Abstract
Background: Hemispheric lateralization of the brain occurs during development and underpins specialized functions. It is posited that aberrant neurodevelopment leads to abnormal brain lateralization in individuals with psychotic illnesses. Here, we sought to examine whether white matter hemispheric lateralization is abnormal in individuals with the psychotic spectrum disorders of schizophrenia and bipolar disorder.
Methods: We examined the white matter microstructure lateralization in patients with schizophrenia, bipolar disorder with psychotic features and healthy controls by measuring the laterality indices of fractional anisotropy (FA) and mean diffusivity (MD). We also correlated the laterality indices with clinical measures.
Results: We included 150 patients with schizophrenia, 35 with bipolar disorder and 77 healthy controls in our analyses. Shared FA lateralization abnormalities in patients with schizophrenia and bipolar disorder were found in the cerebral peduncle and posterior limb of internal capsule, with more extensive abnormalities in patients with bipolar disorder than in those with schizophrenia. The shared MD lateralization abnormalities were more widespread, extending to the subcortical, frontal–occipital, limbic and callosal tracts, with patients with bipolar disorder showing greater abnormalities than patients with schizophrenia. While lateralization was decreased in patients with schizophrenia, the lateralization was reversed in those with bipolar disorder, underpinned by the more pronounced microstructural abnormalities in the right hemisphere. The loss of FA lateralization in patients with schizophrenia was associated with lower quality of life and psychosocial functioning.
Limitations: Owing to the cross-sectional study design, we cannot confirm whether the lateralization abnormalities are neurodevelopmental or a consequence of psychosis onset or chronicity.
Conclusion: Shared and distinct white matter lateralization abnormalities were found in patients with schizophrenia and bipolar disorder. In distinct regions of abnormalities, the lateralization was attenuated in patients with schizophrenia and reversed in those with bipolar disorder.
Introduction
The hemispheres of the human brain are anatomically and functionally asymmetric.1,2 The hemispheric lateralization is important for motor, language, perception and emotional processes.1,3 Hemispheric lateralization occurs early during the perinatal stages and is genetically associated within families.4 As such, the loss of hemispheric lateralization has long been proposed to be a consequence of disrupted neurodevelopment in individuals with psychotic disorders.2,4,5 The interest in hemispheric lateralization in individuals with psychosis dates back more than 3 decades, with the field dominated by theories of preferential left hemisphere dysfunction in people with schizophrenia and right hemisphere dysfunction in those with bipolar disorder (BD).6
The advent of noninvasive neuroimaging MRI techniques ushered in a series of structural and task-based fMRI studies in patients with schizophrenia that demonstrated loss of leftward volume asymmetry of specific brain regions involved in auditory and language processing.2,7,8 More recent cross-modal evidence has also indicated a graded loss of leftward asymmetry in functional connectivity measures of these language processing areas in patients with schizophrenia and their first-degree relatives compared with controls.9 The loss of asymmetry in white matter microstructure in patients with schizophrenia has also been reported.10–12 In patients with BD, neuroimaging evidence of preferential hemispheric abnormalities has been reported as well, although findings are not as conclusive; many converging studies of metabolism, task-based fMRI and white matter revealed preferential right hemispheric disturbances, whereas other studies found left hemispheric disturbances in patients with BD.13–18
Clinically, the boundaries between schizophrenia and BD can be blurred early in the illness and even over time.19,20 Given the overlap of psychotic symptomatology and risk gene loci,21,22 direct comparisons of hemispheric lateralization in patients with schizophrenia and BD can yield insight into common and different pathophysiology underlying the shared and unique clinical features of these disorders. More specifically, if we build on the assumption that hemispheric abnormalities reflect neurodevelopmental aberrancy in individuals with psychosis,2 then shared lateralization abnormalities in neural substrates across schizophrenia and BD could represent a trait rather than a state feature of psychosis.
Neuroimaging studies that make direct comparisons of hemispheric lateralization in patients with schizophrenia and BD remain limited. An earlier MRI study of regional cerebral volume asymmetry across the psychosis spectrum disorder indicated a continuum of asymmetry abnormalities.23 An fMRI study of patients with psychotic depression, psychotic BD and schizophrenia found cross-diagnostic decreases in language network lateralization across the patients with psychosis compared with healthy controls.24 Another fMRI study found reduced lateralization in language networks in patients with schizophrenia but not in those with BD; no grey matter volume differences in the language networks were found between patients with schizophrenia and those with BD.25 To our knowledge, no dedicated study comparing hemispheric lateralization of the white matter microstructure in patients with schizophrenia and BD has been performed to date.
