Abstract
Background: Psychopathic offenders inevitably violate interpersonal norms and frequently resort to aggressive and criminal behaviour. The affective and cognitive deficits underlying these behaviours have been linked to abnormalities in functional interhemispheric connectivity. However, direct neurophysiological evidence for dysfunctional connectivity in psychopathic offenders is lacking.
Methods: We used transcranial magnetic stimulation combined with electroencephalography to examine interhemispheric connectivity in the dorsolateral and motor cortex in a sample of psychopathic offenders and healthy controls. We also measured intracortical inhibition and facilitation over the left and right motor cortex to investigate the effects of local cortical processes on interhemispheric connectivity.
Results: We enrolled 17 psychopathic offenders and 14 controls in our study. Global abnormalities in right to left functional connectivity were observed in psychopathic offenders compared with controls. Furthermore, in contrast to controls, psychopathic offenders showed increased intracortical inhibition in the right, but not the left, hemisphere.
Limitations: The relatively small sample size limited the sensitivity to show that the abnormalities in interhemispheric connectivity were specifically related to the dorsolateral prefrontal cortex in psychopathic offenders.
Conclusion: To our knowledge, this study provides the first neurophysiological evidence for abnormal interhemispheric connectivity in psychopathic offenders and may further our understanding of the disruptive antisocial behaviour of these offenders.
Introduction
Psychopathic individuals show profound impairments in affective functioning and live a life characterized by impulsivity, irresponsibility and recurrent criminal behaviour. Although most research on psychopathy has focused on the amygdala and ventromedial regions of the prefrontal cortex, available evidence suggests that the corpus callosum is also affected.1–3 However, interhemispheric connectivity is often overlooked as a potential important contributor to the pathogenesis of antisocial personality disorder (ASPD) and psychopathy. Interhemispheric connectivity reflects the process of exchange and integration of information between the cerebral hemispheres in which the corpus callosum plays a vital role. It has been suggested that the corpus callosum is involved in functional cerebral specialization, that is the lateralization of cognitive functions, in which individuals with psychopathy show profound impairments.1–3 In support of this notion, psychopathic offenders demonstrate unusual lateralization of functions such as language and divided attention.3–5 In general, cognitive functions involve a complex interplay of facilitation and inhibition of contralateral activity. Specifically, although some functions involve facilitation of interhemispheric connectivity, other functions (e.g., language, motor-related behaviours) are more lateralized and involve the upregulation of activity in 1 hemisphere while simultaneously inhibiting activity in the contralateral hemisphere.6 Abnormalities in connectivity mechanisms may thus lead to impaired cognitive functioning, which is often observed in psychopathic offenders.4,7
Consistent with the idea of abnormal interhemispheric connectivity, patients with ASPD show bilateral reductions in white matter integrity in the genu, the anterior part of the corpus callosum.8 Additional evidence for interhemispheric connectivity abnormalities in psychopathic individuals has been provided by Raine and colleagues,9 who showed that psychopathic individuals have increased callosal white matter and increased callosal length, but reduced callosal thickness. Indirect evidence from functional magnetic resonance imaging (fMRI) in the same study suggests that functional interhemispheric connectivity is increased in psychopathic offenders.9 Using behavioural measurements, Hiatt and Newman10 suggest a possible directional component to these connectivity abnormalities, as psychopathic offenders have a prolonged inter-hemispheric transfer time. In addition, the results also suggest that signal transfer from the right to the left frontal cortex connectivity may be specifically affected in psychopathic offenders.10 However, direct neurophysiological evidence providing substantiation of right to left interhemispheric connectivity abnormalities in psychopathic offenders is lacking.
