Abstract
Background: Neuroimaging studies have revealed abnormal functional interaction during the processing of emotional faces in patients with major depressive disorder (MDD), thereby enhancing our comprehension of the pathophysiology of MDD. However, it is unclear whether there is abnormal directional interaction among face-processing systems in patients with MDD.
Methods: A group of patients with MDD and a healthy control group underwent a face-matching task during functional magnetic resonance imaging. Dynamic causal modelling (DCM) analysis was used to investigate effective connectivity between 7 regions in the face-processing systems. We used a Parametric Empirical Bayes model to compare effective connectivity between patients with MDD and controls.
Results: We included 48 patients and 44 healthy controls in our analyses. Both groups showed higher accuracy and faster reaction time in the shape-matching condition than in the face-matching condition. However, no significant behavioural or brain activation differences were found between the groups. Using DCM, we found that, compared with controls, patients with MDD showed decreased self-connection in the right dorsolateral prefrontal cortex (DLPFC), amygdala, and fusiform face area (FFA) across task conditions; increased intrinsic connectivity from the right amygdala to the bilateral DLPFC, right FFA, and left amygdala, suggesting an increased intrinsic connectivity centred in the amygdala in the right side of the face-processing systems; both increased and decreased positive intrinsic connectivity in the left side of the face-processing systems; and comparable task modulation effect on connectivity.
Limitations: Our study did not include longitudinal neuroimaging data, and there was limited region of interest selection in the DCM analysis.
Conclusion: Our findings provide evidence for a complex pattern of alterations in the face-processing systems in patients with MDD, potentially involving the right amygdala to a greater extent. The results confirm some previous findings and highlight the crucial role of the regions on both sides of face-processing systems in the pathophysiology of MDD.
Introduction
Major depressive disorder (MDD) is one of the most severe and prevalent mental illnesses, with significantly impaired social functioning.1–3 The processing of emotional human faces plays a vital role in social interaction.4 Elucidating the neural basis underlying the processing of emotional human faces in people with MDD can help improve our understanding of the neural mechanisms involved in abnormal social functioning in patients with the disorder.
In terms of face processing, both core and extended face-processing systems are important.5,6 The core system, including the occipital regions and fusiform face area (FFA), plays a crucial role in processing facial features and incorporating them into a holistic representation.7,8 On the other hand, the extended system, including the amygdala, dorsolateral prefrontal cortex (DLPFC), and ventromedial prefrontal cortex (VMPFC), is responsible for combining this fundamental information with higher-level functions.9–11 Therefore, it is crucial to examine the functional interaction among regions in both the core and extended face-processing systems.12 This will provide a more comprehensive understanding of how information is integrated to process emotional faces and how any abnormalities in the face-processing systems may manifest in patients with MDD.
Neuroimaging studies on emotional face processing in people with MDD have reported abnormal activity in brain regions within the core and/or extended face-processing systems, such as the amygdala, FFA, and DLPFC.13–16 The FFA engages in early perceptual processing of facial stimuli17,18 and is a major entry node in the cortical network that mediates face perception.19 Previous studies have documented an aberrant pattern of fusiform gyrus activation in patients with MDD when responding to different expressions in emotional processing tasks.14,20 The amygdala is a brain region that is sensitive to facial emotion stimuli.21 Previous studies commonly indicated abnormal amygdala activation in patients with MDD when processing facial expressions, such as happy, fearful, angry, sad, and even neutral expressions.22–24 Both the DLPFC and VMPFC appear to be important in integrating basic information with higher-order functions.11 The DLPFC is known to recruit cognitive control functions that are relevant to emotion regulation,25 and the VMPFC is more likely to play a mediatory role between the amygdala and DLPFC, serving as an interface between emotion and other aspects of cognition.26 Aberrant activation in the PFC has been observed in patients with MDD.27,28 However, inconsistent findings have also emerged, with some studies reporting no statistically significant differences in activation in specific regions, such as the amygdala, FFA, and PFC, between healthy and depressed individuals.29–31
Moreover, abnormal functional connectivity among the core and extended face-processing systems have also been reported in patients with MDD. For example, a study reported decreased amygdala functional connectivity with the fusiform gyrus through face-matching conditions in patients with depression compared with healthy participants.31 Several studies have identified decreased PFC–amygdala functional connectivity during emotional face processing in MDD.32–35 However, functional connectivity does not assess the directionality of these functional interactions. Therefore, it is unknown how the directionality of functional interactions among regions in the core and extended face-processing systems change in patients with MDD.
