Animal Center of Xuzhou Medical University. The mice were housed in plastic cages lined with corncob bedding, with 6 mice per cage, under controlled conditions of room temperature (22°C–26°C), a 12-hour light–dark cycle and ad libitum access to food and water. We established 3 groups of mice, namely a control group (saline injection), an MPTP group (MPTP and saline injection) and an MPTP plus GDNF injection group. We injected mice intraperitoneally with a 7 mg/mL solution of MPTP (M0896, Sigma-Aldrich, 30 mg/kg) at a fixed time (900) every day for 5 consecutive days. The control group of mice received an equivalent volume of ordinary saline (0.9% sterile saline) via intraperitoneal delivery. On day 6, we administered GDNF directly into the mPFC brain region via stereotaxic injection (0.5 ng/μL, 0.2 μL) at the following coordinates: anterior–posterior 2.00 mm, medial–lateral 0.35 mm and dorsal–ventral −2.10 mm. We conducted behavioural tests assessing depression and motor ability over the subsequent 9 days. After intraperitoneal injection of the DRD1 antagonist (SCH-23390 MedChemExpress) at a dose of 0.1 mg/kg in the MPTP plus GDNF injection group, the mice were sacrificed within 30 minutes.
Sucrose preference test
To assess depressive-like phenotypes, we employed a 4-day sucrose preference protocol. After the mice acclimated to the environment, they were given 2 drinking bottles containing normal drinking water and 1% sucrose water, respectively. After a 24-hour water deprivation, the mice were presented with 1 bottle of 1% sucrose and 1 water bottle for a 2-hour period. We then switched the positions of the bottles after 1 hour. We measured the fluid weight from each bottle to determine the amount of sucrose consumption and water consumption. We calculated the relative sucrose preference for each mouse on each day the proportion of sucrose consumption relative to total consumption of sucrose and water, multiplied by 100%.19
Rotarod test
The rotarod test is a standard method for assessing motor coordination, balance and fatigue in rodents. We placed 5 mice on the rotarod (diameter 3 cm) simultaneously, after a 30-second adaptation period. Subsequently, the mice were allowed to move freely around the rotarod at a constant speed of 5 rpm for 90 seconds. After this duration, the rotarod fatigue metre was activated. An infrared device meticulously monitored the latency, or time duration, of the mouse as it descended from the rotarod. The timing sequence started upon placing the mouse onto the rotating rod and concluded when the infrared device detected the mouse’s descent from the rotating rod. Simultaneously, the system recorded the latency while adjusting the rotarod speed to 16 rpm. Each mouse underwent 3 trials, and we used the average of those measurements to calculate the overall running time for the experiment.20
Forced swimming test
After a 1-hour rest, we gently placed each group of mice inside a glass jar containing water maintained at a controlled depth of 15 cm and a temperature of 25°C. The entire 5-minute session was recorded on video, and we subsequently calculated the immobility time during the session. Behavioural despair was characterized by movements on the water’s surface without extending beyond the range needed to keep the nose above the water.19
Tail suspension test
To isolate the mice from their surroundings, we employed a white barrier to prevent visual stimuli. In addition, we securely taped the rear third of the mouse’s tail to a horizontal metal rod, causing it to hang upside down with its head positioned 15 cm above the ground. Throughout the entire experimental procedure, we recorded videos in a quiet environment to ensure minimal disturbance to the mice. The recording duration was 6 minutes, followed by a 4-minute analysis period. We defined immobility as the absence of limb movement, except for essential respiration, while the mouse was suspended. To avoid any visual or auditory interference, we assessed each mouse independently during the experiment.21
Exclusion and inclusion criteria for mice with depression phenotype
We identified depressive behaviour among MPTP-induced Parkinson disease mice models based on the results of the sucrose preference test, forced swimming test and tail suspension test, compared with controls. In the forced swimming test, we determined evaluation indices based on the duration and frequency of motionlessness, climbing, and swimming. We identified depressive behaviours by a significant increase in inactivity time, a decrease in swimming time and distance, or a decrease in climbing time. The climbing time refers to the duration during which the mouse engages in upward movements along the wall of the barrel in an effort to escape the water in a forced swimming test. Essentially, it represents the period in which the mouse endeavours to overcome the aquatic environment. The tail suspension test measured immobility and movement struggle time. Depressive behaviour was identified by a significantly higher inactivity time or lower struggle time compared with controls. The sucrose preference test used sucrose preference as an indicator, and a sucrose preference lower than 0.4, or a significant decrease compared with the control group, was considered indicative of depression. For all behavioural statistics, we included data from all mice. However, for subsequent staining and protein experiments, we employed specific criteria for identifying depressive mice. In the forced swim test, we considered mice with immobility times exceeding 150 seconds (control group: mean + 1.5 standard deviation [SD]) as meeting the depression criterion. In the sucrose preference test, we included mice with a preference ratio below 0.5 (control group: mean – 1.5 SD). In the tail suspension test, we included mice with immobility times exceeding 125 seconds (control group: mean + 1.5 SD). We applied these criteria to include 5–6 of the 8 mice in each behavioural study as meeting the criteria for depression.
