Abstract
Background: Deficits in error processing are reflected in an inability of people with externalizing problems to adjust their problem behaviour. The present study contains 2 meta-analyses, testing whether error processing — indexed by the event-related potentials error-related negativity (ERN) and error positivity (Pe) — is reduced in children and adults with externalizing problems and disorders compared to healthy controls.
Methods: We conducted a systematic search in PubMed (1980 to December 2018), PsycInfo (1980 to December 2018) and Scopus (1970 to December 2018), identifying 328 studies. We included studies that measured error processing using the Eriksen flanker task, the go/no-go task or the stop-signal task in healthy controls and in adults or children with clearly described externalizing behavioural problems (e.g., aggression) or a clinical diagnosis on the externalizing spectrum (e.g., addiction).
Results: Random-effect models (ERN: 23 studies, 1739 participants; Pe: 27 studies, 1456 participants) revealed a reduced ERN amplitude (Hedges’ g = 0.44, 95% confidence interval [CI] 0.29 to 0.58) and a reduced Pe amplitude (Hedges’ g = −0.27, 95% CI −0.44 to −0.09) during error processing in people with externalizing problems or disorders compared to healthy controls. Type of diagnosis, age and the presence of performance feedback or comorbidity did not moderate the results. The employed cognitive task was a moderator for Pe but not for ERN. The go/no-go task generated a greater amplitude difference in Pe than the Eriksen flanker task. Small-sample assessment revealed evidence of publication bias for both event-related potentials. However, a p curve analysis for ERN showed that evidential value was present; for Pe, the p curve analysis was inconclusive.
Limitations: The moderators did not explain the potential heterogeneity in most of the analysis, suggesting that other disorder- and patient-related factors affect error processing.
Conclusion: Our findings indicate the presence of compromised error processing in externalizing psychopathology, suggesting diminished activation of the prefrontal cortex during performance monitoring.
Introduction
Externalizing problem behaviour has been associated with problems in cognitive control,1 of which error processing is an important component.2,3 People with externalizing problems or disorders are characterized by disruptive and problematic behaviour that is directed outward to the environment and are further referred to as externalizing samples. Specific diagnoses and behaviours that belong to the category of externalizing samples include attention-deficit/hyperactivity disorder (ADHD), oppositional defiant disorder, psychopathy, conduct disorder, aggression, antisocial personality disorder, substance use disorder (SUD) and delinquency.4 Error-processing, which refers to the ability to detect errors and evaluate performance, allows for the adaptation of behaviour to correctly react to stimuli from the environment.5 Deficits in error processing can be reflected in a failure to adjust behaviour, which is indicative of externalizing psychopathology. Deficits in error processing can be detected by electroencephalography (EEG) and are reflected in a diminished amplitude of the event-related potentials (ERP) error-related negativity (ERN) and error positivity (Pe). Several studies have investigated differences in the ERN (e.g., Lo6 in children and adolescents with externalizing problem behaviour) and Pe (e.g., Luijten and colleagues7 in substance use disorder), but a systematic review is lacking in children and adults that combines externalizing samples and includes comparison with healthy controls. This study investigates the ERN and Pe across different externalizing samples to determine error processing deficits in children and adults with externalizing problems or disorders compared to healthy controls.
Error-related negativity
The ERN8,9 (or negativity error; Ne)10 is a negative deflection that occurs approximately 50 ms to 100 ms after commission of an error.11,12 This ERP waveform peaks at the frontocentral electrodes, reflecting the neuronal activity of the anterior cingulate cortex13 during error processing.3,5,14 The ERN is a robust and reliable15,16 neurobiological marker that reflects the brain’s initial reaction to an error and the start of error processing, whether or not the person is conscious of the error.17,18 Several theories outline the functional significance of the ERN (for an overview, see Loo and colleagues19 and Olvet and Hajcak20), including mismatch theory, motivational significance theory, reinforcement and learning-based theory, and conflict monitoring theory. These theories describe different processes of error and conflict detection, as well as the role of the dopaminergic system of the brain and the anterior cingulate cortex. Previous work has suggested that the ERN can serve as a candidate endophenotype for psychopathology, especially for internalizing disorders. Meta-analyses of internalizing samples show that the ERN appears to be increased in patients with anxiety,21 obsessive–compulsive disorder22 and depression disorders23 compared to healthy controls. A recent meta-analysis concluded that the ERN can serve as a transdiagnostic marker for internal and externalizing disorders.24 The current study is an extension of this meta-analysis, including child samples as well as adult samples, and also including psychopathy samples. In addition, we have used effect size calculation rather than effect size estimation, and included the late error processing component Pe as well as the ERN.