Transcranial magnetic stimulation (TMS) is a valuable research tool for measuring physiologic aspects of interhemispheric connectivity.11,12 Interhemispheric signal propagation (ISP) represents a novel and reliable approach to measuring interhemispheric connectivity in motor and nonmotor regions of the cortex through combined TMS with electroencephalography (EEG).13,14 In this paradigm, magnetic pulses are applied to the cerebral cortex, after which excitatory fibres that run through the corpus callosum are activated. These excitatory fibres terminate on inhibitory interneurons of the contralateral hemisphere.12,15,16 Particularly, TMS-induced activation of the frontal cortex induces a contralateral response in homotopical regions, which recently has been shown using diffusion tensor imaging (DTI) to depend on the microstructural integrity of the corpus callosum.14 Importantly, the contralateral response is smaller than the ipsilateral activation, which is proposed to reflect the contralateral inhibitory processes previously described.14 Measuring ISP, therefore, provides an index of interhemispheric connectivity from the dorsolateral prefrontal cortex (DLPFC),13,14 a cortical region that is more closely associated with social behaviour and cognition.13,17
The combination of TMS and EEG provides a means to directly tap into intercortical circuits and study potential abnormalities in functional interhemispheric connectivity. The aim of the present study was to explore functional interhemispheric connectivity in psychopathic offenders using TMS and to provide the first neurophysiological data on inter-hemispheric (dys)function in individuals with psychopathy. As functional interhemispheric connectivity only partly relies on signal transfer through the corpus callosum, the aim of our study was to assess functional interhemispheric connectivity rather than corpus callosum functioning. In keeping with earlier findings, we hypothesized that psychopathic offenders would demonstrate functional deficiencies in right to left interhemispheric connectivity.
Methods
Participants
Right-handed18 male psychopathic offenders were recruited through posters displayed in halfway houses in the Greater Toronto Area and through the Law and Mental Health Program at the Centre for Addiction and Mental Health. Offenders were informed about the study and screened with a standard clinical interview to check for contraindications to TMS,19 medication usage and neurologic disorders. Next, we asked them to sign a release of information, after which we reviewed case notes and psychological assessments to retrieve the Hare Psychopathy Checklist-revised, second edition (PCL-R), score.20 The PCL-R interview and concomitant file review was administered by trained, certified forensic psychologists and psychiatrists for clinical and risk assessment purposes. Exclusion criteria were age younger than 18 years or older than 65 years and diagnosis of schizophrenia, schizophreniform/psychotic disorders, mood disorders, anxiety disorders, or any comorbid personality disorders (e.g., borderline personality disorder). These conditions were ruled out by way of an interview and confirmed with a review of previous psychological assessments completed by an experienced clinician (T.S.). All psychopathic offenders had previously been administered the Shipley Institute of Living Scale and the Wechsler Adult Intelligence Scale-R (WAIS-R).21 The Shipley Institute of Living Scale screens for organic brain damage and has a high correlation (r = 0.85) with the full scale WAIS-R.22
We recruited healthy male control participants, who were screened with a standard clinical interview assessing contraindication to TMS19 and in whom right handedness was confirmed.18 Current forms of psychopathology were screened using the Personality Assessment Screener (PAS; Psychological Assessment Resources Inc.). The PAS is a self-administered inventory of adult personality and psychopathology (e.g., personality, depression, somatic disorders, anxiety, anxiety-related disorders, schizophrenia). It is composed of nonoverlapping clinical, treatment, interpersonal and validity scales. Specifically, the PAS measures manifestation of clinical syndromes, providing information to assist diagnosis, treatment and screening for all psychopathology corresponding to DSM-IV categories.23
Exclusion criteria for both psychopatic offenders and controls were substance abuse or dependence in the 6 months preceding the study, as per the DSM-IV-TR. All psychopathic offenders were subjected to regular drug screening as part of the terms of their parole and these drug tests indicated that none had been using drugs at or around the time of testing.
The ethics committee at the Centre for Addiction and Mental Health approved our study, and we obtained written informed consent from each participant. All participants were paid for taking part in this study.
The study consisted of a 6-hour TMS-EEG session and a 2-hour TMS only session, which were performed on separate days. In the TMS only session, we measured cortical inhibition and facilitation from the left and right motor cortex.