Dynamic causal modelling (DCM) is a popular method to measure effective connectivity.36 Using this method, researchers can determine the direction of influence and the valence (i.e., excitatory or inhibitory signalling) of the influence among regions. A few studies have investigated effective connectivity among regions in the core and extended face-processing systems in patients with MDD. Sacu and colleagues found decreased effective connectivity from the left amygdala and left DLPFC to the fusiform gyrus in patients with MDD compared with relatives and controls.37 However, the influence of antidepressants was not excluded in their study, and they found that these alterations did not survive when the effect of antidepressants was included. The use of antidepressants may be associated with more inhibitory connection from higher-order regions to the fusiform gyrus. Additionally, their study focused only on intrinsic connectivity, without considering the modulation effect of emotional face processing. In contrast, 2 other studies specifically recruited unmedicated patients to examine effective connectivity while excluding the confounding effect of antidepressant treatment. Jamieson and colleagues found a reduced negative modulation effect from the DLPFC to the VMPFC as well as reduced positive modulation from the DLPFC to the amygdala during the processing of sad faces in unmedicated adolescents and young adults (aged 15–25 yr) with depression.38 Another study reported lower intrinsic connectivity from the amygdala to the subgenual anterior cingulate cortex (sgACC) in the right hemisphere among unmedicated adolescents with MDD compared with healthy controls.39 While these 2 studies excluded the effect of antidepressant treatment on effective connectivity by recruiting unmedicated patients, it is important to consider that the findings were obtained in youth or adolescents with depression. Therefore, caution should be exercised when generalizing these findings to adults with MDD, as there may be developmental changes of the functional interaction between brain regions across the lifespan.40–42 Furthermore, in previous studies exploring effective connectivity among brain regions involved in face processing in patients with MDD, the DCM analysis typically focused on brain regions within the right hemisphere, 38,43 or the DCM model was constructed separately for each hemisphere.39 The deliberate emphasis on the right hemisphere was primarily motivated by concerns of insufficient activation in the left hemisphere.19 However, a previous study had shown that the left amygdala is more frequently activated than the right amygdala during emotion processing.44 Moreover, aberrant interactions have been observed in left-sided brain regions45 or in bilateral hemispheres in patients with MDD.37 These findings strongly suggest the need for further exploration of directed functional interactions between regions of interest (ROIs) in both hemispheres. Therefore, it is of great value to investigate the abnormal directional interactions among regions in the face-processing systems in unmedicated adults with MDD, taking into consideration both hemispheres within a single DCM model.
To investigate altered effective connectivity in the face-processing systems in patients with MDD, we enrolled a group of adults with MDD who were carefully screened to ensure they were either not taking any antidepressant medications or that they had taken antidepressants for no more than 7 days within the 14 days before enrolment. This criterion was implemented to minimize the confounding effect of antidepressants on effective connectivity and to ensure the enrolment of patients was feasible. We instructed them to complete a face-matching task while undergoing scanning. This task was designed to elicit emotional responses and activate face-related processing implicitly. In this study, we specifically examined the bilateral amygdala, bilateral DLPFC, bilateral FFA, and VMPFC, as these regions are essential components of the core and extended emotional face-processing systems. Using DCM and a recently developed Parametric Empirical Bayes (PEB) pipeline,46 we assessed differences in effective connectivity among these regions between patients with MDD and healthy controls. Based on previous literature, we hypothesized that aberrant interaction patterns in face-processing systems would be present in both hemispheres, and we expected to observe effective connectivity abnormalities in the prefrontal regions, amygdala, and FFA in patients with MDD.
Methods
Participants
Participants were recruited via public notices or referrals from their psychiatrists. Data collection took place between March 2021 and April 2022 at Beijing Anding Hospital. Eligible participants had to be native Chinese Han, between the ages of 18 and 45 years, right-handed, and have secondary education or above. Patients were assessed by clinicians using the MINI-International Neuropsychiatric Interview 7.0 (M.I.N.I. 7.0) based on Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-V) criteria for major depressive episodes. We also used the M.I.N.I. 7.0 to rule out any kind of pre-existing or concurrent Axis I disorder among healthy controls. The Hamilton Rating Scale for Depression (HAMD-17) and the Hamilton Rating Scale for Anxiety (HAM-A) were used to evaluate the severity of patients’ depression and anxiety, respectively. Only patients with scores higher than 17 on the HAMD-17 were enrolled in the study. Additional inclusion criteria for patients with MDD were either absence of any antidepressant medications, or duration of antidepressant treatment of no more than 7 days within the 14 days preceding enrolment; duration of the current episode less than 2 years; and absence of modified electroconvulsive therapy in the 6 months preceding enrolment. Individuals with any substance dependence (except for tobacco), who were pregnant, or who had metal implants in their bodies were ineligible to participate in the study. We asked all participants to complete the Patient Health Questionnaire (PHQ-9). Those who scored less than 5 on the PHQ-9 and had no personal or family history of mental illness were enrolled as healthy controls. All participants provided informed consent before study inclusion, and the ethics committees of Beijing Anding Hospital and Capital Medical University approved our study protocol.
