Abstract
Background: Posttraumatic stress disorder (PTSD) is associated with medial frontal and amygdala functional alterations during the processing of traumatic material and frontoparietal dysfunctions during working memory tasks. This functional magnetic resonance imaging (fMRI) study investigated the effects of trauma-related words processing on working memory in patients with PTSD.
Methods: We obtained fMRI scans during a 3-back task and an identity task on both neutral and trauma-related words in women with PTSD who had been sexually abused and in healthy, nonexposed pair-matched controls.
Results: Seventeen women with PTSD and 17 controls participated in the study. We found no behavioural working memory deficit for the PTSD group. In both tasks, deactivation of posterior parietal midline regions was more pronounced in patients than controls. Additionally, patients with PTSD recruited the left dorsolateral frontal sites to a greater extent during the processing of trauma-related material than neutral material.
Limitations: This study included only women and did not include a trauma-exposed non-PTSD control group; the results may, therefore, have been influenced by sex or by effects specific to trauma exposure.
Conclusion: Our results broadly confirm frontal and parietal functional variations in women with PTSD and suggest a compensatory nature of these variations with regard to the retreival of traumatic memories and global attentional deficits, respectively, during cognitively challenging tasks.
Introduction
Posttraumatic stress disorder (PTSD) is associated with altered processing of emotional material with a strong attentional bias toward trauma-related information,1 which is likely to facilitate stimulus detection2 and interfere with concomitant cognitive processing.3 Brain correlates of such traumatic material effect have been extensively studied in patients with PTSD through symptom provocation protocols; results have mostly indicated medial frontal hypoactivation and amygdala hyperactivation (for a review, see Liberzon and Sripada4) and a negative correlation between the activations of these regions in patients perceiving aversive stimuli.5 The medial frontal lobe is implicated in high-level cognitive processes, such as working memory, and the amygdala is implicated in negative emotions. These results may therefore support the idea of an inhibitory relation between emotional amygdala activations and frontal cognitive activations.6
Aside from trauma processing specificities, a wide range of global cognitive impairments have been related to PTSD state with predominant attention and verbal memory deficits.1,7 Of these, working memory has been found to be particularly affected.8 Neuroimaging studies have suggested frontal and parietal dysfunction as a source for working memory alterations in patients wtih PTSD with an under-recruitment of dorsolateral frontal9,10 and posterior parietal sites,10 which are classically associated with the n-back task in healthy participants.11 Moreover, frontoparietal functional connectivity appears to be altered when patients perform working memory tasks,12,13 showing a lack of differentiation between the networks implicated in the maintenance and the updating of information in patients with PTSD.
Whereas the effects of PTSD on working memory and trauma-related material processing have been separately evaluated, little is known about the effect of traumatic memories on working memory. This point may be particularly relevant to pathology because the maintenance and manipulation of short-term traumatic memories could explain an important part of the behavioural and cognitive symptoms of PTSD (e.g., traumatic intrusions, difficulty inhibiting recurrent thoughts, difficulty concentrating1). To our knowledge, only 1 study14 has investigated the effect of trauma-related material in a working memory neuroimaging model, demonstrating an increased activation of the amygdala, ventrolateral prefrontal cortex and fusiform gyrus in patients with PTSD who were presented reminders of the trauma. Importantly, in that study trauma-related items were used as distractors, whereas task-relevant material was neutral. As a consequence, the results do not provide direct information on the neural correlates of the cognitive manipulation of traumatic material in working memory.
We sought to investigate the direct effect of trauma-related material on an n-back verbal working memory task in women with abuse-related PTSD compared with healthy controls. Considering the attentional biases that could influence this task in patients with PTSD, our study involved a separate attentional task with neutral and traumatic material. In line with the literature, we expected a decrease in frontal and parietal activations during working memory tasks in patients with PTSD compared with controls. We hypothesized that the introduction of traumatic material would elicit an increase in amygdala response and an alteration of frontal activation only in the PTSD group that would be negative (as seen in symptom provocation studies) or positive.14 However, owing to the exploratory nature of this experiment, we analyzed data sets without assumptions (i.e., on the whole brain, with bilateral tests).