Interhemispheric signal propagation
We applied single monophasic pulses to the left and right motor cortex and DLPFC using a figure-8 coil with a 7 cm loop diameter connected to the Bistim Module. The Bistim Module was connected to 2 Magstim 200 stimulators (Magstim Company). Owing to persistent reoffending among psychopathic offenders in the community, we decided not to perform MRI for localization of the DLPFC to increase the chance that offenders would finish the study. For activation of the DLPFC, electrodes F5 (left hemisphere) and F6 (right hemisphere) were used according to the International 10–20 system for EEG. Stimulation over these electrodes approximates ideal activation of the DLPFC when MRI-based co-registration is not available.24 For the motor cortex, the coil was placed over the site that elicited the strongest motor-evoked potentials (MEPs) from the abductor pollicis brevis (APB). This site was marked to ensure stable placement over the same spot throughout the testing session. The coil was held tangentially on the head with the handle pointing backward at 45° laterally from the midline. Over the left and right motor cortex and DLPFC, we administered 100 suprathreshold pulses with an interstimulus interval (ISI) of 5 seconds. Conditions were counterbalanced between participanst. We recorded EEGs with a 64-channel Synamps2 DC-coupled EEG system (Compumedics). We used 4 electrodes to record eye movement–induced artifacts, 2 on the outer side of the eyes, and 1 above and 1 below the left eye. The reference electrode was placed just posterior to the Cz electrode (vertex). We recorded EEG signals down current at a sampling rate of 20 kHZ, and they were low-pass filtered at 100Hz. Offline processing was done with Neuroscan (Compumedics). Data were first downsampled to 1 kHZ and cut into segments that included 1000 ms prestimulus and 1000 ms poststimulus activity. Segments were baseline-corrected until 110 ms before the test stimulus to ensure a TMS-free baseline. Posttest stimulus intervals (25–1000 msec) that were artifact-free were extracted and digitally filtered using a zero-phase shift 1–100 Hz bandpass filter (48dB/Oct). Hereafter, epochs were visually inspected to exclude movement, eye blink or TMS artifacts. The remaining epochs were averaged and eye blink corrected according to previously described methods.25,26 The average cortical-evoked potentials were bandpass filtered (1–50Hz) for each participant. We measured cortical-evoked potentials (CEPs) over the left hemisphere under electrode C3, while electrode C4 was used to measure CEPs over the right hemisphere. Previous studies have shown that these electrodes are closest to the optimal site for APB activation using TMS.27 To record activation of the DLPFC, we used electrodes AF3 and AF4. In the stimulated hemisphere, the area under the rectified curve was measured 50–150 ms post-stimulus. We chose an onset of 50 ms, as it reflects the earliest time frame of artifact-free data.13,14 We used an offset of 150 ms, as TMS-induced activation of γ-aminobutyric acid (GABA)-B inhibitory neurotransmission lasts on average 150 ms.28 In keeping with previous findings,11,12,14 we used an interhemispheric conduction time of 10 ms; therefore, the propagated signal to the contralateral hemisphere was measured 60–160 ms poststimulus. Interhemispheric signal propagation is calculated as the ratio of cortical evoked potential on the stimulated hemisphere to the contralateral cortical evoked activity (ISP = [area under rectified curve contralateral hemisphere ÷ area under rectified curve stimulated hemisphere] × 100).14
Interhemispheric inhibition
Using a paired pulse paradigm we also indexed a second measure for interhemispheric connectivity: interhemispheric inhibition (IHI). We assessed IHI by applying a conditioning stimulus to the ipsilateral motor cortex followed by a test stimulus to the contralateral motor cortex.11 When compared with an MEP induced by a single pulse to the contralateral hemisphere, the conditioning stimulus attenuates the MEP by about 50%.11,12 The ISI was set at 10 ms. We measured IHI without the EEG cap and without the Bistim Module. For the left hemisphere we used a Magstim figure-8 coil with a 7 cm loop diameter, and for the right hemisphere we used a Magstim figure-8 coil with an 8 cm loop diameter.