Face-matching task
We used a face-matching task paradigm to investigate the neural correlates of implicit recognition of facial expressions. 47 During this task, participants were asked to match 1 of the 2 simultaneously presented images with the top image on the screen in trios of stimuli. In this task, there are 2 conditions: face-matching and shape-matching conditions. In the face-matching condition, the stimuli are greyscale photos of happy, angry, fearful, and sad faces, counterbalanced for gender, from the Chinese Facial Affective Picture System. In the shape-matching condition, the stimuli are 3 kinds of geometric shapes (triangle, rectangle, and circle). We used a block-design version, in which 1 block contained 6 trials. There were 12 face-matching blocks in the following order: fearful, happy, angry, sad, fearful, angry, happy, angry, sad, fearful, sad, happy. Meanwhile, the other 12 blocks of shape-matching were interspersed in the face-matching blocks. From the beginning of each block, there was a short introduction (3 s) followed by 6 trials. During each trial, there were 2 seconds for matching and 1 second for an interval. The total experiment lasted 514 seconds, including a 6-second preparing time, and a 4-second ending time. We differentiated only the neural mechanism of face-matching and shape-matching; we did not explore the effects of valence of emotional faces on effective connectivity.
Behavioural and characteristic analysis
Differences across groups and conditions for reaction times and accuracy rate were assessed separately using analysis of variance (ANOVA) with condition (face-matching v. shape-matching) as a within-subjects factor and group (patients v. controls) as a between-subjects factor. Owing to the non-normal distribution of the percentage of correct responses, we transformed results into z-scores, then we calculated the main effects and interactions of the 2 factors.
We performed 2-sample t tests and χ2 tests to compare the demographic characteristics and PHQ-9 score between the groups.
Image acquisition and preprocessing
All participants underwent T1-weighted structural scans, field map images and task-based functional MRI (fMRI). The images were collected using a 3.0 T Siemens MAGNETOM Prisma MR D13D scanner at Beijing Anding Hospital. The task was presented using E-Prime 2.0 and was projected onto a flat screen mounted in the scanner bore. Participants viewed the screen using a mirror mounted on a 64-channel head coil.
Preprocessing was implemented in DPABI V6.1,48 including field map correction, slice timing, realignment, normalization, and smoothing. For a more detailed description of image acquisition parameters and preprocessing, see Appendix 1, available at https://www.jpn.ca/lookup/doi/10.1503/jpn.230123/tab-related-content.
Brain activation analysis
After completing the preprocessing procedures, we performed general linear model (GLM) analysis using Statistical Parametric Mapping 12 (SPM12; available at www.fil.ion.ucl.ac.uk/spm) to assess the brain responses at the individual level and group level, concerning the task conditions (see Appendix 1 for more details).
ROI selection and time-series extraction
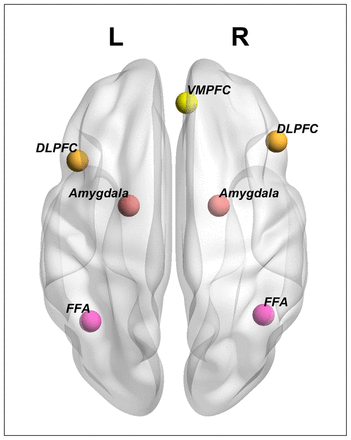
We selected the FFA, amygdala, DLPFC, and VMPFC as the ROIs, in accordance with previous studies using face-matching tasks.37 All ROIs were bilateral, except the VMPFC (Figure 1). The bilateral FFA, amygdala, and DLPFC were identified by the face-matching > shape-matching contrast across groups, whereas the VMPFC was identified by the inverse contrast across groups.12,49 We first generated anatomic masks for each ROI. Then, we searched for the local maxima within the anatomic mask in the activation map of each participant to ensure that regional time series fell within the anatomic boundaries of a given ROI. Subsequently, we created a 6 mm radius sphere centred at each local maxima for each ROI. Furthermore, we performed an intersection of the suprathreshold of individuals’ activation maps, the anatomic mask, and the 6 mm sphere to extract the time series for the ROI of each participant.37,50 In cases when the subject-specific maximum could not be found within the predefined region, we extracted the time series of the ROI from a 6 mm radius sphere centred at the group-level maximum.50 To create the anatomic masks of the bilateral amygdala, bilateral DLPFC, and VMPFC, we used the Anatomic Automated Labelling (AAL) library in the Wake Forest University (WFU) PickAtlas toolbox (https://www.nitrc.org/projects/wfu_pickatlas/). However, for the FFA, we found that the anatomic mask in AAL contained an excessive number of voxels, not all of which were associated with face processing. Therefore, we drew a 10 mm radius sphere centred at the coordinate derived from the maximum of the meta-analytical coactivation map, which was generated by searching “fusiform face area” on Neurosynth (https://neurosynth.org/). This sphere served as the anatomic mask for the FFA (see Appendix 1 for more details).
Seven regions of interest centred on participant-level peak coordinates across groups. Render visualized using BrainNet Viewer. DLPFC = dorsolateral prefrontal cortex; FFA = fusiform face area; L = left; R = right; VMPFC = ventromedial prefrontal cortex.