White matter structures are largely composed of axonal fibres and myelin sheaths, which wrap around the axons to facilitate quick and efficient transfer of electrical impulses. White matter degradation or destruction, such as the loss of myelin and axonal damage, could hence affect the transfer of nerve impulse between distributed brain regions. Diffusion tensor imaging (DTI) provides information on the diffusion properties of white matter microstructure (i.e., the translational displacement of water molecules in the thousands of axonal fibres and myelin contained within each image voxel).26 Fractional anisotropy (FA) is the most widely used DTI index to capture white matter microstructural changes.26 For instance, reduced FA may reflect local changes, such as thinner myelin sheaths, or changes in connectivity, such as decreased density of axonal fibres or decreased coherence/increased crossing fibres.27 Two other indices, axial diffusivity (AD) and radial diffusivity (AD), complement FA measures; they have been suggested to reflect axonal and myelin disruptions, respectively.15,28–30 Another widely used DTI index, mean diffusivity (MD), is used to quantify the average magnitude of diffusivity in a particular voxel or region in all directions. Increased MD may indicate axonal disruptions due to degeneration, myelin degeneration or edema (increase in tissue swelling).26
The DTI studies directly comparing white matter between patients with schizophrenia and BD have so far yielded inconsistent findings, with several studies showing no differences31–34 and others finding distinct differences between the disorders.15,35,36 The inconsistent findings could be explained by participant inhomogeneity. One study focused on first-episode patients,15 while others included patients with BDI with and without psychosis,33,35 or did not specify whether the participants with BD had history of psychosis.36 The DTI methodologies among these studies vary too, including voxel-based analysis of all white matter voxels of the whole brain,15,36 whole brain voxel-based approach followed by a priori regions of interest (ROI) analyses31,32,33 and an ROI approach.35 A recent study — the only study to our knowledge that examined shared white matter abnormalities in patients with schizophrenia and BD34 — adopted tract-based spatial statistics (TBSS), a whole-brain voxel-based approach focusing on the major white matter tracts that are common and created from the cohort studied.37
Hence in the present study, we sought to examine 3 hypotheses using complementary white matter diffusion measures in our cohort of patients with schizophrenia, patients with BD with a history of psychosis and healthy controls. First, we sought to identify whether there is a common feature of disrupted lateralization in the white matter tracts of patients with schizophrenia and BD. We hypothesized lateralization abnormalities in the white matter tracts of the language networks, which are functionally abnormal across psychotic disorders24,38 (i.e., the fronto–temporal–occipital associational fibres),39 as well as subcortical regions of the external capsule and posterior limb of the internal capsule, which were shown to be attenuated in a dedicated study of white matter lateralization in patients with schizophrenia.12 Second, based on findings of left hemispheric abnormalities in a meta-analysis of voxel-based studies of schizophrenia,40 the earlier lesion and functional findings of preferential left hemispheric dysfunction in patients with schizophrenia and preferential right hemispheric dysfunction in patients with BD,6 which were echoed in more recent neuroimaging studies of first-degree relatives of patients with schizophrenia41 and BD,42 we hypothesized differential patterns in white matter hemispheric lateralization abnormalities in patients with these disorders. Third, we hypothesized an association between white matter lateralization abnormalities and the severity of psycho-pathology in patients with schizophrenia and BD. As hemispheric lateralization subserves higher-level cognition, we predicted that the white matter lateralization abnormalities would also be associated with functional indices of daily functioning and quality of life.
Methods
Participants
Our cross-sectional study sample included patients with schizophrenia, patients with BD with psychotic features and matched healthy controls. The schizophrenia and BD samples were clinically stable outpatients recruited at the Institute of Mental Health, Singapore. All diagnoses were confirmed by a psychiatrist (K.S.) using information obtained from the existing medical record, clinical history, mental status examination, interviews with the patients and their relatives as well as the administration of the Structured Clinical Interview for DSM-IV disorders — Patient Version (SCID-I/P).43 We recruited demographically matched healthy controls within the hospital community and affiliated academic centres as well as through word of mouth. We screened controls by administering the SCID — Non-Patient Version (SCID-I/NP).44 To be included in our study participants had to have no history of head trauma or neurologic disorders, no current or history of substance use or alcohol use disorder, and no contraindications for MRI scans and major medical conditions.
