Abstract
Background: Impaired functional connectivity between the bilateral hemispheres may serve as the neural substrate for anxiety and depressive disorders, yet its role in comorbid generalized anxiety disorder (GAD) and depression, as well as the effect of treatment on this connectivity, remains unclear. We sought to examine functional connectivity between homotopic regions of the 2 hemispheres (voxel-mirrored homotopic connectivity [VMHC]) among people with GAD with and without comorbid depression at baseline and after a 4-week paroxetine treatment.
Methods: Drug-naïve patients with GAD, with or without comorbid depression and healthy controls underwent functional magnetic resonance imaging and clinical assessments at baseline and after treatment. We compared VMHC and seed-based functional connectivity across the 3 groups. We performed correlation analysis and support vector regression (SVR) to examine the intrinsic relationships between VMHC and symptoms.
Results: Both patient groups (n = 40 with GAD only, n = 58 with GAD and depression) showed decreased VMHC in the precuneus, posterior cingulate cortex and lingual gyrus compared with healthy controls (n = 54). Moreover, they showed decreased VMHC in different brain regions compared with healthy controls. However, we did not observe any significant differences between the 2 patient groups. Seeds from abnormal VMHC clusters in patient groups had decreased functional connectivity. Voxel-mirrored homotopic connectivity in the precuneus, posterior cingulate cortex and lingual gyrus was negatively correlated with cognitive impairment among patients with GAD only and among all patients. The SVR analysis based on abnormal VMHC showed significant positive correlations (p < 0.0001) between predicted and actual treatment responses. However, we did not observe significant differences in VMHC or functional connectivity after treatment.
Limitations: A notable dropout rate and intergroup somatic symptom variations may have biased the results.
Conclusion: Patients with GAD with or without comorbid depression exhibited shared and distinct abnormal VMHC patterns, which might be linked to their cognitive deficits. These patterns have the potential to serve as prognostic biomarkers for GAD.
Clinical trial registration: ClinicalTrials.gov NCT03894085.
Introduction
Generalized anxiety disorder (GAD) is a prevalent anxiety disorder, with patients often experiencing a range of somatic discomforts. In addition, comorbid depression is frequently observed among people with GAD and may be a fundamental feature of the nature and progression of GAD.1,2 A meta-analysis showed that people with a current anxiety disorder often have comorbid current (63%) or lifetime (81%) major depressive disorder (MDD).3
A functional magnetic resonance imaging (fMRI) study reported aberrant amplitude low-frequency fluctuation, regional homogeneity and functional connectivity involved in the postcentral and precentral gyrus, the posterior cingulate cortex and the frontal–limbic regions of patients with GAD.4–7 Moreover, a meta-analysis concluded that the pathology of GAD involves various brain networks, including the default mode network, affective limbic network and sensorimotor network.5 However, exploration of the neural mechanism of GAD using voxel-mirrored homotopic connectivity (VMHC), referring to functional connectivity between homotopic regions of the 2 hemispheres, has been infrequently reported in the literature.8 Recently, impaired connectivity between the bilateral hemispheres has been frequently reported in MDD, schizophrenia and autism, implying that interhemispheric communication may be the neural substrate to facilitate coherent cognition, emotion and behaviour control.9–11
Furthermore, structural MRI studies have observed hypergyrification in the fusiform, frontal, temporal and parietal lobes of people with GAD.12 The process of gyrification is thought to occur during neurodevelopment. This finding can explain why GAD often emerges during adolescence and early adulthood. It is worth noting that during neural development, some neural functions in healthy individuals tend to exhibit lateralization. For example, language lateralization increases between the ages of 5 and 20 years, and plateaus between age 20 and 25 years.13 Similarly, different activation patterns in the left and right hemispheres during the perception and processing of emotional stimuli have been reported. 14 According to the findings of Deng and colleagues,15 stronger and weaker VMHC may, respectively, indicate increased or decreased interhemispheric communication and lateralized processing. As neural development plays an important role in both GAD and MDD, patients with GAD, with or without depression, may exhibit aberrant VMHC.
Almost 50% of people with GAD do not respond to first-line treatments such as antidepressants, underscoring the need for improved tools to predict patient response to drug therapy.16 Functional MRI is a nondestructive, high–spatial resolution tool that has shown promise in this regard. Moreover, machine learning algorithms, such as support vector regression (SVR), have been used to explore the potential value of neuroimaging as a prognostic biomarker.17 The SVR algorithm, using neuroimaging indicators, has shown the ability to predict short-term responses in studies of obsessive–compulsive disorder,18 schizophrenia19 and GAD.20 Thus, we sought to conduct an SVR analysis based on VMHC to predict treatment response in patients with GAD.