Dynamic causal modelling
Effective connectivity was conducted using the DCM 12.5 implementation in SPM12. Dynamic causal modelling treats the brain as a deterministic, nonlinear, dynamic system that receives inputs and produces outputs.36 It aims to estimate and make inferences about effective connectivity among brain areas and how that effective connectivity is influenced by changes in an experimental context. In a DCM, there are 3 matrices: A, B, and C. Parameter matrix A specifies the average effective connectivity across experimental conditions and thus reflects how the effective connectivity behaves intrinsically, independent of task modulation. Parameter matrix B specifies the modulation of effective connectivity due to experimental conditions. In both matrices A and B, the off-diagonal elements represent between-region connectivity, in hertz. The values reflect the influence one region has on another, with positive values indicating excitation and negative values indicating inhibition.51 The diagonal elements of the matrices represent self-connections, which implies self-inhibition regulating a region’s response to its inputs and maintaining the balance between excitation and inhibition.50 The self-connections are unitless log scaling parameters that scale the default value of −0.5 Hz. Positive values of self-connections indicate stronger self-inhibition in response to stimuli, leading to decreased sensitivity to inputs from the rest of the network. Negative values of self-connections indicate reduced self-inhibition (i.e., disinhibition) in response to stimuli, resulting in increased sensitivity to inputs from the network.51 We set up a fully connected model of intrinsic connectivity about matrix A, representing all connections between selected regions and self-connections within regions. We entered task conditions into the model as modulatory inputs on each region’s self-connection by setting the prior parameters in the diagonal elements of matrix B, similar to previous studies.50,51 Zeidman and colleagues51 put forward that limiting modulatory effects to the self-connections, rather than including the between-region connections, adds biological interpretability (as changes in the excitatory–inhibitory balance of each region) and generally improves parameter identification. Additionally, our initial analysis showed a lower absolute value of free energy when matrix B included only self-connection, compared with a model with modulatory inputs on all connections. Therefore, we focused on self-connections in matrix B. Finally, matrix C represents the driving inputs. In previous studies, FFA was considered to be a driven input region influenced by various visual stimuli, including faces and shapes, while the amygdala was recognized as part of the visual pathway, especially involved in processing facial expressions.12,52 Therefore, we entered the bilateral FFA and bilateral amygdala into matrix C for the effect of face-matching, but we entered only the bilateral FFA for the effect of shape-matching. We conducted diagnostic tests to verify the quality of the DCM fit and to confirm the success of the model inversion using the spm_dcm_fmri_check function. We excluded participants whose variance was less than 10% from further analyses according to previous studies.37,53,54
PEB model for group DCM
After the estimation of DCM models, group effects on the DCM parameters were analyzed using the PEB model to find commonalities and differences between healthy controls and patients with MDD.55 For a detailed description of the PEB model, see Appendix 1.
The specific procedures of the PEB used in this study are as follows: the PEB was specified for each field (A, B, C) separately; the design matrix specification was set to define the hypotheses about commonalities and group differences (controls v. MDD); and Bayesian Model Reduction (BMR) was used with an automatic search form to test the estimated PEB models. Results of PEB were reported based on the posterior probability (free energy with v. without parameter) greater than the 0.95 criterion.
Results
A total of 121 participants (65 patients with MDD, 56 healthy controls) responded to the public recruitment notices or were referred by their psychiatrists to participate in this study. We ruled out 11 controls and 17 patients with MDD owing to some unexpected data conditions (Appendix 1), leaving 92 participants (48 patients, 44 controls) for inclusion in the subsequent behavioural and imaging analyses. Table 1 shows the demographic and clinical characteristics of the MDD and healthy control groups.
Demographic and clinical characteristics of the study sample
Sample and behavioural data
There were no significant differences between healthy controls and patients with MDD in terms of age, sex, education level, or tobacco use; however, PHQ-9 scores differed significantly between the groups (Table 1).
Regarding the accuracy rate and reaction time, the main effects of the condition were both significant (F = 39.245, p < 0.001 and F = 1363.143, p < 0.001, respectively); however, the group effects were not significant. The interactions between condition and group were also nonsignificant. Specifically, the shape-matching condition had a higher percentage of correct responses across all participants (controls: 99% ± 1%; MDD: 99% ± 1%) than face-matching (controls: 97% ± 4%; MDD: 97% ± 3%). Reaction times were similar, with shape-matching having shorter reaction times (controls: 620.83 ± 78.52 ms; MDD: 628.23 ± 77.66 ms) than face-matching (controls: 1143.33 ± 153.33 ms; MDD: 1149.85 ± 155.75 ms).