Tissue sampling and preparation
After the behavioural testing, we euthanized the mice under isoflurane anesthesia during the daytime. The brains of mice from each group were promptly removed. Using the brain mould and combining it with the positioning of the brain atlas, we extracted fresh brain tissue from the mPFC and substantia nigra.
For immunohistological experiments, we perfused the mice from each group transcardially with phosphate-buffered saline (PBS), followed by 4% paraformaldehyde (pH 6.9). We then removed the brains and post-fixed them in the 4% paraformaldehyde at 4°C overnight. We subsequently immersed in 20% and 30% sucrose solutions at 4°C for 2–3 days to achieve dehydration until an isotonic state was reached.
In addition, we used some mice for Golgi staining. Large brain samples, such as whole brain tissue, were sectioned into chunks about 10 mm thick using a sharp blade. We incubated these sections with a Golgi staining kit (FD Rapid Golgi Stain TM Kit, FD NeuroTechnologies). Subsequent staining involved preparing brain tissue slices with a thickness of 100–200 μm using a vibrating microtome (Leica VT1000s).
For Western blot analysis, we stored the brain tissue samples in a −80°C freezer for later protein content detection. We measured the total protein concentration using a bicinchoninic acid protein assay kit. The absorbance was read at 562 nm using an ultraviolet spectrophotometer.
Immunohistochemisty imaging and image analysis
After achieving an isotonic state, we sectioned frozen brains into 40 mm coronal slices using a freezing microtome at −20°C. These brain slices were placed in an antifreeze solution and stored at −20°C. When ready for analysis, the prepared sections were taken out from the freezers, warmed for 30 minutes at 37°C in an incubator and then rinsed 3 times, for 5 minutes each time, with PBS.
We immersed brain slices in a mixture of 10% goat serum and 0.3% Triton X-100 (diluted in PBS) at room temperature for 1 hour to block nonspecific binding sites. Subsequently, we carefully removed the excess blocking solution and immersed the brain slices in the prepared primary antibody diluent at 4°C overnight. Afterward, we rewarmed the sections in a 37°C incubator for 30 minutes, followed by 3 washes with PBS (5 min each time) to remove any unbound antibodies. Next, we immersed the brain slices in fluorescent secondary antibody diluent and incubated them at room temperature for 2 hours in the dark. After the incubation period, we rinsed the slices 3 times with PBS (5 min each time) to remove excess secondary antibodies. To visualize cell nuclei, we dipped brain slices in 4′,6-diamidino-2-phenylindole (DAPI) staining solution, incubated them at room temperature for 15 minutes in the dark and then washed them 3 times with PBS (5 min each time). Finally, we mounted the brain slices on slides with an anti-fluorescence quencher and observed and photographed them using an upright fluorescent microscope.
For quantitative immunohistochemical analysis, we acquired cross-sectional images, viewed under microscopes (Olympus VS120). We quantitatively imaged the number of cells positive for DRD1, postsynaptic density protein 95 (PSD95), dopamine transporter (DAT) and tyrosine hydroxylase (TH) antibodies (Appendix 1, Table 1, available at https://www.jpn.ca/lookup/doi/10.1503/jpn.230079/tab-related-content, provides information on primary and secondary antibodies). Two sections were randomly selected from each mouse, and 6 mice in each group were imaged and quantified. We used ImageJ software for further analysis, such as cell counts and location analysis.
