Abstract
Background: Earlier contributions have documented significant changes in sensory, attention-related endogenous event-related potential (ERP) components and θ band oscillatory responses during working memory activation in patients with schizophrenia. In patients with first-episode psychosis, such studies are still scarce and mostly focused on auditory sensory processing. The present study aimed to explore whether subtle deficits of cortical activation are present in these patients before the decline of working memory performance.
Methods: We assessed exogenous and endogenous ERPs and frontal θ event-related synchronization (ERS) in patients with first-episode psychosis and healthy controls who successfully performed an adapted 2-back working memory task, including 2 visual n-back working memory tasks as well as oddball detection and passive fixation tasks.
Results: We included 15 patients with first-episode psychosis and 18 controls in this study. Compared with controls, patients with first-episode psychosis displayed increased latencies of early visual ERPs and phasic θ ERS culmination peak in all conditions. However, they also showed a rapid recruitment of working memory–related neural generators, even in pure attention tasks, as indicated by the decreased N200 latency and increased amplitude of sustained θ ERS in detection compared with controls.
Limitations: Owing to the limited sample size, no distinction was made between patients with first-episode psychosis with positive and negative symptoms. Although we controlled for the global load of neuroleptics, medication effect cannot be totally ruled out.
Conclusion: The present findings support the concept of a blunted electroencephalographic response in patients with first-episode psychosis who recruit the maximum neural generators in simple attention conditions without being able to modulate their brain activation with increased complexity of working memory tasks.
Introduction
Working memory refers to the temporary online maintenance of new information in a form that can be manipulated to support ongoing cognitive processing. It includes the active storage in a visual short-term memory buffer and a phonologic loop under the control of an executive system that allocates cognitive resources.1 Working memory deficits represent one of the core features of schizophrenia.2 They start as early as the prodromal state3 and may remain relatively stable over time after the first episode of psychosis.4 Despite substantial research efforts, the origin of working memory disturbances in patients with schizophrenia remains obscure. In fact, these disturbances cannot be fully explained by clinical factors, such as duration of illness, antipsychotic medication or symptom profile.5 Physiologically, this cognitive deficit is thought to reflect at least partly early deficits in the activation of the pre-frontal network.6,7 However, discordant functional magnetic resonance imaging (fMRI) data were reported in this respect mainly owing to the variability of the experimental designs.8,9 For instance, decreased activation of the dorsolateral prefrontal cortex (DLPFC) was described in patients with schizophrenia during working memory tasks, indicating the presence of information processing deficits, whereas no difference or even increased DLPFC activation was reported in other studies.9 Studies on the relation between task performance and DLPFC activation revealed that high-performing patients tend to increasingly activate this area as well as the parietal cortex, whereas poor performers have consistently lower DLPFC activation compared with controls.10,11 In line with these results, functional network studies showed that a stronger prefrontal–posterior parietal coupling predicts better working memory performance in patients with schizophrenia.12,13
In contrast to fMRI, event-modulated electroencephalographic (EEG) dynamic analysis may represent a more sensitive method to explore the pathologic effects when addressing cognitive functions that occur in the time range of 100 ms. Event-related potentials (ERPs) represent a series of EEG events that reflect the progressive activation of neuronal sub-populations in the course of cognitive processing. In the 0–150 ms after stimulus onset, ERPs are related to physical characteristics of stimulus (i.e., exogenous ERPs) and reflect sensory treatment (mainly predominant in occipital areas for visual stimuli), whereas the later ERPs or endogenous ERPs are task-dependant. The brain oscillatory activity recorded with EEG or magnetoencephalography has also aroused a growing interest as a potential functional support for the development of ERPs. In controls, the successful performance of the n-back working memory task is associated with significant changes of ERPs and brain rhythm oscillations. In particular, a significant decrease of P200 and N200 latencies is observed after stimulus onset during the 2 working memory conditions.14,15 This decrease may correspond to the encoding and retrieval phases of working memory processing that follow sensory processes.16,17 Among brain oscillations, earlier reports pointed to the reactivity of the θ frequency bands (4–8 Hz) to various memory paradigms reflecting the corticohippocampal interactions needed in case of increasing memory load.18,19 In particular, a frontal θ power increase in initial phases of encoding with demanding attentional tasks followed by a late sustained oscillatory activity for higher working memory load was also described during n-back activation.20,21 Whereas the phasic θ synchronization supports the activation of neural networks involved in allocation of attention,20,21 the later sustained power appears related to item retention and active maintenance for further task requirements.22