Deviating activation patterns in the brain with respect to cognitive control have been found in externalizing behaviour.20,25 Some studies have reported decreased ERN amplitude in ADHD (e.g., Wiersema and colleagues26) and addiction (e.g., Zhou and colleagues27), but other reports have found no differences in the ERN compared to healthy controls (e.g., in addiction28 and ADHD29). Some studies have even reported increased ERN amplitude in ADHD30 and addiction31 compared to controls. The presence of comorbid internalizing problems has been suggested as a possible explanation for these mixed results, as illustrated by the study of Schellekens and colleagues31 in a sample of patients with alcohol dependence. Although medication (e.g., Groom and colleagues32), age6 and the experimental paradigm15 have been studied as moderating factors for ERN amplitude, it remains unclear whether these variables influence ERN results across externalizing samples. Furthermore, several studies have reported that performance feedback during tasks can influence error processing.33–35 When participants receive feedback on performance, they become cautious of their response accuracy, inducing greater reactions to errors.
By conducting a meta-analysis, we were able to integrate inconsistent findings to shed light on the role of the ERN in externalizing behaviours. Moreover, by explicitly testing medication use, age, comorbidity, experimental paradigm and performance feedback as moderators, we were able to investigate whether or how they account for variability in ERN studies.
Error positivity
Another ERP component relevant for performance monitoring is Pe amplitude. The Pe is a slow, positive deflection, peaking at approximately 200 ms to 600 ms after an error; it is measured across the centroparietal area.10,36,37 The Pe is said to reflect the conscious awareness of errors and error processing.37 It is an independent ERP component, despite the fact that it follows directly after the ERN and shows similarities with the P300 component (for example, the latency window). (For further reading on the similarities and differences between these components, see Arbel and Donchin,36 Overbeek and colleagues,37 Davies and colleagues38 and Ridderinkhof and colleagues.39) The functional significance of the Pe has been described in several hypotheses,12,37 including the affective processing hypothesis (in which the Pe reflects the emotional appraisal of the error), the behaviour-adaptation hypothesis (in which the Pe indicates performance adjustment after error) and the error awareness hypothesis (in which the Pe reflects the conscious recognition of the error committed). Although empirical evidence is needed to support these hypotheses (specifically in terms of the neural generators of Pe), they suggest that diminished Pe could be related to deviant activity of the rostral part of the anterior cingulate cortex.14,40
Compared to controls, Pe reductions in error processing for people with externalizing problems or disorders have been observed more consistently than ERN reductions, although some discrepancies have been found across studies. For instance, some studies have found diminished Pe amplitudes in people with ADHD (e.g., Albrecht and colleagues41) and substance use (e.g., Franken and colleagues42), but these were not replicated in subsequent studies (e.g., addiction studies28,43). Moreover, other studies have found a reverse effect, indicating that increased Pe amplitudes are related to externalizing behaviour (e.g., in addiction44 and ADHD45). This is the first meta-analysis to summarize Pe findings in externalizing samples compared to healthy controls. We have examined the moderators suggested for the ERN, above, to try to better understand these discrepancies in study findings.
In the current study, we aimed to investigate whether the ERN and Pe were different in children and adults with externalizing problems or disorders compared to healthy controls. We use meta-analysis and focused on the mean amplitude of the ERN at the midline frontocentral electrode (FCz; for subsequent analyses to investigate the effect of other midline electrode sites Fz and Cz, see Appendix 1, available at jpn.ca) and the Pe at the midline central electrode (Cz). We expected that both the ERN and Pe amplitudes would be reduced in the externalizing groups, indicating deficits in error processing. To explain the mixed results found in this field of research, we investigated potential heterogeneity by adding type of diagnosis, presence of comorbidity, experimental paradigm, age and medication use as moderators in both analyses. Except for comorbidity (comorbid internalizing or externalizing symptoms or a combination of both) and performance feedback, we expected that the moderators would not influence ERP amplitudes. We did not expect that effect size variability would be explained by the experimental paradigm, because these tasks often elicit highly correlated amplitudes and have high construct validity.15,46 In cases of comorbidity, we expected that differences in ERN and Pe between the clinical and control groups would be smaller for samples that had internalizing comorbid symptoms, and greater for samples with externalizing symptoms. We also expected that the presence of performance feedback would elicit a greater ERN and Pe than no performance feedback.
