Abstract
Background: Attention deficit/hyperactivity disorder (ADHD) is a highly prevalent childhood disorder. Maternal smoking during pregnancy is a replicated environmental risk factor for this disorder. It is also a robust modifier of gene methylation during the prenatal developmental period. In this study, we sought to identify loci differentially methylated by maternal smoking during pregnancy and relate their methylation levels to various behavioural and physical outcomes relevant to ADHD.
Methods: We extracted DNA from blood samples from children diagnosed with ADHD and deeply phenotyped. Genome-wide DNA methylation was assessed using Infinium MethylationEPIC BeadChip. Maternal smoking during pregnancy was self-declared and assessed retrospectively.
Results: Our sample included 231 children with ADHD. Statistically significant differences in DNA methylation between children exposed or not to maternal smoking during pregnancy were detected in 3457 CpGs. We kept 30 CpGs with at least 5% of methylation difference between the 2 groups for further analysis. Six genes were associated with varied phenotypes of clinical relevance to ADHD. The levels of DNA methylation in RUNX1 were positively correlated with the CBCL scores, and DNA methylation in MYO1G correlated positively with the score at the Conners rating scale. Methylation level in a CpG located in GFI1 correlated with birthweight, a risk factor for ADHD. Differentially methylated regions were also identified and confirmed the association of RUNX1 methylation levels with the CBCL score.
Limitations: The study has several limitations, including the retrospective recall with self-report of maternal smoking during pregnancy as well as the grouping of individuals of varying age and developmental stage and of both males and females. In addition, the correlation design prevents the building of causation models.
Conclusion: This study provides evidence for the association between the level of methylation at specific loci and quantitative dimensions highly relevant for ADHD as well as birth weight, a measure that has already been associated with increased risk for ADHD. Our results provide further support to public health educational initiatives to stop maternal smoking during pregnancy.
Introduction
Attention-deficit/hyperactivity disorder (ADHD) is one of the most common early-onset neurobehavioural disorders, with a worldwide prevalence of 5.9%–7.1% in the school-aged population.1 It is a complex disorder involving both genetic and environmental factors.2 The genetic factors include common DNA sequence variants (single nucleotide polymorphisms [SNPs]) with small effect size as well as rare copy number variants with larger effect size.3 Genetic studies have reported the overall heritability of ADHD to be around 74%.4 It is now well established that multiple genes contribute to the overall risk for ADHD, each with a small effect. A recent genome-wide association study (GWAS) meta-analysis identified 27 loci that showed significant association with ADHD.5 Fine mapping of these risk loci implicates 76 potential genes with enriched expression in the brain (particularly in the frontal cortex) and during early development.
In addition, environmental factors are known to play an important role in ADHD.6 There are consistent data indicating that the interplay between genetic and environmental factors plays a major role in the pathogenesis of this disorder, but a clear understanding of the implicated mechanisms is still lacking. Among environmental risk factors that are relatively well replicated, maternal smoking has been very widely discussed and studied. Three meta-analyses of more than 20 studies (more than 3 million participants) concluded that maternal smoking during pregnancy is associated with a greater than 50% increase in incidence of ADHD.7–9 However, this association has been discussed by genetically designed studies, suggesting that genetic factors may increase both the risk for smoking and ADHD.10,11
Notwithstanding the question of causality, it is well documented that maternal smoking during pregnancy is associated with harmful effects on the developing fetus.12 In a recent meta-analysis of studies conducted in the Americas, it was reported that active maternal smoking during pregnancy was associated with low birth weight (odds ratio [OR] 2.00, 95% confidence interval [CI] 1.77–2.26, I2 = 66.3%).13 Abraham and colleagues14 conducted a systematic literature review and meta-analysis to better understand the gestational age at which maternal smoking during pregnancy becomes associated with reduced antenatal fetal size and growth. They noted that, by the second trimester, biparietal diameter and femur length were reduced by at least 0.06 standard deviations (SD), and all fetal measurements were reduced by 0.2 SD in the third trimester. Low birth weight has also been consistently associated with risk for ADHD. In a meta-analysis including 88 independent studies (4 645 482 participants), it was reported that low birth weight correlated with severity of ADHD symptoms.15 These studies suggest that maternal smoking during pregnancy may exert pervasive effects on somatic and brain development of the fetus, though the mechanisms implicated in ADHD are still not well understood.