Cortical inhibition and facilitation
To check for possible abnormalities in local hemispheric processing, we measured baseline cortical inhibition and facilitation over the left and right motor cortex. For this we indexed the cortical silent period (CSP), short interval intracortical inhibition (SICI) and intracortical facilitation (ICF). The CSP taps into GABAB receptor–mediated neurotransmission, whereas SICI is more commonly associated with GABAA receptor functioning.29 The ICF relates to N-methyl-d-aspartate (NMDA) receptor–mediated mechanisms30 (see the Appendix, available at cma.ca/jpn, for a schematic representation of the different TMS paradigms). We applied TMS to the hand area of the left and right motor cortex with a 70 mm loop figure-8 magnetic coil and 2 Magstim 200 magnetic stimulators (Magstim). The coil was held tangentially on the head with the handle pointing backward at 45° laterally from the midline.
The cortical silent period (CSP) is measured after motor cortical stimulation in a moderately contracted (hand) muscle:31 stimulation induces an MEP after which electromyographic activity is momentarily absent, commonly referred to as the CSP.31 Measurement of the CSP duration was obtained in moderately tonically active APB (i.e., 20% of maximum contraction) by stimulating the motor cortex with intensities of 140% of resting motor threshold. We performed 10 trials. The CSP duration was the time from the MEP onset to the return of any voluntary electromyography (EMG) activity. This is referred to as the absolute CSP and ends with a deflection in the EMG waveform.32 For SICI and ICF, a subthreshold conditioning stimulus (80% of resting motor threshold) preceded the suprathreshold test stiumuls, which was adjusted to produce an average MEP of 0.5–1.5 mV peak-to-peak amplitude in the contralateral APB muscle.33 We applied the conditioning stimulus to the motor cortex before the test stimulus at 1 of 5 ISIs: 2 ms and 4 ms for the SICI and 10 ms, 15 ms and 20 ms for the ICF. We performed 72 trials, 12 for each condition. For the SICI and ICF, changes in the test stimulus MEP amplitude at each ISI were expressed as a percentage of the mean unconditioned MEP amplitude.34 The SICI and ICF were averaged over the different ISIs. The order of administration of the 2 paradigms was counterbalanced over hemispheres and between participants.
Electromyography recording
We recorded a surface EMG from the right and left first APB muscles with disposable disc electrodes placed in a tendon-belly arrangement. The participant maintained relaxation throughout the experiment, and the EMG was monitored on a computer screen and via speakers at high gain. The signal was amplified (Intronix Technologies Corporation Model 2024F), filtered (band-pass 2 Hz to 5 kHz), digitized at 5 kHz (Micro 1401, Cambridge Electronics Design) and stored in a laboratory computer for offline analysis.
Resting motor threshold
We measured resting motor threshold before testing with and without the EEG cap on the left and right motor cortex. The resting motor threshold was defined as the minimum intensity that would evoke an MEP of at least 50 μV in 5 of 10 consecutive trials.33 Prior to testing, we determined the intensity needed for 1 mV MEPs with and without the EEG cap and from both hemispheres.
Statistical analyses
To test for differences in ISP, we conducted a repeated-measures general linear model with region (motor cortex v. DLPFC) and direction (left to right v. right to left) as within-subjects variables and group (psychopathic offenders v. control) as a between-subjects variable. Separate independent sample t tests were applied to check for differences in CEPs on the left and right motor cortex and DLPFC.
For IHI, we conducted a repeated-measures general linear model with direction (left to right v. right to left) as a within-subjects variable and group (psychopathic offenders v. controls) as a between-subjects variable. As intensity of the conditioning stimulus and test stimulus have been shown to influence IHI,12 we checked the conditioning stimulus and test stiumuls for differences between conditions and groups.
For the CSP, SICI and ICF, we conducted 3 separate repeated-measures general linear models with hemisphere (left v. right) as s within-subjects variable and group (psychopathic offenders v. controls) as a between-subjects variable. Additional independent samples t tests were conducted to check for differences in test pulses in the SICI and ICF paradigm.