We aimed to investigate the interhemispheric synergy in patients with GAD with or without comorbid depression, identify common and distinct VMHC patterns, explore the impact of comorbid depression on VMHC and evaluate the potential of VMHC as a prognostic biomarker for GAD using SVR. In addition, we sought to use seed-based functional connectivity analysis based on the results of the VMHC analysis to comprehensively assess region-related functional connectivity in GAD, and investigate the associations between clinical manifestations and VMHC values. Finally, we sought to explore the short-term effects of drug treatment on VMHC and seed-based functional connectivity patterns in patients with GAD.
Methods
Participants
The study enrolled people with GAD among the outpatients who visited the Department of Psychiatry at the Second Xiangya Hospital between January 2019 and August 2022. We recruited healthy controls from the local community through advertising. Before participating in the research, all participants received a complete explanation of the study’s purpose and procedures and provided written informed consent.
Two psychiatrists used the Structured Clinical Interview for Diagnostic and Statistical Manual of Mental Disorders-5 (DSM-5, patient version) to diagnose GAD in the outpatients, while we selected healthy controls using the Structured Clinical Interview for DSM-5 Axis I Disorders (non-patient edition). We assigned patients with 17-item Hamilton Depression Rating Scale (HAMD-17) scores of 14 or higher to the comorbidity group, and assigned those with scores less than 14 to the pure GAD group. After the first clinical assessment and fMRI scan, patients were started on medication (paroxetine 20 mg/d), and a second clinical assessment and fMRI scan were conducted at the 4-week follow-up visit. Healthy controls provided demographic information and underwent an fMRI scan at baseline.
The clinical assessments for patients included the Hamilton Anxiety Rating Scale (HAMA) and HAMD-17 to assess the severity of depression and anxiety, the Social Disability Screening Schedule (SDSS) to measure daily living activities and social function,21 the Simplified Coping Style Questionnaire (which measures active and passive coping dimensions) to assess coping styles using22 and the Brief Cognitive Assessment Tool for Schizophrenia to assess cognitive function using; this tool includes 4 subscales, namely the Trail Making Tests A and B (TMT-A and TMT-B), the Digit Symbol Substitution Test and category fluency.23 We also used the Chinese version of the Eysenck Personality Questionnaire (EPQ) to evaluate personalistic characteristics.24 The EPQ includes 4 subscales for psychoticism, extraversion, neuroticism and lying.
MRI scanning and VMHC and functional connectivity calculation
We acquired functional images using a 3.0 T brain scanner (Philips Achieva, Philips Medical System). The data were collected through an echo-planar imaging sequence with a repetition time of 2000 ms, an echo time of 30 ms, 33 slices, 64 × 64 matrices, 90° flip angle, 22-cm field of view, 4-mm slice thickness, no gap and a total of 240 volumes.
We applied standard preprocessing procedures to the fMRI data. Specifically, to eliminate nuisance signals, we used the Friston 24-parameter model in combination with white matter and cerebrospinal fluid signals for covariate regression. However, we chose not to regress out global signal in alignment with the findings of Zhang and Northoff25 that global signal may have an as-yet-undefined physiologic role in brain function. Nevertheless, we reported the results with global signal regression in Appendix 1, available at www.jpn.ca/lookup/doi/10.1503/jpn.230091/tab-related-content. A more comprehensive description of the data acquisition and preprocessing steps can be found in the supplementary document.
We computed VMHC using the Data Processing Assistant for Resting-State fMRI (DPARSF) toolbox by calculating the Pearson correlation coefficient between each voxel and its mirrored counterpart to measure homologous functional connectivity for each participant.26 We converted the resulting correlation maps to z-value VMHC maps using the Fisher r-to-z conversion. Subsequently, we performed an integrated approach combining VMHC and seed-based functional connectivity analysis using the same toolbox.27 We defined regions of interest (ROIs) for the functional connectivity analysis based on the clusters of VMHC that showed significant group differences. We computed the reference time course as the mean time series of the abnormal VMHC cluster, and performed a correlation analysis between this seed reference time course and the time series of all other voxels in the brain. To enable subsequent group comparisons, we converted the resulting correlation maps to z-value functional connectivity maps using the Fisher r-to-z conversion.