Methods
Participants
We recruited women aged 18–40 years with sexual abuse–related chronic PTSD from the University Hospital, and through local advertisement to the general population we recruited controls pair-matched for sex, age and educational level (< 1 yr difference) with no history of sexual abuse.
In both groups, exclusion criteria were history of head injury, illicit substance abuse, claustrophobia, current use of psychotropic medication for more than 21 days, medical disorders affecting brain function (e.g., epilepsy, tumour) and non–magnetic resonance imaging (MRI) compliant material. A clinical neuroradiologist further reviewed the MRI scans with abnormal — especially tumoural and/or vascular —findings to exclude ineligible participants. The PTSD symptomatology, dissociative dimension and psychiatric comorbidities were evaluated by a trained psychiatrist (W.E.H.) using the Clinical Administered PTSD Scale (CAPS),15 the Dissociative Experiences Scale (DES)16 and the Mini International Neuropsychiatric Interview (MINI 5.0.0), respectively.17,18 Participants received full details about the experimental protocol and provided written informed consent before the beginning of the experiments.
This study conforms to the Code of Ethics of the World Medical Association (Declaration of Helsinki) and was approved by the local Ethical Committee (CCPPRB 2005–04). This project was registered on ClinicalTrials.gov (NCT00288314) and supervised by a clinical investigations monitoring committee (INSERM CIC 202). All participants received monetary compensation for time and efforts spent completing the study.
Imaging protocol
Scanning was performed on a 1.5-T Signa LX General Electric scanner. Participants were in the supine position in the scanner, and their heads were secured with foam cushions and a forehead strap to minimize motion. We acquired structural images using a whole-head, T1-weighted, continuous sagittal 3-dimensional spoiled gradient recall (3-D SPGR) sequence (echo time [TE] 5 ms, repetition time [TR] 25 ms, flip angle 10°, field of view [FOV] 240 mm, 124 slices, in-plane resolution 0.9375 mm2). We acquired functional images using a single-shot echo planar imaging (EPI) sequence sensitive to blood oxygen level–dependent (BOLD) contrast (TR 3 s, TE 60 ms, flip angle 90°). The blocked-task paradigm comprised 3 functional runs of 95 whole-brain acquisitions (25 contiguous slides, thickness 5 mm, 3.75 × 3.75–mm in-plane resolution, FOV 240 mm). The first 5 EPI volumes were discarded from analysis to allow stabilization of longitudinal magnetization. Through a mirror attached to the head coil, participants viewed stimuli that were presented on a screen at their feet, and they held a single-button response device in each hand.
Experimental procedure
The paradigm was explained to the participants before scanning. Participants underwent 3 consecutive fMRI runs, each 4 minutes, 45 seconds in length. Each experimental run consisted of 4 activation blocks (identity and 3-back with neutral and trauma-related material, counterbalanced across runs) interspersed with 3 control condition blocks. During the identity task, participants were sequentially presented 15 pairs of words for 2 seconds each, and they were instructed to determine whether words were identical or different on each trial. For the 3-back task, participants were presented 10 words for 3 seconds each (after presentation of 3 “loading” words), and they had to determine whether the item was identical to the one from 3 trials previous. Control condition blocks involved covert sequential reading of 15 words paced at 2 seconds per word. Verbal material consisted of frequent nouns 4–8 letters long. We selected trauma-related material from a list of the most frequently recurrent words across traumatic scripts collected during prior clinical interviews from patients with PTSD (e.g., violence, anguish). We chose neutral words from a previous study19 because they were nontraumatic and nonemotional. Participants were instructed to, after reading each word, press left-or right-hand buttons to provide their responses during activation blocks and to press both buttons during control condition trials.
