Abstract
Background: Previous studies have reported MRI abnormalities of the corpus callosum (CC) in patients with bipolar disorder (BD), although only a few studies have directly compared callosal areas in psychotic versus nonpsychotic patients with this disorder. We sought to compare regional callosal areas in a large international multicentre sample of patients with BD and healthy controls.
Methods: We analyzed anatomic T1 MRI data of patients with BD-I and healthy controls recruited from 4 sites (France, Germany, Ireland and the United States). We obtained the mid-sagittal areas of 7 CC subregions using an automatic CC delineation. Differences in regional callosal areas between patients and controls were compared using linear mixed models (adjusting for age, sex, handedness, brain volume, history of alcohol abuse/dependence, lithium or antipsychotic medication status, symptomatic status and site) and multiple comparisons correction. We also compared regional areas of the CC between patients with BD with and without a history of psychotic features.
Results: We included 172 patients and 146 controls in our study. Patients with BD had smaller adjusted mid-sagittal CC areas than controls along the posterior body, the isthmus and the splenium of the CC. Patients with a positive history of psychotic features had greater adjusted area of the rostral CC region than those without a history of psychotic features.
Limitations: We found small to medium effect sizes, and there was no calibration technique among the sites.
Conclusion: Our results suggest that BD with psychosis is associated with a different pattern of interhemispheric connectivity than BD without psychosis and could be considered a relevant neuroimaging subtype of BD.
Introduction
Bipolar disorder (BD) is associated with widespread abnormalities of white matter tracts, including the corpus callosum (CC).1–3 Microstructural impairments of white matter are increasingly considered a relevant endophenotype for genetic studies in patients with BD,1 reinforcing the importance of large-scale neural network dysfunctions in the etiology and course of the disorder.2
The CC is the major interhemispheric white matter pathway that includes about 200 million axons. Corpus callosum maturation begins in childhood and continues into early adulthood.4 In humans, the extensive bilateral projections of callosal fibres provide a unique pathway for interhemispheric transfer. A meta-analysis has highlighted a reduction of the total mid-sagittal area of the CC in patients with BD.5 However, results in patients with BD are more heterogeneous with respect to the area differences in the CC subregional mid-sagittal areas, including their association with clinical features. Reduced posterior CC area is the most commonly reported difference between patients with BD and healthy controls.5–11 However, several studies have also reported reduced areas of the anterior CC in patients with BD,7,8,10,12,13 while others have reported no such regional difference.14–17 These heterogeneous results may be partly explained by the phenotypic variability of patients with BD, such as the proportion of patients with psychotic features or mean illness duration, across studies.
Among the different clinical features of BD, there is a relative lack of exploration of the neuroimaging patterns associated with psychotic BD, which contrasts with the studies highlighting the relevance of the distinction of psychotic versus nonpsychotic BD in clinical and genetic studies.18–20 In a recent study, we reported that BD with psychosis was associated with greater microstructural CC impairments than BD without a history of psychosis.3 However, to our knowledge only 2 studies have directly compared callosal areas between these 2 subgroups, yielding negative results.15,21 Moreover, these studies included relatively limited samples, possibly leading to insufficient power to detect more subtle differences. Furthermore, they did not consider CC subdivisions. Consequently, it is largely unknown whether BD with psychotic features is associated with a different pattern of macrostructural regional callosal abnormalities than nonpsychotic BD presentations.
In the present study, we thus aimed to compare regional callosal areas in patients with BD-I and healthy controls using a relatively large sample derived from international multicentre sites. We also compared these regional areas in patients with BD with and without a positive history of psychotic features in order to search for specific regional callosal differences associated with such features. We hypothesized that compared with controls, patients with BD would show decreases in CC areas and that these decreases would be more pronounced in patients with psychotic features.
Methods
Participants
We recruited patients with BD-I via clinical referral made by the regional psychiatric services from the 4 participating international, university-affiliated psychiatry departments: Assistance Publique Hôpitaux de Paris (Henri Mondor Hospital, Créteil, France, and Fernand Widal-Lariboisière Hospital, Paris, France), the Western Psychiatric Institute and Clinic, UPMC (Pittsburgh, United States) and the Central Institute for Mental Health (Mannheim, Germany) between 2008 and 2011 and the Clinical Science Institute (Galway, Ireland) between 2006 and 2008. Controls were matched to patients within each site and were recruited from local registry offices and from media announcements.