SVR analysis
We performed the SVR analysis using the Library for Support Vector Machines (LIBSVM) toolbox.28 We defined actual treatment response as the reduction rate in the HAMA or HAMD-17 scores after treatment. For instance, we calculated the reduction rate for HAMA by substracting HAMA scores at 4 weeks from that at baseline, and dividing the total by the baseline scores. The SVR model was trained to predict treatment response based on VMHC using a radial basis function kernel and 5-fold cross-validation. We used a grid search method to optimize the hyperparameters of the SVR model. We evaluated the predictive performance by calculating the mean square error (MSE) and Pearson correlation between the predicted and actual treatment response. To determine if the predictive performance was better than chance, we performed a permutation test was performed. See Appendix 1 for the detailed process of SVR analysis.
Statistical analysis
We analyzed demographic and clinical data using SPSS (version 25.0). We compared age, education level and gender distribution across the 3 groups using analysis of variance (ANOVA) and χ2 tests. We determined differences in illness duration and the scores on the HAMD-17, HAMA, SDSS, Coping Style Questionnaire, Brief Cognitive Assessment Tool for Schizophrenia and EPQ of the 2 patient groups using 2-sample t tests. We used paired t tests to compare the clinical symptoms of patients before and after treatment. Statistical significance was set at p less than 0.05.
We conducted statistical analyses for imaging data using the DPARSF toolbox. We performed ANOVA and post hoc t tests to compare whole-brain VMHC maps among the 3 groups, with mean framewise displacement, age, gender and years of education as covariates. We used paired t tests to compare VMHC values before and after treatment. We defined individuals who had responded to treatment as those who had a reduction rate of at least 30% on the HAMA scores (among patients with GAD only) and at least 30% on either the HAMA or HAMD-17 scores (among patients with GAD and comorbid depression).29 We set the significance levels in the post hoc and paired t tests at p < 0.05 and corrected significance levels for multiple comparisons using Gaussian random field theory, with a cluster significance of p < 0.05 and voxel significance of p < 0.001. We conducted correlation analyses to examine the relationship between VMHC and clinical data at baseline, as well as the relationship between changes in VMHC and changes in clinical data after treatment. We used a significance level of 0.05 and applied a Bonferroni correction for multiple comparisons (p = 0.05/the number of tests). We applied Pearson correlation for normally distributed or approximately normally distributed data, and Spearman correlation for non-parametric data.
Ethics approval
The study was approved by the Research Ethics Committee of the Second Xiangya Hospital (no. 2018025). The clinical trial was registered on ClinicalTrials.gov (NCT03894085). Additional information about the trial’s registration is available at https://clinicaltrials.gov/ct2/show/NCT03894085.
Results
Demographic and clinical characteristics
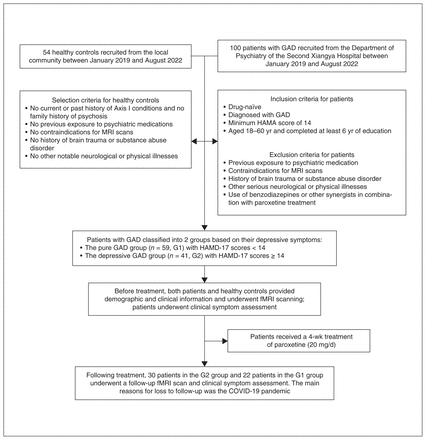
A detailed inclusion and exclusion flow chart is presented in Figure 1. After excluding 2 patients (1 in each patient group) because of excessive head movement, 98 patients (40 with GAD only, 58 with comorbid depression) and 54 healthy controls were enrolled for statistical analysis. Because of the COVID-19 pandemic, only 52 of the 98 patients (22 with GAD only, 30 with GAD and depression) completed the 4-week follow-up, while 16 patients with GAD only and 26 patients with GAD and depression responded to the treatment (Appendix 1, Table S1). Before and after treatment, age, gender and years of education were not significantly different among the 3 groups, and nor was illness duration between the 2 patient groups (Table 1 and Table 2). At baseline, patients with GAD and depression showed higher HAMA (p < 0.001), HAMD-17 (p < 0.001) and SDSS (p = 0.003) scores, and scored higher on the neuroticism scale of the EPQ (p = 0.002) than patients with GAD only (Table 1).
A flow chart. fMRI = functional magnetic resonance imaging; GAD = generalized anxiety disorder; HAMA = Hamilton Anxiety Rating Scale; HAMD-17 = 17-item Hamilton Depression Rating Scale; MRI = magnetic resonance imaging.