Task-related brain activation
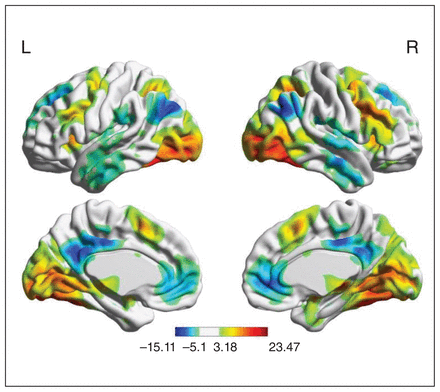
In the face-matching > shape-matching contrast, we observed brain activation in the bilateral visual cortices (bilateral lingual, calcarine, and fusiform gyrus, and occipital areas), sensorimotor cortex (superior motor area, precentral and postcentral cortex), inferior and middle PFC, precuneus, thalamus, superior and inferior parietal lobule, middle and inferior temporal areas, as well as the bilateral amygdala. We observed significant deactivation in the medial PFC, middle and postcingulate cortex, precuneus, angular gyrus, inferior parietal cortex, and middle occipital gyrus across groups (Figure 2 and Appendix 1, Table S3).
Regions showing a main effect of task condition across groups (p < 0.05, whole brain voxel-wise family-wise error correction). Colours indicate T statistics. Warm colours represent greater activation in the face-matching condition, and cool colors represent greater deactivation in the face-matching condition compared with the shape-matching condition. L = left; R = right.
Moreover, we found decreased activation across conditions (face-matching and shape-matching) in 2 clusters in patients with MDD. One cluster encompassed the right insula and right inferior frontal gyrus, and the other cluster involved the left inferior frontal gyrus, left postcentral gyrus and left precentral gyrus (voxel-wise p < 0.001, cluster-wise family-wise error [FWE] p < 0.05) (Appendix 1, Table S3).
There were no significant differences in activation pattern between the control and MDD groups throughout the face-matching > shape-matching contrast, or the face-matching < shape-matching contrast when controlling for age, gender, and education level. We also explored group differences in brain activity in the 7 ROIs, and we did not observe evidence for a differential response between the groups in these ROIs (Appendix 1, Table S4).
Effective connectivity
The overall structure of the DCM included all connections (intrinsic connectivity) within matrix A, self-connections within matrix B, and the neural inputs within matrix C. We found 5 participants (4 controls, 1 patient with MDD) whose explained variance was less than 10%, so we eliminated these participants from the analysis and fitted the data from the remaining 87 participants into the PEB model to identify the commonalities across participants and group differences in effective connectivity.
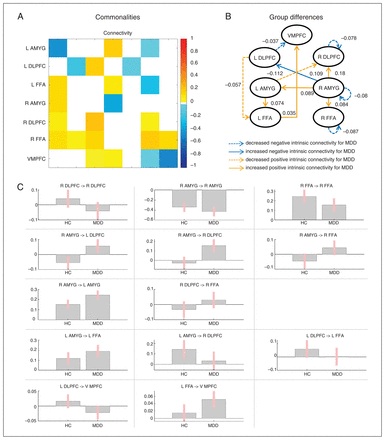
Concerning the commonalities, in matrix A we observed positive (excitatory) intrinsic connectivity from the left amygdala to the left FFA, right amygdala, right DLPFC, and right FFA; from the left DLPFC to the right FFA; from the left FFA to the left DLPFC, right DLPFC, right FFA and VMPFC; from the right amygdala to the left amygdala and left FFA; from the right FFA to the right DLPFC; and from the VMPFC to the right FFA (Figure 3A). Conversely, we observed negative (inhibitory) intrinsic connectivity from the right DLPFC to the left DLPFC; and from the right FFA to the left amygdala, left FFA, and VMPFC. Additionally, we observed self-connection in the left amygdala, left DLPFC, right amygdala, right FFA, and VMPFC in both the MDD and control groups. More importantly, we found group differences after PEB model reduction, and only a restricted set of parameters survived model reduction. Figure 3B and C shows differences in intrinsic connectivity between patients with MDD and healthy controls. Specifically, patients with MDD showed decreased self-connection in the right DLPFC, right amygdala, and right FFA. It is important to note that these parameters are presented on a logarithmic scale, where a negative difference indicates decreased self-inhibition, implying a relative disinhibition in patients.52 Thus, this finding suggests a relative disinhibition in the right side of the face-processing systems in patients with MDD. The patients with MDD also showed increased intrinsic connectivity from the right amygdala to the bilateral DLPFC and the right FFA (negative connectivity in the controls, but positive connectivity in patients with MDD) as well as from the right amygdala to the left amygdala (increased positive connectivity in patients with MDD), suggesting an increased intrinsic connectivity centred in the amygdala in the right side of the face-processing systems. Finally, patients with MDD showed increased positive intrinsic connectivity from the left amygdala to the left FFA, and from the left FFA to the VMPFC, and decreased positive intrinsic connectivity from the left DLPFC to the left FFA and VMPFC, and from the left amygdala to the right DLPFC, suggesting a mixed abnormality in the left side of the face-processing systems.
Findings from matrix A. (A) The group mean across all participants. (B) Connections with strong evidence for group differences in the reduced model. (C) Bars graphs for each of connections showing evidence of group differences on matrix A. The vertical axis represents the connection strength. The self-connections in the model are characterized by log scaling parameters, which can be transformed into hertz using the following equation: y = −0.5 Hz × exp(x), where x represents the log scaling parameter, −0.5 Hz serves as the reference value, and y represents the strength of the self-connection measured in hertz. AMYG = amygdala; DLPFC = dorsolateral prefrontal cortex; FFA = fusiform face area; HC = healthy controls; L = left; MDD = major depressive disorder; R = right; VMPFC = ventromedial prefrontal cortex.