All participants had to be between the ages of 18 and 60 years, and BD-I was diagnosed using DSM-IV criteria. Exclusion criteria were history of neurologic illness; contraindication to MRI; and alcohol use disorder or dependence within the participants’ lifetime (France, Germany), within the 2 months preceding the study (United States), or within the year preceding the study (Ireland). In addition, controls were required to have no personal or family history of axis I mood disorders, schizophrenia or schizoaffective disorder. Participants were assessed by trained raters using the Diagnostic Interview for Genetic Studies (French site)22 and the Structured Clinical Interview for DSM-IV (German, Irish and US sites).23
Symptoms of depression and mania were assessed using the Montgomery-Åsberg Depression Rating Scale (French and German sites)24 or the Hamilton Rating Scale for Depression (HAMD; 25-item version at the US site and 19-item version at the Irish site)25 and the Young Mania Rating Scale (all sites).26 Two hundred participants took part in a diffusion-weighted tractography study in which structural T1 images were used only for registration purposes,3 and 120 took part in a voxel-based structural analysis of grey and white matter.27 We defined symptomatic patients as having a score greater than 7 on the mood scales, and we considered patients to have a positive history of psychotic features if they had at least 1 manic or depressive episode with delusions or hallucinations.
All participants were given full verbal and written information about the aims, methods and risks of this research, and were given the option to decline participation. They were invited to ask questions about the research and about the consent form, with replies contributing to ensuring their informed participation. Patients with enforced hospitalization or under conservatorship were not included in the study, nor were patients in an active psychotic state that may have interfered with their ability to give informed consent. Such processes were assessed through clinical examination and consultation with the medical team involved in the patients’ care, as appropriate. After a complete description of the study each participant gave written informed consent.
The local ethics committees of each centre approved the study: the Ethical Committee of the Medical Faculty Mannheim, Heidelberg University (Germany), the NUI Galway and the University College Hospital Galway research ethics committees (Ireland), the Institutional Review Board of the University of Pittsburgh (United States) and the Comité de Protection des Personnes Ile-de-France (France).
MRI data collection
Each participant underwent high-resolution 3-dimensional T1-weighted structural MRI using a Siemens Magnetom TrioTim 3 T Syngo MR B17 scanner (Siemens Medical Solutions) with a 12-channel head-coil at the French, German and US sites (echo time [TE] 2.98 ms, repetition time [TR] 2300 ms, 160 slices, voxel size 1.0 × 1.0 × 1.0 mm or 1.1 mm) or a 1.5 T Siemens Magnetom Symphony scanner with a standard quadrature head coil at the Irish site (TE 4.38 ms, TR 1140 ms, 160 slices, reconstructed, voxel size 0.9 × 0.9 × 0.9 mm). Two of us (J.H. and S.S.) assessed images visually for artifacts and geometrical distortions affecting the CC.
Data processing
We estimated the regional areas of the CC following a 2-step method: the delineation of the CC on a resampled mid-sagittal slice and then its parcellation into 7 regional areas following Witelson’s scheme.28 To overcome biases related to manual delineations and extensive processing time, we used an automated segmentation software: the Automatic Registration Toolbox (ART) for CC, “yuki” module, version 2.0 for Linux (www.nitrc.org/projects/art/).29
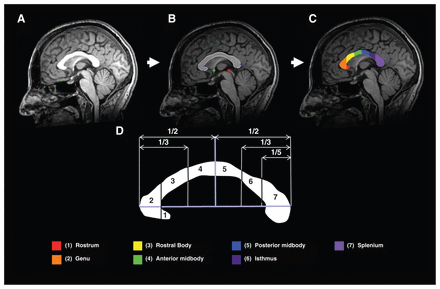
Briefly, the yuki module of the ART for CC follows 4 steps for each participant to delineate the CC. These methods are described in depth elsewhere.29,30 The first step is the localization of the mid-sagittal plane using a method designed to reduce the potential effects of head tilt. The second step is the localization of the anterior and posterior commissures on the mid-sagittal plane. Third, the software reconstructed the mid-sagittal slice via trilinear interpolation and a reorientation (512 × 512 isotropic pixels of 0.5 × 0.5 mm), with the centre of the image being the midpoint between the anterior and posterior commissure and with the top–bottom axis and the left–right axis of the image corresponding to the superior–inferior axis and the anterior–posterior axes, respectively (Fig. 1A). Finally, the software delineated the CC in the mid-sagittal slice with a method based on a multi-atlas model using a nonlinear registration algorithm (Fig. 1B). The last step was the automated delineation of the CC in 7 regions following the geometrical method described previously28 (Fig. 1C and 1D): the rostrum, genu, rostral body, anterior midbody, posterior midbody, isthmus and splenium of the CC (Fig. 1D). One of us (S.S.), who was blind to the diagnosis, checked all segmentations visually, resulting in the exclusion of 1 individual owing to a delineation error in the isthmus region. No manual correction was applied.