A small subgroup of psychopathic offenders were taking medication; therefore, we also conducted analyses excluding these participants.
The PCL-R consists of 2 main factors. Factor 1 represents the affective/interpersonal deficits and factor 2 includes the antisocial lifestyle aspects map.20 We calculated Pearson product-moment correlation coefficients to investigate whether there was a linear association between the PCL-R factors and TMS measures. The α level of significance was set at 0.05, 2-tailed.
Results
We enrolled 18 psychopathic offenders (mean age 33.4 ± standard deviation [SD] 6.8 yr), all of whom scored 25 or higher on the PCL-R (mean 28.8 ± 2.7). All psychopathic offenders scored in the average range on the the Shipley Institute of Living Scale, corresponding to a score of 90–110 on the WAIS-R.21 The spectrum of criminal activity included murder, manslaughter, aggravated sexual assault, (bank) robbery, unlawful confinement, burglary, home invasion, human trafficking and a number of fraudulent crimes. We also enrolled 15 healthy controls (mean age 32.1 ± 9.1 yr). Sixteen psychopathic offenders participated in the TMS-EEG session, and 12 participated in the TMS only session. All healthy controls participated in both the TMS-EEG and the TMS only session.
All participants tolerated TMS well, and no adverse events occurred. The TMS-EEG ISP data of 1 control partipant were excluded owing to inferior signal quality, and the TMS-EEG ISP data of 1 psychopathic offender were excluded owing to the data being outliers (> 3 SDs above the mean). No differences were found in motor threshold or in the intensity needed for 1mV MEPs (Table 1). The groups did not differ in age (F1, 32 = 0.066; p = 0.80). Unfortunately we did not have precise IQ scores (only estimates based on the SILS); therefore, we were not able to test them statistically. For the motor threshold with and without cap, the hemisphere × group interactions were not significant (F1,30 = 0.915, p = 0.35 and F1,33 = 0.203, p = 0.65, respectively). With regards to 1 mV intensity with and without cap, the hemisphere × group interactions were not significant (F1,30 = 1.217, p = 0.28 and F1,33 = 1.125, p = 0.30, respectively).
Comparison of the motor threshold and intensity needed to achieve a 1 mV motor-evoked potential amplitude of controls and psychopathic offenders
Interhemispheric signal propagation
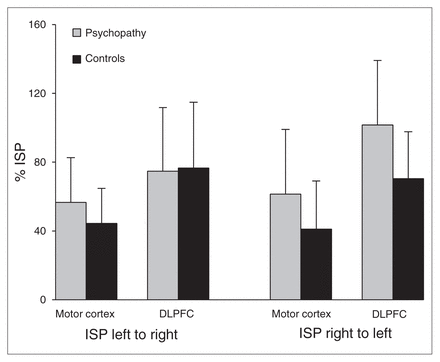
The repeated-measures general linear model yielded a significant direction × group interaction (F1,25 = 5.375, ηp2 = 0.177, p = 0.029), and showed that psychopathic offenders have a global increase in right to left ISP (Fig. 1). The region × direction × group interaction was not significant, (F1,25 = 0.924, p = 0.35). No difference was observed between psychopathic offenders and healthy controls in the CEPs on either hemisphere (left motor cortex: F1,25 = 0.001, p = 0.97; left DLPFC: F1,25 = 0.379, p = 0.54; right motor cortex: F1,25 = 0.191, p = 0.66; right DLPFC: F1,25 = 0.252; p = 0.62).
Interhemispheric signal propagation (ISP) per cortical area in psychopathic offenders and healthy controls. DLPFC = dorsolateral prefrontal cortex.
Interhemispheric inhibition
The repeated-measures general linear model did not generate a significant direction × group interaction (F1, 25 = 1.335, p = 0.26), and there was no main effect for direction (F1,25 = 0.283, p = 0.60). Test pulses on either hemisphere did not differ between psychopathic offenders and healthy controls (left hemisphere: F1,25 = 0.29, p = 0.10; right hemisphere: F1,25 = 0.31, p = 0.90). Conditioning stimuli also did not differ (left hemisphere: F1,25 = 2.8, p = 0.10; right hemisphere: F1,25 = 0.073, p = 0.79).
