Abstract
Background: Interhemispheric cooperation is one of the most prominent functional architectures of the human brain. In patients with schizophrenia, interhemispheric cooperation deficits have been reported using increasingly powerful neurobehavioural and neuroimaging measures. However, these methods rely in part on the assumption of anatomic symmetry between hemispheres. In the present study, we explored interhemispheric cooperation deficits in schizophrenia using a newly developed index, connectivity between functionally homotopic voxels (CFH), which is unbiased by hemispheric asymmetry.
Methods: Patients with schizophrenia and age- and sexmatched healthy controls underwent multimodal MRI, and whole-brain CFH maps were constructed for comparison between groups. We examined the correlations of differing CFH values between the schizophrenia and control groups using various neurotransmitter receptor and transporter densities.
Results: We included 86 patients with schizophrenia and 86 matched controls in our analysis. Patients with schizophrenia showed significantly lower CFH values in the frontal lobes, left postcentral gyrus and right inferior temporal gyrus, and significantly greater CFH values in the right caudate nucleus than healthy controls. Moreover, the differing CFH values in patients with schizophrenia were significantly correlated with positive symptom score and illness duration. Functional connectivity within frontal lobes was significantly reduced at the voxel cluster level compared with healthy controls. Finally, the abnormal CFH map of patients with schizophrenia was spatially associated with the densities of the dopamine D1 and D2 receptors, fluorodopa, dopamine transporter, serotonin transporter and acetylcholine transporter.
Conclusion: Regional abnormalities in interhemispheric cooperation may contribute to the clinical symptoms of schizophrenia. These CFH abnormalities may be associated with dysfunction in neurotransmitter systems strongly implicated in schizophrenia.
Introduction
Interhemispheric cooperation allows for more efficient information processing during complex cognitive tasks.1–4 Schizophrenia is considered a disconnectivity syndrome characterized by disrupted functional connectivity between brain areas, including areas in opposite hemispheres.5 These abnormalities in interhemispheric connectivity have been confirmed at the behavioural level and by brain activity measures using various neuroimaging methods.6–9
Behavioural studies have reported that healthy individuals show an advantage in processing information presented bilaterally compared with the same information presented initially to a single hemisphere.10 However, this bilateral advantage is generally absent in patients with schizophrenia, suggesting a deficit in interhemispheric cooperation.11–13 Accumulating evidence from resting-state functional magnetic resonance imaging (rs-fMRI) also supports the hypothesis of regional interhemispheric cooperation abnormalities in both early-onset and adult schizophrenia, regardless of medication use.9,14–16 Moreover, these interhemispheric cooperation impairments have been strongly correlated with symptom severity and, along with clinical symptoms, have been shown to improve with drug therapy.17,18 In addition, it has been reported that interhemispheric cooperation increased significantly in specific brain regions compared with healthy controls.19 Therefore, interhemispheric cooperation abnormalities appear throughout the course of schizophrenia and contribute to pathogenesis.
In previous fMRI studies, interhemispheric cooperation was typically quantified by the correlation between a given voxel in 1 hemisphere and its mirrored voxel in the opposite hemisphere.20 However, the bilateral hemispheres are not anatomically symmetric.21 Regardless, investigators have normalized individual brain connectivity maps using a standard symmetric template, which may produce unexpected biases. The advantages of using functional rather than anatomic correspondence have also been clearly shown by the construction of precise cross-hemispheric cortical maps.22 Thus, a better option would be to define homotopic regions based on function rather than structural features.
In the present study, we examined changes in interhemispheric cooperation among patients with schizophrenia relative to matched healthy controls using a newly developed index, connectivity between functionally homotopic voxels (CFH), in which the functional homotopic region of a given voxel is defined as the point with the largest functional connectivity (FC) value in the contralateral hemisphere. Thus, regions with higher CFH values communicate more extensively across hemispheres. Recently, CFH was used to measure the interhemispheric cooperation in patients with Parkinson disease.23 To facilitate the development of future treatments, we also examined whether the CFH differences were associated with neurotransmitter receptor and transporter densities.