Several studies have suggested that epigenetic factors may also be involved in the pathophysiology of ADHD. They may explain why exposure to environmental stressors during the pre- and postnatal periods increases susceptibility to adverse health outcomes later in life. There is much scientific evidence that suggests that epigenetic modification may be an important mechanism linking environmental exposure to the onset of neurodevelopmental disorders.16 Of the varied epigenetic mechanisms known to be involved, DNA methylation has been the most extensively examined within the ADHD context. 17 Several studies have investigated DNA methylation in relation to ADHD diagnosis or symptoms using candidate gene approaches or epigenome-wide association studies in peripheral blood and saliva tissue. Candidate-gene approaches have focused primarily on the monoaminergic system, given that blunted dopamine reward/motivation pathways in the striatum and prefrontal cortex have been implicated in the etiology of ADHD. Alterations in DNA methylation have been noted in studies examining the dopamine transporter gene (DAT1),18–24 genes encoding dopamine receptors,18,25–27 the serotonin transporter (5-HTT),25,28 the norepinephrine transporter,29 and catechol-O-methyltransferase (COMT).25,30 However, as with genetic studies, epigenome-wide association studies (EWAS) have been favoured since the genome can be probed in a hypothesis-free manner.
The published EWAS on ADHD have largely been case–control studies comparing peripheral DNA methylation among different age groups: children,31–33 adolescents34 and adults.35 Of these, only 1 detected genome-wide significant differences in DNA methylation between groups.35 However, some promising targets have been identified, with the most notable example being VIPR2 methylation. This gene encodes a receptor for vasoactive intestinal peptide, a small neuropeptide that plays a role in neuronal function. While the methylation of this gene has been implicated in ADHD in multiple clinical studies,31–33 there have been inconsistent findings.34,35 It has been suggested that the association is complex in its sex-dependence: boys with ADHD showed lower VIPR2 methylation and girls with ADHD had higher methylation relative to controls.33
In addition, several studies provide evidence that the DNA methylation patterns correlated with ADHD show significant variability with age. Examining data from more than 800 children in the Avon Longitudinal Study of Parents and Children (ALSPAC), it was noted that DNA methylation patterns at birth differed between children who went on to follow a chronic high versus low ADHD symptom trajectory from age 7–15 years. However, none of the 13 sites that met genomewide significance thresholds at birth were significant when methylation was measured at 7 years of age.36 Similar findings were noted in a recent EWAS meta-analysis.37 In addition, a large-scale EWAS over the first 2 decades of life noted that half of DNA methylation sites changed significantly in a nonlinear way over time.38
In contrast to the inconsistent findings between epigenetic markers and ADHD, many studies have reported a strong and consistent effect of smoking on the methylation of a large number of CpG sites. The largest EWAS meta-analysis to date evaluating the association of maternal smoking during pregnancy with cord blood DNA methylation at birth was conducted within the Pregnancy And Childhood Epigenetics (PACE) consortium and included 13 cohorts.39 These analyses showed that 6073 CpGs had significant differential methylation with sustained maternal smoking during pregnancy. The CpGs most widely identified across EWAS from diverse ancestries are within coding or regulatory regions of AHRR, GFI1, CYP1A1 and MYO1G.40–44 Differential methylation within these genes persisted from birth to 17 years of age when the ALSPAC cohort was examined.42
In an earlier study, we reported that maternal smoking during pregnancy was associated with differential methylation at specific sites within AHRR, GFI1 and CYP1A1 among children with a diagnosis of ADHD.45 Furthermore, hypomethylation of these GFI1 sites correlated with lower birth weight and an increase in the number of ADHD symptoms. In a recent study, Miyake and colleagues46 explored the association between prenatal smoking exposure, ADHD symptoms at preschool age and cord blood DNA methylation using the Hokkaido Study on Environment and Children’s Health, a prospective birth cohort study. They selected the CpG sites of 5 genes: AHRR, CYP1A1, ESR1, MYO1G and GFI1. They confirmed the finding that hypomethylation at sites within GFI1 was correlated with low birth weight.