Previous contributions documented the presence of early changes in sensory ERPs,23,24 attention-related endogenous ERPs25,26 and θ band oscillatory responses24,27 during working memory activation in patients with chronic schizophrenia. Whether these changes are a simple epiphenomenon or a similarly biologic marker of schizophrenia is still unclear. In this respect, studying patients with first-episode psychosis may be crucial since it allows the investigation of the presence of early EEG activation deficits that may precede the structural and metabolic deficits observed in chronic forms of the disease. Previous EEG studies using working memory activation in patients with first-episode psychosis are scarce and mostly focus on auditory sensory processing.25,28 Similarly, the impacts of first-episode psychosis on working memory–related brain oscillations have rarely been addressed.29,30 The present study reports the analysis of endogenous and exogenous ERPs and θ rhythm synchronization in healthy controls and patients with first-episode psychosis during 2 visual n-back working memory tasks compared with oddball detection and passive fixation tasks.
Methods
Participants
We recruited patients with first-episode psychosis from the specialized program for young adult psychiatric patients (JADE) in Geneva, Switzerland. Patients were recruited after a first contact with mental health services for psychotic symptoms (defined as hallucinations, delusions or formal thought disorders). The diagnosis was made according to DSM-IV criteria31 by 2 experienced psychiatrists independent of the study. We excluded patients with drug addictions; however, we did not exclude those with some previous recreational use of cannabis. We also recruited for participation in this study a group of healthy, matched controls without history of sustained head injury or other neurologic or psychiatric disorders.
Patients were clinically rated for symptom severity using items of the Brief Psychiatric Rating Scale (BPRS).33 Consistent with Ventura and colleagues,34 we used the blunted affect and emotional withdrawal items to assess negative symptoms, and we used the bizarre behaviour, unusual thought content, hallucinations and suspiciousness items to assess positive symptoms. After careful assessment by the JADE program of patients’ ability to understand the proposed project, we obtained informed, written consent from all patients before final inclusion in the study. Controls also provided informed consent. The study was approved by the Ethics Committee of the University Hospitals of Geneva, Switzerland, and the study protocol was in line with the Helsinki Declaration.
Experimental design
Participants watched a computer-controlled display screen at a distance of 57 cm. They viewed pseudorandom sequences of consonants and vowels common to the French alphabet, and pressed a computer-controlled button with their right index fingers as soon as a target appeared (response trials). For non-target stimuli, no motor response was required (no-response trials). Targets were defined either according to the oddball task (rare event) or to the n-back task. Stimuli consisted of white letters in Arial font (2° × 2.5° visual angle), with 10% grey noise, embedded in a 50% random-noise grey rectangular background patch (6° × 6.7° visual angle). They were presented for a duration of 0.5 s, separated by 5-s intervals (onset to onset) during which a dot helped participants maintain fixation. Participants were instructed to remain quiet and to move only their right index fingers in accordance with the nature of the task to minimize any muscle artifacts.
In a simple detection task (control), sequential letters or background patches without letters were presented. Participants responded as fast as possible when patches with letters appeared. In the 1-back task, the target was any letter identical to the one immediately preceding it. In the 2-back task, the target was any letter that was identical to the one presented 2 trials back. Thus, working memory load increased from detection (memory-free) to 1-back (moderately demanding) and 2-back (highly demanding) conditions. In the passive fixation task, letter series identical to the 2-back task were presented, but participants were unaware of the order of the letters and were asked to fix the screen and watched the series passively. Each task was tested in 3 stimulus sequences composed of 30 images each (7 targets). Before each sequence, participants were informed of the nature of the task. The protocol started with 3 sequences of the passive task. A first detection task sequence was followed by 1 sequence of the 1-back, 3 sequences of the 2-back, 2 sequences of the 1-back and 2 sequences of the detection task. Reaction time and performance were systematically recorded. Before and after the experimental paradigm, an open- and closed-eyes EEG session without task was conducted (3 min). We performed EEG assessments in the morning.