Methods
We did not preregister this study, but to enhance reproducibility and accommodate the open science community, our data and code are available at the Open Science Framework (https://osf.io/dkxtp/). We determined a search strategy and inclusion and exclusion criteria before our literature search. Secondary to the steps undertaken as described in this report, we reviewed the relevant literature,47 consulted experts and compiled study-related factors to ensure that we were informed about the state of art in this field. We intended to identify as many EEG studies that evaluated ERN and Pe magnitudes in case–control (externalizing samples v. healthy volunteers) studies.
Search strategy
We conducted the literature search using 3 databases: PsycInfo (1980 to December 2018), PubMed (1980 to December 2018) and Scopus (1970 to December 2018). Search terms included the following: inhibit*, cognitive or inhibitory control, error processing or monitoring, external* symptoms, disorders and problems, alcohol, cocaine, stimulants, heroin, smoking, cannabis, substance abuse, substance use-, dependence-, misuse, alcoholism, ADHD, ADD, antisocial personality disorder, oppositional defiant disorder, aggression, psychopathy, intermittent explosive disorder, conduct disorder, antisocial behaviour, behavioural problems or disorders, psychopathic traits and callous-unemotional traits. We cross referenced the above terms with the following: error-related negativity, error positivity, ERP, EEG, Eriksen flanker task, go/no-go task and stop-signal task. For complete search strategy queries by database, see Appendix 1.
Eligibility criteria
We assessed studies identified from the literature search, together with studies identified from other sources, according to the following inclusion criteria: studies were published in peer-reviewed journals in English and performed in human volunteers of any age; studies addressed error processing using the EEG components ERN (at Fz, FCz and Cz) or Pe (Cz), irrespective of the latency window; the ERN and Pe were measured during the Eriksen flanker task,48 the go/no-go task or the stop-signal task; studies included a healthy control group of participants with no clinical or neurologic diagnosis; and participants in the patient groups were recruited because they had a clinical diagnosis of an externalizing disorder (based on the Diagnostic and Statistical Manual of Mental Disorders, fifth edition [DSM-5] or the International Classification of Diseases, 10th revision [ICD-10], or earlier versions) or they showed subclinical levels of externalizing problems. Studies were excluded according to the following criteria: means and standard deviations of the ERN or Pe amplitude for both groups (derived from averaging ERN and Pe epochs where the peak was the maximum from error trials) were unavailable from the published report or after contact with the authors; studies used adjusted paradigms (e.g., lack of neutral stimuli presented) or stimuli that were not presented visually; and studies used the continuous performance task or error awareness task (despite including go/no-go elements).
Data extraction
Records identified through the literature search were imported to Mendeley. In this program, we screened titles and abstracts using our inclusion criteria. To avoid unwanted exclusion, all articles in which the abstract did not contain full information were kept for further reading. Next, we downloaded and read the full text of articles that had passed the screening stage, and we reviewed their reference lists to identify additional studies for potential inclusion. The first author (ML) extracted relevant information from the included studies, retrieving sample characteristics such as sample size, age, sex ratio, disorder (ADHD, addiction or other externalizing disorders), condition (clinical v. subclinical), the presence of comorbidity (if known, coding for externalizing, internalizing or mixed problems), the use of medication and pretesting group differences. We assessed patients’ diagnostic status (clinical or subclinical) by extracting the diagnostic tools used (DSM III or IV or ICD-10) and details about the informants (specialist, self-report, parent, teacher, or medical or legal reports). A study was considered clinical when the diagnosis was obtained by a trained psychologist or psychiatrist, or when participants were recruited from inpatient treatment facilities. Offenders incarcerated for serious crimes were also considered to be clinical. We categorized the studies into 5 diagnosis groups: child and adult ADHD, clinical and subclinical addiction (adults only) and “other.” Studies with samples of offenders, people with multi-problem behaviour and people with high scores on psychopathy or aggression measures were considered “other,” leaning toward a sample with forensic characteristics. Participants were considered subclinical when no diagnosis was determined, but when diagnostic tools or self-reports indicated heightened levels of externalizing problems. To be included in the final analysis, the eligible study had to report a cut-off score or level for the diagnostic tool. People were considered healthy controls when no clinical or neurologic diagnosis or disabilities were reported. Comorbidity was coded as any co-occurring symptom or (sub)clinical level of other internalizing (e.g., anxiety) or externalizing (e.g., conduct) problems.