Methods
Participants
We recruited right-handed patients with schizophrenia aged 17–53 years from the Anhui Mental Health Center, Hefei, China. Schizophrenia was diagnosed by 2 psychiatrists using the Structured Clinical Interview for the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (SCID-IV). All patients were assessed for symptom severity using the Positive and Negative Syndrome Scale (PANSS). We excluded patients with a history of significant head trauma or neurologic disorders, history of addiction (including alcohol or drugs), focal brain lesions on T1- or T2-weighted fluid-attenuated inversion-recovery images and any contraindications to MRI. Age- and sex-matched healthy controls were recruited from the local community. The healthy controls had no history of psychiatric or neurologic disorders and met the safety criteria for MRI.
This study was approved by the Ethics Committee of Anhui Medical University. All participants provided written informed consent before examination.
MRI data acquisition
Functional and structural MRI data were acquired at the University of Science and Technology of China (Hefei, Anhui Province) using a 3.0 T scanner (Discovery 750, GE Healthcare) with the same parameters as in our previous studies.24 Briefly, functional images (217 volumes) were acquired using a single-shot gradient-recalled echo planar imaging sequence (repetition time [TR] 2400 ms, echo time [TE] 30 ms, flip angle 90º). Images of 46 transverse sections (field of view [FOV] 192 × 192 mm2, 64 × 64 in-plane matrix, section thickness 3 mm without intersection gap) were acquired parallel to the anteroposterior commissure line. High spatial resolution T1-weighted anatomic images were then acquired in the sagittal orientation using a magnetization-prepared rapid gradientecho sequence (TR 8.16 ms, TE 3.18 ms, flip angle 12º, FOV 256 × 256 mm2, 256 × 256 matrix, section thickness 1 mm without intersection gap, voxel size 1 × 1 × 1 mm2, 188 sections). Foam fillers and earplugs were used to minimize head motion and attenuate scanner noise during image acquisition. All participants were instructed to keep their eyes closed and rest without falling asleep during scanning.
Resting state-MRI data preprocessing
All fMRI data preprocessing steps were performed using the WhiteMatter toolkit (https://github.com/jigongjun/Neuroimaging-and-Neuromodulation), which includes selected functions of AFNI,25 SPM12 (http://www.fil.ion.ucl.ac.uk/spm/software/spm12) and FSL (http://fsl.fmrib.ox.ac.uk/fsl). Briefly, preprocessing consisted of the following steps: deletion of the first 5 functional volumes; despiking; slice timing correction and realignment; coregistering individual functional data to structural images and segmenting structural images into grey matter, white matter and cerebrospinal fluid (CSF); regressing out 27 nuisance signals (average white matter, CSF and whole-brain signals as well as 24 head motion parameters, including the 6 head motion parameters, 6 head motion parameters 1 time point before and 12 corresponding squared items26); smoothing of functional images using a 4-mm isotropic Gaussian kernel; temporal band-pass filtering (0.01–0.1 Hz); and spatial normalization to the Montreal Neurological Institute (MNI) space using the matrix produced by structural image segmentation and the DARTEL algorithm of SPM12. Whole scan images were discarded if head motion exceeded 3 mm of translation or 3º of rotation during scanning.
CFH calculation
We developed a new index to measure interhemispheric cooperation, CFH. The method for calculating CFH was described in detail in our previous study.23 Briefly, each voxel was used as a seed to calculate whole-brain FC maps, which were then averaged across all participants. The voxel with the maximal resting-state FC value in the contralateral hemisphere was defined as the homotopic voxel for the seed voxel. Pearson correlations between seed and homotopic voxels were then Z-score transformed to improve the normality. The resulting values were defined as the CFH value used to quantify the interhemispheric cooperation and were used for subsequent group-level analyses. A higher CFH value is considered indicative of stronger communication with the opposite hemisphere.
The homotopic regions of a given voxel were defined as its mirrored voxel in the opposite hemisphere in most previous fMRI studies. However, we define homotopic regions based on function rather than structural features in our study. Here, we calculate the distances between the anatomically homotopic voxels and functionally homotopic voxels (Appendix 1, available at https://www.jpn.ca/lookup/doi/10.1503/jpn.230037/tab-related-content).
