Serotonin (5-HT) neurons constitute a tiny fraction of neurons in the brain (300 000 out of a billion neurons in human brain [0.03%]), yet they send projections to virtually all regions of the brain.1 The 5-HT system has been the target of multiple therapeutic drugs, like selective serotonin reuptake inhibitors (SSRIs), approved to treat major depressive disorder by the US Food and Drug Administration since 19872 and with indications for several mental health conditions including anxiety, compulsions and eating disorders.3 More recently, psychedelic drugs targeting 5-HT receptors have been receiving attention for their behavioural actions, which are powerful yet variable among individuals.4 It is perhaps because of this breadth of actions that it has been difficult to define how 5-HT modulates specific brain regions to alter behaviour. The underlying complexity of the serotonin system can lead to contradictory findings, as argued by Moncrieff and colleagues.5 Yet, as discussed in this editorial, through consideration of how the 5-HT system works and the limitations of methods used we can also understand why different groups may obtain different or even contradictory results.
Moncrieff and colleagues5 present a systematic umbrella review of studies reviewing different aspects of 5-HT related to depression. Unfortunately, the umbrella approach tends to muddy the waters. A good example is the inconsistent association of lower regional 5-HT1A receptor levels with depression. 6–9 Without knowing the specifics of the probe (antagonist v. agonist10), method (positron emission tomography [PET] v. postmortem),7 use of a reference region compared with arterial input (e.g., in PET9) and regions affected (e.g., presynaptic v. postsynaptic 5-HT1A that have opposite effects on 5-HT transmission11,12), it is expected that substantial heterogeneity in results would ensue. Regarding the type of probe, antagonist probes appear insensitive to 5-HT because they are not displaceable by enhanced 5-HT levels, but some evidence suggests that agonist probes may be sensitive to 5-HT concentrations.13 Furthermore, antagonist probes bind equally to high- and low-affinity forms of the receptor, while agonist probes bind only to high-affinity receptors resulting in low binding capacity.14 They suggest that lower 5-HT1A receptor density, presumably the postsynaptic ones, would indicate higher levels of 5-HT in depression, but no justification for this argument has been presented. Since most 5-HT1A receptors are postsynaptic, with autoreceptors restricted to the raphe, lower 5-HT1A expression likely reflects reduced binding of postsynaptic receptors.12,15 Contrary to what Moncrieff and colleagues5 write, there is good evidence that use of arterial input for calculating the PET outcome yields replicable evidence of elevated 5-HT1A receptors.16 A combination of higher inhibitory autoreceptors and lower extracellular 5-HT may lead to compensatory upregulation of postsynaptic 5-HT1A receptors. On the other hand, in patients on a sustained regimen of 5-HT reuptake inhibitors that enhance synaptic levels of 5-HT, the elevated synaptic 5-HT may reduce 5-HT1A receptor density by desensitization, most notably the autoreceptors on the cell body of 5-HT neurons.17 Thus, while the umbrella review highlights numerous studies, their conclusions are as varied as the studies because so many variables can yield very different results. The sensitivity of this approach is thus very limited, ultimately able to detect only the effects of long-term administration of medications used to treat depression, not the effects associated with the illness per se. It is a weakness of large overviews that critical details are often overlooked.
Another example is that of studies on acute tryptophan depletion. These are currently conducted rarely in samples with depression, or in patients who have recovered from an episode, where the strongest evidence for mood lowering and relapse is seen.18,19 Historically, few effects have been observed in healthy individuals, and the current studies simply confirm this finding, although healthy women appear preferentially vulnerable.20 While healthy individuals without a family history of psychiatric illness show no response to tryptophan depletion, both men and women with a positive family history tend to show some depressive symptoms.21–23 This can simply reflect a difference in vulnerability in people at risk for depression or who have “recovered” from depression compared to those without depression. Similarly, reduced levels of the 5-HT metabolite 5-hydroxy-indole acetic acid (5-HIAA) in cerebrospinal fluid have rarely been associated with depression; rather, the stronger association is with suicidality and aggression.24,25 This probably reflects the variability of serum 5-HIAA, which is predominantly determined by the gut as it contributes 95% of 5-HT/5-HIAA in the periphery. Furthermore, sustained administration of 5-HT reuptake inhibitors decreases whole blood 5-HT by more than 90%, with a lesser effect in plasma,26,27 which is not the same in the corticospinal fluid, in which no changes have been detected.28 Therefore, this measure by no means reflects synaptic 5-HT in the brain, which is the measure that is needed but not feasible in living humans at present. While it is not yet possible to measure synaptic 5-HT concentrations in human brain, there is preliminary evidence of reduced cortical 5-HT release in people with a mood disorder using a displaceable 5-HT2A receptor PET tracer, 11C-Cimbi-36.29