Golgi staining
The intact brain tissue was carefully removed from the mice after they had been thoroughly stripped and perfused with 0.9% saline at 4°C. We immersed the brain tissue in an impregnation solution (mixture of equal volumes of solutions A and B from the Golgi Stain Kit) mixture, which had been made at least 24 hours beforehand (FD Rapid Golgi Stain Kit, FD NeuroTechnologies). After replacing the brain tissue with fresh AB mixture, it was kept in the dark at room temperature for 2 weeks. Subsequently, we immersed the brain tissue in an appropriate amount of solution C, and replaced the C solution the next day. We then stored the brain tissue at room temperature away from light for 4 days. Following this, we rinsed the sections with deionized water for 5 minutes twice, then submerged them for 10 minutes in a solution composed of 1 part solution D, 1 part solution E and 2 parts deionized water (prepared and used at that moment). Next, we rinsed the sections with deionized water for 5 minutes twice, and subjected them to an alcohol gradient dehydration for 4 minutes. Lastly, we rinsed the sections 3 times with clear xylene for 4 minutes. The slide was finally sealed with neutral gum, and the brain sections were photographed using a digital scanning section system (Olympus VS120).12
Western blot
After the mice were killed by dislocation, the intact brain tissue was stripped off on an ice box, and the brain tissue of the medial PFC and substantia nigra was quickly removed. We added whole-cell protein lysate (composed of radioimmunoprecipitation assay buffer, phenylmethylsulfonyl fluoride and phosphatase inhibitor, in a volume ratio of 100:1:1) to 100 mg of tissue plus 1 mL of whole-cell protein lysate. The protein sample can be balanced at the appropriate concentration. Based on the volume ratio of the sample, we used the protein loading buffer to dilute the protein at a ratio of 5 parts buffer to 1 part protein). We used a boiling water bath for about 5–10 minutes to denature the protein. We separated protein samples (20–30 μg) by sodium dodecyl sulfate polyacrylamide gel electrophoresis (10%–12%) for 90 minutes. Subsequently, we transferred samples to nitrocellulose filter membranes at 80–120 V for 1 hour. After blocking with 5% skim milk for 2 hours at room temperature, the membrane was incubated overnight at 4°C with primary antibodies (Appendix 1, Table 1), including GDNF antibody (1:1000), TH antibody (1:200), DRD1 antibody (1:1000), PSD95 antibody (1:1000), protein kinase A (PKA) antibody (1:20 000), phospho-PKA (pPKA) antibody (1:1000), cyclic adenosine monophosphate (cAMP) response element binding protein (CREB) antibody (1:1000), and phosphorylated CREB antibody (1:1000). Subsequently, we applied peroxidase-labelled secondary antibody (1:500, Thermo Scientific) at room temperature for 90 minutes. We visualized protein bands using an imaging system (LI-COR Biosciences), analyzed with ImageJ software (National Institutes of Health).
Statistical analysis
We used SPSS version 19.0 statistical analysis software for the statistical analysis of experimental data. Quantitative data were expressed as means and standard errors of mean (SEM). We used Student t tests to compare 2 groups, and a 1-way analysis of variance (ANOVA) followed by the least significant difference (LSD) or the Dunnett T3 post hoc test when comparing the differences between more than 2 groups. We used the normalized positive area calculation method to subtract the average of the control data from the data of the control group and the experimental group at the same time. We used a 2-factor repeated ANOVA to determine whether there were statistically significant differences in motor function between the control and MPTP groups. The 2 factors considered were time and inter-group variations, with a focus on identifying any noteworthy differences within these parameters. We used an α of 0.05 as the test level, and considered a p value of less than 0.05 statistically significant. We drew graphs using GraphPad Prism version 9 software.
Ethics approval
All experimental procedures involving animals were conducted in accordance with the ethical regulations of the Ethics Committee of the Experimental Animal Center of Xuzhou Medical University (no. L20210226140 and no. L20210226312), as well as the guidelines provided by the National Institutes of Health for the Care and Use of Laboratory Animals.
Results
Establishment of mouse model of Parkinson disease with depression-like behaviours
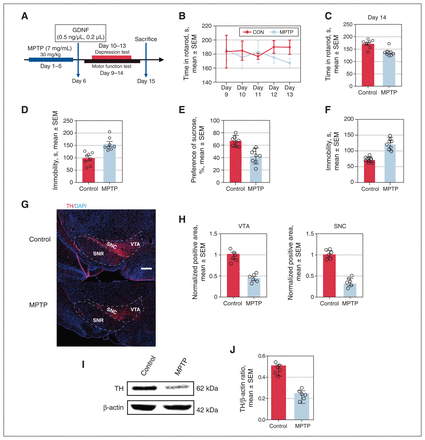
Figure 1 depicts control (n = 8) and MPTP (n = 8) groups. Figure 2 illustrates control (n = 8), MPTP (n = 8) and MPTP plus GDNF (n = 8) groups. Figure 3 showcases MPTP plus GDNF (n = 8) and MPTP, GDNF and antagonist (n = 8) groups. We took rigorous measures to ensure that the mice within each group originated from the same batch and week when executing a particular experiment. The timeline of the experiments for Parkinson disease mouse models is shown in Figure 1A. Between day 9 and day 13, the mice did not exhibit any significant motor impairment, as determined by 2-way repeated-measures ANOVA (Figure 1B). After pairwise comparison between the 2 groups at different time points, we found a statistically significant difference between the mice on day 14 (Figure 1C). As a result, we collected data from day 10 to day 13 and analyzed the results of depression tests during this period, where no significant motor impairment was observed. During the forced swimming test, mice injected with MPTP exhibited a longer immobility time than controls, reaching 150 seconds (Figure 1D). In addition, these mice demonstrated a significantly reduced preference for sucrose (Figure 1E). Furthermore, the tail suspension test found a significantly higher immobility time in the MPTP group than the control group (Figure 1F).