Participant characteristics
Characteristics of patients who finished follow-up
After treatment (Table 2), both patient groups showed statistical improvement in HAMA, HAMD-17, SDSS and TMT-A scores. In addition, patients with both GAD and depression showed significant improvement in the scales of the TMT-B (p = 0.012), and patients with GAD only had increased active coping scores (p = 0.032). However, the change in the scores of Digit Symbol Substitution Test differed by patient group, with an increase among patients with both GAD and depression (p = 0.002) and a decrease among those with GAD only (p = 0.003). Except for HAMD-17 scores (p = 0.036), we did not observe any differences in scale scores between the 2 patient groups after treatment.
VMHC differences between groups
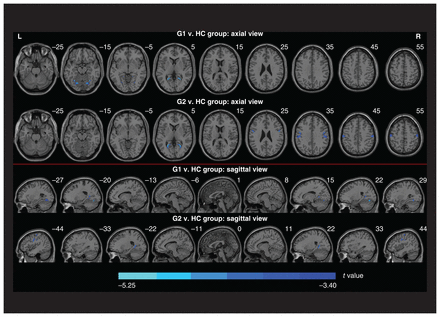
Compared with healthy controls, patients with GAD only had decreased VMHC in the fusiform, lingual gyrus and cerebellum VI, and in the precuneus, posterior cingulate cortex and lingual gyrus; patients with both GAD and depression had decreased VMHC in the postcentral and precentral gyrus and the precuneus, posterior cingulate cortex and lingual gyrus (Figure 2 and Table 3). However, there was no difference in VMHC between the 2 patient groups. After treatment, there were no significant changes in VMHC relative to baseline when comparing either all patients who completed the follow-up or those who showed a response to treatment.
Brain regions exhibiting abnormal voxel-mirrored homotopic connectivity among patients with generalized anxiety disorder (GAD) only (G1) and those with both GAD and depression (G2), compared with healthy controls (HC). The corresponding statistical t values are represented by the colour bars.
Regions with abnormal voxel-mirrored homotopic connectivity (VMHC) values among patients with GAD at baseline and alterations of VMHC values after treatment
As shown in Appendix 1, Figure S1, the results with global signal regression were similar to those without global signal regression.
Seed-based functional connectivity analysis
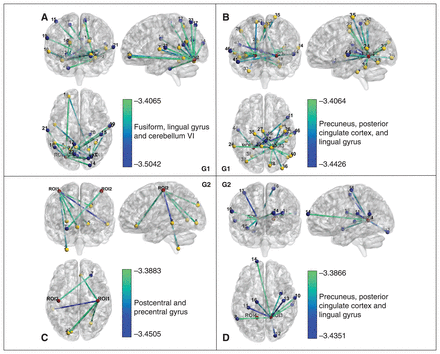
We defined the ROIs for the functional connectivity analysis based on the clusters of VMHC that exhibited significant group differences (4 clusters in each patient group). We observed decreased functional connectivity in all of the defined seeds in the patient groups, primarily distributed in the temporo-parieto-occipital cortex (TPO), sensorimotor regions (including the supplementary motor area, paracentral lobule, postcentral gyrus and precentral gyrus), visual association cortex (comprising the fusiform, lingual and calcarine gyrus) and insula, relative to healthy controls. More detailed information regarding these findings can be found in Figure 3 and Appendix 1, Table S2.