Electrophysiologic recordings
Continuous EEG (Micromed) was recorded, using 20 surface electrodes placed over the scalp according to the 10–20 international electrode placement system35 with linked earlobes as a reference. Skin impedance was kept below 5 KOhms. Electrophysiologic signals were sampled at 1024 Hz, with a lower cut-off of 0.33 Hz and an upper cut-off of 120 Hz (DC amplifiers, Micromed). The electro-oculogram (EOG) was recorded using 2 pairs of bipolar electrodes. Single pulses (TTL) synchronized with stimulus onset were recorded and used offline to segment the continuous EEG data into epochs time-locked to stimulus onset.
Data processing
We analyzed EEG data using NeuroScan software. The EEG signals were corrected for ocular artifacts using an off-line threshold reduction algorithm (NeuroScan Inc.). This method operates by subtracting EOG signals from EEG channels using a linear derivation approach with a spatial filter transformation. The EEG signals were automatically cleared of movement artifacts in which voltage exceeded 100 μV criteria, and the remaining trials were inspected visually to control for minor artifacts. Electrodes with artifacts were interpolated using the Launch Montage Editor (NeuroScan Inc.). In the present study, a maximum of 2 electrodes located on the side of the scalp were interpolated only when necessary. The remaining trials were inspected visually to control for minor artifacts. The EEG data were detached into epochs of 4800 ms, starting 1300 ms before stimulus onset. To exclude the confounding effect of motor action on EEG measures, we analyzed data from correct nonmotor response trials according to the task condition. Then, the EEG data were analyzed with 3 different types of electrophysiologic analysis: ERPs, spectral power analysis, and event-related desynchronization and synchronization (ERD/ERS).
Event-related potentials
We averaged ERPs over a time window of 600 ms with a 200-ms prestimulus onset, and they were then band-pass filtered between 0.33 Hz and 30 Hz (slope 24 dB per octave for a low-pass filter). We measured peak latencies of the ERP components as a function of time from stimulus onset. Latencies of visual N75, P100 and N145 components were measured at the occipital (O1-Oz-O2) electrode sites in the 2 groups. Analyses of the later P200 and N200 ERP components were restricted to the anterior (F3-Fz-F4-C3-Cz-C4) electrode locations.
Spectral power analysis
We converted artifact-free open-eye EEG recordings at rest into the frequency domain using a fast Fourier transform function computed on overlapping 2-second windows (10% Hanning filter). For resting EEG data, we measured slow oscillations in θ frequency band power at frontal (F3, Fz, F4) electrode sites, with the average power between 4 Hz and 7.5 Hz.
Event-related desynchronization and synchronization
To obtain synchronization data, the epoch was digitally filtered in the 4–7.5 Hz (θ) frequency band using a narrow band-pass filter (−48 dB per octave). The filtered epochs were then squared to obtain a signal proportional to the power of the EEG frequency. Within each task and for each electrode, the power from the stimulus onset to 3000 ms was determined as the power during the experimental condition (A), and the mean power from −1000 to 0 ms relative to stimulus onset was determined as the reference period power (R). Then, the averaged data over trials were computed as the percentage power increase (ERS) or decrease (ERD) for a particular time interval in the θ frequency band using the following traditional formula: %ERS = [(A–R)/R] × 100.
For each participant, we measured the culmination peak (ms) and magnitude (%) of the phasic θ ERS and the percentage (%) of sustained θ ERS, respectively, in the 0–500 ms and 650–1400 ms after stimulus onset over frontal (F3, Fz, F4) electrode sites. Amplitudes of the phasic θ ERS were measured at the maximum peak.
As reported previously, a rapid development of sustained θ synchronization occurs during the 500–1600 ms time interval after stimulus onset for the 2-back task in frontal electrodes.20,21 As for ERPs, the sustained θ ERS was assessed in a time period defined by the time lag (500–1600 ms) after subtraction of 150 ms (15% of the total sustained θ) corresponding to rise and fall time.