We also gathered information about the experiment, including the cognitive task used, whether the task was adjusted (e.g., instructions the participants received) and latency windows. For studies that used multiple experimental manipulations, we systematically selected the first or baseline time point, the neutral stimuli trials and, when multiple tasks were presented, the Eriksen flanker task. We requested the mean and standard deviation of ERN and Pe amplitudes of error trials by contacting authors when articles did not provide this information. We also requested unpublished data, but those requests did not lead to viable data for our analysis.
Two authors (I.V. and M.M.) independently extracted information from the manuscripts to verify the work of the first author. For categorical variables, Cohen’s κ was between 0.79 and 0.81, indicating strong level of agreement. For continuous variables, intraclass correlation was between 0.97 and 0.99, which was near-perfect agreement. We evaluated the selected studies primarily on their choice of sample and their experimental design. We (M.L., I.V., M.M. and I.F.) discussed whether the selected studies adhered to our inclusion and exclusion criteria and whether they were similar enough to be compiled.
Data analysis and small sample bias assessment
For both the ERN and Pe meta-analyses, we assumed a random model because of variance in the estimates due to different clinical disorders and experimental tasks administered.49 We used restricted maximum likelihood estimation to estimate between-study variance.50 As recommended by Veroniki and colleagues,51 we ran analyses with the DerSimonian–Laird and Sidik and Jonkman estimators to determine sensitivity, but restricted maximum likelihood estimation resulted in a better model fit. We computed standardized mean difference (SMD; Hedges’ g52) from the means and variances of the ERN and Pe amplitudes, including factor J to reduce overestimation of the bias induced by small sample sizes. For studies with multiple externalizing groups, we adjusted the weights appointed to effect sizes by splitting the N of the control group.53 This was to avoid unit-of-analysis errors or double-counting problems evoked by multiple testing of the control groups. For the ERN, a positive SMD indicated reduced amplitude for the externalizing group. For the Pe, a negative SMD indicated reduced amplitude for the externalizing group. Both SMDs indicated a diminished electrocortical reaction after the error. Effect sizes of 0.2 to 0.3 were considered small; effect sizes of approximately 0.5 were considered medium; and effect sizes of 0.8 and higher were considered large.54 We investigated influential or outlier studies based on the recommendations of Viechtbauer and Cheung.55 We evaluated the degree of heterogeneity using I2, where a larger value indicated increasing variety in effect sizes.56,57 We performed subgroup or moderation analyses when heterogeneity (Cochran’s Q) was significant. Moderation analyses for clinical disorder, comorbidity, medication use, experimental task and a meta-regression of age were determined a priori. Reviewers also suggested that we test the effect of performance feedback and electrode site (Appendix 1, section 4) as additional moderators.
We examined small sample study bias by assessing asymmetry in funnel plots, applying Egger’s test of the intercept58 and Duval and Tweedie’s trim-and-fill procedure.59 To detect whether small samples distorted the funnel plots created using SMD,60 we performed a sensitivity analysis using adjusted funnel plots with 1 √ n on the y axis as a precision estimate, rather than the standard error. In the end, the adjusted funnel plots also detected asymmetry as a result of publication bias, not only small sample size. We also evaluated the robustness of the effects we found with the fail-safe N calculation using the Orwin approach.61 However, these assessments have their limitations,62 so we also performed a p curve analysis62–65 (Appendix 1, section 5) to inspect whether significant p values (p < 0.0562) provided proof of evidential value (for a full description and application of this assessment, see Harrer and colleagues53). As recommended by van Aert and colleagues,66 a p curve analysis is conducted only when I2 is less than 50% and studies’ effects are in one direction, to allow for robust conclusions. All analyses were performed in R (version 1.3.959), using meta (version 4.12.0),67 metafor (version 2.4.0)68 and dmetar (version 0.0.9000),53 guided by the instructions of Harrer and colleagues.53 All significance tests were conducted at a significance level of 5%.