The Institutional Review Boards of the Institute of Mental Health, Singapore, and the National Healthcare Group, Singapore, approved this study. We obtained written informed consent from each participant following a detailed explanation of the study procedures.
Clinical and quality of life measures
We used the Positive and Negative Syndrome Scale (PANSS) to assess the severity of psychotic phenomenology,45 the Young Mania Rating Scale (YMRS) to assess symptoms of mania in patients with BD46 and the Global Assessment of Functioning (GAF) scale to rate the patients’ general level of psychosocial functioning.47 In addition, the participants filled in a self-rated World Health Organization Quality of Life Assessment — Brief Form scale that reflected their quality of life (QOL) in the social, psychological, physical and environmental domains.48 In cases where participants had missing quality of life measures, their data were excluded from subsequent neuroimaging clinical correlational analyses.
Image acquisition and derivation of white matter lateralization indices
We obtained diffusion-weighted images from all participants using the same 3 T scanner (Philips Achieva). All preprocessing and analyses were performed using FSL (www.fmrib.ox.ac.uk/fsl). The DTI data were preprocessed using similar steps as those used in our previous studies.49,50 We then used TBSS to carry out a voxel-wise analysis of the DTI metrics (FA, MD, AD and RD) of the “skeleton” (i.e., major white matter pathways throughout the whole brain) of each individual.37 The diffusion-based laterality indices for each individual were generated from the original skeleton (asymmetric) and the left side of the skeleton that was flipped to the right (symmetric).51 The laterality index (LI) was calculated as follows: LI = (original – flipped) ÷ (original + flipped).
As a secondary analysis, we created an asymmetry index (i.e., images of the left – right hemisphere). Further details can be found in Appendix 1, available at jpn.ca.
Statistical analyses
Before testing our a priori hypotheses of white matter lateralization, we first carried out voxel-wise statistics on the skeletonized FA/MD/AD/RD images using 2-sample t tests to obtain the group differences in DTI metrics. We then applied 1-sample t tests for all participants within each group to generate group-level white matter laterality maps of FA/MD.
To test our primary hypotheses of shared and unique abnormalities in the white matter lateralization in patients with schizophrenia and BD, we performed voxel-wise 2-sample t tests on FA/MD LI maps (schizophrenia–control, BD– control and schizophrenia–BD). Unique abnormalities in patients with either schizophrenia or BD were elucidated by the schizophrenia–BD t test. Shared abnormalities between patients with schizophrenia and BD were identified by the conjunction of results from the schizophrenia–control and the BD–control t tests. As lithium may have a neuroprotective effect, we performed a secondary analysis with the same tests, adjusting for lithium medication. To rule out variances in the primary findings that were associated with handedness and race, we repeated the analyses on a subset of right-handed Chinese participants (the largest racial group in the study cohort). A posteriori testing to interrogate the unique lateralization abnormalities in patients with BD and schizophrenia were then performed on both the DTI metrics and LI.
To evaluate the association between white matter lateralization abnormalities and clinical measures in patients with schizophrenia and BD, we performed voxel-wise regression of FA/MD/AD/RD LI against each clinical measure (PANSS subscales, YMRS, GAF, QOL scores and antipsychotic dosages) in each patient group separately.
All the group-level analyses were performed using non-parametric permutation testing, which is available as a software tool “Randomise” in FSL.52 Age, sex, handedness and race were included as covariates in all analyses. To control for family-wise error (FWE) rate among the multiple comparisons, we ran the permutations 5000 times. We used a threshold-free cluster enhancement (TFCE) approach, omitting the need to prespecify initial peak height and size of the cluster, as the significance of the target voxel is defined by magnitude and spatial extent of neighbouring voxels.53
Results
Sample cohort characteristics
Our sample comprised a total of 262 participants: 150 patients with schizophrenia, 35 patients with BD with psychotic features and 77 healthy controls. There were no group-based differences among the groups in terms of age, race or handedness (Table 1). There were no differences in number of hospital admissions between the schizophrenia and BD groups; however, compared with patients with BD, those with schizophrenia had a relatively longer duration of illness (p < 0.010), and received greater amounts of daily antipsychotic medication (p = 0.032).
Demographic and clinical characteristics of the study sample
Seven of the 150 patients with schizophrenia had missing QOL measures, hence their data were excluded from subsequent neuroimaging clinical correlational analyses. In terms of QOL, the patients with schizophrenia reported a poorer sense of well-being in all 4 domains than healthy controls (p < 0.001) and poorer psychological QOL than patients with BD (p = 0.017). Patients with BD reported worse physical health (p = 0.030) and social relationships (p = 0.027) than healthy controls.