Illustration of the automated processing pipeline for 1 participant. (A) Reoriented midsaggital slice. (B) Automatic localization of the anterior commissure (green cross) and of the posterior commissure (red cross). Automatic delineation of the corpus callosum based on a multiatlas model (white border, blue crosses). (C and D) Automatic segmentation of the corpus callosum based on the geometrical method described by Witelson and colleagues.28
To check the reliability of the automatic segmentation technique, we performed manual regional parcellations of the CC in 20 random scans using ITK-SNAP 2.431 and following the geometrical method described previously.28 We found a high agreement among the areas of the CC that were automatically29 and manually determined (intraclass correlation coefficient = 0.98). Using the Brain Extraction Tool of the FMRIB FSL toolbox,32 we computed a skull-stripped image for each Participant. One of us (S.S.) inspected each brain extraction visually, and we calculated the total brain volume (TBV) based on this skull-stripped image for each participant.
Statistical analysis
We used linear mixed models to compare the area of each region of the CC between patients with BD and controls and between patients with and without a positive history of psychotic features. Diagnosis (BD v. control or BD with v. without psychotic features) was included as a factor of interest. We considered the following variables that are potentially associated with callosal area differences: sex; handedness; antipsychotic and lithium medication status (yes v. no);33 history of alcohol abuse/dependence (yes v. no);34 and acute mood symptoms, defined as scores greater than 7 on mood rating scales (yes v. no). These variables were included as fixed-effect factors. Age and TBV were included as fixed-effect covariates, while site was included as a random-effect factor in order to control for specific site-related effects. In addition, illness duration was included as a fixed-effect covariate in the model when comparing patients with and without a history of psychosis.
We tested differences between continuous and categorical demographic variables using Student t tests and Pearson χ2 tests, respectively, if appropriate assumptions were met. Correlations between the callosal areas and the duration of illness were assessed using the Spearman partial correlation test while controlling for TBV. We used PASW software version 18 for data analysis. We considered results of comparisons of demographic data and CC areas to be significant at p < 0.05, false-discovery rate (FDR)–corrected. We calculated corrected p values using R 2.15.1, Multitest package (www.r-project.org). All the analyses were performed after outlier detection using leaf plots. Deviation from normality was detected for 3 regional areas. To respect the statistical assumption of normal distribution of residuals when using linear mixed models, we performed analyses on transformed areas using natural logarithmic transformation,35 which we checked for the normality of residues using the Kolmogorov–Smirnov test and qq plots. To further enable reader clarity, raw mean areas are presented. To exclude an effect of the transformation, untransformed data are also shown (Appendix 1, available at jpn.ca).
Results
Sample
The sample included 174 patients with BD and 146 controls. Information about alcohol lifetime abuse/dependence was missing for 2 patients; therefore, the final sample comprised 172 patients and 146 controls. Demographical data are summarized in Table 1.
Demographic and clinical characteristics of the study sample
Patients and control groups did not differ significantly in age, sex, handedness and TBV. Mean years of education was 16.4 ± 3.4 for controls and 14.7 ± 3.2 for patients at the Galway and Mannheim sites. Categorical data about level of education were missing for 16 patients at the US and Créteil sites. Level of education was at least high school diploma in 64 (97.0%) patients and 39 (95.1%) controls and below high school diploma in 2 (3.0%) patients and 2 (4.9%) controls. Regarding medication on the day of scanning, 84 (48.3%) patients were being treated with lithium salts, 91 (52.3%) with first- or second-generation antipsychotics, 88 (50.6%) with anticonvulsants, 61 (35.1%) with antidepressants and 90 (51.7%) with a combination of medications, including at least 2 mood stabilizers.
Data concerning the history of psychotic features was missing for 1 patient, while mood assessment confirmed that 35 (20.1%) patients were symptomatic on the day of scanning.
Comparison of callosal areas
We found significant reductions in the mid-sagittal areas of the posterior body, isthmus and splenium of the CC in patients versus healthy controls (Table 2). Patients with a positive history of psychotic features had a significantly larger CC rostrum area than patients without a history of psychotic features (Table 3).