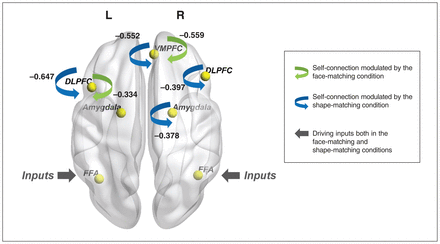
In matrix B, where only task modulations on self-connections were considered in the model, we found commonalities across groups but no differences between groups (Figure 4). Across groups, the face-matching condition modulated the self-connections in the left DLPFC and VMPFC; however, the shape-matching condition modulated the self-connections in the bilateral DLPFC, right amygdala, and VMPFC. We noted that the parameters were negative, indicating there was a reduction in self-inhibition (i.e., disinhibition) in response to the stimuli and thus an increased sensitivity to inputs from the network. We did not find strong evidence to support any group differences between patients with MDD and healthy controls in the modulatory effect of task conditions on self-connections.
Findings from matrix B and matrix C. The blue arrows indicate the brain regions whose self-connections were modulated by the face-matching condition, and the green arrows indicate the brain regions whose self-connections were modulated by the shape-matching condition in matrix B. The parameters that survived the threshold of posterior probability > 95% are presented. The grey arrows indicate the brain regions in which the task stimuli entered the model as a driving input region both in the face-matching and shape-matching conditions in matrix C. DLPFC = dorsolateral prefrontal cortex; FFA = fusiform face area; L = left; R = right; VMPFC = ventromedial prefrontal cortex.
In matrix C, only the bilateral FFA was considered as an input area for both the face-matching and shape-matching conditions (Figure 4). This suggests that the bilateral FFA showed sensitivity to driving inputs regardless of task conditions. However, no input was observed in the bilateral amygdala, whether during the face-matching condition or the shape-matching condition.
Discussion
By leveraging recent advancements in DCM, we found that, compared with healthy controls, patients with MDD showed aberrant effective connectivity in both sides of the face-processing systems, composed of the VMPFC and the bilateral FFA, amygdala, and DLPFC, while performing a face-matching task. The main findings suggest that patients with MDD had decreased self-inhibition in the right FFA, amygdala, and DLPFC; increased intrinsic connectivity centred in the amygdala in the right side of the face-processing systems; and a mixed abnormality in intrinsic connectivity in the left side of the face-processing systems. These findings highlight the altered effective connectivity within the face-processing systems in patients with MDD, providing insight into the neural mechanisms underlying the disorder. Importantly, these alterations were observed irrespective of task conditions.
Similar to previous studies using a similar task,31,37,56,57 we did not observe significant differences in behavioural performance between the groups. The lack of between-group differences in our behavioural results may be attributed to a ceiling effect associated with the task. However, it is worth noting that several studies have reported contrasting findings. For example, Fisher and colleagues29 found that patients with MDD had slower reaction times in the face-matching condition and higher accuracy in the shape-matching condition than healthy controls. We believe that the large sample size in their study (89 depressed and 115 healthy participants) contributed to these positive findings, as the numerical values of behaviour performance were comparable to ours. Furthermore, Nagy and colleagues58 found that patients with MDD who had a history of childhood maltreatment were significantly less accurate in matching facial emotions, especially when matching sad faces. This finding suggests that specific experience of childhood adversity may contribute to impairments in facial emotion recognition. Based on these observations, we speculate that the impairment in facial emotion recognition in patients with MDD may be subtle and detectable only with a large sample size or in patients with a specific subtype of MDD.
In our study, no significant group differences in brain activity were observed when contrasting the face-matching condition with the shape-matching condition. Our findings differ from those of previous studies that reported abnormal brain activity in regions such as the amygdala, FFA, and DLPFC.13–16 However, it is worth noting that several prior studies that examined the contrast between face-matching and shape-matching without focusing on specific emotions, also reported negative findings, even with larger sample sizes.29,31 Additionally, a meta-analysis investigating emotional processing tasks in individuals with depression and controls also indicated a lack of substantial differences in brain activation.30 We believe that these discrepancies may be attributed to variations in experimental design, the specific contrasts used, or the heterogeneity of clinical populations involved.