Statistical analysis
To normalize the variance of the EEG data (i.e., magnitude of θ power at rest and %ERS), we used a logarithmic transformation. We verified the normality of data distribution with skewness and kurtosis tests. Group (controls and patients), task condition (passive, detection, 1-back, 2-back), age, medication (chlorpromazine equivalent units) and a group × task interaction were included as independent variables in a repeated analysis of covariance (ANCOVA) to analyze their respective influence on each of the dependent variables (reaction time, performance and EEG measures). Group, age and medication were the between-subject factors; age and medication were treated as continuous variables. Task condition was treated as a within-subject factor. The significance values of the repeated factors in the ANCOVA were determined using Box’s conservative correction factor. We performed post-hoc analysis using the Scheffé test.36 The statistical threshold was set at p < 0.05. We performed statistical analyses using the Stata software package, version 11.1.
Results
Participants
Fifteen patients with first-episode psychosis (12 men, 3 women; mean age 21.9 [standard deviation (SD) 2.6], range 18–26 yr) participated in this study. Among them, 6 patients had schizophreniform psychotic disorder and 9 had schizophrenia. Five patients had a history of recreational cannabis use. All but 6 patients were taking second-generation antipsychotic medications converted into chlorpromazine equivalent units32 (mean 262 [SD 202]). Of the 6 patients who were not taking antipsychotic medication, 1 was treated with escitalopram. The clinical and pharmacologic characteristics of the patients are summarized in Table 1. We included 18 right-handed controls (9 men, 9 women; mean age 24.4 [SD 1.5], range 22–27 yr) in this study. Controls had no recent history of substance abuse, epilepsy or other neurologic disorders or head injuries. All participants had normal or corrected-to-normal visual acuity.
Clinical and pharmacologic characteristics of patients with first-episode psychosis
Behavioural data
During the n-back testing, reaction times increased as the tasks became more difficult in both controls (F2,34 = 17.7, p < 0.001) and patients with first-episode psychosis (F2,28 = 14.5, p < 0.001). Participants responded the fastest in the detection task, followed by the 1-back (p = 0.015) and 2-back tasks (p < 0.001), independent of their diagnoses (Table 2). In line with a widely accepted hypothesis,37,38 performance in the 2-back task was affected by first-episode psychosis (p = 0.005). It is, however, noteworthy that even during the 2-back task, patients with first-episode psychosis responded correctly in more than 92% of the trials.
Reaction times and performances for the detection, 1-back and 2-back tasks in controls (n = 18) and patients with first-episode psychosis (n = 15)*
Electrophysiologic data
Exogenous event-related potential components
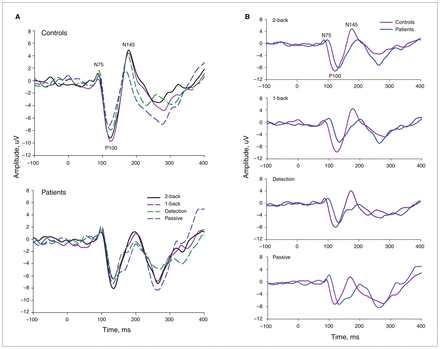
In Figure 1A, waveform ERPs were plotted over the occipital region for each task in each group. As expected, the visual stimuli elicited distinct occipital negative–positive–negative responses at about 75 ms, 100 ms and 145 ms after stimulus onset (Fig. 1B). These sensorial ERP components occurred significantly later for all conditions in patients than in controls (N75, F1,123 = 16.4; P100, F1,123 = 44.43; and N145, F1,123 = 33.0; p < 0.001; Fig. 1B). These differences persisted when we controlled for medication. No other main effect or interaction was observed (Table 3).
(A) Grand average event-related potentials (ERPs) from occipital (O1, Oz, O2) electrode sites for the passive, detection, 1-back and 2-back tasks averaged over no-response trials for controls and patients with first-episode psychosis. (B) Grand average ERPs for the 2 groups during the 4 experimental tasks. Note the delayed latency of the 3 sensory visual ERP components (N75, P100 and N145) for the 4 tasks in patients with first-episode psychosis.