While the association of GFI1 methylation and birth weight appears to be a well-replicated finding, there seems to be a gap in the literature on the association between maternal smoking during pregnancy, DNA methylation and behavioural outcomes in children with ADHD. Earlier we had presented a framework to help break down the complexity of ADHD in which we underscored the importance of examining quantitative phenotypes of clinical relevance instead of looking exclusively at the disorder as a diagnostic category.47 The objective of the present study was to conduct an EWAS to systematically examine the effects of maternal smoking during pregnancy with behavioural and clinical phenotypes of direct relevance to ADHD.
Methods
Participants
Children with a diagnosis of ADHD were selected from the pharmaco-behavioural genetic (PBG) study (n = 780) conducted at the Douglas Mental Health University Institute (DMHUI, Montréal, Canada), which has been described previously. 48 Briefly, children aged 6–12 years were referred by schools, community social workers, family doctors and pediatricians to the Disruptive Behaviour Disorders Program and pediatric outpatient clinics of the DMHUI. The research protocol was approved by the Research Ethics Board of the DMHUI. After explanation of the study, parents provided written consent, and children gave their verbal assent.
Diagnosis of ADHD was made by a child psychiatrist, according to the DSM-IV criteria, based on clinical interviews of the child and at least 1 parent. The diagnosis was supported with a comprehensive structured clinical interview (with the parents), the Diagnostic Interview Schedule for Children-version IV (DISC-IV), that covers 36 mental health disorders for children.49
In addition to ADHD diagnosis, the child’s behaviour at home and at school were evaluated by parents using Conners’ Global Index-Parents version (Conners’-P) and by teachers using Conners’ Global Index-Teacher version (Conners’-T), respectively.50 For a child to be included in the PBG study, at least 1 Conners’-P/T subscore had to be 65 or higher. The Child Behaviour Checklist (CBCL) was used to quantify several behavioural dimensions. The CBCL comprises 118 items and measures 3 dimensions (internalizing, externalizing and total problems). Internalizing behaviours refer to mood disturbance, including anxiety, depression and social withdrawal. Externalizing behaviours reflect conflict with others and violation of social norms. Finally, full IQ was measured using the Wechsler Intelligence Scale for Children (WISC).
For the present study, children with ADHD were selected based on the extreme end of the distribution: in the maternal smoking during pregnancy category, only children whose mothers had smoked 10 or more cigarettes per day throughout the duration of the pregnancy were selected. Final inclusion in our study sample was based on the DNA availability. There were no selection criteria based on symptom or behavioural severity.
Assessment of maternal smoking during pregnancy
Assessment of maternal smoking during pregnancy occurred at the baseline evaluation using the Kinney Medical and Gynaecological Questionnaire, which is a global assessment of exposure to pre- and perinatal environmental risk factors.51 Mothers retrospectively reported smoking during each trimester of the pregnancy. The mother’s report was corroborated in a separate interview with the child’s father or by others who were present during the pregnancy, including grandparents, whenever possible. In order to obtain information on the number of cigarettes smoked per day, the Fagerström Test, a validated questionnaire for nicotine dependence, was added.52 For birth weight, the vaccination booklet issued by the hospital at the birth of the child was used in the analysis.
Methylation
Genomic DNA extracted from peripheral blood was sent to the McGill University and Génome Québec Innovation Centre for treatment with sodium bisulfite. Genome-wide DNA methylation was assessed using Infinium MethylationEPIC BeadChip, which interrogates the DNA methylation profile of 867 532 CpG loci across the genome at a single-locus resolution.
Data preprocessing and clean up
Illumina GenomeStudio software was used to extract signal intensities for each probe (IDAT files). A probe corresponds to a CpG. All computations and statistical analyses were performed within the R x64 4.0.0 statistical analysis environment (http://www.r-project.org).
