Abstract
Background: Schizophrenia is a disorder of basic self-disturbance. Evidence suggests that people with schizophrenia may have aberrant experiences of body ownership: they may feel that they are not the subject of their own body experiences. However, little is known about the development of such disturbances.
Methods: Using a rubber hand illusion paradigm, we assessed body ownership in patients with schizophrenia (n = 54), healthy controls (n = 56), children/adolescents at increased familial risk of developing schizophrenia (n = 24) or mood disorders (n = 33), and children/adolescents without this risk (n = 18). In this paradigm, a rubber hand (visible) and a participant’s real hand (invisible) were stroked synchronously and asynchronously; we then measured subjective illusory experiences and proprioceptive drift.
Results: All groups showed the expected effect of the rubber hand illusion: stronger proprioceptive drift and increased subjective illusory experiences after synchronous versus asynchronous stroking. The effect of synchronicity on subjective experiences was significantly weaker in patients with schizophrenia than in healthy controls, and subjective ratings were positively correlated with delusions in patients. We found no significant differences between children/adolescents with and without increased familial risk.
Limitations: Large individual differences raised questions for future research.
Conclusion: We found subtle disturbances in body-ownership experiences in patients with schizophrenia, which were associated with delusions. We found no evidence for impairments in children/adolescents at increased familial risk of developing schizophrenia or a mood disorder. Longitudinal data might reveal whether impairments in body ownership are predictive of psychosis onset.
Introduction
To most of us, distinguishing between self and others may seem to be an automatic and fluent process. However, schizophrenia is seen as a disorder of basic self-disturbance in which the relation between the self and the world is unstable and complex.1,2 Such self-disturbances are evident in self-agency experiences (the feeling that we cause our own actions and consequences) and in body-ownership experiences (the feeling that we are the subject of our own body experiences).3–5 Although abnormal experiences of body ownership are well-described psychotic symptoms, we still know surprisingly little about their etiology and development.
Body-ownership experiences result from a multisensory integration process, in which visual, tactile and proprioceptive information is integrated.6,7 Matching sensory information from different modalities creates a sense of ownership over body parts;8 for example, when we see and feel the touch of someone’s hand on our arm, we simply know that it is our own arm being touched. However, a mismatch in this sensory information can lead to the illusion that one is not the owner of a body part. The process of multisensory integration may be disturbed in schizophrenia, such that patients are more likely to integrate multisensory signals, even if they do not logically belong together. Consequently, patients with schizophrenia are more susceptible to body-ownership illusions than healthy controls.9–11
Impairments in body ownership have been assessed using a variety of rubber hand illusion (RHI) paradigms.9–14 In the original RHI experiment, the participant’s own invisible hand and a visible rubber hand (positioned near the hidden hand) are stroked synchronously or asynchronously.8 Because the illusion materializes primarily when stroking is synchronous, periods of asynchronous stroking can be used as a control condition.8,15 During the experiment, the body’s internal model is constantly adjusted to match seeing the touch on the rubber hand and feeling the touch on the person’s own hand,16 leading to ownership illusions over the rubber hand. This illusion is thought to depend on the temporal binding window, in which stimuli from different modalities are perceived as occurring together.17 More specifically, a larger time window allows for integration of more incoming stimuli, which could lead to a stronger RHI.
Most studies using the RHI in schizophrenia have measured the subjective experience of the illusion and showed that, compared with healthy controls, people with schizophrenia rated the illusion as more intense, possibly indicating a more flexible sense of ownership.9,10,13 The strength of these illusory experiences has been positively correlated with the severity of positive psychotic symptoms, such as hallucinations or delusions of reference, although the evidence is sparse.9,10 These studies suggest that multisensory integration deficits might underlie specific psychotic symptoms. In addition to measuring subjective RHI experiences, it is possible to quantify the illusion by assessing proprioceptive drift: the perceived shift of the location of one’s own hand toward the rubber hand after stroking. In one study, the effect of synchronous versus asynchronous stroking on proprioceptive drift was greater in people with schizophrenia than in healthy controls.10 Although this finding confirmed body-ownership disturbances, it has been the only study to report proprioceptive drift in this group so far.