Data analysis
We excluded the data for 2 participants (1 from each group) from functional analyses owing to excessive motion during scanning (> 3 mm) and technical issues resulting in low signal-to-noise ratio. We analyzed functional data using Freesurfer and FS-FAST software packages (version 4.05, http://surfer.nmr.mgh.harvard.edu). The fMRI data were motion-corrected and realigned on the first image of the acquisition using AFNI20 and then slice-timing corrected. We rescaled the voxel intensities such that the in-brain grand mean was 1000 for all participants, and images were spatially smoothed using a 3-D Gaussian filter with a full-width at half-maximum of 10 mm. Finally, individual functional volumes were coregistered with corresponding anatomic (T1-weighted) volumes using 6 degrees of freedom transformation.
The hemodynamic response (HDR) was modelled as a γ function with a delay of 2.25 seconds and a dispersion of 1.25 seconds. Movement parameters from realignment corrections and linear drift in the BOLD signal were removed by estimating this component with the amplitude of the HDR.
Individual analysis
We performed a general linear model analysis for each voxel to determine which ones demonstrated significant paradigm-related changes of HDR amplitude. Each participant’s data were registered to FreeSurfer’s average template brain in Montreal Neurological Institute space (MNI305 — the first version of the template). Statistical parametric maps were generated from linear contrasts between the different conditions in each participant.
Group analysis
A second-stage weighted random-effects analysis was then performed in which individual data sets were weighted by the inverse of their noise. We performed 1-sample (within-group) and 2-sample (between-group) t tests on contrast images obtained from each participant for each comparison of interest. We performed contrasts of interest across the whole brain using unilateral threshold criteria of significant activation at a voxel-wise level of p < 0.005 and a Gaussian random field cluster-wise correction for multiple comparisons at p < 0.05. For cluster coordinates reporting of single-task effects (identity and 3-back tasks v. control condition), results were extra-thresholded (p < 10−5, uncorrected) to obtain subclusters. To report activation peaks, we transformed MNI coordinates into Talairach coordinates.
We conducted distinct analyses of variance (ANOVAs) on hits and reaction times in each task, with group as independent factor and material type (neutral, trauma-related) as a repeated measure.
Results
Participants
We recruited 17 patients with PTSD (mean age 24.9 [standard deviation (SD) 4.8] yr; mean years of education 13.7 [SD 2.8]) and 17 pair-matched controls (mean age 24.8 [SD 4.7] yr; mean years of education 13.8 [SD 2.6]). All the patients had severe chronic PTSD (mean CAPS score 73.4 [SD 20.4]). Some patients with PTSD also had a current major depressive episode (n = 8), suicidal thoughts (n = 11), symptoms of agoraphobia (n = 5) or alcohol abuse (n = 1). The dissociative symptoms were significantly higher in the PTSD group than in the control group (mean 26.8 [SD 12.9] v. 4.9 [SD 7.0]; t32 = 5.86, p = 0.001). Results from a structural MRI experiment using the same participants demonstrated no brain morphologic differences between these groups that could explain functional variations.21
Behavioural results
For the identity task, the PTSD group performed less accurately (F1,32 = 5.1, p = 0.031) and had a tendency to respond more slowly (F1,32 = 3.2, p < 0.10) than controls. Overall, trauma-related items were answered less accurately (F1,32 = 4.9, p = 0.033) and more slowly (F1,32 = 48.5, p < 0.001) than neutral items though no interaction between group and material was found.
For the 3-back task, we found no overall group effect on accuracy or reaction time. Trauma-related items were answered less accurately (F1,32 = 9.9, p = 0.004) and more slowly (F1,32 = 14.0, p < 0.001) than neutral items. We observed a trend toward a group × material interaction on reaction time, with significant slowing of reaction time in the control group, but not the PTSD group, for traumatic versus neutral material (t16 = 3.5, p = 0.003; Table 1).