Compared with the healthy controls, brain regions with abnormal seed-based functional connectivity among patients with generalized anxiety disorder (GAD) only (G1) and those with both GAD and comorbid depression (G2). The colour bars indicate the statistical t value. In the G1 group (A and B), the locations of the numbered sites are as follows: region of interest (ROI) 1, right fusiform gyrus, lingual gyrus and cerebellum VI; ROI2, left fusiform gyrus, lingual gyrus and cerebellum VI; ROI3, right precuneus, posterior cingulate cortex and lingual gyrus; ROI4, left precuneus, PCC and lingual gyrus; 1, right calcarine gyrus, precuneus; 2, right fusiform gyrus, parahippocampal gyrus; 3, left precuneus, cingulate gyrus; 4, left superior frontal gyrus; 5, left fusiform gyrus; 6, left lingual gyrus and fusiform gyrus; 7, left superior temporal gyrus; 8, left middle temporal gyrus; 9, left calcarine gyrus; 10, right superior temporal gyrus; 11, left and right calcarine gyrus and cuneus gyrus; 12, left precuneus and paracentral lobule; 13, right fusiform gyrus; 14, right middle occipital gyrus and superior occipital gyrus; 15, right postcentral gyrus and precentral gyrus; 16, left superior and middle orbital frontal gyrus; 17, right cuneus and superior parietal gyrus; 18, left middle temporal gyrus and superior temporal gyrus; 19, right postcentral gyrus and superior temporal gyrus; 20, right fusiform gyrus and cerebellum IV/V; 21, left superior temporal gyrus; 22, right lingual gyrus; 23, left precuneus cortex; 24, left middle temporal gyrus and superior temporal gyrus; 25, right superior temporal gyrus and temporal pole; 26, right postcentral gyrus and precentral gyrus; 27, right and left supplementary motor cortex; 28, right cuneus and superior occipital gyrus; 29, right inferior temporal gyrus and cerebellum crus1; 30, right postcentral gyrus and precentral gyrus; 31, left fusiform gyrus and cerebellum VI; 32, left precuneus and middle cingulate gyrus; 33, right fusiform gyrus and cerebellum IV/V/VI; 34, right insula; 35, left paracentral lobule and precentral gyrus; 36, right middle occipital gyrus; 37, left middle occipital gyrus; 38, left fusiform gyrus and lingual gyrus; 39, right middle temporal gyrus; 40, right middle temporal gyrus; 41, right temporal pole and superior temporal gyrus; 42, right paracentral lobule; 43, right Rolandic operculum cortex; 44, left superior temporal gyrus; 45, right thalamus; and 46, right superior temporal gyrus. In the G2 (C and D) group, the locations of the numbered sites are as follows: ROI1, right postcentral gyrus and precentral gyrus; ROI2, left postcentral gyrus and precentral gyrus; ROI3, right precuneus, posterior cingulate cortex and lingual gyrus; ROI4, left precuneus, posterior cingulate cortex and lingual gyrus; 1, right medial frontal orbital gyrus; 2, right lingual gyrus and calcarine gyrus; 3, right cerebellum VIII; 4, right medial frontal orbital gyrus and anterior cingulate gyrus; 5, left superior occipital gyrus; 6, left lingual gyrus; 7, left middle temporal gyrus; 8, left superior temporal gyrus and middle temporal gyrus; 9, right superior temporal gyrus and middle temporal gyrus; 10, right postcentral gyrus; 11, left insula and Rolandic operculum; 12, right cerebellum crus1; 13, right precentral gyrus; 14, left superior and middle frontal gyrus; 15, right insula; and 16, left precentral gyrus and paracentral lobule.
Correlations between VMHC and clinical characteristics
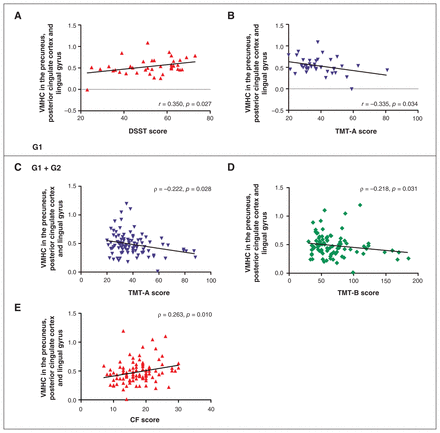
As shown in Figure 4 and Appendix 1, Table S3, among patients with GAD only, we observed a positive correlation between VMHC in the precuneus, posterior cingulate cortex and lingual gyrus cluster and the Digit Symbol Substitution Test score (r = 0.350, p = 0.027), and a negative correlation between this VMHC cluster and the TMT-A score (r = −0.335, p = 0.034). Furthermore, among all patients, VMHC in this cluster exhibited a negative correlation with the TMT-A score (ρ = −0.222, p = 0.028) and TMT-B score (ρ = −0.218, p = 0.031), and a positive correlation with the category fluency score (ρ = 0.263, p = 0.010). However, these correlations did not survive the Bonferroni correction. We did not observe any significant correlations between VMHC and clinical data among patients with both GAD and depression. Similarly, we did not observe significant correlations between changes in VMHC and changes in clinical data after treatment in the patient groups.
The correlation between abnormal voxel-mirrored homotopic connectivity (VMHC) and clinical symptoms of patients. Among patients with generalized anxiety disorder (GAD) only (G1), abnormal VMHC in the precuneus, posterior cingulate cortex and lingual gyrus cluster had a (A) positive correlation with Digit Symbol Substitution Test (DSST) scores (r = 0.350, p = 0.027) and a (B) negative correlation with Trail Making Test A (TMT-A) scores (r = −0.335, p = 0.034). The same brain regions among all patients with GAD, with and without depression (G1 + G2) exhibited a (C) negative correlation with TMT-A scores (ρ = −0.222, p = 0.028) and (D) Trail Making Test B (TMT-B) scores (ρ = −0.218, p = 0.031), and a (E) positive correlation with category fluency (CF) scores (ρ = 0.263, p = 0.010).