Results
Selected studies
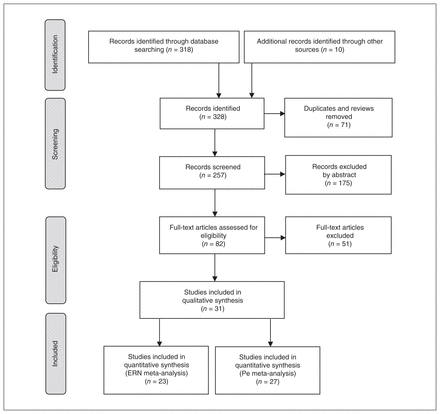
Figure 1 shows a flow chart of the literature search. Where applicable and possible, we adhered to PRISMA guidelines (Appendix 1, section 2). The search of databases and additional sources yielded a total of 328 records. After removing duplicates and reviews (n = 71), we screened the abstracts of 257 studies. We then assessed the full text of the 82 articles that met our inclusion criteria. We included 31 articles for qualitative analysis, of which 23 were ERN studies at the FCz electrode (27 effect sizes; n = 1739) and 27 were Pe studies at the Cz electrode (31 effect sizes; n = 1456). We found no studies that used the stop-signal task. Descriptive information for the included studies is shown in Table 1; further details of the included studies are shown in Appendix 1.
PRISMA flow diagram. ERN = error-related negativity; Pe = error positivity; PRISMA = Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
Characteristics of included studies
ERN summary effect
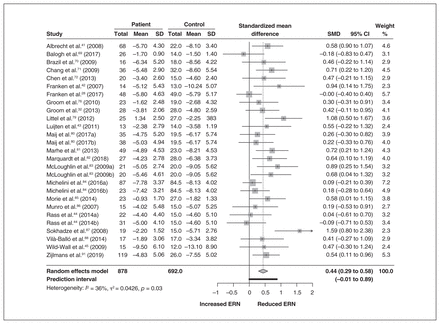
The ERN meta-analysis included 23 studies and 1739 participants. We found a small to medium overall effect size (g = 0.44, 95% CI 0.29 to 0.58, p < 0.01). This indicated that in the patient group, the ERN had a decreased negative amplitude compared to healthy controls. Between-study variability was 36%, indicating a low to moderate amount of variability in effect sizes. The test for heterogeneity was significant (Q26 = 40.69, p = 0.03), which gave us cause to perform moderation analysis. We identified the studies of Sokhadze and colleagues87 (high effect size) and Michelini and colleagues84 (large sample size) as influential cases. However, we kept these studies in the overall analysis because they did not influence the overall model. A forest plot for the ERN is presented in Figure 2.
Overall ERN meta-analysis, including a forest plot. CI = confidence interval; ERN = error-related negativity; SD = standard deviation; SMD = standardized mean difference.
ERN subgroup analyses
Moderation analyses revealed no significant difference in ERN amplitudes between diagnosis groups (Q4 = 0.66, p = 0.96). Comorbidity did not significantly influence the ERN amplitudes (Q3 = 5.11, p = 0.16), and the type of experimental paradigm was not a moderator (Q1 = 0.01, p = 0.91). The presence of performance feedback also did not account for variability in ERN effect size (Q1 = 0.08, p = 0.78). We had initially intended to test the effect of medication, but this variable was confounded in the sample of ADHD participants, making further investigation futile. For study details, see Appendix 1 (medication, section 4; electrode site, section 5). Table 2 provides an overview of the moderation results for the categorical variables. A meta-regression with age as a predictor revealed that age was not associated with the effect sizes (F1,25 = 2.30, p = 0.14).