White matter microstructural abnormalities in patients with schizophrenia and bipolar disorder
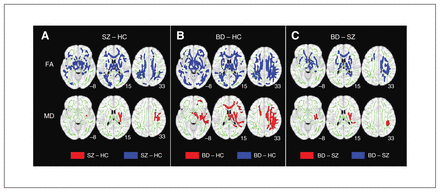
To derive the patterns of white matter hemispheric lateralization, we first determined the DTI indices in patients with schizophrenia and BD and in controls. Further between-group comparisons (thresholded at p < 0.05, TFCE FWE-corrected) indicate widespread reductions of FA in both patients with schizophrenia and BD compared with controls (Fig. 1A and B, Appendix 1, Table S1A). Patients with schizophrenia exhibited a localized pattern of increased MD in the right subcortical and frontal–occipital/parietal tracts. Patients with BD showed increased MD in similar regions of the fornix, corpus callosum, corona radidata, internal capsule and external capsule reported in patients with schizophrenia (corroborated by conjunction analysis; Appendix 1, Table S2) as well as commissural and associational fibres in the right hemisphere.
White matter microstructural abnormalities in patients with schizophrenia (SZ) and bipolar disorder (BD). Between-group comparisons revealed that (A) compared with healthy controls (HC), patients with schizophrenia exhibited reduced fractional anisotropy (FA) that is widespread throughout the frontal, limbic, subcortical and parietal/occipital regions, and more circumscribed increased mean diffusivity (MD) in the right subcortical and frontal/occipital tracts. (B) Compared with healthy controls, patients with BD showed reduced FA that is widely distributed throughout the brain and increased MD in commissural fibres and right association fibres. (C) Compared with patients with schizophrenia, those with BD showed reduced FA in the commissural, subcortical and right association fibres as well as increased MD in the right projection and association fibres (internal capsule, external capsule, corona radiata and superior longitudinal fasciculus). Results were thresholded at p < 0.05 (using a threshold-free cluster enhancement method that controls for family-wise error) and thickened with the “tbss_fill” command (FSL) for better visibility.
Compared with patients with schizophrenia, those with BD exhibited decreased FA in bilateral frontal and subcortical regions, the right parietal/occipital tracts and commissural tracts (Fig. 1C). Patients with BD also demonstrated increased MD compared with patients with schizophrenia in subcortical tracts circumscribed to the right hemisphere. Further probing of the regions showing decreased FA in patients with BD versus those with schizophrenia revealed increased RD (but not AD) in the right hemisphere in patients with BD compared with those with schizophrenia (Appendix 1, Table S1B).
White matter lateralization abnormalities in patients with schizophrenia and bipolar disorder
The FA and MD white matter LI patterns of each group are presented in Appendix 1, Fig. S1. In controls, different white matter tracts exhibited varying leftward and rightward lateralization (Appendix 1, Fig. S1A).
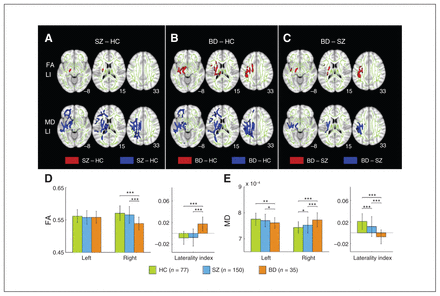
Compared with controls (at a threshold of p < 0.05, TCFE FWE-corrected), patients with schizophrenia showed increased leftward FA LI in 1 cluster, which encompassed parts of the cerebral peduncle and the posterior limb of the internal capsule (Fig. 2A, Appendix 1, Table S3A). Compared with controls, patients with BD showed increased leftward FA LI in similar regions as patients with schizophrenia (Appendix 1, Table S4A) as well as in regions that extended to the frontal tracts (corona radiata), subcortical tracts (internal capsule, external capsule), fornix, frontal-occipital tracts (superior longitudinal fasciculus) and the body of the corpus callosum (Fig. 2B).