Comparison of callosal regional areas between patients with bipolar disorder I and healthy controls*
Comparison of callosal regional areas between patients with a positive history of psychotic features and patients without such a history*
Post hoc analyses
After controlling for TBV, there were no significant partial correlations between the duration of illness and the areas of the posterior midbody (partial correlation = 0.019, p = 0.80), isthmus (partial correlation = −0.032, p = 0.67) and splenium (partial correlation = 0.052, p = 0.50) in patients with BD. When examining the subgroups and controlling for TBV, there was a significant positive correlation between the area of the rostrum and the duration of illness in patients with a positive history of psychotic features (partial correlation = 0.242, p = 0.010) but not in those without a history of psychotic features (partial correlation = −0.054, p = 0.69).
Discussion
We found a reduced area in 3 posterior parts of the CC in patients with BD compared with controls: the posterior midbody, isthmus and splenium. Patients with a positive history of psychotic features had an increased area of the CC rostrum compared with patients who never experienced psychotic symptoms during illness phases. To our knowledge, this is the first multicentre study and the largest sample used to explore callosal areas in patients with BD using structural T1 MRI and the first to report a difference in callosal morphology between patients with with and without a history of psychosis.
Comparisons of callosal areas between patients with BD and controls
In accordance with previous findings, patients with BD had reduced areas of 3 posterior CC regions compared with healthy controls.5–9,11 As the CC follows a topographical organization,4 reductions of the posterior areas suggest that BD may be associated with decreased interhemispheric connectivity between homologous regions wired through the posterior callosum (i.e., parietal, temporal and occipital cortices).36 Interestingly, a growing number of diffusion MRI studies have independently reported microstructural CC alterations in patients with BD,1–3,27,37 suggesting that callosal abnormalities are identifiable both at a macrostructural and microstructural level in these patients. This may overlap with data in patients with callosal agenesis, who can exhibit severe behavioural symptoms, including manic or depressive symptomatology.38 Taken together, these and similar results suggest an important role for CC structural abnormalities in driving impaired interhemispheric integration in patients with BD.
Despite such growing bodies of evidence, however, the functional consequences of these alterations have remained relatively poorly explored, resulting in little attempt to integrate interhemispheric dysconnectivity in most recent BD neurobiological models.2 Future functional connectivity studies based on emotional and cognitive tasks that involve interhemispheric transfer are warranted in order to improve our understanding of the clinical manifestations of decreased interhemispheric integration in patients with BD.
Comparisons of callosal areas between patients with and without a history of psychotic features
A comparison of patient subgroups according to their history of psychotic features showed greater CC rostral areas in patients with a history of psychotic features than in those without such a history. Previous studies have directly compared psychotic and nonpsychotic patients with BD, although these studies were limited by comparisons restricted to total CC area, where no significant differences were evident.15,16 Walterfang and colleagues21 compared callosal metrics between 46 patients with and 24 patients without psychosis and found no difference in total callosal area, length or curvature. These authors subsequently replicated this result15 in a smaller sample of 10 patients with and 11 patients without psychosis. Our results are from a larger sample and suggest a different pattern of anterior interhemispheric connectivity in patients with BD that depends on their history of psychotic symptoms.
The rostral part of the CC is thought to include white matter fibres that link the orbitofrontal cortices (Brodmann area [BA] 11/47), the lateral prefrontal cortices (BA 45/746) and the medial prefrontal cortices (BA 10) bilaterally.36 Interestingly, all of these regions are highly involved in emotion processing and regulation and have been associated with BD pathology in functional neuroimaging studies.2 At a cellular level, increased CC areas have been associated with higher axonal density, particularly in anterior and posterior areas where small unmyelinated fibres predominate.4 However, whether this increased connectivity of the rostral CC is specific to BD with psychotic features or whether it is shared with other diagnoses across the psychosis spectrum requires further investigation. To our knowledge, only 1 study has looked at this question, directly comparing patients with a first episode of affective psychosis (BD with psychosis or major depressive disorder with psychosis) with patients with schizophrenia-spectrum disorder, reporting no between-group difference of the total callosal area.16 However, future studies comparing callosal subregional areas using a dimensional measure of psychosis across different diagnoses will be necessary to clarify whether the results here are typical of psychosis more generally. Moreover, histological studies are warranted to understand the cellular and molecular underpinnings of the differences in callosal areas between subgroups of patients with BD.