Results of multiple moderator analyses for ERN
ERN small sample study bias
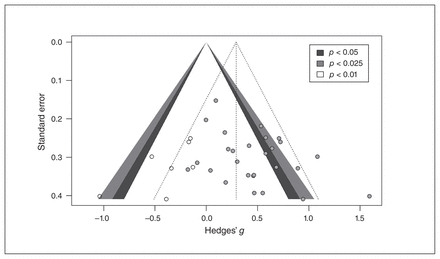
To investigate publication bias, we visually inspected the funnel plots of the effect sizes. The Egger’s intercept of the funnel plot was significant (B = 2.09, p = 0.03), indicating evidence of publication bias. The funnel plot in Figure 3 applies Duval and Tweedie’s trim-and-fill procedure. Application of this procedure revealed that by filling 7 studies, the overall effect would be reduced to small (g = 0.29, 95% CI 0.12 to 0.45, p = 0.001), indicating the presence of bias. Despite this bias, 27 studies were needed to get to an unweighted average effect size of 0.24 using the fail-safe N test. However, a p curve analysis (k = 9) revealed the presence of right skewness of the significant p values, and of evidential value (half: Z = −2.33, p = 0.009; full: Z = −1.43, p = 0.08). The flatness test was not significant (half: Z = 2.73, p = 0.99; full Z = −0.32, p = 0.37). Although the analysis was underpowered (25%), typical in this field, it indicated that there was most likely no selective reporting of p values. The p curve estimate of the average “true” effect size was 0.32, which was similar to the trim-and-fill result of 0.29 and lower than the combined effect size of the overall analysis (0.44). For a full report, including the disclosure table, a p value distribution figure and results of the p curve analysis, see Appendix 1, section 6.
Funnel plot including filled studies for error-related negativity.
Pe summary effect
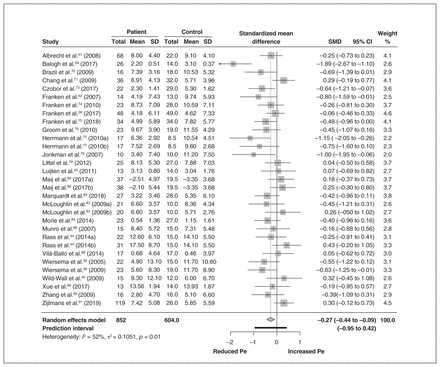
The Pe meta-analysis included 27 studies, incorporating 31 effect sizes and 1456 participants. We found a small to medium overall effect size (g = −0.27, 95% CI −0.44 to −0.09, p = 0.004), indicative of decreased amplitude of the Pe waveform for the externalizing group compared to controls. We observed a moderate degree of heterogeneity (I2 = 52%, Q30 = 62.74, p = 0.004), which gave us cause for further exploration of effect size variability through subgroup analysis. A forest plot for the Pe is presented in Figure 4.
Overall Pe meta-analysis, including a forest plot. CI = confidence interval; Pe = error positivity; SD = standard deviation; SMD = standardized mean difference.
Pe subgroup analysis
Diagnosis was not a moderator for the Pe effect sizes (Q4 = 5.17, p = 0.22), nor was comorbidity (Q3 = 1.61, p = 0.66). The presence of feedback did not account for variability in Pe effect size (Q1 = 2.58, p = 0.12). The experimental paradigm was a moderator in this meta-analysis. The go/no-go task generated a greater difference in Pe amplitudes (SMD = −0.54, k = 9) than the Eriksen flanker task (SMD = −0.15, k = 22; Q1 = 4.17, p = 0.041). Similar to the ERN, medication use was confounded in the ADHD sample, making further moderation analysis ineffective (Appendix 1, section 4). Age did not explain the variability in effect sizes for Pe (F1,27 = 0.02, p = 0.88). Table 3 shows the results of the moderation analysis for the categorical variables.
Results of multiple moderator analyses for Pe
Pe small sample study bias
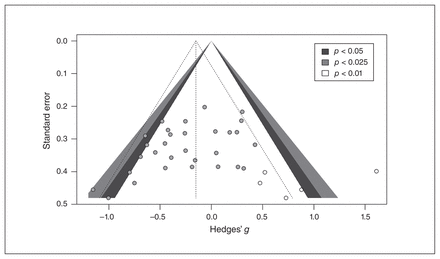
The Egger’s intercept (B = −2.45, p = 0.019) was significant, indicating asymmetry in the funnel plot for the Pe studies. The funnel plot in Figure 5 includes studies that needed to be added to make the plot symmetrical using Duval and Tweedie’s trim-and-fill procedure. This procedure revealed that by filling 5 studies, the overall effect was reduced (g = −0.15, 95% CI −0.35 to 0.05), meaning that the Pe meta-analysis could be contaminated by publication bias. However, the fail-safe N assessment revealed that there needed to be 31 effect sizes to achieve the unweighted effect size of −0.16. We performed a preliminary p curve analysis, but it was inconclusive (see Appendix 1, section 6, for explanation).