Spatial correlation with receptor and transporter density
We also examined whether CFH abnormalities were associated with specific transmitter systems implicated in schizophrenia by comparing the pattern of group differences in CFH values to the densities of various neurotransmitter receptors, transporters and synaptic markers from density atlases. These atlases were obtained from previous positron emission tomography (PET) studies of healthy individuals. Density maps of the following molecules were selected based on known or hypothesized associations with schizophrenia pathology: dopamine receptors (D1R,27 D2R28), serotonin receptors (5-HT1AR,29 5-HT1BR,30 5-HT2AR,30 5-HT4R30), dopamine transporter (DAT),31 serotonin transporter (5-HTT),30 fluorodopa (F-DOPA; a reflection of presynaptic dopamine synthesis capacity),32 γ-aminobutyric acid type A receptor (GABAAR),31 norepinephrine transporter (NAT),33 vesicular acetylcholine transporter (VAChT)28 and metabotropic glutamate receptor (mGluR).28 Subsequently, density values were extracted from each PET atlas, and averaged within 360 regions based on the the HCP-MMP 1.0 atlas (https://cjneurolab.org/).34 We calculated the t values for CFH differences between the schizophrenia and control groups (independent sample t tests) and then extracted the t values and averaged them within 360 regions based on the HCP-MMP atlas. Finally, we used Spearman correlation analysis to investigate the associations between CFH differences (t values) and neurotransmitter receptor and transporter density values. The significance threshold was set at 0.05 (2-tailed) with false discovery rate (FDR) correction.
Statistical analysis
Continuous variables were compared between the schizophrenia and control groups with the Mann–Whitney U test, and categorical variables were compared with the Fisher exact test. Group differences in CFH values at a given location were compared using permutation testing with age and sex as covariates. This comparison test was performed within a grey matter mask using the Statistic nonParametric Mapping (SnPM13) toolbox of SPM12.35 To control for family-wise error in multiple comparisons, we first set a cluster-defined threshold of t = 3.14 (corresponding to p = 0.001 at the voxel level). Only clusters surviving the cluster-level correction at pcorr < 0.01 are reported. Pearson correlation analysis was then used to test the association between imaging data and clinical symptoms in schizophrenia. The significant threshold of correlation analysis was set at 0.05 (2-tailed), uncorrected. For visualization of MRI results we used Surf Ice (https://www.nitrc.org/projects/surfice/), MRIcroGL (https://www.nitrc.org/projects/mricrogl/) and BrainNet software (http://www.nitrc.org/projects/bnv/).36
Results
Participants
We included 86 patients with schizophrenia and 86 healthy controls in the final analysis. There was no significant difference in age or gender ratio between groups. The demographic and clinical characteristics of the schizophrenia and healthy control groups are summarized in Table 1.
Demographic and clinical characteristics of study participants
Group differences in the CFH
The CFH maps of the schizophrenia and healthy control groups exhibited similar patterns (Figure 1 A and B). However, patients with schizophrenia showed significantly lower CFH values in the left middle orbitofrontal cortex (OFC), left postcentral gyrus, right superior OFC, right medial OFC and right inferior temporal gyrus (ITG; Figure 1C). Patients with schizophrenia also showed significantly greater CFH values in the right caudate nucleus (Figure 1C). The details of the 6 clusters with significantly altered CFH values in patients with schizophrenia are summarized in Table 2.
Differences in interhemispheric cooperation between (A) patients with schizophrenia and (B) age- and sex-matched healthy controls. (C) Regions showing significantly weaker interhemispheric cooperation (low connectivity between functionally homotopic voxels) in patients with schizophrenia relative to healthy controls are shown in blue, and regions with stronger interhemispheric cooperation in patients with schizophrenia are shown in red (pcorr < 0.01).
Significant group differences in CFH
To exclude the effect of head motion, we reanalyzed the group differences by adding mean frame displacement (FD) as a covariate. The results were very similar to our original findings. The peak MNI coordinates of the 6 clusters with significantly altered CFH values were the same. Only the number of voxels and the peak t value of the clusters changed slightly (Appendix 1, Table S1).