Previous studies have overlooked the significance of self-connections in the face-processing systems in patients with MDD. In this study, we found decreased self-connections of intrinsic connectivity in the right side of the face-processing systems, including the right amygdala, right DLPFC, and right FFA, in patients with MDD. From a biological perspective, these parameters of self-connections can be interpreted as controlling the region’s excitatory–inhibitory balance, which is mediated by the interaction of pyramidal cells and inhibitory interneurons.59 A more negative self-connection parameter indicates a lower level of inhibition (i.e., relative disinhibition) within the region, allowing for increased responsiveness to inputs.36,51 In line with this, our findings suggest a relative disinhibition across conditions in the right side of the face-processing systems in patients with MDD, suggesting a potential disruption in the excitatory–inhibitory balance within these regions. These findings align with accumulating evidence highlighting the involvement of imbalances in the γ-aminobutyric acid (GABA) neurotransmitter and glutamate in the pathophysiology of MDD, particularly through disturbances in the excitation–inhibition balance within the PFC and basic sensory cortices.60,61
We observed increased effective connectivity across conditions from the right amygdala to the bilateral DLPFC, right FFA, and left amygdala. The amygdala is involved in the appraisal of emotional stimuli and plays a crucial role in facilitating perceptual processing by directing or biasing attention. 62,63 Previous studies have highlighted the significant involvement of the right amygdala, which might trigger an autonomic activation outside of conscious awareness in bottom–up processing.64,65 Many studies have reported abnormal frontolimbic functional connectivity in patients with MDD during emotional face processing.32–35 Furthermore, using DCM analysis, Willinger and colleagues reported reduced intrinsic connectivity from the sgACC to the amygdala in adolescents with MDD,43 while Sacu and colleagues observed no PFC–amygdala alterations in patients with MDD.37 In our study, we did not observe any aberrant connections from the PFC to the amygdala; however, our findings suggest that in patients with MDD, intrinsic connectivity from the amygdala to the DLPFC was increased in response to perceptual stimuli, beyond just facial context. This highlights the significance of bottom–up circuits in overall processing, potentially associated with automatic attention mechanisms.
Moreover, several studies have shown that the amygdala can modulate the visual encoding of socially charged and salient information in the FFA.66,67 Notably, the FFA has consistently shown aberrant connectivity patterns with other regions during emotion processing in patients with MDD.57,68 In the study by Sacu and colleagues, a decreased effective connectivity (interpreted as increased inhibition) from the left amygdala to the right fusiform gyrus was reported in patients with MDD, resulting in diminished attention toward visual stimuli.37 However, our study showed increased positive effective connectivity from the right amygdala to the right FFA and from the left amygdala to the left FFA, reflecting heightened synchronization of visual inputs and attentional processes. Both studies suggest that the altered modulation of fusiform gyrus activity by the amygdala may contribute to atypical attentional processing of stimuli in patients with MDD. The observed discrepancy may be attributed to variations in the MDD populations under investigation. In contrast to the study by Sacu and colleagues, where approximately 70% of recruited patients with MDD were taking antidepressant medication,37 our study specifically focused on unmedicated patients or those who had taken antidepressants for no more than 7 days within the 14 days preceding enrolment. Moreover, the increased intrinsic connectivity from the right amygdala to other brain regions probably reflects higher excitability of the right amygdala in patients with MDD, which is consistent with the observation of weakened self-inhibition within the right amygdala.
Previous studies using DCM analysis to explore neural differences during facial emotion processing between individuals with MDD and healthy controls often constrained the regions of interest to the right side of the brain;12,38,43 however, in our study, we also observed aberrant effective connectivity in the left side of the face-processing systems in patients with MDD, including decreased connectivity from the left DLPFC to the left FFA and the VMPFC, as well as increased positive connectivity from the left amygdala to the left FFA, and from the left FFA to the VMPFC. During the processing of affectively significant items, the amygdala can enhance sensory processing not only from a subcortical source of visual input (amygdala–visual cortex), but also from its broad connectivity with the cortex (amygdala–PFC–visual cortex).69 The DLPFC, on the other hand, is known for its capacity to regulate the activation of sensory and association cortices.70 Based on these functions, we hypothesized that increased positive connectivity observed from the left amygdala to the left FFA could indicate a greater focus on generic stimuli in patients with MDD, whereas the decreased positive connectivity from the left DLPFC to the left FFA in patients with MDD may be related to less regulation of attention. However, it is worth noting that Sacu and colleagues reported increased inhibitory influence from the lateral prefrontal regions to the fusiform gyrus in patients with MDD.37 As previously discussed, the inconsistent findings between our study and that of Sacu and colleagues may be attributed to variations in medication usage within the MDD populations. It is possible that the use of antidepressants could be associated with a greater inhibitory connection from higher-order regions to the fusiform gyrus. As for the VMPFC, it plays a vital role in organizing the physiologic components of negative emotions through its projection to the hypothalamus and amygdala.71 Moreover, it is widely acknowledged that the VMPFC, coordinating with the DLPFC, modulates outputs from subcortical limbic structures.72 Therefore, the decreased connectivity between the left DLPFC and the VMPFC may indicate weaker integral modulation of lower-level cortical structures, such as limbic and visual regions. Furthermore, the VMPFC has also been found to contribute to more rapid, precursory stages of social and emotional stimuli,73 and its damage may impair visual attention to faces.74 As a result, we speculated that increased intrinsic connectivity from the left FFA to the VMPFC might reveal stronger visual perception and emphasize the important role of the VMPFC in the extended face-processing system in patients with MDD.