Cortical inhibition and facilitation
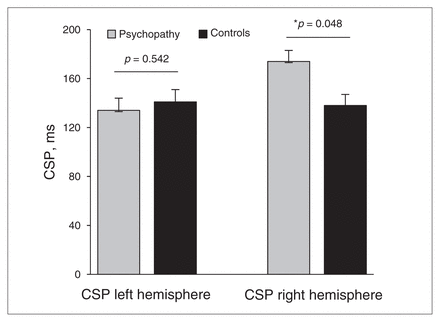
For the CSP, we observed a significant hemisphere × group interaction effect (F1,26 = 10.350, ηp2 = 0.270, p = 0.003. Post hoc 1-way analyses of variance showed that compared with healthy controls, psychopathic offenders exhibited significantly longer CSPs in the right hemisphere (F1,26 = 4.267, p = 0.048), but not in the left hemisphere (F1,26 = 0.381, p = 0.54; Fig. 2). The CSPs measured from the left and right motor cortex did not differ significantly in healthy controls (F1,14 = 0.018, p = 0.90), but did differ in psychopathic offenders (F1,14 = 19.66, p = 0.001). For SICI, the crucial hemisphere × group interaction was not significant (F1,26 = 0.319, p = 0.58]. Also, the hemisphere × group interaction for ICF was not significant (F1,26 = 0.708, p = 0.41]. No group differences in the magnitude of the test pulses were observed for SICI and ICF, (left hemisphere: F1,26 = 2.002, p = 0.17; right hemisphere: F1,26 = 0.263, p = 0.61).
Psychopathic offenders demonstrate an increase in cortical silent period (CSP) in the right motor cortex.
We found no correlations between Factors 1 and 2 of the PCL-R and TMS measures (all p > 0.09, uncorrected for multiple comparisons).
Medication
Two psychopathic offenders were prescribed methadone for a previous heroin addiction. Two other offenders received quarterly intramuscular injections of leuprolide owing to repeated sexual offenses. Post hoc repeated-measures general linear models for ISP and CSP were conducted to exclude the potential influence of these confounds. The direction × group interaction for ISP remained significant (F1,21 = 4.798, p = 0.039). The direction × group interaction for CSP also remained significant (F1,22 = 11.248, p = 0.003).
Discussion
In the present study we examined interhemispheric connectivity in psychopathic offenders compared with healthy controls and found abnormalities in right to left interhemispheric connectivity, as evidenced by higher right to left ISP in psychopathic offenders. In addition, psychopathic offenders showed increased cortical inhibition of the right, but not the left, motor cortex.
Functional and structural interhemispheric connectivity deficits have been reported in individuals with psychopathy9 and patients with ASPD.8 Our study supports and extends these findings by showing that right to left connectivity is affected in psychopathic offenders, whereas left to right connectivity is intact. In psychopathic offenders, TMS-induced neural activation in the right frontal hemisphere results in a similarly large CEP in the left hemisphere, especially in prefrontal areas. Whereas in healthy controls TMS-induced activation is propagated to the contralateral hemisphere and subsequently inhibited, our data suggest that in psychopathic offenders, the left hemisphere does not adequately process input from the right hemisphere. While specific contributions to cognition and behaviour of the left and right DLPFC have been identified,17,35–37 much less is known about the connectivity between these regions that presumably underlie cognition and behaviour. Some evidence suggests that connectivity between homotopical prefrontal regions may be implicated in approach versus withdrawal behaviour38,39 in which psychopathic offenders may have subtle deficits.40 Approach-related behaviour (i.e., behaviour directed at the attainment of rewards) recruits the left prefrontal cortex, whereas withdrawal-related behaviour (i.e., behaviour intended to avoid potential punishment) is associated with the right prefrontal cortex.41 Once individuals with psychopathy initiate goal-directed behaviour, they have difficulty reallocating attention and regulating their behaviour based on new peripheral information.42 In line with this, evidence shows that psychopathic offenders have superior selective attention, but are unresponsive to information that is peripheral to the attainment of their goal, which may account for the poor response modulation that has been observed in individuals with this disorder.42–44 In other words, when engaged in approach-related behaviour, psychopathic offenders tend to persevere their initial responses45 and are unable to modulate their behaviour.40 One could argue that this is explained by the suboptimal processing of input from the right prefrontal cortex mediating behavioural withdrawal.8,40,41