Funnel plot including filled studies for error positivity. Standard error
Discussion
This meta-analysis collated current EEG studies on error processing to test whether the ERN and Pe amplitude were different in people with externalizing problems or disorders compared to controls. As expected, we found diminished ERN and Pe amplitude for people with externalizing problems or disorders compared to controls. These findings confirmed compromised error processing in the externalizing spectrum, regardless of a specific diagnosis or problem behaviour. For both ERP components, we found a considerable degree of heterogeneity. The variation in results was not explained by comorbidity, the presence of performance feedback, age or type of clinical disorder. The experimental paradigm was a moderator for the Pe studies, but not for the ERN studies. Our results for the ERN were in line with a recent meta-analysis by Pasion and Barbosa24 and studies that described error processing deficits in separate externalizing disorders (such as Luijten and colleagues7 and Olvet and Hajcak20 for substance use disorders; Shiels and Hawk92 for ADHD; and Lo6 for children with externalizing symptoms). This was the first meta-analysis to explore age effects in error processing and to confirm deficits in the late error processing component (Pe) for children and adults with externalizing problems and disorders.
Diminished ERN and Pe imply a deviant activation pattern of the dorsal anterior cingulate cortex.93 More specifically, when we consider reinforcement and learning-based theories of the function of the ERN, decreased ERN amplitude could be indicative of abnormal dopamine activity in the midbrain, affecting processes of error and conflict detection. Dysfunction in the dorsal anterior cingulate cortex is also indicative of deficits in inhibitory control and conflict monitoring.3,7,85 Problems in inhibitory control and conflict monitoring (among other cognitive impairments) have been related to symptomatology in externalizing disorders, such as craving in addiction.94 Based on the proposed hypotheses for Pe,37 affected Pe amplitudes reflect differences in conscious recognition of the error committed, differences in emotional appraisal of the error and its consequences, or distinct behavioural adjustment after errors. In turn, such deviation in the processing of errors could imply reduced insight in aberrant and unwanted behaviour for externalizing samples.
The data revealed a considerable amount of heterogeneity for both ERPs, but type of diagnosis, age and the presence of performance feedback or comorbidity did not moderate the results. Studies that controlled for medication use in their samples appeared mostly to be in ADHD, preventing us from examining medication use across other externalizing samples. Although medication use is evident in externalizing samples, many studies have not reported or controlled for medication. Future experiments and systematic reviews should consider the effect of medication on error processing components for different externalizing samples. The experimental task did moderate the association of externalizing problems with Pe and not ERN: we found a greater Pe amplitude difference between the patient group and the control group in the go/no-go task than in the Eriksen flanker task. Although both tasks are known to reliably elicit an electrophysiological reaction after an error,15,46 it is possible that the go/no-go task elicits a stronger reaction to an error than the Eriksen flanker task. As well, it is possible that the Eriksen flanker task allows the participant to be more unconscious of an error than the go/no-go task, because the Pe is said to reflect conscious awareness of the error.37 Finally, contrary to our expectations, comorbidity did not affect the ERN or Pe amplitude in this sample. Although we proposed to test whether internalizing comorbid problems influenced the ERPs, not enough studies were included to properly test this hypothesis and draw firm conclusions.
We performed small bias assessment to investigate the effect of the published data in this study. For the ERN, the result of the trim-and-fill procedure remained significant even after adding 5 studies. The estimated “true” effect size was included in the confidence interval of the overall model, along with evidence of the evidential value from the p curve analysis, indicating that the combined effect size for the ERN was robust. However, for the Pe, we found evidence of publication bias (although an inconclusive p curve analysis), because the trim-and-fill analysis reduced the effect size to nonsignificant. Although this nonsignificant effect could have been because of large heterogeneity, reflected in the broad confidence interval, we should be cautious in drawing firm conclusions about the Pe meta-analysis.