Task effects versus control condition (neutral material) on identity and 3-back tasks completed by women with PTSD related to sexual abuse and pair-matched, nontraumatized controls
Functional MRI results
Within-group comparisons
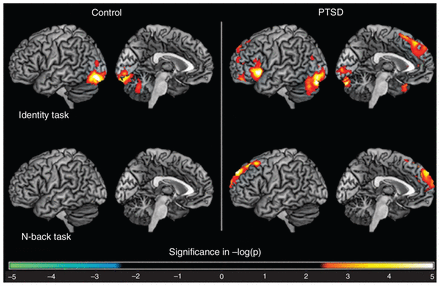
The identity task with neutral material versus the control condition elicited large bilateral activations in controls and patients with PTSD. These activations were mostly localized in posterior parts of the brain, with consistent activations of the parietal lobules extending to the transverse superior and middle occipital gyrus and to inferior posterior parts of the temporal hemisphere. Frontal activations appeared bilaterally in the supplementary motor areas, premotor cortex and middle frontal gyrus and extended to the insula (Table 1).
In the identity task with traumatic versus neutral material, traumatic material effect was associated with increased occipital and cerebellar activations in both groups (Table 2). The PTSD group also demonstrated an important overactivation, which was absent in controls, of the left pars orbitalis (Brodmann area [BA] 47) and left superior frontal gyrus.
Within-group comparisons of material type effect (trauma-related v. neutral material) among women with PTSD related to sexual abuse and pair-matched, nontraumatized controls
For the 3-back task using neutral material versus the control condition, as expected, neutral working memory was characterized by large, bilateral activations of dorsolateral (middle frontal gyrus) and dorsomedial frontal areas (supplementary motor areas), the intraparietal sulcus, postero-inferior temporal lobes and cerebellum in both groups.
For the 3-back task using traumatic versus neutral material, traumatic material effect elicited an increased activation of the left superior frontal gyrus in the PTSD group. Although this pattern was noticeable among controls, it did not survive the clusterwise threshold correction (Table 2, Fig. 1).
Material type effects (traumatic v. neutral material) in both the control and posttraumatic stress disorder (PTSD) groups during identity and n-back tasks (left hemisphere only).
Between-group comparisons
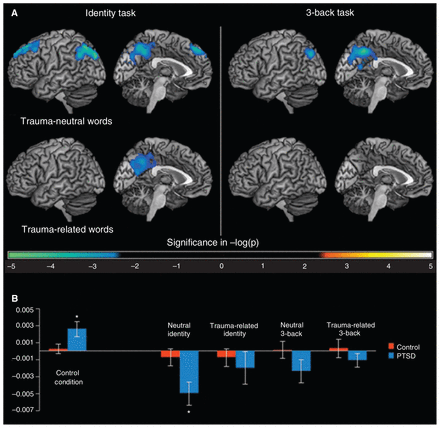
Analyses revealed a significantly greater deactivation in the PTSD group on the precuneus and posterior cingulate in neutral identity and 3-back tasks and in the identity task using traumatic words. Though this effect was noticeable on traumatic 3-back contrast, it did not survive the clusterwise threshold correction. We found an additional left superior frontal negative cluster for the neutral identity task (Table 3).
Between-group comparisons of effect on all tasks among women with PTSD related to sexual abuse and pair-matched, nontraumatized controls
We found no interaction between groups and material using our threshold. However, when restricting analyses to frontal and mediotemporal sites with no clusterwise correction, we found a deactivated cluster in the dorsal anterior cingulate for the identity task and another one in the left posterior inferior frontal gyrus (BA 44) for the 3-back task; these clusters tended to be more activated by trauma-related material in controls than in patients with PTSD (Table 3 and Fig. 2).
(A) Group effects (posttraumatic stress disorder [PTSD] group v. control group) during identity and n-back tasks according to the type of material (left hemisphere only). (B) Mean size effects (error bars represent standard error; *significant difference from 0 at bilateral p < 0.05) in the parietal cluster identified for the between-group comparison of all activation conditions versus control condition contrast.