It has been suggested that basic self-disturbances, including disturbances in self-awareness and presence, can predict transition to psychosis in people at ultra-high risk.18 Regarding body ownership specifically, healthy controls with schizotypal personality traits or psychotic-like experiences showed altered RHI measures, suggesting that RHI measures might be related to a vulnerability for psychosis.10,19–21
In the current study, we administered the RHI in 2 cohorts. The first cohort consisted of patients with schizophrenia and healthy controls. Our aim in this cohort was to replicate previous findings in patients (i.e., in people with a more flexible sense of body ownership) using a well controlled experimental procedure. We applied synchronous and asynchronous stroking conditions, and we measured subjective experiences and proprioceptive drift in the stimulated and unstimulated hands. Based on previous studies, we expected a positive relationship between RHI measures and psychotic symptoms. As well, to our knowledge, no studies have investigated the relevance of RHI measures as a marker of increased risk of developing schizophrenia. In line with the search for markers that predict future conversion to psychosis, and to expand our knowledge of the development of body-ownership disturbances, in the second cohort we explored whether increased familial risk of developing schizophrenia (i.e., in offspring of patients with schizophrenia) was related to alterations in RHI measures compared to controls and people at increased familial risk of developing a mood disorder.
Methods
Participants
The study included 2 cohorts. The first cohort consisted of 54 patients with a DSM-IV diagnosis of schizophrenia (SZ) and 56 healthy controls (HC), aged between 18 and 50 years. We confirmed diagnoses using the Comprehensive Assessment of Symptoms and History.22 We recruited participants for the SZ group from the psychiatry department of the University Medical Centre Utrecht, other psychiatric institutions in the Utrecht area and patient associations. We recruited participants for the HC group using notice boards and an online recruiting company for scientific research (www.proefpersonen.nl). Overall exclusion criteria were an IQ less than 80 and drug or alcohol use over the preceding 6 months. Further exclusion criteria for the SZ group were the presence of a psychotic episode at the time of testing and the chronic use of a medication other than a psychiatric medication. Further exclusion criteria for the HC group were a history of psychiatric illness, first-degree relatives with a psychotic illness and chronic use of medication.
The second cohort was part of the second measurement of an ongoing longitudinal offspring study and consisted of 24 offspring of patients with a schizophrenia-spectrum disorder (SZO), 33 offspring of patients with bipolar disorder (BPO) and 18 controls (CO). At baseline, participants in the SZO and BPO groups had at least 1 first-degree or 2 second-degree relatives with a schizophrenia-spectrum disorder or bipolar disorder, respectively (confirmed by the Structured Clinical Interview for DSM-IV23). Participants in the CO group had no history of psychiatric illness, no first-degree relative with a psychotic disorder or affective disorder and no psychotropic medication use. All participants in the second cohort were aged between 11 and 22 years and had an IQ greater than 70. We recruited participants for the SZO and BPO groups at the psychiatry department of the University Medical Centre Utrecht, other psychiatric institutions in the Netherlands and patient associations. We recruited participants for the CO group from primary and secondary schools in the Utrecht area.
All participants were Dutch-speaking. Informed consent was provided by participants themselves and by the parents of participants younger than 18 years. The Human Ethics Committee of the University Medical Centre Utrecht approved both studies. All participants were financially compensated for study participation.
Measurements
The rubber hand illusion
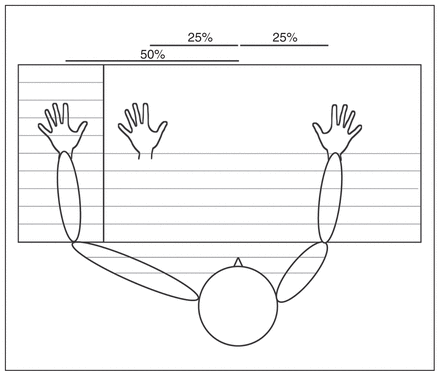
We measured body ownership using the RHI.8 Participants placed their hands in a box, and a cape covered their arms and wrists (Fig. 1). The right hand and rubber hand were at equal distances from the middle of the body.15 To prevent the left and right hands of young participants from being too far apart, we corrected the baseline positions of the hands for arm length.
Rubber hand illusion, experimental setup. Thin lines represent parts of the setup that were invisible to the participant during stroking of the left and rubber hand. Percentages indicate the proportion of arm length.