White matter laterality index (LI) abnormalities in patients with schizophrenia (SZ) and bipolar disorder (BD). Between-group comparisons revealed (A) greater fractional anisotropy (FA) LI in the cerebral peduncle and posterior limb of the internal capsule, and smaller mean diffusivity (MD) LI in patients with schizophrenia along similar regions as well as the external capsule and superior longitudinal fasiculus compared with healthy controls (HC); (B) greater FA LI and smaller MD LI in patients with BD compared with controls in the callosal, limbic, corticospinal and superior longitudinal fasicular tracts; and (C) greater FA LI and smaller MD LI in patients with BD than in those with schizophrenia in similar tracts. Results were thresholded at p < 0.05 (using a threshold-free cluster enhancement method that controls for family-wise error) and thickened with the “tbss_fill” command (FSL) for better visibility. We then parsed the white matter regions found to show FA and MD lateralization differences between patients with schizophrenia and BD. (D) We measured the within-group average of FA values in the left and right hemispheres. The mean FA values of the regions (cerebral peduncle, internal capsule, superior corona radiata, and superior longitudinal fascisculus) are shown in the coloured bars (controls in green, schizophrenia group in blue and BD group in orange). Patients with BD demonstrated decreased FA in the right hemisphere compared with controls and patients with schizophrenia. In addition, we compared the mean FA LI among the 3 groups and found a pattern of reversed LI in patients with BD compared with controls and patients with schizophrenia. (E) The within-group averaged MD values of the regions (cerebral peduncle, internal capsule, external, corona radiata, fornix/stria terminalis, and superior longitudinal fasciculus) in the left and right hemisphere of the different groups are shown. Patients with BD showed decreased MD in the left hemisphere and increased MD in the right hemisphere compared with controls and patients with schizophrenia. In addition, the mean MD LI among the groups are compared. Overall, patients with schizophrenia showed attenuated MD LI compared with controls. Conversely, patients with BD showed reversed FA and MD LI. The mean and standard deviation of each group are presented by the bar charts. *p < 0.05; **p < 0.01; *** p < 0.001.
In addition, patients with schizophrenia showed reduced MD LI in the frontal tracts (corona radiata), subcortical tracts (internal capsule, external capsule, posterior thalamic radiation), frontal-occipital/parietal tracts (sagittal stratum, superior longitudinal fasciculus), cerebral peduncle, fornix, cingulum and corpus callosum compared with controls. Patients with schizophrenia and BD share many regions of MD LI abnormalities (Appendix 1, Table S4A), but were even more extensive, extending to the corticospinal tract. Notably, many regions of reduced MD LI overlapped with those of increased FA LI.
In terms of magnitude, patients with BD exhibited larger leftward FA LI in the cerebral peduncle, superior corona radiata, internal capsule and superior longitudinal fasciculus (Fig. 2C) and lower MD LI in the frontal tracts, subcortical tracts, frontal-occipital/parietal tracts and fornix than patients with schizophrenia.
Secondary analysis accounting for lithium medication taken by a subset of patients with schizophrenia and BD showed that the unique and shared regions of lateralization abnormalities in these patients remained largely unchanged (Appendix 1, Table S3B and Table S4B). Secondary analysis on a subset of right-handed Chinese participants (schizophrenia, n = 122; BD, n = 30; controls, n = 60) also indicated similar patterns in the findings of implicated white matter regions (Appendix 1, Table S3C and Table S4C).
Subsequently, to directly compare the laterality patterns in patients with schizophrenia and BD, we performed post hoc analyses of the white matter regions exhibiting FA or MD lateralization differences between the 2 groups. The FA values were smaller in patients with BD than in patients with schizophrenia and healthy controls, and this was localized to the right hemisphere (BD < control and BD < schizophrenia, p < 0.001; Fig. 2D), accounting for the reversed mean FA lateralization patterns observed in the patients with BD. In addition, both the decreased MD in the left hemisphere and the increased MD in the right hemisphere in patients with BD compared with healthy controls and patients with schizophrenia resulted in extensive reversed mean MD lateralization (left: BD < controls, p = 0.008 and BD < schizophrenia, p = 0.048; right: BD > controls, p < 0.001, BD > schizophrenia, p < 0.001 and schizophrenia > controls, p = 0.034; Fig. 2E). Of note, the increased MD in the right hemisphere of patients with schizophrenia contributed to the loss of MD laterality compared with healthy controls. The overall patterns of reduced lateralization in patients with schizophrenia and reversed lateralization in those with BD in general were also replicated in a subset of right-handed Chinese participants (Appendix 1, Fig. S2).
The percentage of voxels of the white matter regions showing attenuated and reversed FA and MD laterality in patients with schizophrenia and BD, respectively, are presented in Appendix 1. Similar findings of asymmetry abnormalities in patients with schizophrenia and BD were found when we used the index of asymmetry (Appenxix 1, Fig. S3 and Fig. S4).