The subacute model of Parkinson disease with depression-like behaviour established by intraperitoneal injection of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP). (A) Experimental timeline, including injection of glial cell–derived neurotrophic factor (GDNF) in mice in the MPTP plus GDNF group. (B) Performance in rotarod test on days 9–13 among control mice and mice treated with MPTP (pinteraction = 0.3924, ptime = 0.9420, pgroup = 0.1226). (C) Performance in rotarod test on day 14 among control mice and mice treated with MPTP (t = 4.188, p = 0.0009). (D) Performance of control and MPTP-treated mice in forced swimming test on day 10 (t = 3.4268, p = 0.002). (E) Performance of control and mice MPTP-treated mice in the sucrose preference test on day 12 (t = 3.411, p = 0.009). (F) Performance of control and MPTP-treated mice in the tail suspension test on day 11 (t = 4.750, p = 0.0014). (G) Representative images of tyrosine hydroxylase (TH) immunofluorescence staining (red) of dopaminergic neurons in the substantia nigra pars compacta (SNC) and ventral tegmental area (VTA) of control and MPTP-treated mice (4′,6-diamidino-2-phenylindole [DAPI] in blue; scale 200 μm). (H) Normalized positive area in the VTA and SNC from control and MPTP-treated mice (VTA: t = 20.59, p < 0.001; SNC: t = 42.37, p < 0.0001). (I, J) Western blot analysis of TH expression in the substantia nigra of control and MPTP-treated mice (t = 9.002, p < 0.0001). SEM = standard error of the mean; SNR = substantia nigra pars reticulata.
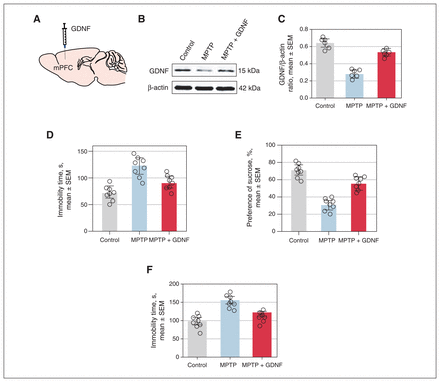
Effects of injecting exogenous glial cell–derived neurotrophic factor (GDNF) into the medial prefrontal cortex (mPFC) on depression-like behaviour in Parkinson disease mouse models. (A) Schematic diagram of GDNF injection site. (B, C) Western blot analysis of GDNF expression in the mPFC of control mice, mice treated with 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) and mice treated with MPTP plus GDNF (MPTP v. control: p < 0.0001; MPTP v. MPTP + GDNF: p < 0.0001). (D) Performance of control, MPTP-treated and MPTP plus GDNF–treated mice in the forced swimming test (MPTP v. control: p < 0.0001; MPTP v. MPTP + GDNF: p = 0.0003). (E) Performance of control, MPTP-treated or MPTP plus GDNF–treated mice in the sucrose preference test, compared with 1-way ANOVA, followed by LSD test (MPTP v. control: n = 8, p < 0.0001; MPTP v. MPTP + GDNF: n = 8, p = 0.0039). (F) Performance of control, MPTP-treated or MPTP plus GDNF–treated mice in the tail suspension test (MPTP v. control: p = 0.0006; MPTP v. MPTP + GDNF: p = 0.0612). SEM = standard error of the mean; SNC = substantia nigra pars compacta; SNR = substantia nigra pars reticulata; VTA = ventral tegmental area.