A first quality check was performed using the R package minfi. Samples with a bad ratio between methylated and unmethylated probes were removed (Appendix 1, Figure 1, available at www.jpn.ca/lookup/doi/10.1503/jpn.230062/tab-related-content). Then, the R package Chip Analysis Methylation Pipeline (ChAMP) was used (https://www.bioconductor.org/packages/release/bioc/vignettes/ChAMP/inst/doc/ChAMP.html). The fraction of failed positions per sample was checked (mean = 0.00034; maximum = 0.0008). The multidimensional scaling (MDS) plot is shown in Appendix 1, Figure 2, and the density plot in Appendix 1, Figure 3. No bias was detected. Filtering probes with a detection p value greater than 0.01 in 1 or more samples has removed 6183 probes from the analysis. Filtering probes with a sbeadcount less than 3 in at least 5% of samples removed 169 probes from the analysis. Filtering non-CPG probes removed a further 2977 from the analysis. According to Zhou,53 96 190 probes corresponding to SNPs were removed; 11 supplementary probes that align to multiple locations54 were removed. Filtering probes on the X or Y chromosome removed 16621 CpGs from the analysis. Further analyses proceeded with 743 767 probes and 231 samples.
Normalization was performed using the Beta-Mixture Quantile (BMIQ) method. The singular value decomposition method (SVD)55 for methylation data was used to identify the most significant components of variation, in particular to detect batch effect. Slide and array effects were detected and corrected using Combat package (Appendix 1, Figure 4). Cell type heterogeneity was corrected using the RefbaseEWAS method that infers changes in the distribution of white blood cells between different subpopulations using DNA methylation signatures, in combination with a previously obtained external validation set consisting of signatures from purified leukocyte samples. All scripts are available on request from the authors.
Association analysis
An association analysis was conducted with the limma R package to identify differentially methylated probes (DMP) with a linear model adjusted on age and sex. Multiple testing correction using false discovery rate (FDR) was performed. Manhattan plots and QQ plots were drawn in R using the qqman package. Differential methylated regions (DMR) were detected using bumphunter and ProbeLasso, both with the default parameters. Multiple testing correction use the family-wise error (FWE) method.
Correlation analysis with clinical data
The corrplot R package was used to test the Pearson correlation of each significant DMP and the mean of the DMR with the Conners’ scale (parent and teacher versions), the CBCL (internalized, externalized and total scores), the total number of items in the DISC, the total IQ and the birth weight. The correction for multiple testing was done using FDR correction (290 tests).
Results
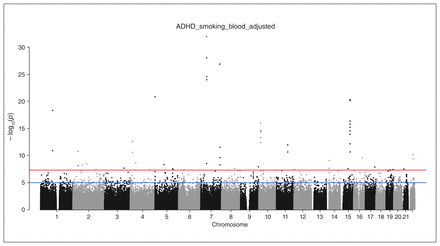
We selected 232 children with ADHD for inclusion in our study. One child was excluded owing to DNA samples with a bad ratio between methylated and unmethylated probes (Appendix 1), leaving a final sample of 231 children. Methylomic data from 80 children exposed to maternal smoking during pregnancy was compared with methylomic data from 151 nonexposed children (Table 1). We identified 3457 CpGs significantly associated with smoking during pregnancy (q < 0.05 after correction for multiple testing; Manhattan plot in Figure 1; qq-plot in Appendix 1, Figure 5). We kept 30 CpGs harboring a methylation difference greater than 5% or less than −5% between the 2 groups for further analyses (Table 2). Then, we tested these 30 CpGs with the clinical data (Table 3; see an example of correlation in Appendix 1, Figure 6). Birth weight was significantly and positively correlated with the methylation levels in 1 CpG located in GFI1 and 3 CpGs in NRP2 and negatively correlated with 1 CpG in ZNF536. The significant CpGs were submitted to the web application BECon (Blood-Brain Epigenetic Concordance; https://redgar598.shinyapps.io/BECon/) to test the concordance between blood and 3 brain areas: Brodmann area (BA)10 (frontal cortex), BA20 (temporal cortex) and BA7 (parietal cortex) (Appendix 1, Figure 7). The best correlations were obtained for CpGs located in CYP1A1 and in RUNX1 (correlation coefficient > 0.7).
Manhattan plot of the methylation epigenome-wide association study comparing children with attention-deficit/hyperactivity disorder (ADHD) who were exposed or not exposed to maternal smoking during pregnancy.