Finally, we found modulation effects on the self-connections within the left DLPFC and the VMPFC in the context of face-matching, and on the self-connections within the bilateral DLPFC, right amygdala, and VMPFC in the context of shape-matching. However, we did not identify any evidence to support group differences in the effect of task conditions. The lack of group differences in the task modulation effect suggests that the self-connections of these regions were modulated by the task conditions to a similar extent between patients with MDD and healthy controls. This finding does not contradict those on intrinsic connectivity, which suggests the intrinsic connectivity did show a between-group effect. The same modulatory effect would have a larger or smaller consequence in each group, depending on the baseline connectivity.50 Similar to our findings, previous studies reported no differences in modulation effect between patients with MDD and healthy controls.39 However, it is worth noting that a previous study reported aberrant regulatory connections in patients with MDD, specifically reduced negative modulation from the DLPFC to the VMPFC and reduced positive modulation from the DLPFC to the amygdala during the processing of sad faces.38 We believe that the discrepancy between our findings and those of this previous study can be attributed to the difference in the specific cognitive processes under investigation. The previous study focused primarily on the functional aspects of specific emotional valence, whereas we examined the modulation of face processing, regardless of emotional valence. Future studies should pay more attention to biased emotional processing by quantifying patterns of behaviour and abnormal neural activation/connectivity across different emotion conditions or abnormal responses to neutral faces.
Limitations
Our study had several limitations. First, as it was a cross-sectional study, we could not determine whether the alterations observed could predict or be influenced by specific types of therapy, such as antidepressants or psychotherapy. For instance, Vai and colleauges observed effective connectivity between the amygdala, ACC, and ventrolateral PFC (VLPFC) during a gender discrimination task before treatment could successfully account for the antidepressant response.75 Thus, future studies should aim to replicate the present findings and assess their potential as biomarkers for prediction of prognosis by conducting longitudinal research. Second, it may be beneficial to incorporate additional ROIs in the construction of DCMs to identify further abnormalities in patients with MDD. For example, the superior temporal sulcus (STS) is known to play a crucial role in normal face processing, alongside the FFA.76,77 Therefore, in future studies, it would be useful to include more regions associated with face processing, including the STS, as ROIs to gain a comprehensive understanding of the altered functional interactions in patients with MDD. Third, we enrolled the participants who took antidepressants for no more than 7 days within the 14 days preceding their enrolment to minimize the potential effect of antidepressants on brain function.78 However, it is important to note that the precise onset of antidepressant action remains uncertain,79,80 and it is unclear whether brain function may be affected during a specific time frame. In future studies, researchers can consider keeping records of the antidepressants taken by the patients to ensure operational feasibility or recruit only individuals who are medication-naïve to rule out the potential effect of antidepressants. Fourth, it is important to acknowledge that the moderate sample size in this study may not be representative of the entire population. Future studies should aim to include a larger number of participants to replicate and validate the current findings.
Conclusion
Our study showed that patients with MDD had altered effective connectivity within the face-processing systems, as evidenced by decreased self-inhibition in the right FFA, amygdala, and DLPFC; increased intrinsic connectivity on the right side; and a mixed alteration on the left side of the face-processing systems. Notably, we did not find any significant group differences in behaviour or activation patterns when contrasting the face-matching condition with the shape-matching condition between patients with MDD and healthy controls. However, the DCM analysis revealed group differences in intrinsic connectivity across conditions, rather than modulation effect of task conditions on effective connectivity. Our findings suggest that patients with MDD may show altered general information processing rather than specific abnormalities in face processing. These findings provide further neuroimaging evidence for a complex pattern of alterations among face-processing systems in patients with MDD, with a potential emphasis on greater involvement of the right amygdala. Our findings support and expand on previous research, highlighting the importance of investigating both sides of face-processing systems to understand the underlying pathophysiology of MDD.
Acknowledgements
The authors extend their gratitude to all the study participants and their families and friends.
Footnotes
Competing interests: None declared.
Contributors: Ling Zhang, Yuan Zhou, and Gang Wang designed the study. Yun Wang, Ruinan Li, Xiaoya Li, Xiongying Chen, Zhifang Zhang, Rui Liu, and Ling Zhang acquired the data, which Fangrui Sheng and Yun Wang analyzed. Fangrui Sheng wrote the article, which all authors reviewed. All authors approved the final manuscript to be published and agreed to be accountable for the work.
Funding: This work was supported by the Beijing Municipal Science & Technology Commission (grant no. Z181100001518005); the National Natural Science Foundation of China (grant nos. 82101597 and 82071531); the Beijing Hospitals Authority Youth Programme (grant no. QML20231901); and the Beijing Scholar 2021 (grant no. 063).
- Received August 26, 2023.
- Revision received January 11, 2024.
- Revision received February 22, 2024.
- Accepted February 24, 2024.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/