Effect of glial cell–derived neurotrophic factor (GDNF) in the medial prefrontal cortex (mPFC) and regional dopamine receptor D1 (DRD1) pathway activity in relieving depression-like behaviour in Parkinson disease mouse models. (A) Experimental timeline. (B) Western blot analysis of DRD1 expression in the mPFC of control mice, mice treated with 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) and mice treated with MPTP plus GDNF (MPTP v. control: p = 0.0048; MPTP v. MPTP + GDNF: p = 0.0053). (C) Immunofluorescent staining showed immunoreactive puncta (red) of DRD1 in the mPFC of control, MPTP-treated and MPTP plus GDNF–treated mice (4′,6-diamidino-2-phenylindole [DAPI] in blue; scale 50 μm) (normalized positive area: MPTP v. control: p < 0.0001; MPTP v. MPTP + GDNF: p < 0.001). (D–F) Western blot analysis of protein kinase A (PKA), and phospho-PKA (pPKA) expression in the mPFC of control, MPTP-treated or MPTP plus GDNF–treated mice (PKA: MPTP v. control: p = 0.5826; MPTP v. MPTP + GDNF: p = 0.7182,) (pPKA: MPTP v. control: p = 0.0005; MPTP v. MPTP + GDNF: p = 0.0029), (pPKA/PKA, MPTP v. control: p = 0.0003, MPTP v. MPTP + GDNF: p = 0.0011). (G, H) Western blot analysis of pPKA and PKA expression in the mPFC of mice treated with MPTP plus GDNF or with MPTP, GDNF and DRD1 antagonist (pPKA: t = 4.273, p = 0.0238) (pPKA/PKA: t = 7.328, p = 0.0429). (I) Performance of mice treated with MPTP plus GDNF or with MPTP, GDNF and DRD1 antagonist in the forced swimming test (p = 0.0028) and (J) the sucrose preference test. (p = 0.0051). (K–P) Western blot analysis of dopamine receptor D2 (DRD2), arrestin, glycogen synthase kinase-3β (GSK-3β) and protein phosphatase 2A (PP2A) expression in the mPFC of control, MPTP-treated and MPTP plus GDNF–treated mice (DRD2: MPTP v. control: p = 0.3152; MPTP v. MPTP + GDNF: p = 0.4269) (arrestin: MPTP v. control: p = 0.0065; MPTP v. MPTP + GDNF: p = 0.0267) (PP2A: MPTP v. control: p = 0.4127; MPTP v. MPTP + GDNF: p = 0.0381) (GSK-3: MPTP v. control: p = 0.0039; MPTP v. MPTP + GDNF: p = 0.0058). ACC = anterior cingulate cortex; IL = infralimbic cortex; PL = prelimbic cortex; SEM = standard error of the mean.
Meanwhile, immunofluorescence staining and Western blot analysis found that the expression of TH protein from the midbrain substantia nigra decreased by more than 2 times in the MPTP group (Figure 1G–J). In summary, the Parkinson disease mouse model was successfully constructed. The outcomes of the forced swimming test, sucrose test and tail suspension test in Parkinson disease mice revealed increased immobility and reduced preference for sucrose. These findings suggest the manifestation of depression-like behaviours in the Parkinson disease mice.
Exogenous GDNF and depression-like behaviour in mouse model of Parkinson disease
For the batch of mice used to investigate the effects of GDNF on depression-like behaviour in Parkinson disease mice exhibiting depressive symptoms, we administered GDNF injections into the mPFC brain region of the Parkinson disease mice. The injection location is illustrated (Figure 2A). The timeline of the experiments is shown in Figure 1A. Western blot analysis showed a significantly lower amount of GDNF content in the mPFC region of the MPTP group, compared with the control group. However, after the exogenous replenishment of GDNF, there was an increase in GDNF (Figure 2B and 2C). To evaluate depression, we conducted the forced swimming test, the sucrose preference test and the tail suspension test (Figure 2D–2F). The results showed that the MPTP plus GDNF group had significantly lower immobility time than the MPTP group, indicating a reduction in depression-like behaviour. In addition, the MPTP plus GDNF group exhibited a higher preference for sucrose.
Increased GDNF content in the mPFC and regional DRD1 pathway activity
To elucidate the mechanism underlying GDNF’s impact on depression-like behaviour in Parkinson disease mice, we focused on the regulation of DRD1-dependent signalling. The establishment of Parkinson disease in mice involved intraperitoneal injections of MPTP over a period of 1–5 days. On day 6, GDNF was injected into the mouse mPFC brain region using stereotactic injection. Subsequently, behavioural tests assessing depression were conducted over the next 9 days. We administered DRD1 antagonists intraperitoneally half an hour before the start of the behavioural tests. Through Western blotting and immunofluorescence analysis, we observed a significant increase in the expression of DRD1, pPKA, arrestin, protein phosphatase 2A (PP2a) and glycogen synthase kinase-3β (GSK-3β) in the mPFC region after GDNF treatment, compared with the MPTP group (Figure 3A–3F). To further validate the importance of DRD1, we employed DRD1 antagonists to assess the changes in the downstream pathway after GDNF treatment, which showed a significant 50% reduction in pPKA after using the DRD1 antagonist (Figure 3G and 3H). Moreover, the mice exhibited depression-like behaviour (Figure 3I and 3J). Interestingly, there was no significant difference in the contents of DRD2, indicating that D2 receptors were not involved in this anti-depression process (Figure 3K–3P). We then aimed to investigate the influence of GDNF on DRD1 within the mPFC of control mice under normal physiologic conditions. However, our experiments did not reveal significant effects.