Prediction of treatment response
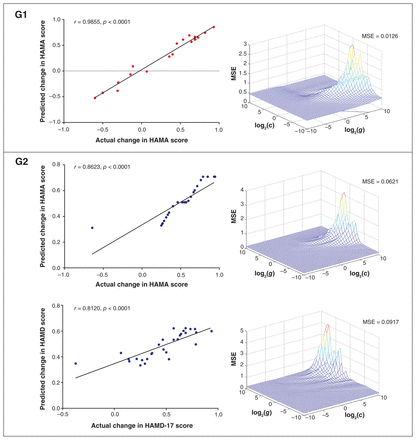
For predicting the improvement of anxiety, the combination of the VMHC in the fusiform, lingual gyrus and cerebellum VI and the precuneus, posterior cingulate cortex and lingual gyrus among patients with GAD only (r = 0.9855, MSE = 0.0126, permutation test p < 0.0001), and the postcentral and precentral gyrus and the precuneus, posterior cingulate cortex and lingual gyrus among patients with GAD and depression (r = 0.8623, MSE = 0.0621, permutation test p < 0.0001) obtained the most favourable outcomes. For predicting the improvement of depression, the VMHC in the postcentral and precentral gyrus among patients with GAD and depression obtained the most favourable outcomes (r = 0.8120, MSE = 0.0917, permutation test p < 0.0001). Detailed information was presented in Figure 5 and Appendix 1, Table S4.
Support vector regression (SVR) to predict the reduction rate of the (A) Hamilton Anxiety Rating Scale (HAMA) score among patients with generalized anxiety disorder (GAD) only (G1) and (B) patients with GAD and comorbid depression (G2), and (C) the reduction rate of the 17-item Hamilton Depression Rating Scale (HAMD-17) score among patients with GAD and comorbid depression, based on abnormal voxel-mirrored homotopic connectivity (VMHC). Among patients with GAD only, the combination of VMHC in the fusiform, lingual gyrus and cerebellum VI and the precuneus, posterior cingulate cortex and lingual gyrus obtained a significant correlation between predicted and actual reduction rate of the HAMA score (r = 0.9855, p < 0.0001; permutation test p < 0.0001) and a low mean square error (MSE = 0.0126, permutation test p < 0.0001). Among patients with GAD and comorbid depression, VMHC in the postcentral and precentral gyrus and the precuneus, posterior cingulate cortex and lingual gyrus obtained significant correlations between predicted and actual reduction rate of both the HAMA score (r = 0.8623, p < 0.0001, permutation test p < 0.0001) and the HAMD score (r = 0.8120, p < 0.0001, permutation test p < 0.0001). The MSE was low for both the HAMA (MSE = 0.0621, permutation test p < 0.0001) and HAMD scores (MSE = 0.0917, permutation test p < 0.0001).
Discussion
In this study, both patient groups had decreased VMHC in the precuneus, posterior cingulate cortex and lingual gyrus cluster than healthy controls. In addition, both patient groups exhibited distinct changes compared with healthy controls, but no significant differences were observed when directly comparing the 2 patient groups. The seeds from the abnormal VMHC clusters in both patient groups exhibited decreased functional connectivity. We found that VMHC in the precuneus, posterior cingulate cortex and lingual gyrus was negatively correlated with cognitive impairment among patients with GAD only and among all patients. The SVR analysis demonstrated the potential of VMHC as a prognostic biomarker.