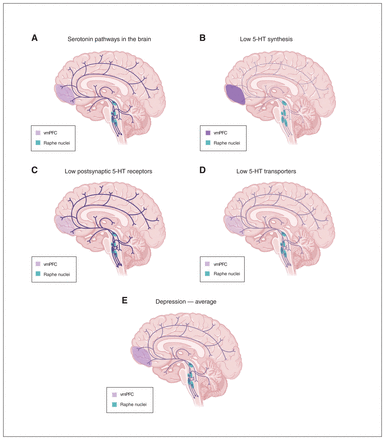
Importantly, the umbrella review by Moncrieff and colleagues5 does not appreciate the complexity of the 5-HT system: multiple different changes could lead to the same outcome of low 5-HT neurotransmission. Let us consider several patients with depression and different characteristics of their 5-HT system (Figure 1). One may have reduced density of postsynaptic 5-HT1A receptors in the cerebral cortex and hippocampus (e.g., due to a genetic change or early life stress) with no change in 5-HT levels, yet less effect on 5-HT transmission due to reduced receptor density. Another patient may show reduced levels or activity of tryptophan hydroxylase type 2 in the brain (whereas it is tryptophan hydroxylase type 1 that controls the synthesis of 5-HT in the periphery), resulting in low 5-HT availability and upregulation of postsynaptic 5-HT receptors to compensate. A third patient may show reduced density or function of 5-HT reuptake transporters due to genetic or environmental factors; by increasing synaptic 5-HT this would lead to reduced 5-HT receptor density, with unchanged total brain 5-HT levels.30 Yet another patient may have a combination of these anomalies. Averaging these together would reveal, not surprisingly, no net effect on the 5-HT system, yet in each case 5-HT transmission would be reduced.
(A) Serotonin (5-HT) pathways in the brain of healthy individual and in 3 scenarios of 5-HT changes in individuals with depression with different characteristics of the 5-HT system: (B) low 5-HT synthesis, (C) low levels of postsynaptic 5-HT receptors, or (D) low levels of 5-HT reuptake transporters. The intensity of colour in projections from the raphe depicts the level of 5-HT, while colour in the ventromedial prefrontal cortex (vmPFC) depicts the level of postsynaptic 5-HT receptors. (E) The average of data from depressed individuals in images B–D showing a similar pattern to the healthy individual in image A. Created with “Serotonin Pathways in the Brain,” BioRender.com (2023) with permission. Retrieved from https://app.biorender.com/biorender-templates
Furthermore, some patients may be taking medications for depression that primarily target 5-HT or other monoamines, which produce compensatory changes that obscure the original deficits that led to depression. For instance, the first patient from our example, with intact 5-HT levels, may respond to treatment with SSRIs, but this could desensitize presynaptic 5-HT receptors.17 The second patient, with deficient 5-HT synthesis, may respond weakly or transiently to chronic SSRI treatment as 5-HT levels become exhausted.31 The third patient, lacking 5-HT transporters, the target of SSRIs, 32 would be expected to be resistant to SSRI treatment, with a further downregulation of the transporter.33 Studies including patients with different treatment doses and times further complicate the ability to decipher depression-related changes in components of the 5-HT system.
Finally, it is suggested by Moncrieff and colleagues5 that there is “a possibility that long-term antidepressant use reduces serotonin concentration.” This is irreconcilable with the therapeutic benefit of monoamine oxidase inhibitors that increase 5-HT concentration in the brain and blood. Furthermore the implication that withdrawal of medications efficacious in the treatment of depression may be beneficial is not borne out, as their abrupt cessation results in severe relapse.34
Considering solely the complexity of elements controlling 5-HT transmission in the central nervous system, it must be recognized that 5-HT is likely only 1 contributor to depression. Given the large placebo effect in the treatment of depression and the reciprocal interactions between monoamine and glutamate neurons, it is likely that the contribution of other systems, including dopamine that is implicated in placebo effect, may be greater than that of 5-HT. In contrast in most animal studies, the placebo effect is not at play, allowing for a clearer opportunity to examine unobstructed mechanisms involving 5-HT. Perhaps by better understanding how to harness the mechanisms in placebo effect in humans could lead to more effective treatments.35
Footnotes
The views expressed in this editorial are those of the author(s) and do not necessarily reflect the position of the Canadian Medical Association or its subsidiaries, the journal’s editorial board or the Canadian College of Neuropsychopharmacology.
Competing interests: P. Blier declares grants from AbbVie, Otsuka and Lundbeck and has received consulting fees from AbbVie, Lundbeck and Pfizer. No other competing interests were declared.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/