GDNF content in the mPFC and indicators of synaptic connections
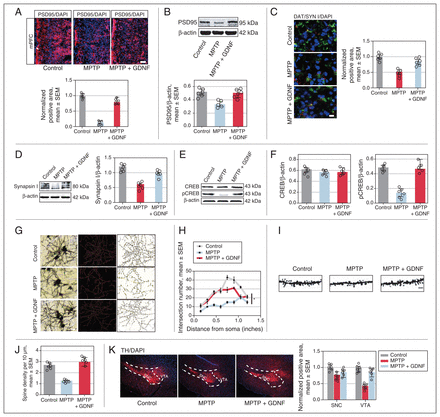
Postsynaptic density protein 95 is a critical protein marker for synapses that promotes dendritic spine growth and plays a crucial role in synaptic plasticity. We used Western blotting and fluorescence analysis to investigate changes in PSD95 content in the mPFC, which showed a significant decrease in PSD95 content in the MPTP group compared with the control group (p < 0.05), while the MPTP plus GDNF group exhibited a significant increase in PSD95 content (Figure 4A and 4B). Subsequently, we explored CREB, a protein related to the PSD95 pathway. Western blotting results confirmed a significant decrease in phosphorylated CREB after MPTP treatment. However, the levels recovered to an average level after the addition of GDNF (Figure 4E–4F). Moreover, immunofluorescent staining showed an increase in immunoreactive puncta of synapsin 1, a marker for presynaptic components, and dopaminergic presynaptic membrane co-localized in the mPFC of the MPTP plus GDNF group (Figure 4C). In addition, Western blotting assays showed a significant increase in synapsin 1 expression after GDNF intervention in MPTP-induced mice (Figure 4D).
Effect of increasing glial cell–derived neurotrophic factor (GDNF) content in the medial prefrontal cortex (mPFC) on synaptic connections and functions. (A) Immunofluorescent staining (scale 10 μm) showed immunoreactive puncta (red) of postsynaptic density protein 95 (PSD95) in the mPFC of control mice, mice treated with 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) and mice treated with MPTP plus GDNF (MPTP v. control: p < 0.0001; MPTP v. MPTP + GDNF: p < 0.001). (B) Western blot analysis of PSD95 expression in the mPFC of control, MPTP-treated or MPTP plus GDNF–treated mice (MPTP v. control: p = 0.0496, MPTP v. MPTP + GDNF: p = 0.0327). (C) Representative images of immunostaining (scale 50 μm) for dopamine transporter (DAT, red), synapsin 1 (SYN 1, green) and 4′,6-diamidino-2-phenylindole (DAPI, blue) in the mPFC of control, MPTP-treated and MPTP plus GDNF–treated mice (MPTP v. control: p = 0.0029; MPTP v. MPTP + GDNF: p = 0.0041). (D) Western blot analysis of SYN 1 expression in the mPFC of control, MPTP-treated and MPTP plus GDNF–treated mice (MPTP v. control: p = 0.0058; MPTP v. MPTP + GDNF: p = 0.0075). (E, F) Western blot analysis of cyclic adenosine monophosphate (cAMP) response element binding protein (CREB) and phosphorylated CREB (pCREB) expression in the mPFC of control, MPTP-treated and MPTP plus GDNF–treated mice (CREB: MPTP v. control: p = 0.5478; MPTP v. MPTP + GDNF: p = 0.8720) (pCREB: MPTP v. control: p = 0.0073; MPTP v. MPTP + GDNF: p = 0.0269). (G) Representative images of neuronal dendrites by Golgi–Cox staining (scale 10 μm). (H) Comparisons of intersection number of control, MPTP-treated and MPTP plus GDNF–treated mice (MPTP v. control: p = 0.0039; MPTP v. MPTP + GDNF: p = 0.0381). (I) Representative images of neuronal dendrites by Golgi–Cox staining. (J) Comparisons of spine density of control, MPTP-treated and MPTP plus GDNF–treated mice (MPTP v. control: p = 0.0167; MPTP v. MPTP + GDNF: p = 0.0074). (K) Immunofluorescent staining (scale 200 μm) showed immunoreactive puncta (red) of tyrosine hydroxylase (TH) in the substantia nigra pars compacta (SNC) and ventral tegmental area (VTA) of control, MPTP-treated and MPTP plus GDNF–treated mice (SNC: MPTP v. control: p = 0.0284; MPTP v. MPTP + GDNF: p = 0.6728) (VTA: MPTP v. control: p = 0.0029; MPTP v. MPTP + GDNF: p = 0.0072). SEM = standard error of the mean.