Correlations between laterality indices and clinical scores
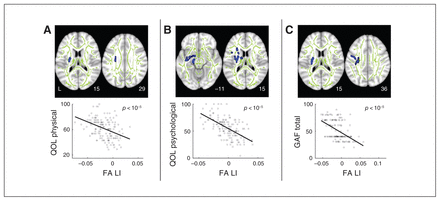
Voxel-wise regression analysis (thresholded at p < 0.05, TFCE FWE-corrected) revealed significant correlations between leftward white matter laterality patterns and clinical measures, but only in the schizophrenia group (Fig. 3, Appendix 1, Table S3). An increase in FA LI mainly in frontal–subcortical white matter tracts was associated with lower physical QOL scores (corpus callosum, posterior limb of internal capsule and superior corona radiata); lower psychological QOL scores (corpus callosum, interior capsule, anterior/superior corona radiata, sagittal stratum, external capsule and superior longitudinal fasciculus); and poorer GAF scores (posterior limb of internal capsule, superior corona radiata, external capsule and superior longitudinal fasciculus; Fig. 3D–F, Appendix 1, Table S3). Taking the negative leftward FA LI indices observed in controls as a normal FA lateralization pattern (refer to Fig. 2D), these findings demonstrated that the increased leftward FA lateralization (or a loss of normal lateralization pattern) in these white matter regions were associated with poorer QOL and psychosocial functioning in patients with schizophrenia.
Indices of white matter lateralization are associated with reduced psychosocial functioning and quality of life (QOL) in patients with schizophrenia. Whole-brain voxel-wise regression of white matter laterality index (LI) indicated a correlation between loss of FA lateralization (i.e., increased FA LI) with poorer (A) self-rated QOL in physical health, (B) QOL in psychological well-being and (C) clinician-rated global assessment of functioning (GAF). The top row presents brain regions in which white matter LI correlated with clinical scores, thresholded at p < 0.05 (using a threshold-free cluster enhancement method that controls for family-wise error). Regions were thickened with the “tbss_fill” (FSL) command for better visibility. Scatter plots illustrate the associations between clinical scores and FA laterality indices averaged across the voxels in regions (blue) in patients with schizophrenia.
No significant correlations were found between the FA LI and symptomatology (PANSS positive, negative and general psychopathology) in the patient groups. Additionally, no significant correlations were found between FA LI and manic symptoms (YMRS) in the BD group. No correlations were found between MD LI and clinical scores in either patient group. Finally, no significant correlations were found between LI and antipsychotic dosages.
Discussion
This is, to the best of our knowledge, the first study to directly compare the brain white matter lateralization patterns between patients with schizophrenia and BD. We found common abnormalities of FA hemispheric lateralization in the cerebral peduncle and posterior limb of the internal capsule as well as common abnormalities of MD lateralization in concordant regions and in the frontal/occipital, limbic and callosal tracts in both patient groups. Additionally, regions of abnormal lateralization in patients with BD were more extensive than in patients with schizophrenia. Overall, white matter lateralization was attenuated in patients with schizophrenia and reversed in patients with BD, driven by the greater white matter microstructure abnormalities in the right hemisphere in those with BD compared with controls and patients with schizophrenia. The loss of normal hemispheric lateralization in patients with schizophrenia correlated with the reductions in the levels of physical and psychological domains of QOL as well as global psychosocial functioning.
White matter lateralization abnormalities in patients with schizophrenia
The overall patterns of leftward lateralization in the associational and subcortical tracts of healthy controls, as revealed by the mean FA and MD laterality indices, were similar to findings in the language networks and major associational tracts of healthy participants.54,55 In patients with schizophrenia, we observed loss of FA hemispheric lateralization in a cluster encompassing the posterior limb of the internal capsule and cerebral peduncle and white matter tracts that transverse across the striatum, thalamus and midbrain. Although our initial analysis of FA indices revealed extensive decreases throughout many white matter tracts in the frontal, temporal, occipital, subcortical and brainstem regions (consistent with earlier studies), tracts on both hemispheres of the brain — except for this cluster — were affected to a similar extent.
The losses of MD lateralization were more extensive than those of FA lateralization, spanning into the frontal, commissural and frontal–parietal/occipital tracts, as a consequence of more widespread right hemispheric tracts MD abnormalities observed in patients with schizophrenia.
The present findings run counter to our hypothesized predominant left hemisphere dysfunction in patients with schizophrenia, but they are consistent with the findings of other meta-analyses of white matter tract or volumes56,57 that did not find preferential abnormalities in either hemisphere.