Association between callosal areas and illness duration
Previous reports have suggested that callosal abnormalities may be linked to a disease-related process rather than being a BD vulnerability trait. Several studies have suggested that morphological12,21 and microstructural callosal abnormalities39 may be patient-specific. Other groups have proposed that the CC is more susceptible to aging in patients with BD.6–9,11 Here, in a relatively large sample, we found a positive association between the CC rostral area and illness duration, but only in the BD subgroup with a positive history of psychosis, adjusted for TBV. This correlation was absent in other CC regions. Importantly, rostral area differences between patients with and without psychosis remained significant even after adjusting for illness duration. This could suggest a different disease-related callosal remodelling across the course of the illness between these 2 subgroups. Further, it could suggest the differential involvement of the rostral CC in patients with BD with and without psychotic features. However, longitudinal studies including the period before illness onset are needed to characterize the evolution of structural CC rostral area changes in patients with BD with and without psychotic features, as highlighted by our results.
Effects of potential confounders
Several studies have explored the influence of lithium salts and antipsychotics on callosal measurements, yielding conflicting results.12,16,21 In our sample there was no evidence of a confounding effect of lithium or antipsychotics on the results. Neurotoxic effects of alcohol abuse may bias assessments of white matter volumes;34 however, this was controlled for in the statistical analysis, as was symptomatic status on the day of MRI acquisition, thereby avoiding any effect of acute symptomatology on our measurements.
Strengths and limitations
Several strengths of our study should be emphasized. First, to our knowledge, this is the first multicentre assessment of regional CC areas in a large sample of patients with BD and healthy controls. Second, the analyses accounted for a number of potentially confounding variables — site of inclusion, sex, age, handedness and TBV of the participants — with the significance of the results FDR-corrected. Third, we used an automatic parcellation technique, thereby reducing biases related to manual CC delineation. Fourth, we investigated the influence of several clinical variables, such as a history of alcohol abuse, effect of mood on the day of scanning, and lithium or antipsychotic medications, all of which might have biased the results.
Despite these advantages our study has several limitations. We found only small to medium effect sizes, in contrast to other studies based on smaller samples. However, small samples may lead to overestimated effect sizes.40 Methodologically, there was no calibration technique among the sites. However, we corrected for site in our statistical analysis. Finally, although the confounding effect of current lithium and antipsychotic medications was ruled out, the effects of past medication cannot be excluded.
Conclusion
We conducted a structural assessment of regional callosal areas in patients with BD and controls using T1 MRI in a relatively large sample. We identified reduced areas in 3 contiguous posterior regions of the CC in patients with BD compared with controls. The BD subgroup with psychotic features showed an increased CC rostral area. These findings support the hypothesis of impaired interhemispheric connectivity in temporal and posterior parts of the brain in patients with BD. Furthermore, our results provide neuroimaging evidence that BD with psychotic features may be a relevant neuroimaging subtype of BD with a different pattern of callosal abnormalities. Future neuroimaging studies assessing the histological correlates of these differences (e.g., using axonal calibration techniques) and the functional consequences of these abnormalities (e.g., functional connectivity during emotional tasks with cross–uncrossed conditions) are needed to clarify the cellular underpinnings and the functional consequences of interhemispheric connectivity impairments in patients with BD.
Acknowledgements
We thank all participants for agreeing to enrol in this study. We also thank the personnel of the participating centers for their help with data collection. We thank David Kupfer, MD for providing logistical and administrative support to this study. We also thank George Anderson at CRC Scotland & London for English editing of this manuscript. This work was supported by public funding from the Alliance pour les Sciences de la Vie et de la Santé (ITMO Neurosciences), the French Agence Nationale pour la Recherche (ANR MNP 2008 and ANR-11-IDEX-0004-02), the German Deutsche Forschungsgemeinschaft (SFB636/C6 and We3638/3-1), the NIMH R01 MH076971 and the National Alliance for Research on Schizophrenia and Depression.
Footnotes
Competing interests: None declared.
Contributors: S. Sarrazin, M. Delavest, J. Almeida, J.-F. Mangin, M. Leboyer and J. Houenou designed the study. S. Sarrazin, M.-A. d’Albis, C. McDonald, J. Linke, M. Wessa, M. Delavest, L. Emsell, A. Versace, J. Almeida, C. Poupon, K. Le Dudai, C. Daban, N. Hamdani and J. Houenou acquired the data, which S. Sarrazin, J. Linke, M. Wessa, M. Phillips and J. Houenou analyzed. S. Sarrazin, M. Phillips and M. Delavest wrote the article, which all authors reviewed and approved for publication.
- Received September 10, 2014.
- Revision received December 17, 2014.
- Accepted December 22, 2014.