Demographic and clinical characteristics of the study sample
Differential methylation probe analysis between children who have been exposed to maternal smoking during pregnancy and those who have not been exposed
Correlation between clinical dimensions and the level of methylation of the significant differential methylated CpG
The externalizing CBCL score was significantly and positively correlated with the methylation levels in 7 CpGs located in RUNX1 and 4 CpGs located in MYO1G. The level of methylation of 6 of the 7 CpGs located in RUNX1 also correlated positively with the internalizing CBCL score. These 7 CpGs in RUNX1 were positively correlated with the total score on the CBCL. The CpG located in RUNX1 and harboring a very good correlation between the frontal cortex and the blood (cg00994804) was significantly associated with the score on the CBCL (Table 2 and Appendix 1, Figure 7). The CpG located in ETS2 was negatively correlated with both the externalizing CBCL score and the total CBCL score. Regarding the Connors’ scale (parent or teacher versions), the total score was significantly and positively correlated with the level of methylation of MYO1G. There was no correlation between the methylation levels in the DMP and IQ, nor with the number of DISC items.
Seven DMRs were identified (Table 4). We tested the mean level of methylation of the DMR with the score at the clinical scales. The mean methylation level in the DMR encompassing RUNX1 was associated with the externalizing, internalizing and total CBCL score. Birth weight was negatively correlated with the methylation of BHMT2 and positively correlated with the methylation of NRP2. The Conners’ scale was significantly and positively correlated with the mean methylation level in FRMD4. There was no correlation between the mean level of methylation in the DMR and IQ, nor with the number of DISC items.
Differential methylated regions analysis between children exposed to maternal smoking during pregnancy and those who were not exposed, and correlation with the clinical scales
Discussion
Using an epigenome-wide association study, we first confirmed that maternal smoking during pregnancy has an effect on DNA methylation in the children. We replicated some findings (hypomethylation of NRP2, hypermethylation of BHMT2),39 while findings for other genes, including RUNX1, were novel. In an earlier report with a small sample, we had noted an association between maternal smoking during pregnancy and differential methylation at specific sites within AHRR, GFI1 and CYP1A1.45 Again, we identified these genes among others and we further investigated the clinical consequences of maternal smoking during pregnancy by correlating the DNA methylation levels with dimensional phenotypes.
The association of birth weight with methylation at GFI1 confirms previous reports.45,46 Here we also report the novel association with birth weight where there was exposure to prenatal smoking during pregnancy, with NRP2 and ZNF536. NRP2 encodes a transmembrane protein, belonging to the neuropilin family of receptor proteins. It is a receptor for specific class 3 semaphorins and interacts with vascular endothelial growth factor. Semaphorins are polypeptides that are essential for axonal guidance, have been implicated in a broader role in the maintenance and stability of neuronal networks. 56 ZNF536 is expressed in the developing central nervous system and encodes a highly conserved zinc finger protein that is most abundant in the brain, showing negative regulation of neuron differentiation.57 Given the important role of NRP2 and ZNF536 in neuronal networks, molecular studies are warranted to help elucidate the epigenetic modulation that results from prenatal tobacco exposure and its connection to birth weight and ADHD.
Analysis with behavioural phenotypes implicated RUNX1 with CBCL scores. Runx1 is a transcription factor playing an important role in the developing nervous system and has been shown to be involved in the development of selected motor neurons, as well as gene expression programs regulating neuronal subtype specification and axonal connectivity.58 A gene from the same family, RUNX2, has been associated with maternal smoking during pregnancy in a meta-analysis conducted by the PACE consortium.39 RUNX1 is a very interesting candidate, as the DNA methylation levels correlated well between blood and the prefrontal cortex. A correlation between methylation level and CBCL and Conners’ scores was also noted with MYO1G. This gene has been recently highlighted as differentially expressed in ADHD by a transcriptomic analysis in the anterior cingulate cortex.59
The remarkable consistency of DNA methylation marks related to maternal smoking during pregnancy are in stark contrast to the more dynamic changes observed in analysis of DNA methylation and ADHD in the literature. Knowledge about the environmental factors is an asset to detect relevant DNA methylation marks and correlate them with clinical symptoms. Notwithstanding the causality issue of maternal smoking during pregnancy and ADHD, there is a need to better understand the effects of this important prenatal exposure on the developing fetus for its long-term effects. Previously we had presented a framework to help break down the complexity of ADHD and suggested that stratification of a sample based on exposure to environmental factors, including maternal smoking during pregnancy, could help provide unique insight into the disorder.47 We also underscored the importance of examining quantitative phenotypes of clinical relevance. The present study encapsulates both these features, and sheds novel insight of pathways that may be involved in the etiology of ADHD, in the subgroup of children whose mothers smoked during pregnancy. One of the specific advantages of this study is that the EWAS was conducted with multiple quantitative assessments of ADHD diagnosis, parent/teacher reports of behaviour using the Conners’ and the CBCL. These analyses extend our understanding of pathways that may be affected in children who were exposed to maternal smoking during pregnancy and hint at implicated mechanisms in adverse behavioural outcomes.