White matter lateralization abnormalities in patients with bipolar disorder
In patients with BD, we found abnormalities in FA lateralization in similar regions as patients with schizophrenia, but the abnormalities extended to the frontal, subcortical, callosal and brainstem regions. This was accompanied by abnormalities in MD lateralization in largely concordant regions. The FA lateralization abnormalities were driven by FA abnormalities that were more extensive in the right hemisphere, specifically in the cingulum, corpus callosum and superior longitudinal fasciculus. Likewise, the MD lateralization abnormalities were largely driven by MD abnormalities in the right hemispheric tracts.
The predominant right dysfunction in patients with BD agreed with our hypothesis and was consistent with the trend of right lateralization of white matter abnormalities shown in a meta-analysis of patients with BD.58 It has been suggested that the right hemisphere is preferentially involved in negative emotions59 and that disturbances of the right hemisphere underlie mood dysregulation.13 Hence, our present findings of preferential white matter abnormalities in the right hemisphere in patients with BD could likely be associated with the affective features of this disorder.
Comparisons of lateralization abnormalities in patients with schizophrenia and bipolar disorder
Common features in abnormal FA lateralization in patients with schizophrenia and BD were localized to the posterior limb of internal capsule and cerebral peduncle, whereas common abnormal MD lateralization spanned the corticospinal projection, interwoven with callosal, frontal/occipital and subcortical/limbic tracts. These common regions of lateralization abnormalities overlapped, to a certain extent, with our hypothesized regions.12
The cerebral peduncle and posterior limb of the internal capsule transverse the thalamus, lentiform nucleus and midbrain.60 They are part of the corticospinal tract, the largest pyramidal pathway that is responsible for psychomotor actions. The superior longitudinal fasciculus is the largest associational fibre that connects the auditory and speech areas. The skewed hemispheric disruptions of these tracts may likely give rise to imbalances in perceptual processing and integration as well as psychomotor processing that could underlie the auditory hallucinations, speech symptoms and deficits in processing speed often found in individuals with psychosis.60 The corpus callosum is the major conduit for information transfer between the left and right hemispheres.1 Anomalies in this region could have disrupted interhemispheric connectivity and communication, resulting in the abnormal lateralization.
Interestingly, we found larger and more distributed regions of abnormal lateralization in patients with BD than in patients with schizophrenia. In these regions, the microstructural abnormalities were more prominent in the right than in the left hemisphere in patients with BD. The finding of more severe microstructural abnormalities is not consistent with the results of studies that found lower FA in patients with schizophrenia than BD31,35 or those of studies that found no changes between these groups.32,33,36 However, our finding is similar to that of a study comparing medication-naive patients with first-episode BD and schizophrenia that reported lower FA in the callosal, subcortical and fronto-occipital white matter regions in patients with BD.15 Our secondary finding of predominant right RD abnormalities but lack of AD abnormalities (a finding also reported in a previous study15) in patients with BD compared with patients with schizophrenia suggests that there may be relatively more severe demyelination or neuroinflammation in those with BD.
The present pattern findings of attenuated lateralization in patients with schizophrenia and reversed lateralization in patients with BD have also been observed in non-neuroimaging studies. The alterations in epidermal ridge development are thought to be linked to deviations in neurodevelopment, as the epidermis and nervous system have the same ecotodermal origins during the perinatal stages.61 In a study of finger-print asymmetry, patients with BD showed greater asymmetry than healthy controls and patients with schizophrenia.62 Another study using a tactile line bisection task, which is commonly used to test hemispheric dysfunction in patients with neurologic deflcits, found that patients with schizophrenia showed attenuated lateralization bias, whereas those with BD showed reversed lateralization.63
Association between white matter lateralization indices and clinical measurements
We noted that the white matter lateralization abnormalities in patients with BD and schizophrenia were not associated with psychotic symptoms. We did not observe any association between lateralization indices and QOL/global functioning in the patients with BD, who showed little variability in QOL or functioning outcome measures. However, in our schizophrenia cohort, who demonstrated lower QOL and psychosocial functioning than the BD cohort, the loss of normal FA lateralization in the posterior limb of the internal capsule and superior corona radiata was correlated with lower physical QOL and level of psychosocial functioning. Additionally, the loss of normal FA lateralization in many cortico-cortical associational bundles (anterior limb of internal capsule, external capsule and superior longitudinal fasciculus) was associated with lower psychological QOL in patients with schizophrenia.