Correlations with clinical characteristics
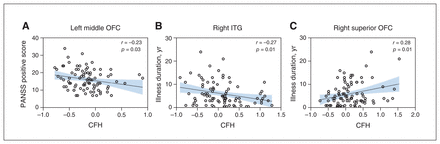
The PANSS positive score was negatively correlated with CFH values of the left middle OFC (r = −0.23, p = 0.03; Figure 2A), while illness duration was positively correlated with CFH values in the right superior OFC (r = 0.28, p = 0.01; Figure 2B) and negatively correlated with CFH values in the right ITG (r = −0.27, p = 0.01; Figure 2C). No significant correlations were found between other CFH values with significant group differences and PANSS total, positive, negative and general scores or illness duration (Appendix 1, Table S2).
Correlations between regional CFH values and clinical characteristics. (A) PANSS positive subscale score was negatively correlated with CFH values in the left middle OFC. Illness duration was (B) negatively correlated with CFH values in the right ITG and (C) positively correlated with CFH values in the right superior OFC. The significance threshold was set at 0.05 (2-tailed), uncorrected. CFH = connectivity between functionally homotopic voxels; ITG = inferior temporal gyrus; OFC = orbitofrontal cortex; PANSS = Positive and Negative Symptom Scale.
Correlations with receptor and transporter densities
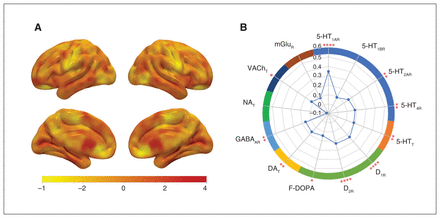
The group difference in CFH (Figure 3A) was positively correlated with the densities of 5-HT1AR (r = 0.35, p < 0.0001), 5-HT2AR (r = 0.16, p = 0.006), 5-HT4R (r = 0.18, p = 0.002), 5-HTT (r = 0.19, p = 0.002), D1R (r = 0.24, p < 0.0001), D2R (r = 0.23, p < 0.0001), F-DOPA (r = 0.15, p = 0.011), DAT (r = 0.17, p = 0.005), GABAAR (r = 0.16, p = 0.006) and VAChT (r = 0.12, p = 0.04) (Figure 3B), but not with densities of the other receptors and transporters examined (Appendix 1, Table S3).
Correlations of group CFH differences with neurotransmitter receptor and transporter densities. (A) The CFH difference (t value) map without correction. (B) The CFH difference was positively correlated with the mean densities of 5-HT1AR, 5-HT2AR, 5-HT4R, 5-HTT, D1R, D2R, F-DOPA, DAT, GABAAR and VAChT. The vertical axis of the radar map indicates the correlation coefficient (r value). 5-HT1AR = serotonin receptor 1A; 5-HT2AR = serotonin receptor 2A; 5-HT4R = serotonin receptor 4; 5-HTT = serotonin transporter; D1R = dopamine receptor 1; D2R = dopamine receptor 2; F-DOPA = fluorodopa; DAT = dopamine transporter; GABAAR = γ-aminobutyric acid type A receptors; mGluR = metabotropic glutamate receptor; NAT = norepinephrine transporter; VAChT = vesicular acetylcholine transporter.*p < 0.05, **p < 0.01, *** p < 0.001 (false discovery rate–corrected, 2-tailed).
Discussion
Interhemispheric cooperation was significantly altered in patients with schizophrenia compared with matched controls, and these abnormalities were correlated with clinical variables and with the densities of multiple neurotransmitter signalling proteins implicated in schizophrenia pathogenesis. Moreover, we found substantially reduced FC within the frontal lobes of patients with schizophrenia. These findings suggest that sub-optimal interhemispheric cooperation, especially in the bilateral OFC, contributes to the cognitive and behavioural deficits in schizophrenia. Further, these interhemispheric cooperation deficits may involve dysfunction of dopamine, serotonin and cholinergic transmitter systems.