Exogenous GDNF and dendritic field branches and spine densities in the mPFC
Dendritic spines are crucial for the formation and structural plasticity of synapses, facilitating information transmission between neurons. In mice treated with MPTP, the number of intersection points decreased by half compared with the rates observed in mice treated with saline. In addition, Golgi–Cox staining of fixed brain tissues confirmed a reduction in the density of dendritic spines in the dendrites of MPTP-treated mice. However, the results showed that mice treated with GDNF exhibited a significant increase in the number of crossing points in the dendritic branches of neurons and an increased density of dendritic spines compared with the MPTP group (Figure 4G–4J). These findings suggest that increased GDNF can effectively prevent the loss of dendritic branches and dendritic spine density in the mPFC of Parkinson disease mouse models. After GDNF treatment of the mPFC brain region, further observation of TH results in the ventral tegmental area of mice showed that the damage was reduced compared with the MPTP group, suggesting the existence of retrograde protective effect of GDNF (Figure 4K).
Discussion
In this study, we established a Parkinson disease mouse model to investigate the effects of GDNF in the frontal cortex on the depression-like behaviour of Parkinson disease mice. The behavioural experiments revealed a decrease in depression-related emotions after GDNF intervention. We found that GDNF activated the adenylyl cyclase–cAMP–PKA pathway, mediated by DRD1 in the mPFC, contributing to its antidepressant effects. Furthermore, GDNF increased the expression content of PSD95 and synapsin 1, and enhanced the density of dendritic branches and dendritic spines, providing retrograde protection.
Briefly, MPTP is a highly lipophilic drug with the ability to penetrate the blood–brain barrier. Its metabolite, 1-methyl-4-phenyl pyridine ion (MPP+), damages dopaminergic neurons by inhibiting the expression of the rate-limiting enzyme in dopamine synthesis, leading to a reduction in dopamine content. The MPTP-induced animal model exhibits symptoms and pathological changes similar to human Parkinson disease, and these manifestations remain relatively stable.22–24 In this experiment, C57BL/6 mice received intraperitoneal injections of MPTP and then performed in the forced swimming test, sucrose preference test and tail suspension test. The results indicated the presence of depression-like symptoms. A commonly used neurotrophic factor, GDNF, can improve Parkinson disease symptoms in animal models induced by either MPTP or 6-hydroxydopamine.25–27 Our experiments further confirmed that increasing the level of GDNF in the mPFC can alleviate depression. However, we observed that the effect of intracranial GDNF injection was short-lived. Therefore, it is crucial to take into account the drug’s residence duration and plasma concentration for effective treatment. Depression often precedes the development of motor symptoms in Parkinson disease.28 The precise mechanism linking depression and Parkinson disease remains incompletely understood. However, reduced dopamine levels in specific brain regions, such as the mPFC and nucleus accumbens, have been associated with decreased volitional activity, apathy and anhedonia.29 The mPFC, which is involved in emotion regulation, relies on top–down regulation.30 Studies have shown that people with depression exhibit compensatory activity in the left prefrontal cortex, as detected by magnetoencephalography.31 The nucleus accumbens, a key brain region involved in depression and addiction, has been extensively studied.32 The projections from the mPFC to the nucleus accumbens are believed to influence social behaviour, and stimulating these synapses has shown antidepressant effects.33 Therefore, comprehending the PFC–nucleus accumbens top–down regulation model is crucial for developing effective treatments targeting the mPFC.
Our previous report found that GDNF enhanced the synaptic transmission of dopamine in the PFC.12 However, the specific receptor responsible for the antidepressant effect remains unclear. Dopamine receptors belong to the G-protein–coupled receptor family and are divided into 2 subtypes, DRD1 and DRD2. Our findings indicate that DRD1 in the mPFC plays a crucial role in the antidepressant effects of GDNF by activating the adenylate cyclase–cAMP–PKA pathway. Other studies have also identified abnormally low activity of PKA and PKC in patients with severe depression. In laboratory animals, the drug rolipram potentiates activation of the adenylate cyclase–cAMP–PKA pathway to improve depressive behaviour.34 In mice, DRD1 agonists have been shown to improve the decrease in dopamine levels and hopelessness induced by chronic unpredictable stress.35 The D1-like receptor attaches to G-coupled proteins, stimulating adenylate cyclase and increasing intracellular cAMP levels, which in turn activates PKA. On the other hand, the D2-like receptor binds to Gi/o proteins, inhibiting PKA activity.36 Activation of DRD1-expressing pyramidal cells in the mPFC has been shown to produce rapid and persistent antidepressant and anxiolytic effects. In contrast, the activation of DRD2-expressing pyramidal cells did not affect anxiety-like and depression-like parameters.6 Downstream of DRD2 activation, biologically verified arrestin–PP2A–protein kinase B complexes have been identified.37 These findings are consistent with our results and further highlight that GDNF exerts its antidepressant effect by activating D1 receptors in the mPFC.