The patterns of the present findings are consistent with findings from recent cross-modal studies. First, no correlations between abnormal lateralization observed in the caudate nucleus and thalamus (subcortical regions flanking the internal capsule) and symptom severity were found in a resting-state hemispheric lateralization study in patients with schizophrenia.64 Second, a link between reduced global functioning and decreases in volume of the internal capsule of antipsychotic-naive patients with first-episode schizophrenia was reported.65 Third, an association between shared white matter abnormalities and impaired social and occupational functioning, but not florid psychotic symptoms, across patients with BD and schizophrenia was reported.34 Taken together, these findings suggest that lateralization abnormalities in these structural connections do not reflect the present state of psychosis, which can be managed to a certain extent with medications and other interventions. Rather, the lateralization abnormalities in these cortical–subcortical connections, which subserve higher-order cognitive functions,1,3 may be associated with the traits of psychosis that impede psychosocial functioning in the long run.
Limitations
There are several limitations to our study. First, the patients were medicated with antipsychotics and mood stabilizers, which have been shown to be associated with changes in white matter.66,67 Although we found no correlations between white matter hemispheric lateralization and antipsychotic dosages, the possible effects of history and current exposure to different classes of medication on white matter lateralization could not be discounted. Second, we could not completely ascertain that the shared white matter lateralization abnormalities observed underlie the feature of psychosis; further comparative studies of other psychotic spectrum disorders, such as delusional disorders or brief psychotic disorder, or cross-diagnostic nonpsychotic disorders, such as BD with no psychosis, would be required for a more complete picture. Third, our study was cross-sectional in design. Although hemispheric lateralization occurs during perinatal stages,4 we could not fully establish whether the white matter hemispheric lateralization anomalies found across participants with schizophrenia and BD were due to neurodevelopmental aberrance or whether they were a consequence of the onset or chronicity of illness. To confirm its neurodevelopmental origins, prospective longitudinal studies on prodromal and first-episode psychosis patients would be required to determine that the white matter hemispheric abnormalities are present before psychosis onset. Fourth, our DTI acquisition protocol was relatively limited in spatial resolution and diffusion contrast. Additionally, because of the inherent ambiguity of the diffusion tensor, the interpretation of the DTI values is not straightforward.30 For instance, voxels that contain complex but not uniform fibrous tissue can also result in lower FA values. Hence, we cannot fully attribute the abnormalities found in our primary analyses of FA and secondary analyses of RD and AD to disease-related abnormalities in myelin or axonal disruptions. Future studies that acquire diffusion data using multifold diffusion directions, higher spatial and angular resolution,68,69 or advanced diffusion modelling as well as comparative histopathology studies of schizophrenia and bipolar disorder are needed to better characterize the underlying pathophysiology of abnormal white matter measures and lateralization.
Conclusion
We found common abnormalities of subcortical white matter lateralization in patients with schizophrenia and BD, which may underlie their common clinical features. We also found distinct lateralization abnormalities between patients with schizophrenia and BD. In these distinct regions of abnormalities, lateralization was attenuated in patients with schizophrenia and reversed in patients with BD.
Acknowledgements
This research was supported by the Agency for Science, Technology, and Research (A*STAR) Singapore under the Biomedical Research Council (13/1/96/19/687 awarded to J. Zhou) and the Duke-NUS Graduate Medical School Signature Research Program, funded (awarded to J. Zhou) by the Ministry of Health, Singapore. This research was also supported by funding from the National Medical Research Council Centre Grant (Institute of Mental Health; awarded to N.F. Ho), the National Healthcare Group (NHG 11003, 12003 awarded to K. Sim) and the Agency for Science, Technology, Research/Singapore BioImaging Consortium (ASTAR/SBIC 009/2006 awarded to K. Sim). The authors thank all the research staff and patients who participated in this study.
Footnotes
↵* Share first authorship;
↵† share senior authorship.
Competing interests: None declared.
Contributors: Z. Li, K. Sim and J. Zhou designed the study. F. Ji, C. Kuswanto, M.-Y. Sum, Y.-Y. Sitoh, K. Sim and J. Zhou acquired the data, which N.-F. Ho, Z. Li, M. Wang, H.-Y. Tng, K. Sim and J. Zhou analyzed. N.-F. Ho, Z. Li, K. Sim and J. Zhou wrote the article, which all authors reviewed and approved for publication.
- Received May 11, 2016.
- Revision received September 21, 2016.
- Accepted November 6, 2016.