Abnormal interhemispheric cooperation in schizophrenia
Hemispheric cooperation in the right caudate nucleus was significantly greater in patients with schizophrenia than healthy controls, in accordance with the results of a previous study.37 The striatum and associated cortical structures contribute to many cognitive and affective processes that are affected in schizophrenia,38–40 and the caudate nucleus is an important node within corticostriatal circuits. Consistent with this finding, several studies have also reported abnormalities in caudate anatomy.41–43 Moreover, the classic dopamine hypothesis posits that the positive symptoms of schizophrenia are associated with hyperactive dopaminergic transmission within the caudate,44,45 and mean dopamine system densities were correlated with the alteration in mean CFH in the current study. Taken together, these results provide evidence that abnormal FC within the right caudate nucleus, possibly involving dopaminergic system dysfunction, contributes to schizophrenia pathology.
Patients with schizophrenia also showed significantly reduced interhemispheric cooperation in a large span of the frontal lobes encompassing the left middle OFC, right superior OFC and right medial OFC. Damage to the OFC affects cognitive functions, emotion regulation and social behaviours, resulting in a host of psychiatric syndromes.46–48 Structural and functional impairment of the OFC have been reported in patients with schizophrenia. For example, studies have provided evidence of volume and thickness reductions of the OFC in medication-naïve patients with schizophrenia.49,50 Previous fMRI studies have shown altered OFC activation during social decision-making in patients with schizophrenia.51 People with OFC damage have been shown to make disastrous life choices, such as bad investments or breaking the law.46 Finally, using voxel-mirrored homotopic connectivity, interhemispheric cooperation in the OFC has been found to be significantly decreased in schizophrenia.18 While our current findings support those of previous neuroimaging studies, the characterization of the role of the OFC is complicated by multiple factors. More research is therefore needed to demonstrate the role of the OFC in psychiatric disorders such as schizophrenia.
We also found significantly reduced CFH values in the right ITG and left postcentral gyrus. The temporal cortex is a key structure for auditory and language processing, both of which are impaired in schizophrenia.52,53 Consistent with our findings, several previous studies have reported impaired interhemispheric cooperation in the temporal cortex of patients with schizophrenia.15,16 In addition, several morphometric studies have reported structure deficits in the temporal gyrus of patients with schizophrenia.54–57 Our findings suggest that impaired connectivity with the right ITG may contribute to schizophrenia pathology. Furthermore, this dysfunction appears to worsen with disease progression, as we found a negative correlation between interhemispheric cooperation and disease duration. The postcentral gyrus receives and processes somatosensory information,58–60 and reduced interhemispheric cooperation of the postcentral gyrus has also been reported in schizophrenia.9,15 A growing body of literature indicates that the somatosensory cortex contributes to emotional and cognitive processing, and impaired somatosensory processing is a common symptom of schizophrenia.61 Future studies are warranted to examine whether sensory impairments in schizophrenia contribute to deficits in higher-order cognition.
Correlations with molecular substrate densities
Neurotransmitter receptors and transporters support the propagation of signals and shape network-wide communication in the human brain. The dopamine hypothesis was the first neurochemical theory of schizophrenia, and it is still widely accepted to explain some symptoms of the disorder.62 We found that group differences in CFH between patients with schizophrenia and healthy controls were significantly correlated with the densities of D1R, D2R, DAT and F-DOPA. The dopamine system is implicated in motor behaviour, cognition and emotion, all of which are affected by schizophrenia.63 The classic formulation of the dopamine hypothesis posits that hypoactive dopamine transmission in the prefrontal cortex leads to negative symptoms such as flat affect and anhedonia, while hyperactivity of mesolimbic dopamine neurons leads to positive symptoms.45,64 This theory is strongly supported by the efficacy of dopamine receptor antagonists to treat behavioural and cognitive symptoms in patients with schizophrenia.