At the beginning of the experiment, the box was covered by a wooden panel. Before participants saw the rubber hand and before any tactile manipulation by the experimenter, participants estimated the position of their own hands as a baseline measure. To obtain this estimate, the experimenter slowly moved their own index finger along the top of the experimental setting (twice: once from each side of the box) and instructed the participant to say “stop” when they felt that the experimenter’s index finger was aligned with their own.15 A ruler was attached at the bottom of the experimental setup to measure participants’ estimate.
Participants then closed their eyes, and the wooden panel was placed vertically between their left hand and the rubber hand, so that only the right hand and the rubber left hand were visible to the participant. The rubber hand and the participant’s left hand were stroked with a soft paintbrush, either synchronously (same timing and direction on the participant’s hand and the rubber hand) or asynchronously (different timing and direction). Stroking was done on the back of the hands and the index fingers. Participants watched the rubber hand during stroking.
In both synchronicity conditions, participants estimated the position of their own left and right index fingers after 4 periods of stroking: first after 2 minutes, and 3 more times at intervals of 20 seconds.24 The side from which the experimenter moved their index finger along the top of the experimental setting to measure the estimated position of the hands was counterbalanced for each participant (i.e., twice from the left and twice from the right). The order in which the synchronous and asynchronous conditions were administered was counterbalanced between participants.
We measured proprioceptive drift by calculating the difference between the mean of the 4 post-manipulation measures and the mean baseline measure; a positive value indicated drift toward the rubber hand. For each participant, we calculated drift 4 times: after the synchronous and asynchronous conditions in both the stimulated and unstimulated hand. Because the position of the hands was based on the participant’s arm length, we calculated drift as a percentage of this length.
We measured subjective RHI experiences after the synchronous and asynchronous condition with 2 questions: “When I was stroking with the paintbrush, did it sometimes feel as if you could feel the touch of the brush where the fake hand was?” and “When I was stroking with the paintbrush, did it sometimes feel as if the fake hand was your hand, or belonged to you?”24 These questions were rated on a 7-point scale, ranging from 0 (no, not at all) to 6 (yes, very). In most studies a more extensive questionnaire is used,8 but we used this shorter questionnaire to ensure that young participants could understand the questions correctly and differentiate between them.
Symptomatology
In the SZ group, we used the Self-Experience Lifetime Frequency Scale (SELF) to assess disturbances in self-experience.25 This self-report questionnaire measures the presence and frequency of lifetime disturbances of self-awareness and experiences of depersonalization (e.g., “Have you felt unreal or like a stranger to yourself?”). Scores were rated on a scale of 0 (never/not distressed) to 4 (all the time/severely distressed). We summed all SELF items (scores for presence and frequency) to obtain a total score.25 We also assessed SZ participants’ current level of psychotic symptoms using the Positive and Negative Syndrome Scale (PANSS).26
In the SZO, BPO and CO groups, we assessed for the presence of psychotic symptoms using the Schedule for Affective Disorders and Schizophrenia for School-Age Children–Present and Lifetime Version (K-SADS-PL).27 Symptoms were scored as absent, subthreshold or threshold.
Statistical analysis
To determine group differences in age, years of education and/or parental years of education, and sex, we performed independent sample t-tests (HC/SZ), analyses of variance (ANOVA; CO/BPO/SZO) and χ2 tests.
In both samples, ratings on the 2 questionnaire items were highly correlated in both the synchronous and asynchronous conditions (rs = 0.52–0.65, all p < 0.001) and had good internal consistency (Cronbach α = 0.72–0.78). Therefore, we combined the 2 items by calculating the mean to represent the strength of subjective RHI experiences.
Because we used asynchronous stroking as a control condition, the effect of synchronicity (synchronous relative to asynchronous stroking) on subjective RHI experiences and proprioceptive drift was the main outcome measure.