We also observed that psychopathic offenders demonstrated increased CSP, but not SICI or ICF, in the right motor cortex. It is likely that CSP reflects GABAB-mediated inhibitory neurotransmission, while SICI is more commonly associated with GABAA-receptor functioning.12,46 The exact mechanisms of interhemispheric connectivity remain the subject of debate,16 but evidence suggests that GABAB receptors are strongly involved in interhemispheric connectivity.12,16 Therefore, the proposed increase in GABAB-mediated neurotransmission in the right motor cortex in psychopathic offenders may contribute to the changes in interhemispheric connectivity. By contrast, the increase of GABAB receptor–mediated inhibitory neurotransmission in the right motor cortex of psychopathic offenders may also reflect a local response to increased interhemispheric connectivity. Both are conceivable and have been reported in the literature,6 but the current data cannot provide conclusive evidence. Given the possible neurodevelopmental aspect of interhemispheric connectivity deficits in individuals with psychopathy,9 research in children or adolescents with psychopathic traits may provide more conclusive evidence. In line with this, Finger and colleagues47 have shown that adolescents with conduct disorder or oppositional defiant disorder combined with psychopathic (i.e, callous-unemotional) traits have intact microstructural integrity of white matter tracts. However, deficits in functional connectivity are already present in these youths.47 If neurodevelopmental abnoralities in functional interhemispheric connectivity are indeed part of the pathogenesis of psychopathy, research in children and adolescents with psychopathic traits would greatly further our knowledge on potential interhemispheric connectivity abnormalities in individuals with this profoundly disruptive disorder.
Limitations
Several limitations of this study should be mentioned. Although not significantly different, ISP from right to left was higher than ISP from left to right in prefrontal areas in psychopathic offenders, suggesting that right to left connectivity abnormalities may be more pronounced between the prefrontal cortices (Figs. 1, 3 and 4). However, because of the relatively small sample, we could not demonstrate significantly impaired connectivity in these prefrontal areas. Owing to the relatively small sample size, we also did not correct for multiple comparisons. In addition, we were not able to demonstrate any correlations with the total score on the PCL-R or its subfactors. Furthermore, another control group consisting of nonpsychopathic offenders would constitute an important addition to the possible self-selection bias in the recruitment of healthy controls via advertisements. This would also account for potential influences of drug abuse, as we cannot completely rule out that some participants may have used cannabis or alcohol in the months before testing or that there was an influence of drug withdrawal on the current findings. In addition, decreases in cortical inhibition are often detected in psychiatric patients.30,48–50 In contrast, increases in cortical inhibition have also been reported, for instance, in abstinent cocaine-dependent patients.51 Similar to cocaine, drugs that target the dopamine (DA) system (e.g., pergolide, L-DOPA, clozapine) prolong CSP.52–55 As disruption of DA and the mesolimbic reward pathway has been observed in individuals with psychopathic traits,56–58 one could speculate that, to a certain extent, cortical dysfunction in psychopathic offenders could be a corollary of aberrant subcortical dopaminergic activity impacting negatively on interhemispheric connectivity. Also, we recently showed decreased cortical inhibition in the left DLPFC59 in psychopathic offenders. Whether, similar to the right motor cortex, cortical inhibition in the right DLPFC is overly inhibited remains unclear, but these findings could suggest that psychopathic offenders show a unique neurophysiological profile in which the left frontal cortex shows signs of decreased inhibition, while the right hemisphere appears overly inhibited. This exciting possibility warrants further investigation with research tools that provide evidence other than what traditional methods, such as fMRI, are able to generate. Finally, abnormalities in frontal interhemispheric connectivity in psychopathic offenders may relate to the decrease in cortical inhibition that we recently observed in the DLPFC in psychopathic offenders.59 However, as opposed to inhibitory effects, facilitatory effects could also explain our data. At present, our data do not provide concrete evidence favouring inhibitory or facilitatory effects. In fact, the sometimes fast transition from inhibitory to facilitatory effects should be emphasized. For example, a TMS study involving subthreshold stimulation showed that at very short intervals (~7 ms) the ventral premotor cortex has inhibitory effects on the primary motor cortex.60 This inhibitory effect changes into a facilitatory one when the participant performs a task that recruits both the ventral premotor and primary motor cortex.60 Therefore, it should be noted that if neuronal flexibility is already present at short latencies, one would expect such mechanisms to also be present at the longer latencies used in this study. To disentangle structural callosal contributions to interhemispheric connectivity from local inhibitory or facilitatory mechanisms involving GABA/NMDA, future studies should address this issue by combining DTI with TMS-EEG measures of interhemispheric connectivity.