Reduced homotopic connectivity in patient groups
Both patient groups exhibited decreased VMHC in the precuneus, posterior cingulate cortex and lingual gyrus before treatment. The precuneus and posterior cingulate cortex are known to play crucial roles in various cognitive tasks,30 including emotional processing, episodic memory retrieval, cognitive processing and attention regulation.31,32 The precuneus and posterior cingulate cortex are essential components in the default mode network.33 Previous studies have reported structural and functional abnormalities in these regions among patients with GAD, suggesting a neural basis for cognitive impairment in these individuals.4,34,35 Specifically in the context of anxiety, the neurocardiac desynchronization is linked to alterations in serotonergic and noradrenergic neurotransmission. 36 These changes, in turn, contribute to a decrease in default mode network connectivity, potentially giving rise to an unstable sense of self.36 Consistent with this issue, we observed a significant correlation between the severity of cognitive impairments and VMHC in the precuneus, posterior cingulate cortex and lingual gyrus among patients with GAD only and among all patients. The findings of this study suggest that the impaired homologous functional connectivity in the precuneus, posterior cingulate cortex and lingual gyrus is not a specific alteration in patients with GAD but rather a shared pathological mechanism of patients with pure GAD and patients with comorbid GAD and depression.37 Specifically, the deficits of VMHC in the precuneus, posterior cingulate cortex and lingual gyrus may be the underpinning of the cognitive impairments among these patients.
In addition, patients with GAD displayed abnormal VMHC only in the fusiform, lingual gyrus and cerebellum VI, which are areas that are commonly affected in patients with GAD and are involved in facial perception, including facial identity recognition and eye contact.38 Notably, alterations in these regions are fundamental changes in GAD and also potential therapeutic targets. A study demonstrated that acupuncture can partially improve anxiety symptoms by modifying the activities of the fusiform gyrus and cerebellar vermis.39
Patients with both GAD and depression showed exclusive changes in VMHC in the postcentral and precentral gyrus compared with healthy controls. These regions are primarily involved in processing sensory and motor information, and their aberrant activity and functional connectivity have been speculated to be related to increased muscle tension, feelings of being on edge and hypervigilance.5 Moreover, the postcentral and precentral gyrus are crucial for the processing of emotional information.40 Both MDD and GAD feature an impaired ability to identify the emotional stimulus and generate suitable emotional states, which has been found to be associated with aberrant activities in the postcentral and precentral gyrus.4,41–43 Thus, the distinctive decreased VMHC in the postcentral and precentral gyrus among people with comorbid GAD and depression embodies the feature of comorbidity.
Comorbid depression may affect the psychopathological process of patients with GAD,44 but the changes in the psychopathological process of patients with GAD cannot be solely attributed to depression. For instance, patients in the comorbidity group had higher anxiety scores, which may modulate activation in response to divergent emotional information. 45 Temperament and personality are known to mediate internal and external factors contributing to psychiatric disorders.46 In our study, the comorbid group had higher scores on the neuroticism scale than the non-comorbid group, indicating that depressive symptoms among patients with GAD may result from the interaction between neurotic personality and GAD. In sum, the absence of significant differences in VMHC between the 2 patient groups could be attributed to the combined effects of comorbid depression, higher anxiety levels and higher neuroticism.
Decreased seed-based functional connectivity in patient groups
The 2 patient groups exhibited widespread functional connectivity decreases between the ROIs and brain regions. The regions with significantly reduced functional connectivity in the patient groups were primarily distributed in the TPO regions, sensorimotor regions and visual association cortex relative to healthy controls. These scattered brain regions have been reported to have abnormal activities among patients with GAD.5,34
An event-related fMRI study revealed that TPO regions are interactive with the posterior cingulate cortex and associated with top–down attentional-control processes that are active in response to instructive cues, like rightward- or leftward-pointing cues.7 Furthermore, the TPO regions play a role in integrating auditory, somatosensory and visual information, which contributes to the formation of a heteromodal association area.47,48 This area lays a fundamental base for human high-cognitive functions, including information processing, emotion regulation and cognitive control.49 Therefore, the impaired communication between all the ROIs (the precuneus, posterior cingulate cortex and lingual gyrus; the fusiform gyrus, lingual gyrus and cerebellum VI; and the postcentral and precentral gyrus) and TPO regions reflects the abnormal emotional and sensory information processing, self-awareness and attention among patients with GAD.13,50
In addition to their involvement in processing motor and emotional information, somatosensory areas were involved in perceiving interoceptive and exteroceptive sensory stimuli with the visual association cortexes. Aberrant activity in these brain regions may indicate a low perception threshold among patients with GAD, which means that a large amount of sensory information cannot be effectively filtered. Referred to a heartbeat perception task during fMRI, patients with GAD displayed increased interoceptive awareness, which was thought associated with abnormal functional connectivity from the insula to the postcentral and precentral gyrus, occipital pole and calcarine cortex.51 Abnormal activities in these brain regions may contribute to the physical discomfort of people with GAD.