Early and late sensory event-related potential latencies measured on passive, detection, 1-back and 2-back tasks recorded over occipital electrode locations O1, Oz, O2 for controls (n = 18) and patients with first-episode psychosis (n = 15)*
Endogenous event-related potential components
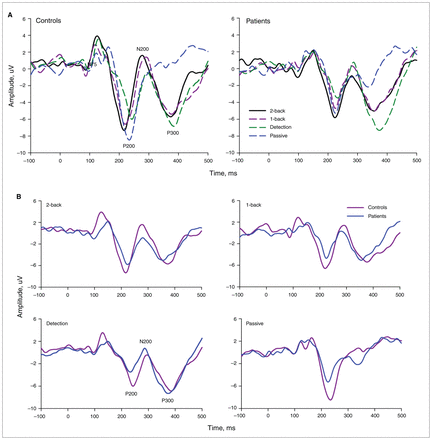
For each task, analysis of averaged ERPs revealed a series of ERP peaks (P200 and N200) over anterior regions in both groups (Fig. 2). P200 and N200 latencies were modulated by task (P200, F3,123 = 10.41; N200, F3,123 = 19.5; p = 0.001). P200 latency was shorter in the 2-back compared with 1-back (p = 0.014), detection and passive tasks (p < 0.001); it was also shorter in the 1-back compared with the detection (p < 0.001) and passive tasks (p = 0.05). N200 latency was shortest in the 2-back, followed by 1-back (p = 0.05), detection and passive tasks (p < 0.001). Unlike P200 latency, a significant group effect was found for N200 latency (F1,123 = 10.1, p = 0.005). In contrast to controls, N200 latency decreased steadily in detection but remained stable between 1-back and 2-back tasks in patients. Both P200 and N200 latencies were free from interaction between group and tasks (Table 4, Fig. 2). The observed differences were also free from medication-related effects. In contrast to latencies, the amplitudes of the previously reported ERPs were free from group-, task- or interaction-related effects.
(A) Grand average waveforms from anterior (F3, Fz, F4, C3, Cz, C4) electrode sites for the 4 experimental tasks, averaged over no-response trials for controls and patients with first-episode psychosis, and (B) grand average event-related potentials for the groups and each experimental task.
Latencies of P200 and N200 endogenous event-related potential components measured on passive, detection, 1-back and 2-back tasks recorded over anterior electrode locations F3, Fz, F4, C3, Cz and C4 for controls (n = 18) and patients with first-episode psychosis (n = 15)*
Spectral power analysis
The magnitude of frontal θ power during a resting state with open eyes was free from group effects (controls, 0.67 ± 0.27; patients, 0.65 ± 0.28 log[μV]; p = 0.95).
Event-related desynchronization and synchronization analysis
θ frequency band
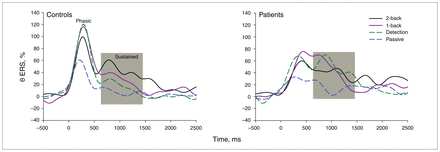
After stimulus presentation, a phasic increase of θ ERS was observed during a 0–500 ms period in all experimental conditions in both groups (Fig. 3, Table 5). In all experimental conditions, the θ ERS culmination peak was delayed in patients compared with controls (F1,123 = 24.4, p < 0.001). There was also a significant task effect (F3,123 = 4.4, p = 0.025) revealing a longer culmination peak latency in each active task compared with the passive task in both groups (p = 0.006). No 2-way interaction between group and task conditions was observed.
Event-related synchronization of the θ (4–7.5 Hz) frequency band as a function of time (ms) for the passive, detection, 1-back and 2-back tasks in controls and patients with first-episode psychosis, measured at frontal (F3, Fz, F4) electrode sites. The Y axis depicts relative (%) power values; positive values stand for synchronization. Visually, controls had significantly higher native phasic θ (4–7.5 Hz) event-related synchronization (ERS) values compared with patients with first-episode psychosis. However, the logarithmic normalization of the data and high interindividual variability led to statistically nonsignificant group differences in phasic θ amplitude. The ERS representation revealed that frontal θ amplitude remained high later in the delay after stimulus presentation. This percentage of induced sustained θ ERS was measured in the 650–1400 ms time window after stimulus onset over frontal (F3, Fz, F4) electrode sites. Note the increase of sustained θ synchronization for the detection and 1-back tasks in patients compared with controls.