Limitations
This study has limitations. The correlative design prevents us from establishing causality and leaves it unclear whether the identified DNA methylation patterns represent an antecedent (e.g., reflecting genetic or environmental risk factors for ADHD), a mere correlate (e.g., due to smoking or other behaviours associated with ADHD) or a consequence (e.g., as a result of medication use or as part of the disease process itself). The other limitation of the study was that the information on maternal smoking during pregnancy was obtained through self-reports and was based on retrospective recall. While there was an attempt to obtain partner and/or family member verification of maternal smoking, the information may still be subject to recall bias. Other potential biases can emerge from the cohort that groups samples of varying age and developmental stage, as well as both males and females. We adjusted for sex and age, and excluded sex chromosome, but we cannot exclude sex-specificity. Moreover, we did not assess the type of medications the children were taking, the duration of the illness or of the drug exposure. All these factors can influence DNA methylation,59,60 but it is unlikely that they dysmethylate the same genes as maternal smoking during pregnancy. Future longitudinal studies should consider detailed environmental recording. In addition, the results obtained here relate to a clinical sample of children with a diagnosis of ADHD; we did not assess a control sample at the same time.
Conclusion
While our study results are interesting, they need to be confirmed in large, independent samples of children with ADHD. It will also be interesting to examine whether methylation changes in these peripheral blood markers are observed across the lifetime, in longitudinal studies of individuals with ADHD. Although further work is warranted, our results provide additional empirical evidence to help underscore the importance of preventing maternal smoking during pregnancy.
Acknowledgments
The authors express sincere gratitude to all the families who participated in this study. This work was supported in part by grants from the Fonds de Recherche du Québec - Santé (FRSQ#6690) and the Canadian Institutes of Health Research (MOP#53309, MOP#84537, MOP#126023) to R. Joober and N. Grizenko. B. Chaumette received a postdoctoral fellowship from the Healthy Brains for Healthy Lives project (Talent program) and a grant from the Fondation Bettencourt Schueller (CCA-INSERM-Bettencourt). W. Fageera received a PhD scholarship from the Ministry of Education of Saudi Arabia. The authors thank past and present members of the ADHD group for technical and clinical assistance.
Footnotes
Competing interests: B. Chaumette has received research funding from the Fondation Bettencourt Schueller (180 000€/4 yr) and speaking fees from Janssen-Cilag, Lundbeck and Eisai, outside the submitted work. He has no direct employment in profit organizations nor consultancies. N. Grizenko reports receiving research funding from the Canadian Institutes of Health Research (CIHR) and is a member of the advisory board for Purdue and Shire. A. Labbe is an associate editor of JPN. She was not involved in the review or decision to accept this manuscript for publication. R. Joober reports having received research funding from CIHR. He is on the advisory boards and speakers’ bureaus of Pfizer, Janssen Ortho, BMS, Sunovion, Otsuka, Lundbeck, Perdue and Myelin. He has received grant funding from them and from AstraZeneca and HLS. He has received honoraria from Janssen Canada, Shire, Lundbeck, Otsuka, Pfizer and from Perdue for CME presentations and royalties for Henry Stewart talks. No other competing interests were declared.
Contributors: N. Grizenko and R. Joober designed the study. W. Fageera, M.-È. Fortier and M. Ter-Stepanian acquired the data, which B. Chaumette and A. Labbe analyzed. B. Chaumette wrote the article. All of the authors revised it critically for important intellectual content, gave final approval of the version to be published and agreed to be accountable for all aspects of the work.
- Received April 25, 2023.
- Revision received June 27, 2023.
- Revision received August 7, 2023.
- Revision received August 10, 2023.
- Accepted August 11, 2023.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/