Treatment effects
After 4 weeks of treatment with paroxetine, patients with GAD exhibited a statistically significant reduction in symptom severity. This finding is consistent with previous studies that have reported significant improvement in cognitive function and anxiety symptoms among patients with GAD after short-term SSRI treatment with selective serotonin reuptake inhibitors.52,53 Furthermore, the lack of difference between the response rates between patients with GAD, with and without depression, suggested that comorbid depressive symptoms did not affect the treatment response rate.
Compared with a meta-analysis that included 5 studies on the efficacy of paroxetine in treating GAD — in which the lowest effect size observed was 1.15 (95% confidence interval [CI] 0.96–1.34) and the highest effect size was 1.60 (95% CI 1.37–1.84) — the pre–post effect size of treatment in this study was higher among patients with both GAD and depression (Cohen d 2.05, 95% CI 1.46–2.76), but not among patients with GAD alone (Cohen d 1.05, 95% CI 0.54–1.63).54 However, when considering the reduction in HAMA scores, our study (mean change scores of 7.58 across all patients) did not show a higher change than the meta-analysis (approximate mean change score of 12).54 Nevertheless, from the perspective of response rates, patients with both GAD and depression (87%) showed a higher response rate than a previous study that reported a response rate of 72%,55 as well as another study that reported response rates of 62% and 68% for patients with GAD receiving 20 mg and 40 mg of paroxetine, respectively.56 In contrast, patients with GAD (73%) did not exhibit a significantly higher response rate. The assessment of paroxetine’s efficacy in treating GAD may be affected by several factors, such as treatment duration, baseline severity of anxiety, response threshold, inclusion of drug-naïve patients, presence of comorbid depression, dose of paroxetine and sample size. In comparison to the studies included in the meta-analysis,54 our study had a shorter treatment duration (4 wk v. 8 wk), a lower baseline severity of anxiety (mean HAMA score 17 v. around 25 in the meta-analysis) and a different threshold for response (30% reduction in HAMA score after 4-wk treatment v. 50% reduction after 8-wk treatment). In addition, we included drug-naïve patients with a relatively small sample size and a group patients with GAD and comorbid depression. These factors could potentially account for the higher response rate we observed than the previous meta-analysis. 54 The patient’s response to the drug may affect the outcome of the imaging study. An extreme example is the difference in imaging findings between refractory and nonrefractory patients.57
We did not observe significant changes in neural activities relative to the baseline in the 2 patient groups using VMHC and functional connectivity analysis. The lack of follow-up studies of VMHC on GAD prevents us from making direct comparisons between our findings and previous research. We speculated that the lack of significant changes in our study may be because of the need for a longer treatment duration to detect changes in VMHC. Furthermore, local changes in the brain area may be possible to observe in the early stage of medication use, but it may not be possible to observe VMHC changes because it reflects long-range interhemispheric functional connectivity. The VMHC may represent the recovery of brain coordination and integration ability, which can be understood as an indicator of long-term treatment of patients.
Limitations
We had relatively high dropout rate. We contacted patients who were unable to participate in the follow-up and found that most of them dropped out because of the inconvenience caused by the COVID-19 pandemic. We did not measure differences in somatic symptoms between the patient groups. Somatic symptoms may mediate the development of comorbidities. 52 Although we established and tested the SVR model, its generalizability to other samples of patients with GAD remains uncertain.
Conclusion
Patients with GAD with or without comorbid depression exhibited shared and distinct abnormal VMHC patterns, which may be linked to their cognitive deficits. These patterns may have the potential to serve as prognostic biomarkers for GAD.
Acknowledgment
The authors thank all participants.
Footnotes
↵* These authors contributed equally to this work and share first authorship.
Competing interests: None declared.
Contributors: All of the authors contributed to the conception and design of the work. Yiding Han, Haohao Yan, Xiaoxiao Shan, Huabing Li and Wenbin Guo contributed to data acquisition. Yiding Han, Haohao Yan contributed to data analysis. All of the authors contributed to data interpretation. Yiding Han and Haohao Yan drafted the manuscript. All of the authors revised it critically for important intellectual content, gave final approval of the version to be published and agreed to be accountable for all aspects of the work.
Funding: This study was supported by grants from the National Natural Science Foundation of China (no. 82171508), Natural Science Foundation of Hunan (no. 2020JJ4784), Science and Technology Program of Hunan Province (no. 2020SK53413) and Natural Science Foundation of Tianjin (no. 18JCQNJC10900).
Data sharing: Data can be made available upon reasonable request.
- Received June 6, 2023.
- Revision received August 1, 2023.
- Revision received August 26, 2023.
- Accepted August 26, 2023.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/