Limitations
We should acknowledge the limitations of this study and the methodological considerations of EEG research in general. The present meta-analysis included studies with ERN amplitudes on the FCz electrode and Pe amplitudes on the Cz electrode, generated by the Eriksen flanker and go/no-go tasks. These criteria allowed for solid results (supplementary analysis revealed no effect of electrode site), but other electrodes (see Arbel and Donchin36 for a summary of reliable electrodes for error processing), neurophysiological measures (e.g., functional MRI) and experimental paradigms can be used to examine error processing. Future research should be directed at investigating whether similar error processing deficits are found at other electrode sites and using other tasks. In addition, investigating deficits in biobehavioural markers of performance monitoring, such as post-error slowing (indicative of response caution for maintaining accuracy) and cortisol involvement95 can shed light on responses and behaviour after errors, elucidating different behaviour patterns. Although we considered a substantial number of potential moderators in this study, other sample characteristics could have accounted for differences in the ERP findings. For example, the global assessment of functioning of patients with a specific psychiatric disorder could influence the magnitude of deficits in error processing. Future studies could examine whether the severity of symptoms within a disorder is related to the degree of diminished reaction (that is, correlational measures with ERP amplitudes). As well, individual differences such as personality traits have been known to influence the ERN and Pe,6,37 and we did not control for these factors in this study. Furthermore, we should be cautious of the results (particularly indicated by the publication bias assessment) because of variations in quality in the EEG experiments. Although the current study evaluated the included experiments, differences between experiments could have influenced our results. We could not assess some aspects of experimental design, such as the manner in which the ERN or Pe were quantified or the effects of task adjustments, because this information was not provided in the reports. To address the possible effect of experimental design differences on the associations between ERP or Pe with externalizing problems in the future, we encourage researchers to disclose the following information: the minimum number of trials or errors for a reliable ERP calculation (e.g., 816 or 696 trials for a reliable ERN); which trials were used (incongruent or error trials) to calculate the ERP; and a clear description of the task instructions and adjustments (e.g., error rate to ensure task difficulty, participant instructions or feedback to influence performance); and other potential confounding variables such as medication use and latency window.
Future work could investigate error processing in specific externalizing disorders that are underexplored in the current literature, such as antisocial personality disorder, specific addictions (e.g., Internet addiction disorder) and double diagnoses (e.g., addiction and a personality disorder). We recommend that future work examine the predictive value of the ERN and Pe using large-scale longitudinal designs to elucidate their role in the etiology of these disorders. We also encourage researchers to assess the feasibility of interventions aimed at improving error processing. To improve error processing abilities in patients, next steps for future experiments could include examinations of the effectiveness of behavioural training, medication and neuromodulation techniques.
Conclusion
Our meta-analysis showed that the neurophysiological correlates of error processing, ERN and Pe, were reduced in children and adults with externalizing problems or disorders. However, we found considerable heterogeneity that could not be explained by the moderators explored in this study; this warrants further exploration and limits strong conclusions. Future research can elucidate the role of individual differences, symptom severity and experimental characteristics in error processing deficits specific to externalizing disorders. With the knowledge that the EEG correlates of error processing are affected in people with internalizing problems and could serve as a possible marker for these disorders, we propose that reduced ERN and Pe could be considered markers for the externalizing spectrum of disorders.
Acknowledgments
Thank you to Judith Gulpers for her comments on my literature search. Thank you to Peter Clayson for his answers on analysis questions. Thank you to Arnout Boot for his help in R. We are grateful to all of the authors who provided with additional information for this manuscript.
Footnotes
Funding: This work was supported by the Erasmus Initiatives for Vital Cities and Citizens, Erasmus University Rotterdam, the Netherlands.
Competing interests: None declared.
Contributors: M. Lutz, R. Kok, S. Koot and I. Franken designed the study. M. Lutz and I. Verveer acquired the data, which M. Lutz, M. Malbec and P. van Lier analyzed. M. Lutz wrote the article, which all authors reviewed. All authors approved the final version to be published and can certify that no other individuals not listed as authors have made substantial contributions to the paper.
- Received February 13, 2020.
- Revision received July 3, 2020.
- Accepted July 4, 2021.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/