Normalized θ event-related synchronization culmination peak and phasic and induced θ event-related synchronization value measured on passive, detection, 1-back and 2-back tasks recorded over frontal F3, Fz, F4 electrode locations for controls (n = 18) and patients with first-episode psychosis (n = 15)*
The phasic θ ERS amplitude was independent from group effect or 2-way interaction between group and task. However, a significant task effect was observed (F3,123 = 6.0, p = 0.019), indicating a lower θ amplitude in the passive task compared with the 2-back (p = 0.027), 1-back and detection tasks (p < 0.001) as well as in the 2-back task compared with the detection task (p = 0.014). The observed culmination peak and amplitude differences were free from medication-related effects.
During the 650–1400 ms interval after stimulus onset, sustained θ was modulated in amplitude according to group (F1,123 = 5.6, p = 0.039) and task (F3,123 = 4.4, p = 0.045). Contrasting with the progressive increase of θ values in controls, patients with first-episode psychosis showed a rapid increase of sustained θ amplitude in the detection and 1-back tasks and a subsequent decline in the 2-back task. The task effect was primarily owing to lower θ ERS amplitude for the passive task compared with active tasks (detection, p = 0.047; 1-back and 2-back tasks, p < 0.001). No interaction between group and tasks was observed.
Discussion
The present data indicate 2 distinct patterns of EEG abnormalities in patients with first-episode psychosis during working memory activation. In line with the early hypothesis of a progressive damage of neural generators involved in memory and attention processes in patients with schizophrenia, patients with first-episode psychosis showed increased exogenous ERP latencies and phasic θ ERS maximums compared with controls. However, they also showed a rapid recruitment of working memory–related neural generators, even in attentional tasks with a low demand on working memory, as evidenced by the decreased N200 latency and increased amplitude of sustained θ ERS in the detection task. In contrast to controls, this recruitment was independent of working memory load in patients with first-episode psychosis. From a clinical viewpoint, this dissociation between the nature of cognitive needs and brain activation should be interpreted within the framework of the perceptual organization impairment theory in patients with schizophrenia. In fact, early abnormalities in the processing by which visual information is structured into coherent patterns at the preattentional and attentional stages are thought to be at the origin of the late development of clinically overt deficits in working memory performances (for a review see Silverstein and Keane39).
As expected, a frontal phasic θ increase was observed in both groups within 500 ms poststimulus, reflecting the encoding of the first visual stimulus that is usually followed by a habituation in the subsequent stimuli.21,24,40 Although the amplitude of this EEG component was preserved in patients with first-episode psychosis, they exhibited a general delay of phasic θ culmination peak independent of the task, suggesting the presence of early deficits in the integration of the incoming visual information for further cognitive process. Supporting this idea, an inability to respond to the first stimulus with an increase in evoked θ activity and to habituate to the second stimulus was described in a visual n-back working memory paradigm24,27 and an auditory sensory modality41–43 in patients with schizophrenia. From a neurophysiologic viewpoint, early frontal θ oscillations reflect initial phases of attention-related encoding include a major phase-locked component to stimuli. This physical property supports a strong relation between early θ activity and visual ERPs.44,45 Importantly, recent investigations revealed that early visual ERP deficits during working memory encoding contribute to the deficits in evoked oscillatory activity in these patients.23 Paralleling this finding, our observations on exogenous sensory ERPs demonstrated the presence of brain activation deficits in the early sensory treatment of all tasks in patients with first-episode psychosis.46