Cohort 1: case–control
To check for effects of group, synchronicity and laterality on proprioceptive drift, we performed a repeated-measures ANOVA with group (HC/SZ) as the between-subjects factor and synchronicity (synchronous/asynchronous stroking) and laterality (stimulated/unstimulated hand) as within-subjects factors. We did the same (without laterality) for subjective RHI experiences. We then analyzed counterbalancing effects and estimated baseline positions and the relation between proprioceptive drift and subjective RHI experiences (Appendix 1, available at jpn.ca/180049-a1). We assessed the relationship between RHI measures and SELF score in the SZ group. Because the total SELF score was not normally distributed, we used Spearman correlation to assess the relationships between the SELF score and drift in the synchronous and asynchronous conditions, subjective RHI experience in the synchronous and asynchronous conditions, and synchronicity effects (i.e., difference between synchronous and asynchronous conditions). Finally, we assessed the relationship between these RHI measures and the PANSS positive subscale, PANSS item 1 (delusions) and PANSS item 3 (hallucinations). We used Bonferroni correction for multiple comparisons (α = 0.05/4 symptom scores = 0.0125).
Cohort 2: offspring
We performed similar analyses to compare RHI measures between the SZO, BPO and CO groups. Next, we assessed the relationship between psychotic symptoms and RHI measures in the high-risk sample only (SZO + BPO), because no symptoms were present in the CO group. We created 2 groups based on the lifetime presence of psychotic symptoms (yes/no). Presence was scored when at least 1 symptom from the K-SADS-PL hallucinations or delusions supplement was scored as “threshold,” because this might indicate an elevated risk of developing psychosis later in life.28 Using Mann–Whitney U tests in the SZO, BPO and SZO + BPO groups, we compared the 2 symptom groups for proprioceptive drift and subjective RHI experience in the synchronous and asynchronous conditions, and for synchronicity effects.
Results
Demographic and clinical characteristics
Table 1 shows demographic and clinical information for both cohorts. The SZ group had fewer years of education than the HC group. Among offspring, the CO group had significantly more males and fewer females than the SZO group. After the experiment, 6 participants (2 HC, 2 SZ, 1 CO and 1 SZO) indicated that they used spatial reference points when proprioceptive drift was measured; they were removed from all analyses of proprioceptive drift. One member of the SZ group was excluded from subjective RHI analyses because they did not understand the questions sufficiently well.
Demographic and clinical characteristics*
Cohort 1: case–control
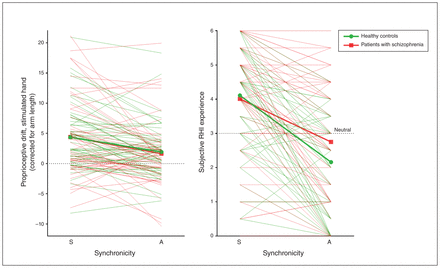
As expected, the effect of synchronicity on proprioceptive drift was more pronounced in the stimulated hand than in the unstimulated hand (F1,104 = 21.73, p < 0.001, ηp2 = 0.17; synchronicity × laterality). Follow-up analyses on both hands separately confirmed that in the stimulated hand only, proprioceptive drift was greater after synchronous versus asynchronous stroking in both groups (Table 2, Fig. 2). As well, subjective RHI ratings were higher after synchronous stroking. Here, the synchronicity effect was significantly smaller in the SZ group than in the HC group. In follow-up analyses, subjective RHI ratings were marginally higher in the SZ group than the HC group only in the asynchronous condition (t107= −1.81, p = 0.07, Cohen d = 0.35).
Individual (thin lines) and mean (bold lines) RHI measures for healthy controls and patients with schizophrenia after synchronous and asynchronous stroking. Lines represent synchronicity effects. Subjective RHI scores ranged from “no, not at all” (0) to “yes, very” (6). A = asynchronous; RHI = rubber hand illusion; S = synchronous.
ANOVA results for the effects of synchronous and asynchronous stroking on the RHI*†
We found no counterbalancing effects, and baseline estimation errors did not differ between groups (Appendix 1). Furthermore, in both groups we found a significant relationship between proprioceptive drift and subjective RHI experiences (Appendix 1).
After multiple comparison correction, we found a significant positive correlation between PANSS delusions and the strength of subjective RHI experiences in the synchronous condition (rs= 0.36, p = 0.01; Appendix 1). At trend level, subjective ratings were positively related to the SELF scale (synchronous condition), the PANSS positive symptom subscale (synchronous and asynchronous condition) and the PANSS delusions item (asynchronous condition).