Discussion
To our knowledge, our study is the first to investigate the effects of trauma-related material manipulation in working memory. We found that PTSD was associated with decreased BOLD effect on the posterior cingulate/precuneus for neutral attentional and memory tasks and the trauma-related attentional task. The same pattern existed for the trauma-related working memory condition but did not remain after cluster-wise threshold correction and did not appear to differ significantly from the corresponding nontraumatic contrast, indicating potential type-II error. We therefore discuss this result as being primarily associated with the cognitive tasks rather than the traumatic status of the material. These areas are also well-known to be central structures of a default mode network,22 which is activated at rest or during self-centred cognitive processes and deactivates with high, external cognitive demands. Precuneal deactivation has been found to covary with cognitive demands in both attentional and working memory tasks23 and to correlate with working memory accuracy.24 In particular, recent evidence suggests a pivotal role for the precuneus in working memory25 that could be explained by the associated attentional processes.26 Owing to the strong relation between precuneal deactivation and cognitive performance identified in the literature, our results could indicate an increased cognitive effort in patients to compensate for pathological brain dysfunction. Interestingly, the functional connectivity of this region has been previously found to be altered in patients with PTSD27 and to predict subsequent PTSD symptom development in recently acutely traumatized patients.28 However, previous studies using a similar paradigm with nontraumatic material did not report medial parietal decreases in patients when performing working memory tasks.9,10,12,13 This discrepancy between our results and those reported in these latter studies may be related to their use of a 1-back task in which participants only had to maintain the last item in memory, whereas in our study participants had to maintain and constantly update the 3 last items perceived. Thus, the previous absence of evidence of parietal midline deactivation may be explained by relatively low cognitive demands (as compared with our 3-back task), resulting in a lesser amount of default mode network mobilization in both groups and subthreshold between-group variations in these regions.
In the present study, trauma-related material elicited increased frontal activations in patients with PTSD, but not in controls, with an overactivation of the ventrolateral prefrontal cortex in the identity task and greater activity in the dorsolateral and medial parts of the prefrontal cortex in both identity and working memory tasks. Rather unexpectedly, we found no medial frontal decrease or amygdala increase in activation (although all participants with PTSD reported re-experiencing symptoms), which may be related to the low signal-to-noise ratio in the mediotemporal regions and to cognitive load effects on emotional limbic activation. A recent study has associated left superior frontal recruitment with emotional regulation of both positive and negative feelings.29 This overactivation in patients compared with nonexposed controls may therefore indicate an adaptative process of coping with increased emotional arousal. Accordingly, the left superior frontal gyrus has also been found to be overactivated by sexually abused women with PTSD during retrieval of emotional compared with nonemotional items, with no amygdala activity increases.30 Moreover, van Dillen and colleagues31 have demonstrated less amygdala activation due to trauma-related material when a cognitive task was used, potentially indicating a downregulation of dorsal cognitive activations on ventral emotional structures. Increased activations of left dorsal frontal areas on trauma-related contrasts during cognitively demanding tasks along with the absence of trauma-related performance decreases in patients with PTSD are broadly consistent with these results, indicating potential compensatory processes in trauma-related cognition.
Accordingly, our finding of a slowing in 3-back reaction times for traumatic versus neutral material in controls but not in patients with PTSD (trend-level interaction) may be related to controls over-recruiting compared with patients under-recruiting the left posterior inferior frontal gyrus in this condition. Since this region (frequently referred to as the Broca area) is implicated in both the phonologic and semantic processing of words, these results may indicate an attentional shift in the control group toward the semantic rather than task-relevant properties of the stimuli; this shift appeared not to occur in the PTSD group, possibly because of their greater investment in the task. This hypothesis may also explain the absence of such differences in the identity task, which requires less high-level cognitive processing than the n-back task.
Limitations
An important aspect of our study was the sex and the type of traumatic event exposure of our PTSD sample. In western countries, women make up the largest part of the PTSD population and are almost twice as likely to have PTSD than men, possibly owing to increased lifetime risk of exposure to sexual crimes.32 We therefore chose to restrict our study participants to a homogeneous group of female patients with PTSD in the aftermath of sexual abuse. Interestingly, the structural brain alterations observed in women with PTSD related to sexual abuse are not consistent with those classically observed in men with PTSD.21 Moreover, recent meta-analytic work has revealed few, if any, deficits in verbal memory for sexual abuse–related PTSD compared with war trauma–related PTSD,7 with no significant effect observed in studies involving trauma-exposed, non-PTSD control groups. Because most victims of sexual abuse are women, this result indicates potential trauma-type or sex effects in neurocognitive manifestations of PTSD that might be further considered through experiments with both men and women who experienced various types of trauma. As a consequence, the results of the present study may not be generalized to men with combat-related PTSD.