Numerous functional imaging studies have revealed that participants performing an n-back working memory task activated distinct cortical networks within the frontal and parietal areas that correspond to each phase of cognitive processing (for a review see Cabeza and colleagues47). Two earlier studies revealed that modulation of P200 and N200 latencies during the n-back task is related to the development of an additional working memory load–related parietal component.14,15 To our knowledge, this study is the first to explore P200 and N200 latencies in patients with first-episode psychosis using working memory paradigms. As expected, P200 latencies decreased as a function of working memory load in patients and controls. The same pattern was evident for N200 latencies in controls but not in patients with first-episode psychosis who displayed a steady decrease of N200 latency in the detection task that was independent of working memory load. It is likely that these patients activate certain working memory neural generators in a nonspecific manner for attentional tasks with a low demand on working memory. This scenario was further supported by our observations on sustained θ ERS, which is known to increase during working memory tasks in young adults.48,49 It occurs during the retention interval and is thought to reflect incremental retention and maintenance of new items for further task requirements.17,50 As expected, its amplitude increased in n-back tasks compared with the detection task in controls. In agreement with the literature,27 patients with first-episode psychosis displayed high sustained θ ERS values in the detection task that remained stable independent of the working memory load. Interestingly, a similar floor effect in the activation of low-frequency band oscillatory activities during maintenance of working memory load was also reported in patients with early-onset schizophrenia.27 Although the authors did not use a control task, they found higher θ amplitudes for the 1-back task that remained stable in the 2-back task in patients compared with controls. Altogether, these findings support the concept of a blunted EEG response in patients with first-episode psychosis who recruit the maximum neural generators in simple attention conditions without being able to modulate their brain activation facing the complexity of increasing working memory needs. It is, however, noteworthy that the patients were able to preserve their n-back task performances. Earlier EEG and fMRI studies also revealed hyperfrontality combined with increased parietal activity in patients with first-episode psychosis during working memory tasks.10,51–54 It is thus likely that the early activation of parietal generators in attentional tasks observed in our study may represent a compensatory mechanism55 to facilitate the maintenance of executive performances in patients with first-episode psychosis.
Limitations
Strengths of the present study include the strict exclusion of patients with other psychiatric comorbidities, control for medication load, and concomitant analysis of exogenous and endogenous ERPs and θ band synchronization. Several limitations should also be considered when interpreting the present data. Clinically, no distinction was made between schizophrenia symptoms (i.e., positive v. negative and mixed symptoms) owing to the small sample size. Given the limited number of women in our patient sample, we cannot draw definite conclusions about the effect of sex on our observations. Although we controlled for the global load of neuroleptics, we cannot formally rule out the qualitative effect of these agents on EEG measures. Physiologically, the present conventional ERD/ERS measure did not allow for distinguishing between evoked and induced θ components.
Conclusion
Future studies in larger series combining multichannel EEG and fMRI investigations for optimal spatial source localization, as well analysis of both evoked and induced components of brain rhythms in well documented clinical subtypes of first-episode psychosis, are needed to further explore the early changes in brain activation patterns that precede the decline in cognitive performances in patients with this condition.
Acknowledgments
This project was funded by the Swiss National Foundation for Scientific Research, grant 3100A0/103770/1, and the Research and Development Hospital, grant 02-1-122.
Footnotes
Contributors: P. Missonnier, P. Giannakopoulos and M.C.G. Merlo designed the study. P. Missonnier, L. Curtis, D. Canovas, F. Chantraine and J. Richiardi acquired the data. P. Missonnier, F.R. Hermann, A. Zanello, M. Badan Bâ, P. Giannakopoulos and M.C.G. Merlo analyzed the data. P. Missonnier, F.R. Hermann, L. Curtis, P. Giannakopoulos and M.C.G. Merlo wrote the article. All authors reviewed the article and approved its publication.
Competing interests: None declared for P. Missoner, F.R. Hermann, A. Zanello, M. Badan Bâ, L. Curtis, D. Canovas, F. Chantraine, J. Richiardi and P. Giannakopoulos. M.C.G. Merlo declares board membership at Sandoz and declares having received speaker fees from AstraZeneca, Bristol-Myers Squibb, Essex Chemie AG, GlaxoSmithKline, Janssen-Cilag, Eli Lilly, Lundbeck, Mepha, Novartis, Pfizer, Sandoz, Sanofi, Servier, Vifor Pharma and Zeller. He also received funding for travel, accommodations and meeting expenses from Eli Lilly and Pfizer.
- Received April 4, 2011.
- Revision received July 25, 2011.
- Revision received September 1, 2011.
- Accepted September 6, 2011.