Intracerebral perfusion of DRD1 agonists has been found to alter the levels of glutamate and γ-aminobutyric acid outside the cells in the prefrontal lobe.38 Building on this evidence, we hypothesized that enhancing D1 mediated by GDNF might also have an impact on the regional concentration of glutamate and excitability; however, the specific mechanism underlying this effect requires further investigation. To assess the long-term plasticity of glutamatergic pyramidal neurons, we performed Golgi staining. The results revealed that increasing GDNF in the mPFC significantly enhanced dendritic branching and the length and density of dendritic spines in Parkinson disease mice, thereby restoring the morphological structure and synaptic plasticity of pyramidal neurons. In addition, Western blotting indicated an increase in regional PSD and synapsin 1 expression, indicating stronger synaptic connections in higher numbers in the region. These findings suggest that GDNF intervention may promote synaptic plasticity in the mPFC, contributing to the development of excitatory microcircuits. Previous studies have shown that impaired microcircuits and an imbalance between excitatory and inhibitory neurons in the PFC can lead to deficits in short-term working memory and depression-like behaviours in early Alzheimer disease.39 In other words, the biology of depression in the early stages of neurodegenerative disease is likely to originate from issues in the top region, such as the PFC, which subsequently affects the functioning of the downstream region, such as the nucleus accumbens. Furthermore, a closer examination of TH results in the midbrain of mice found a reduction in damage when compared with the Parkinson disease group after treatment with GDNF. This finding suggests the presence of a retrograde protective effect of GDNF.40
In this experiment, we identified that DRD1, but not DRD2, in the mPFC plays an essential role in the antidepressant effects of GDNF treatment. This effect is mediated through the adenylate cyclase–cAMP–PKA pathway. In addition, we propose a treatment scheme for depression in Parkinson disease, which has important implications for understanding the cause and mechanism of action of this condition. Furthermore, our findings provide a valuable reference for identifying more effective treatment and prevention targets.
Limitations
A subtle motor phenotype became evident only toward the conclusion of the behavioural testing period. The study exclusively involved male mice, and no separate control group receiving only GDNF treatment was included in the experimental design. Given our nonsignificant findings in investigations of the influence of GDNF on DRD1 in the mPFC of control mice under normal physiologic conditions, we are contemplating the necessity of enlarging the sample size and increasing the GDNF dosage to elucidate the impact of GDNF on dopamine receptor regulation under physiologic conditions in the future.
Conclusion
This study elucidated the protective impact of GDNF on depression-like behaviour in a mouse model of Parkinson disease, specifically within the PFC. Our behavioural experiments revealed a notable decrease in symptoms related to depression and anxiety after GDNF intervention. Further investigations pinpointed the involvement of the adenylate cyclase–cAMP–PKA pathway, which is modulated by DRD1 in the mPFC, as the mechanism responsible for GDNF’s antidepressant effects. Notably, the administration of a DRD1 antagonist induced depression-like behaviour in the mice. In addition, GDNF enhanced the expression of synaptic markers PSD95 and synapsin 1, and promoted increased density in dendritic branches and dendritic spines. Finally, GDNF supplementation in the frontal cortex exerted retrograde neuroprotective effects, leading to an increase in midbrain dopamine neuron survival.
Footnotes
↵* Yehao Liu and Xiaoyu Zhou contributed equally as first authors.
Competing interests: None declared.
Contributors: Xiaoyu Zhou and Chuanxi Tang contributed to study conception and design. Yehao Liu and Ruiao Sun contributed to data acquisition. Ke Xue and Yan Tang contributed to data analysis and interpretation. Yehao Liu, Xiaoyu Zhou, Ruiao Sun and Chuanxi Tang drafted the manuscript. All of the authors revised it critically for important intellectual content, gave final approval of the version to be published and agreed to be accountable for all aspects of the work.
Data statement: All data generated or analyzed during this study are included in this published article and its supplementary information files.
Funding: The study was funded by the National Natural Science Foundation of China (no. 82101263 to Chuanxi Tang), the Jiangsu Province Science Foundation for Youths (no. BK20210903 to Chuanxi Tang) and the Research Foundation for Talented Scholars of Xuzhou Medical University (no. RC20552114 to Chuanxi Tang).
- Received May 19, 2023.
- Revision received August 4, 2023.
- Revision received October 6, 2023.
- Revision received October 17, 2023.
- Accepted October 17, 2023.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/