Although different patterns of material effects have been found separately in PTSD and control groups, subsequent whole-brain ANOVAs (material × group effect) did not demonstrate significant differences between groups in these regions at our threshold, and correlations between activation level and behavioural performance were not possible owing to the relatively small size of the samples and the lack of variance in behavioural performance. Consequently, our study does not provide direct evidence for the compensatory nature of frontal overactivation in patients with PTSD during traumatic material processing. This hypothesis may be addressed in future work with larger samples and/or various levels of cognitive load to investigate the precise relation between traumatic material effect and cognition in patients with PTSD. In addition, since participants were aware of the procedure and had to perform several runs all comprising traumatic material, sustained emotional effects may have influenced all experimental conditions, including neutral and control conditions. Results can therefore only account for transient emotional material effect, and future studies may include independent group design on material type to investigate more sustained effects.
Another limitation concerns the characteristics of our control sample: the control group included healthy participants with no history of traumatic exposure or psychiatric conditions, and most participants with PTSD had comorbid disorders. This difference between groups may have acted as a confounding variable and did not allow us to ascertain the specific relevance of our observations to PTSD rather than traumatic exposure or other psychiatric disorders. This potential effect is owing to the rarity of “pure” PTSD (i.e., with no past or present comorbid disorders) and of sexual abuse with no current or past PTSD. Moreover, for ethical reasons, we chose not to recruit nonclinical participants with a history of sexual abuse; however, the fact that working memory alterations have been found in psychiatric inpatients with traumatic history compared with patients without such history33 (although not all of these traumatized patients had chronic PTSD) may support the specificity of the alteration of the working memory network in patients with PTSD.
Conclusion
Our results broadly confirm different patterns of frontal and parietal task effects in PTSD and control groups; however, rather unexpectedly, we found dorsal frontal sites to be more activated by traumatic than neutral material in the PTSD group, and parietal midline structures were more deactivated during working memory in patients than in controls. We found a similar profile on a nonmnemonic matching task, which calls the specificity of these results into question. Both frontal overactivation and precuneal deactivation may reflect a compensatory recruitment of attentional structures under high cognitive demands. Future studies will therefore be needed to clarify the relevance of these results to symptomatology and cognitive profiles associated with PTSD.
Footnotes
Contributors: L. Landré, C. Destrieux, G. Tapia, M. Isingrini and W. El-Hage designed the study. L. Landré, L. Barantin, N. Jaafari and W. El-Hage acquired the data, which L. Landré, C. Destrieux, F. Andersson, Y. Quideé, N. Jaafari, D. Clarys, P. Gaillard and W. El-Hage analyzed. L. Landré, G. Tapia and W. El-Hage wrote the article, which L. Landré, C. Destrieux, F. Andersson, L. Barantin, Y. Quidé, N. Jaafari, D. Clarys, P. Gaillard, M. Isingrini and W. El-Hage reviewed. All authors approved publication.
Competing interests: None declared for L. Landré, F. Andersson, L. Barantin, Y. Quidé, G. Tapia, N. Jaafari, D. Clarys, P. Gaillard and M. Isingrini. W. El-Hage has received speaker fees from BMS, Eli Lilly, Janssen-Cilag and Lundbeck. He received research grants from Servier, the French Association of Biological Psychiatry and Neuropsychopharmacology (AFPBN) and Lundbeck, which are unrelated to the content of this manuscript. He is coinvestigator on research projects sponsored by Lundbeck and Astra-Zeneca, he receives no remuneration from any of them. The authors report no other competing interests or financial relationships with commercial interests. Supported by a grant from the French Ministry of Health, Hospital Clinical Research Program, PHRC-APR-04 (C. Destrieux, W. El-Hage). ClinicalTrials.gov registry number, NCT00288314.
- Received November 21, 2010.
- Revision received April 23, 2011.
- Revision received June 26, 2011.
- Accepted June 27, 2011.