Cohort 2: offspring
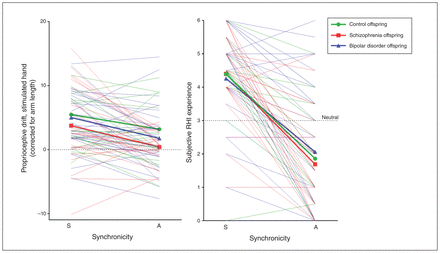
Similar to cohort 1, the synchronicity effect was larger in the stimulated hand than in the unstimulated hand (F1,70 = 13.17, p = 0.001, ηp2 = 0.16). Follow-up analyses on proprioceptive drift showed significance in the stimulated hand only (Table 2 and Fig. 3). We also found a significant synchronicity effect on subjective RHI ratings. We found no significant group differences between the SZO, BPO and CO groups. Results for counterbalancing, baseline estimation errors and the relationship between proprioceptive drift and subjective RHI experiences are reported in Appendix 1. Baseline estimation errors did not differ between groups. The effect of synchronicity on proprioceptive drift was significantly stronger when the synchronous condition was applied first. We found a significant relationship between proprioceptive drift and subjective RHI experiences in the asynchronous condition only.
Individual (thin lines) and mean (bold lines) RHI measures for control offspring and offspring of patients with schizophrenia-spectrum disorder or bipolar disorder after synchronous and asynchronous stroking. Lines represent synchronicity effects. Subjective RHI scores ranged from “no, not at all” (0) to “yes, very” (6). A = asynchronous; RHI = rubber hand illusion; S = synchronous.
Among offspring, 38% of SZO (9/24) and 12% of BPO (4/33) had experienced at least 1 psychotic symptom (lifetime). The RHI measures did not differ between participants with or without these symptoms in the SZO or BPO groups, or in both groups combined (Appendix 1). Additional analyses using the subthreshold (instead of threshold) presence of psychotic symptoms as a cutoff yielded similar conclusions.
Discussion
This study investigated susceptibility to body-ownership illusions in healthy controls, patients with schizophrenia and children/adolescents with or without an increased familial risk of developing schizophrenia or a mood disorder. As expected, we found that synchronous stroking of the participant’s hand and the rubber hand increased both proprioceptive drift of the stimulated hand and subjective ratings in all groups. Interestingly, the synchronicity effect on subjective RHI experiences was significantly less pronounced in patients than in controls, a finding that was explained by increased illusory experiences after asynchronous stroking. Furthermore, subjective RHI ratings were positively related to the severity of delusions in patients. Finally, we found no body-ownership abnormalities in children/adolescents at increased familial risk of developing schizophrenia or a mood disorder.
Despite a low effect size, our finding that the synchronicity effect on subjective ratings of body ownership was significantly smaller in patients than in controls was consistent with the hypothesis that patients would have an increased temporal binding window.13,29 That is, their time window to perceive 2 stimuli as similar might be larger.29 Consequently, asynchronous stroking might have felt more synchronous to patients, which could have led to a more flexible sense of body ownership.10,17 This concept was supported by our finding that in the asynchronous condition, patients rated the strength of the illusion as marginally stronger than controls. Unlike most other studies, we used only a 2-item questionnaire, but our findings were in line with previous suggestions that patients are more susceptible to the illusion.9–11,13 Importantly, this group difference in subjective ratings could not be explained by how much subjective ratings depended on proprioceptive drift or to differences in the estimation of baseline positions.
Although alterations in subjective body-ownership illusions have been reported before in schizophrenia, to our knowledge the current study is only the second to also provide insight into potential abnormalities in proprioceptive drift. We did not find group differences in proprioceptive drift. Our findings were partly in line with an earlier study that used an experimental setup similar to ours: Thakkar and colleagues10 found a significant synchronicity effect in patients. However, in contrast to our findings, they found that the effect in patients was significantly greater than in healthy controls. Importantly, their results could be explained by the absence of an effect of synchronicity on proprioceptive drift in controls. The absence of such an effect was surprising, given the consistent evidence for its presence in studies with healthy participants. 8,30,31 Another explanation for the difference in findings between our study and that of Thakkar and colleagues could be that our sample size was almost twice as large.