However, it has long been appreciated that dopaminergic dysfunction alone cannot account for the full spectrum of psychopathology in schizophrenia.65,66 Consistent with contributions of serotonergic and cholinergic signalling dysfunctions in schizophrenia, we found significant positive correlations between the mean group difference in CFH and the densities of 5-HTT, GABAAR and VAChT. The serotonin system interacts with the dopamine system at multiple levels. Moreover, 5-HTT signalling is critical for proper neurodevelopment, and the appearance of prodromal symptoms before clinical disease suggests neurodevelopmental deficits in schizophrenia. Furthermore, changes in 5-HTT are associated with cognitive deficits in patients with schizophrenia.67 Associations of both serotonergic dysfunction and 5-HTT polymorphisms68 with schizophrenia pathophysiology have also been reported.66,69 Moreover, some newly developed antipsychotic medications preferentially target the serotonin system.70,71 The pathophysiology of schizophrenia and/or symptom expression may also involve altered cholinergic transmission. Tobacco smoking rates are high among patients with schizophrenia, suggesting self-medication with nicotine (a nicotinic acetylcholine receptor [nAChR] agonist).72–75 Reduced expression of α7 ionotropic nAChR expression has been observed in the postmortem hippocampus and cingulate cortex of patients with schizophrenia.74,76 The role of GABA in patients with schizophrenia has also been reported in clinical and basic neuroscience studies.77,78 The GABA system has been shown to be effective in improving symptoms of schizophrenia.79
Recent studies have implicated glutamate and norepinephrine in addition to dopamine, serotonin, GABA and acetylcholine in the pathobiology of schizophrenia.80 However, our findings show that the density maps of these neurotransmitter receptors and transporters were independent of the CFH group differences. These molecular substrates may be involved in other cognitive dysfunction or symptoms of schizophrenia, but not in interhemispheric cooperation. Our findings may provide a host of additional therapeutic targets for schizophrenia. However, further research is needed to elucidate the potential therapeutic mechanisms and roles of different neurotransmitters in the treatment of schizophrenia.
Limitations
This study has several limitations. First, all patients with schizophrenia were treated with antipsychotic medications before the scan, which could have had unpredictable effects on our findings. Therefore, the alterations of interhemispheric cooperation should be investigated in drug-naïve patients with schizophrenia. Second, we used a cross-sectional design to investigate interhemispheric cooperation. A longitudinal study is necessary to assess how abnormalities in interhemispheric cooperation emerge and change with disease progression. Third, the sample sizes of some receptor and transporter atlases were relatively small and came from healthy individuals. Atlases from a large sample of patients with schizophrenia are necessary to validate our findings.
Conclusion
We have shown that interhemispheric cooperation is significantly altered in patients with schizophrenia and that these alterations are associated with clinical symptoms. In addition, region of interest–based analysis showed markedly reduced FC between bilateral frontal lobes in patients with schizophrenia. Finally, abnormal CFH values were correlated with the densities of several neurotransmitter receptors and transporters. Interventions to enhance interhemispheric cooperation, especially in the frontal lobe, may be an effective treatment strategy for schizophrenia.
Acknowledgment
The authors thank the participants for taking part in this study. They also thank the Information Science Laboratory Center of University of Science and Technology of China for measurement services.
Footnotes
↵* These authors contributed equally to this work.
Competing interests: None declared.
Contributors: K. He, Q. Hua, G.-J. Ji and K. Wang designed the study. Y. Zhang, X. Yao, Y. Yang, W. Xu, J. Sun, L. Wang and A. Wang acquired the data, which Q. Hua and Q. Li analyzed. K. He, and Q. Hua wrote the article, which all authors reviewed. All the authors had full access to all the data in the study, take responsibility for the integrity of the data and the accuracy of the data analysis, and approved the final version for publication.
Funding: This study was funded by the National Natural Science Foundation of China, grant/award numbers 31970979, 82090034, and 81971689; the Anhui Provincial Key Research and Development Project (Population Health Special Project, 202104j07020033); the Hefei Comprehensive National Science Center Hefei Brain Project; the Key Laboratory of Philosophy and Social Science of Anhui Province on Adolescent Mental Health and Crisis Intelligence Intervention (SYS2023B10); the key project of applied medicine research in 2021 of Hefei Municipal Health Committee (Hwk2021zd013); and the Collaborative Innovation Center of Neuropsychiatric Disorders and Mental Health of Anhui Province (2020xkjT057).
- Received March 2, 2023.
- Revision received June 26, 2023.
- Accepted September 5, 2023.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/