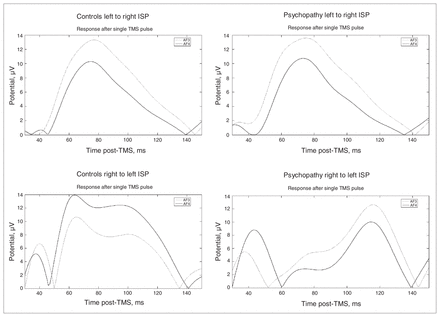
The mean amplitude of the cortical evoked potentials is represented at different times (successive lines) in healthy controls (left 2 columns) and psychopathic offenders (right 2 columns). The mean amplitude is colour coded. DLPFC = dorsolateral prefrontal cortex; TMS = transcranial magnetic stimulation.
Temporal representation of the activity of the prefrontal electrodes, thought to measure dorsolateral prefrontal cortex activity. ISP = inter-hemispheric signal propagation; TMS = transcranial magnetic stimulation.
Conclusion
To our knowledge, this is the first TMS-EEG study to provide direct neurophysiological evidence for abnormalities in right to left interhemispheric connectivity in psychopathic offenders. These novel findings may provide a neurophysiological basis for our understanding of the immensely disruptive antisocial behaviour of psychopathic offenders.
Acknowledgements
We thank all volunteers whose participation was essential to the completion of this work. We also thank the reviewers for their very helpful and insightful comments. Y. Sun declares having received a Master’s Awards from the Canadian Institutes of Health Research (CIHR). Z.J. Daskalakis declares being supported in part by a CIHR Clinician Scientist Award, an operating grant from the Ontario Mental Health Foundation, and a Constance and Stephen Lieber through a National Alliance for Research on Schizophrenia and Depression (NARSAD) Lieber Young Investigator award. D.J.L.G. Schutter declares being supported by an Innovational Research Grant (VIDI 452–07–012) from the NWO.
Footnotes
↵* These authors contributed equally to this work.
Competing interests: Z.J. Daskalakis declares external funding from Neuronetics Inc. and Aspect Medical Inc. He also declares having received travel support from Pfizer Inc. and external funding from Neuronetics Inc., Aspect Medical, Inc. and Pfizer, Inc. No other competing interests declared.
Contributors: S. Hoppenbrouwers, J. McMaster, Z. Daskalakis and D. Schutter designed the study. S. Hoppenbrouwers, D. de Jesus, T. Stirpe and G. Hughes acquired the data, which S. Hoppenbrouwers, Y. Sun and D. Hofman, analyzed. S. Hoppenbrouwers and Z. Daskalakis wrote the article, which all authors reviewed and approved for publication.
- Received March 5, 2012.
- Revision received October 23, 2012.
- Revision received December 10, 2012.
- Revision received February 23, 2013.
- Accepted April 5, 2013.