That we found group differences in multisensory integration processes related to the subjective illusion, but not to proprioceptive drift, suggests different underlying mechanisms. This has been suggested before, but the exact differences remain unclear.32–34 One explanation might come from the distinction between bottom–up (i.e., sensory input) and top–down (i.e., cognitive representation of body schema) mechanisms that influence multisensory integration.30,35 Top–down mechanisms could influence subjective ratings of the illusion, but not proprioceptive drift.36 Consequently, alterations in cognitive representations might underlie the specific impairments we found. To confirm the conclusion that alterations in proprioceptive drift are not present in people with schizophrenia, and to investigate the distinction between bottom–up and top–down mechanisms of multi-sensory integration, additional studies are needed.
The RHI measurements did not differ between children/adolescents with or without increased familial risk of developing psychosis or a mood disorder. This suggests that we found no evidence that familial risk might be a marker of vulnerability for the development of schizophrenia. Because the sample sizes in our offspring groups were small, these findings must be interpreted with caution. The offspring groups included participants who will not experience any mental disorder later in life, and the alterations in patients in our case–control sample were subtle. This led to low statistical power to detect alterations during a possible premorbid stage. To further examine whether impairments in body ownership as measured by the RHI are predictive of developing schizophrenia, longitudinal studies are needed. Such studies would add to previous findings of self-disturbances as a marker of psychosis onset, because altered bodily sensations and anomalous self-experiences have been suggested as predictors of conversion to psychosis.18
Taken together, our findings suggest subtle alterations in embodiment experiences in patients with schizophrenia. Interestingly, the severity of delusions in the week preceding the study (measured by the PANSS) was positively correlated with the strength of subjective RHI experiences in the synchronous condition. These findings were in line with previous studies showing significant associations between the strength of subjective illusory RHI experiences and the severity of hallucinations, delusions of reference, delusions of control and somatic delusions.9,10 Moreover, in healthy people, psychotic-like symptoms (e.g., schizotypy) are associated with body-ownership illusions.10,19–21 These findings confirm the hypothesis that there is a relationship between multisensory integration impairments and the positive symptoms of schizophrenia, suggesting that they may share similar underlying mechanisms. We found no evidence for a relationship between multisensory integration and disturbed self-experiences in patients or lifetime presence of any delusion or hallucination in our high-risk sample. The fact that these symptoms were measured over a participant’s lifetime, whereas PANSS measures current symptoms, might explain this.
Limitations
Some limitations should be considered when interpreting the findings. First, because symptom severity in patients was very low at the time of measurement, results might be different in patients who were more severely ill. Second, large individual differences in RHI responses were present in all groups (Fig. 2 and Fig. 3). People likely differ in their sensitivity to the RHI, and some will not show the effect of synchronicity at all, but it is not clear why in some the effect of synchronicity was reversed. Important questions for future research emerged from these findings: what caused these individual differences, and do they influence our current way of looking into the sensory-integration processes that underlie RHI experiences? For example, it has been suggested that temperament affects proprioceptive drift.37 As well, individual differences might be caused by methodological factors, such as the potential influence of nonsystematic variation in tactile stimulation or the social context in which the illusion was administered.
Conclusion
Our findings suggest that people with schizophrenia have only subtle impairments in multisensory integration processes underlying body ownership. Specifically, this group showed impairments in subjective body-ownership illusions that were related to the severity of delusions. Furthermore, we found no evidence for disturbances of body ownership in those at increased familial risk of developing schizophrenia. The current study contributes to the understanding of body-ownership impairments in psychosis and its development, and it provides suggestions for further research.
Footnotes
Competing interests: M. Hillegers reports grants from the Brain and Behavior Foundation (NARSAD) during the conduct of the study. N. van Haren reports grants from the Netherlands Organization for Scientific Research during the conduct of the study and personal fees from Janssen-Cilag outside the submitted work. No other competing interests declared.
Contributors: M. Prikken, A. van der Weiden, M. Hillegers, H. Aarts and H. van Haren designed the study. M. Prikken, A. van der Weiden and H. Baalbergen acquired the data, which M. Prikken, A. van der Weiden, R. Khan, H. Aarts and N. van Haren analyzed. M. Prikken wrote the article, which all authors reviewed. All authors approved the final version to be published and can certify that no other individuals not listed as authors have made substantial contributions to the paper.
- Received March 27, 2018.
- Revision received June 12, 2018.
- Accepted August 2, 2018.